Chp2- Elements, Compounds and mixtures
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
8 Terms
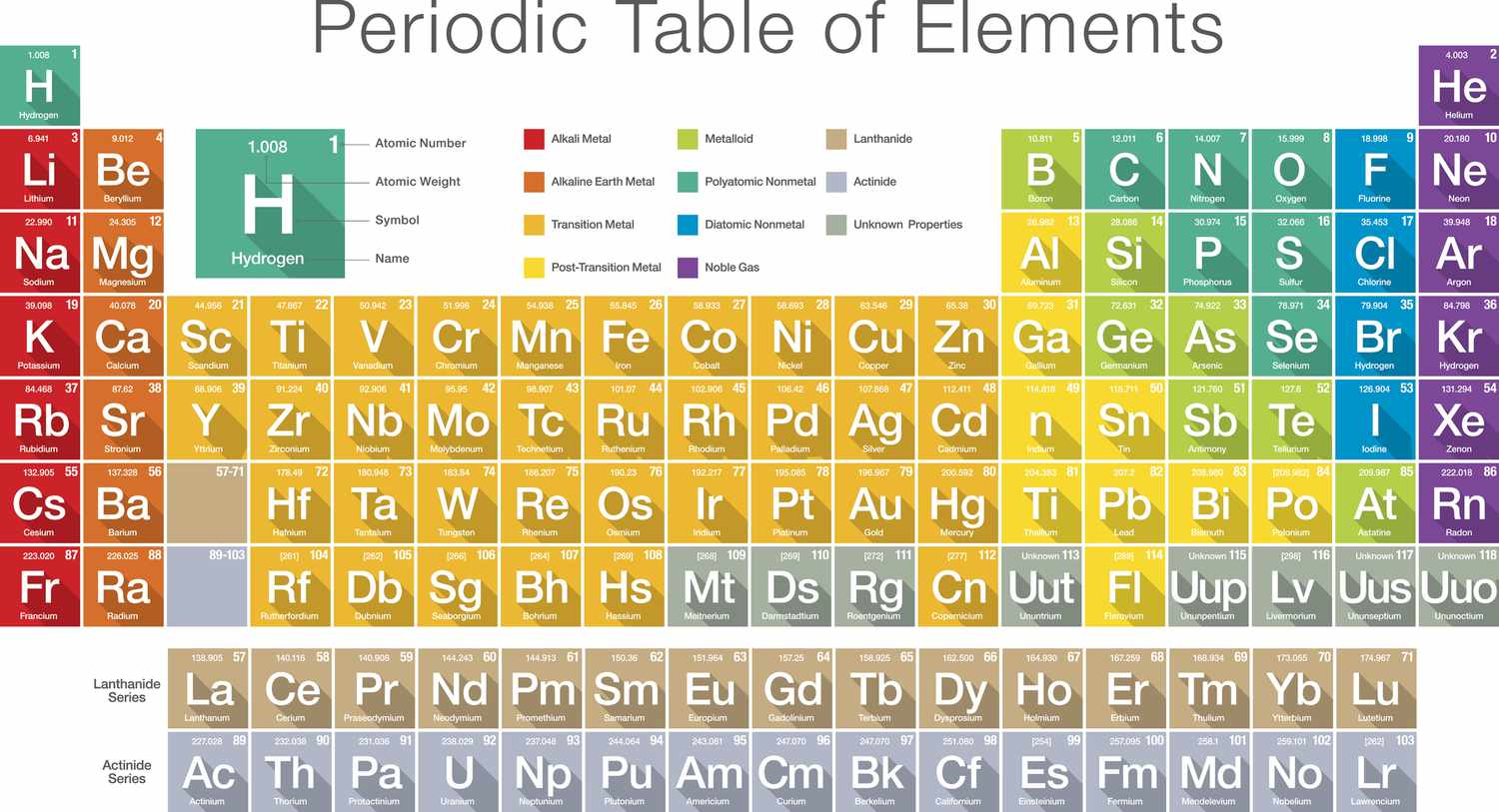
What are Elements?
It is a pure substance that cannot be broken into simpler substances by any means
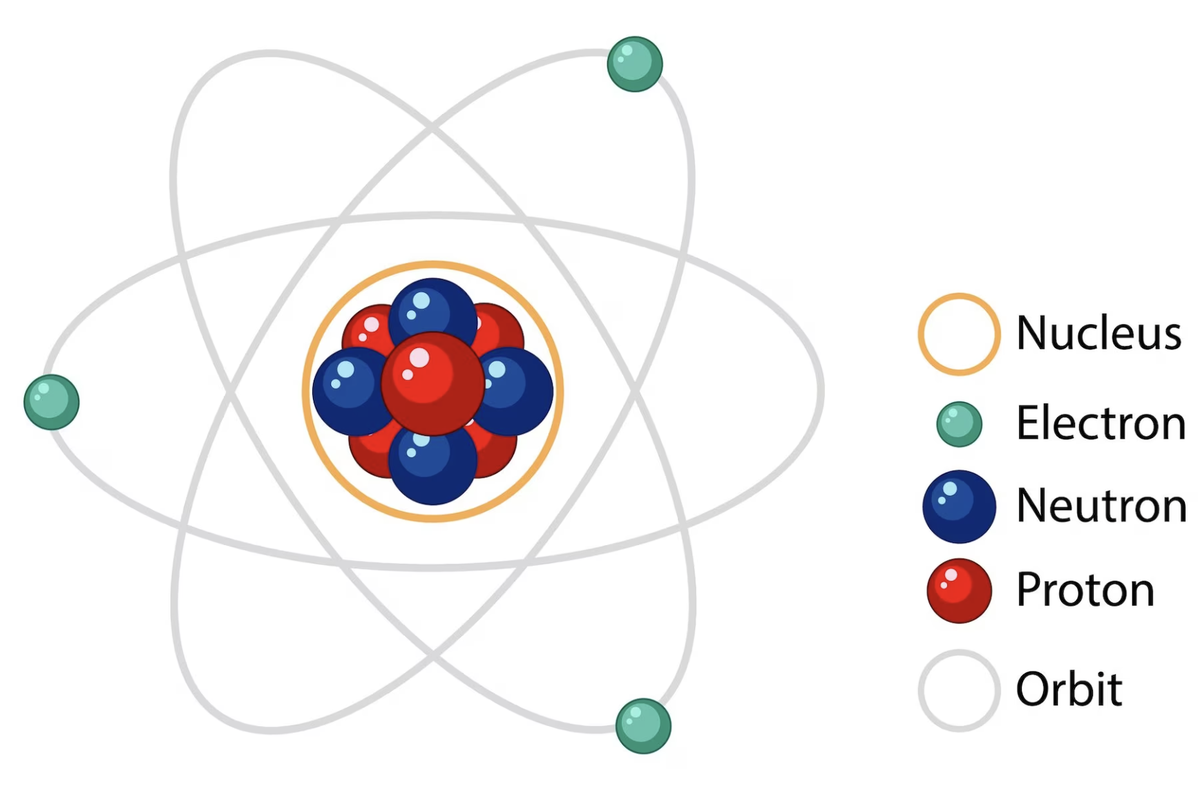
What are atoms?
Smallest particle of an element that have the same chemical properties as the element
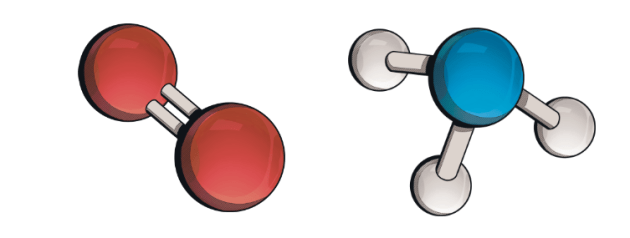
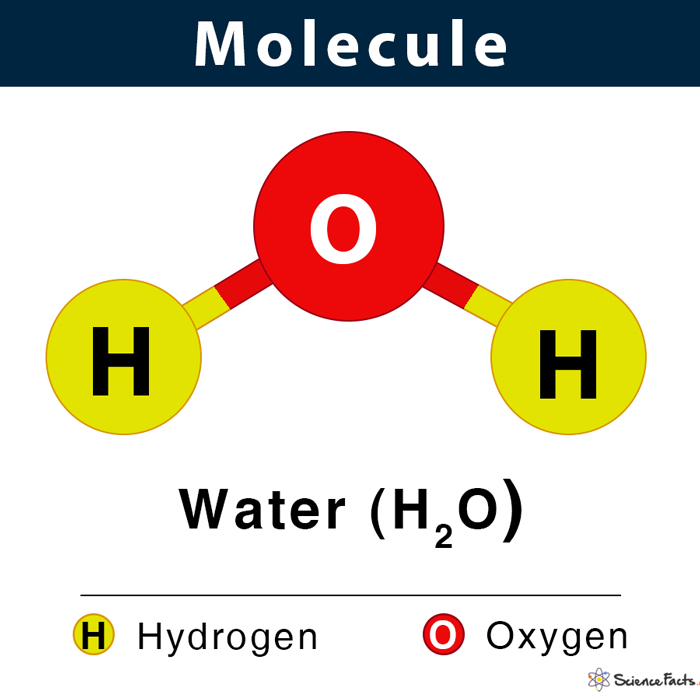
What is a molecule?
It is a group of two or more atoms chemically combined
What is a compound
It is a pure substance made up of two or more elements chemically combined in a fixed ratio
How can you break down compounds?
Thermal Decomposition- heating the compound strongly
Electrolysis- electricity is used to break down a compound
What is the difference between elements and compounds?
Elements only consist of one type of atom. While a Compound consists of two or more atoms chemically combined
The smallest particle in an element is an atom. The smallest particle in a compound is a molecule or ion
Elements can not be broken down. Compounds can be broken down by electrolysis or thermal decomposition
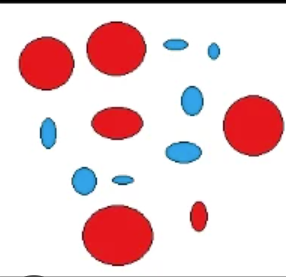
What is a mixture?
It is made up of two or more substances that are not chemically combined.
Difference between mixture and compound
Mixture can be physically separated
Mixtures have the same properties as their components
Compounds have chemical changes
Mixtures are mixed in random proportion