Unit 6: Thermochemistry
1/41
Earn XP
Description and Tags
6.0 Introduction, 6.1 Endothermic and Exothermic Processes, 6.2 Energy Diagrams, 6.3 Heat Transfer and Thermal Equilibrium, 6.4 Heat Capacity and Calorimetry, 6.5 Energy of Phase Changes, 6.6 Introduction to Enthalpy of Reaction, 6.7 Bond Enthalpies, 6.8 Enthalpy of Formation, 6.9 Hess's Law
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
types of energy
kinetic and potential energy
thermal energy
type of kinetic energy involving heat; molecule vibrations
chemical energy
type of potential energy involving stored energy in bonds or IMFs
unit for energy
joules
enthalpy
denoted H, total energy/heat content of system
enthalpy change
denoted ΔH, amount of heat transferred during a reaction
heat
denoted q, form of energy directly measured by changes in temperature
equation for heat
q = mcΔT
assume that q =
ΔH
in calorimetry, often assume
solution has same density (1g/ml) and specific heat capacity (4.18J/g°C) as water
units for q
J
units for ΔH
kJ/mol
+ΔH means reaction is
endothermic
-ΔH means reaction is
exothermic
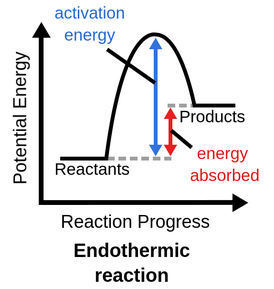
endothermic reaction
products have more PE than reactants; more energy was absorbed to break bonds than what was released to form new bonds
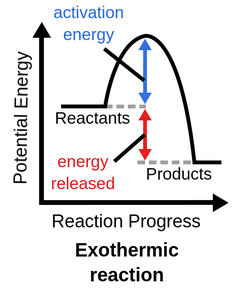
exothermic reaction
reactants have more PE than products; less energy was absorbed to break bonds than what was released to form new bonds
what type of reactions are always exothermic
combustion, condensing, freezing, deposition (molecules release energy to form IMFs)
what type of reactions are always endothermic
melting, evaporation, sublimation (molecules absorb energy to weaken/overcome IMFs)
dissolution in thermodynamics
the ionic solid and water must undergo an endothermic reaction to break apart, then come together and mix in an exothermic reaction; enthalpy of solution is the difference between these processes
how to represent endothermic reactions on a diagram
products have higher energy than reactants
how to represent exothermic reactions on a diagram
reactants have higher energy than products
temperature
measure of average kinetic energy
thermal equilibrium
two substances reach same temperature and average KE
which way does energy flow
from hot to cold
how is KE transferred
through collisions
heat capacity
denoted C, quantity of energy needed to change temperature
specific heat capacity
denoted c, quantity of heat required to raise the temperature of 1g of a specific substance by 1°C
specific heat capacity of water
4.18 J/g°C, characteristically high
specific heat of water vs. metals
metals have lower specific heat than water—easier to change temperature
why do we set q = -q
heat gained = heat lost
what happens during phase changes (assume substance is being heated, so melting/boiling)
PE increases, IMFs are weakened/broken, temperature does not change
what happens in between phase changes as a substance is heated
temperature increases, KE increases
what equation to use when substance is heating/cooling
q = mcΔT
what equation to use when substance is changing phases
q = ΔHm, where ΔH is given in J/g or kJ/mol, m = mass or moles depending on unit of ΔH
standard conditions
room temperature, 1 atm, solutions 1M, substances in pure form
bond enthalpy
enthalpy change required to break/form given bond
enthalpy of bond formation
always exothermic, energy released to form 1 mole of covalent bonds
enthalpy of bond breaking
always endothermic, energy required to break 1 mole of covalent bonds
how to use bond enthalpy to calculate ΔH
given enthalpy of bond breaking
count up number of each specific type of bond in reactants and products
multiply by given values for each type of bond
for products (bond formation) take the negative of given values
add all values together
enthalpy of formation
energy change when 1 mole of substance formed (must maintain 1 mole of product); enthalpy of an element in pure form is 0 kJ/mol
how to use enthalpy of formation to calculate ΔH
given enthalpies of each molecule
use given values to sum up and calculate enthalpy of reactants and products
remember, elements in pure form (e.g. O2) have 0 enthalpy
ΔH = ∑ΔHproducts - ∑ΔHreactants
how to use Hess’s Law to calculate ΔH
given separate rxns and ΔHrxn of each
manipulate equations to create original equation (multiply to get desired coefficient, multiply by -1 to reverse rxn):
start with the substances in the original equation that show up once in the list of equations
manipulate the rest so they cancel each other out
add up resulting enthalpies to get ΔH