Module 12: Other Forms of Metabolism
1/200
Earn XP
Description and Tags
Microbial Physiology
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
201 Terms
Lipids
Structurally heterogeneous group of substances.
Highly soluble in non-polar solvents; relatively insoluble in water.
Predominantly found in membranes of prokaryotic and eukaryotic cells.
Major lipids in bacterial and eukaryotic membranes: phosphoglycerides
phosphoglycerides
Major lipids in bacterial and eukaryotic membranes: _______________________
Carriers in LPS/Peptidoglycan Synthesis
Roles of Lipids
Component of Membranes
Structural integrity and fluidity of cell membranes.
__________________________
Lipids are involved in transporting molecules required for synthesizing lipopolysaccharides (LPS) and peptidoglycans in bacterial cell walls.
Cell Surface Components
Lipids contribute to surface structures such as glycolipids and lipoproteins.
Alternative Forms of Energy
Lipids can be broken down through β-oxidation to generate ATP.
Protective Coating
Forms protective layers in many organisms, preventing desiccation and providing defense.
Lipid Metabolism Pathways
Lipid Digestion and Absorption
Lipids are emulsified by bile salts and hydrolyzed by lipases into fatty acids and glycerol.
Fatty Acid Oxidation (β-Oxidation)
Occurs in mitochondria and peroxisomes, breaking down fatty acids to acetyl-CoA for ATP production.
Lipid Biosynthesis
Involves the synthesis of triglycerides, phospholipids, and cholesterol from acetyl-CoA and other precursors.
Ketogenesis
In conditions of low carbohydrate availability, excess acetyl-CoA is converted into ketone bodies in the liver.
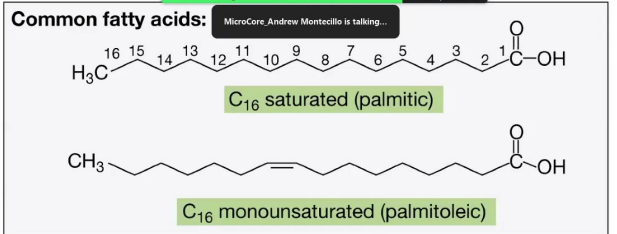
C16 Saturated (Palmitic acid)
Common Fatty Acids
_______________________:
Contains 16 carbon atoms with no double bonds (fully saturated with hydrogen).
C16 Monounsaturated (Palmitoleic acid):
Contains 16 carbon atoms with one double bond, creating a kink in the chain.
C16 Monounsaturated (Palmitoleic acid)
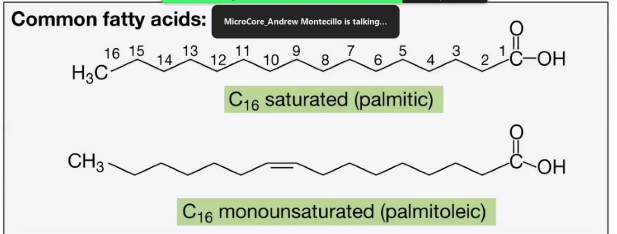
Common Fatty Acids
C16 Saturated (Palmitic acid):
Contains 16 carbon atoms with no double bonds (fully saturated with hydrogen).
__________________________________:
Contains 16 carbon atoms with one double bond, creating a kink in the chain.
Simple Lipids (Triglycerides)
______________________
Structure:
Composed of three fatty acids attached to a glycerol backbone through ester linkages.
The glycerol molecule has three hydroxyl groups (-OH) that form bonds with the carboxyl groups (-COOH) of the fatty acids.
Function:
Main form of energy storage in cells.
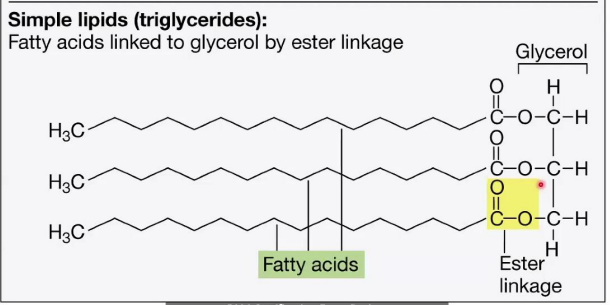
ester linkages
Simple Lipids (Triglycerides)
Structure:
Composed of three fatty acids attached to a glycerol backbone through __________________.
The glycerol molecule has three hydroxyl groups (-OH) that form bonds with the carboxyl groups (-COOH) of the fatty acids.
Function:
Main form of energy storage in cells.
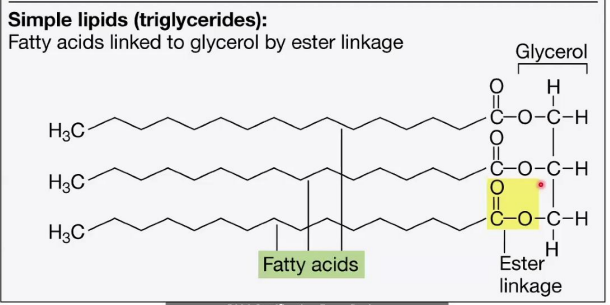
Complex Lipids
______________________________
Phosphatidyl Ethanolamine (Phosphoglyceride):
A complex lipid with a glycerol backbone, two fatty acids, and a phosphate group attached to ethanolamine.
Phosphate group: Hydrophilic (polar) head.
Fatty acid chains: Hydrophobic (non-polar) tails.
Monogalactosyl Diglyceride (Glycolipid):
Contains one sugar molecule (galactose) attached to glycerol and two fatty acids.
Glycolipids are important in cell membranes and signaling.
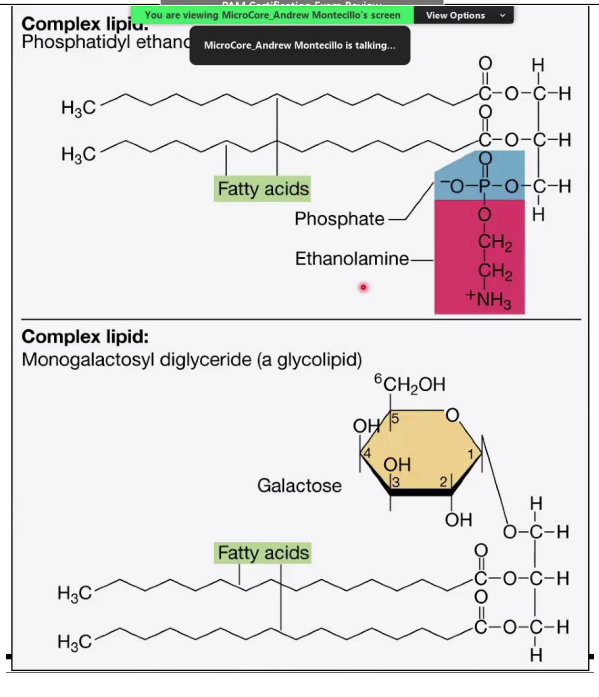
Phosphatidyl Ethanolamine (Phosphoglyceride)
Complex Lipids
__________________________:
A complex lipid with a glycerol backbone, two fatty acids, and a phosphate group attached to ethanolamine.
Phosphate group: Hydrophilic (polar) head.
Fatty acid chains: Hydrophobic (non-polar) tails.
Monogalactosyl Diglyceride (Glycolipid):
Contains one sugar molecule (galactose) attached to glycerol and two fatty acids.
Glycolipids are important in cell membranes and signaling.
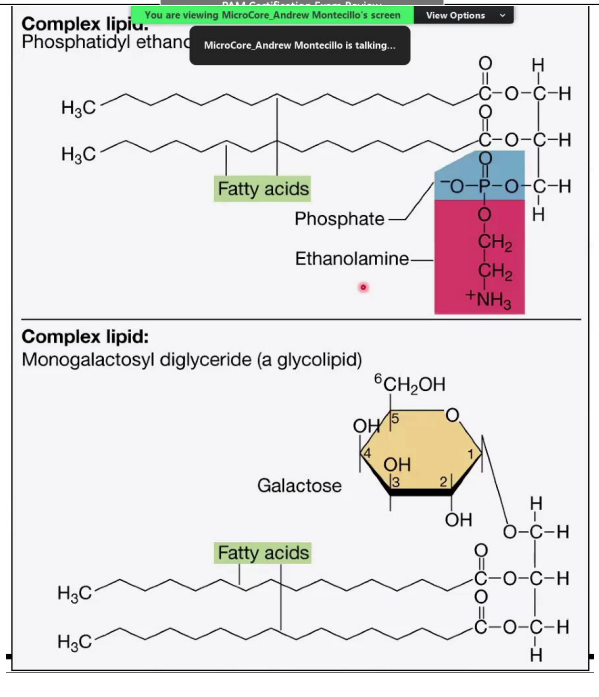
Monogalactosyl Diglyceride (Glycolipid)
Complex Lipids
Phosphatidyl Ethanolamine (Phosphoglyceride):
A complex lipid with a glycerol backbone, two fatty acids, and a phosphate group attached to ethanolamine.
Phosphate group: Hydrophilic (polar) head.
Fatty acid chains: Hydrophobic (non-polar) tails.
___________________________:
Contains one sugar molecule (galactose) attached to glycerol and two fatty acids.
Glycolipids are important in cell membranes and signaling.
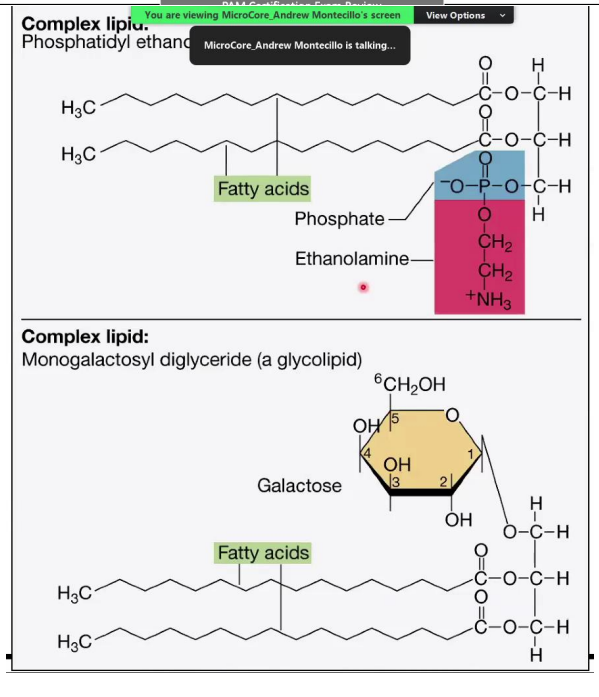
Breakdown of Lipids
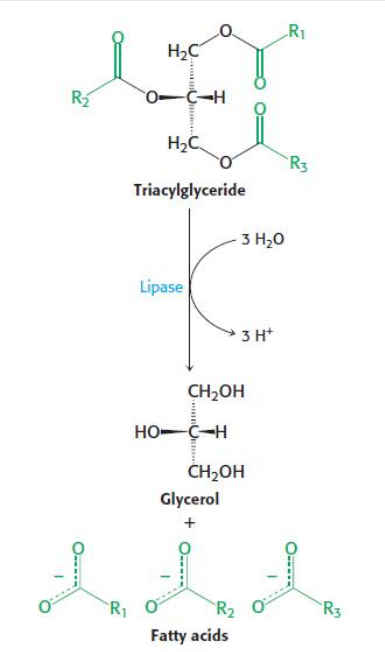
________________________
Process:
Triglycerides are broken down into glycerol and fatty acids by lipases.
Enzymes Involved:
Lipase: Cleaves the ester bonds between glycerol and fatty acids.
Step-by-Step Hydrolysis:
Triacylglycerol → Diacylglycerol → Monoacylglycerol → Glycerol + Fatty Acids.
Glycerol enters glycolysis as glycerol-3-phosphate.
Fatty acids undergo β-oxidation to produce acetyl-CoA for energy production.
Phospholipase A/B/C/D
Key Enzymes:
______________________________: Breaks down phospholipids at different positions on the molecule.
Lipase: Specifically hydrolyzes triglycerides into glycerol and fatty acids.
Lipase
Key Enzymes:
Phospholipase A/B/C/D: Breaks down phospholipids at different positions on the molecule.
_______________: Specifically hydrolyzes triglycerides into glycerol and fatty acids.
β-Oxidation
Lipid Catabolism via ______________________
______________________ is a metabolic process by which fatty acids are broken down to generate acetyl-CoA, which can enter the citric acid cycle (Krebs cycle) for energy production. This process takes place in the mitochondria of eukaryotic cells and involves several steps:
Key Steps of ______________________:
Activation of Fatty Acids:
Fatty acids are activated by conversion to acyl-CoA using ATP and CoA.
Dehydrogenation (Step 1):
Enzyme: Acyl-CoA Dehydrogenase.
Reaction: Acyl-CoA is oxidized, forming a double bond between α and β carbons, producing trans-Δ²-enoyl-CoA.
Byproduct: FADH₂ is produced (enters the electron transport chain).
Hydration (Step 2):
Enzyme: Enoyl-CoA Hydratase.
Reaction: Water is added across the double bond, converting trans-Δ²-enoyl-CoA to L-β-hydroxyacyl-CoA.
Oxidation (Step 3):
Enzyme: L-Hydroxyacyl-CoA Dehydrogenase.
Reaction: L-β-hydroxyacyl-CoA is oxidized to β-ketoacyl-CoA.
Byproduct: NADH is produced (enters the electron transport chain).
Cleavage (Step 4):
Enzyme: Thiolase (β-Ketothiolase).
Reaction: β-Ketoacyl-CoA is cleaved by CoA to generate acetyl-CoA and a shortened acyl-CoA (2 carbons shorter).
This shortened acyl-CoA re-enters the cycle for further oxidation.
Key Points:
Two carbons are removed at a time from the fatty acid chain in the form of acetyl-CoA.
Enzymes Involved:
Acyl-CoA Dehydrogenase
Enoyl-CoA Hydratase
L-Hydroxyacyl-CoA Dehydrogenase
Thiolase
Energy Yield: Each cycle of β-oxidation produces 1 FADH₂ and 1 NADH, which generate ATP through oxidative phosphorylation.
Repeat of the Cycle
The shortened fatty acyl-CoA re-enters the cycle, repeating the process until the entire fatty acid is converted into acetyl-CoA units.
Energy Yield
FADH₂ and NADH generated in steps 2 and 4 enter the electron transport chain, producing ATP.
Each acetyl-CoA enters the citric acid cycle, generating additional energy.
Two carbons
_________________ are removed at a time from the fatty acid chain in the form of acetyl-CoA.
Enzymes Involved:
Acyl-CoA Dehydrogenase
Enoyl-CoA Hydratase
L-Hydroxyacyl-CoA Dehydrogenase
Thiolase
Energy Yield: Each cycle of β-oxidation produces 1 FADH₂ and 1 NADH, which generate ATP through oxidative phosphorylation.
1 FADH₂ | 1 NADH
Two carbons are removed at a time from the fatty acid chain in the form of acetyl-CoA.
Enzymes Involved:
Acyl-CoA Dehydrogenase
Enoyl-CoA Hydratase
L-Hydroxyacyl-CoA Dehydrogenase
Thiolase
Energy Yield: Each cycle of β-oxidation produces __________ and _____________, which generate ATP through oxidative phosphorylation.
Acyl-CoA Dehydrogenase
Enoyl-CoA Hydratase
L-Hydroxyacyl-CoA Dehydrogenase
Thiolase
Two carbons are removed at a time from the fatty acid chain in the form of acetyl-CoA.
Enzymes Involved:
_________________________
Energy Yield: Each cycle of β-oxidation produces 1 FADH₂ and 1 NADH, which generate ATP through oxidative phosphorylation.
Activation of Fatty Acid
______________
Reaction: A fatty acid reacts with ATP and Coenzyme A (CoA) to form fatty acyl-CoA.
Enzyme: Fatty acyl-CoA synthetase.
Key Input/Output: ATP is used, producing AMP and pyrophosphate (PPi).
This step prepares the fatty acid for β-oxidation.
Steps in β-Oxidation
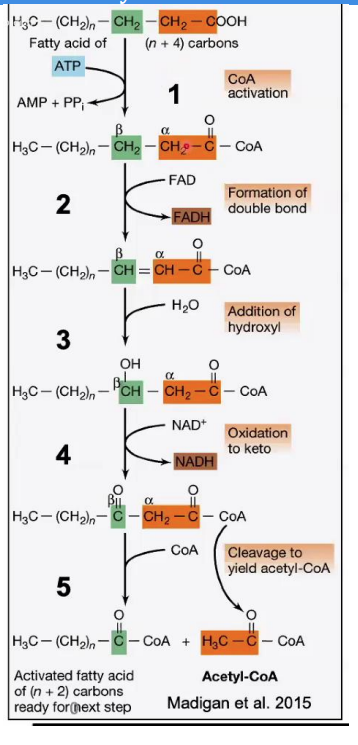
Fatty acyl-CoA synthetase
Activation of Fatty Acid
Reaction: A fatty acid reacts with ATP and Coenzyme A (CoA) to form fatty acyl-CoA.
Enzyme: __________________.
Key Input/Output: ATP is used, producing AMP and pyrophosphate (PPi).
This step prepares the fatty acid for β-oxidation.
Steps in β-Oxidation
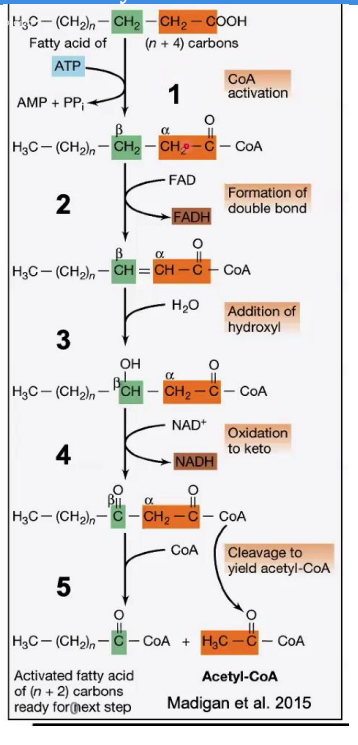
Formation of Double Bond (Oxidation)
___________________________
Reaction: The fatty acyl-CoA is oxidized to form a trans-Δ²-enoyl-CoA. A double bond forms between the α and β carbons (C2 and C3).
Enzyme: Acyl-CoA dehydrogenase.
Key Input/Output: FAD is reduced to FADH₂.
Steps in β-Oxidation
Acyl-CoA dehydrogenase
Formation of Double Bond (Oxidation)
Reaction: The fatty acyl-CoA is oxidized to form a trans-Δ²-enoyl-CoA. A double bond forms between the α and β carbons (C2 and C3).
Enzyme: ______________________.
Key Input/Output: FAD is reduced to FADH₂.
Steps in β-Oxidation.
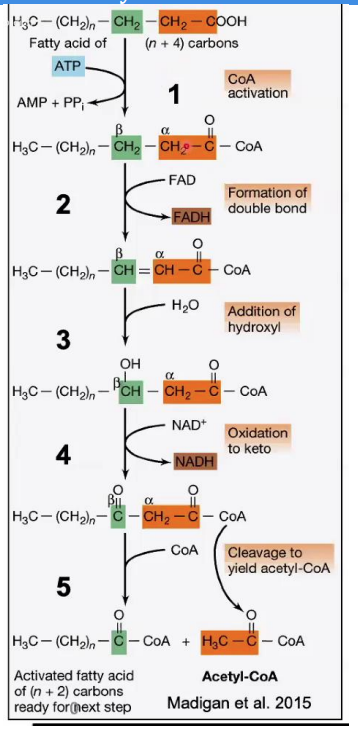
Hydration of Double Bond
_____________________
Reaction: Water (H₂O) is added across the double bond, resulting in the formation of β-hydroxyacyl-CoA.
Enzyme: Enoyl-CoA hydratase.
This adds a hydroxyl (-OH) group to the β-carbon.
Steps in β-Oxidation.
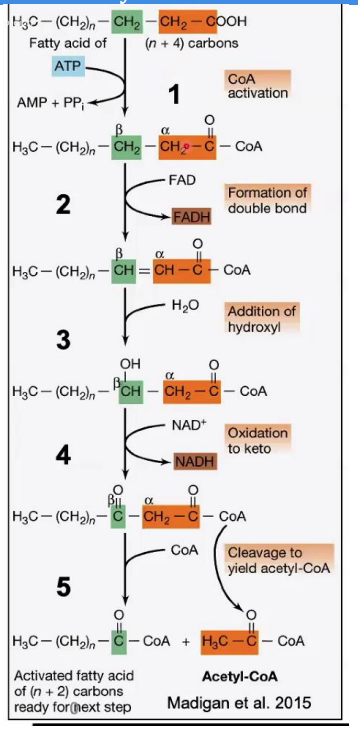
Enoyl-CoA hydratase
Hydration of Double Bond
Reaction: Water (H₂O) is added across the double bond, resulting in the formation of β-hydroxyacyl-CoA.
Enzyme: __________________.
This adds a hydroxyl (-OH) group to the β-carbon.
Steps in β-Oxidation.
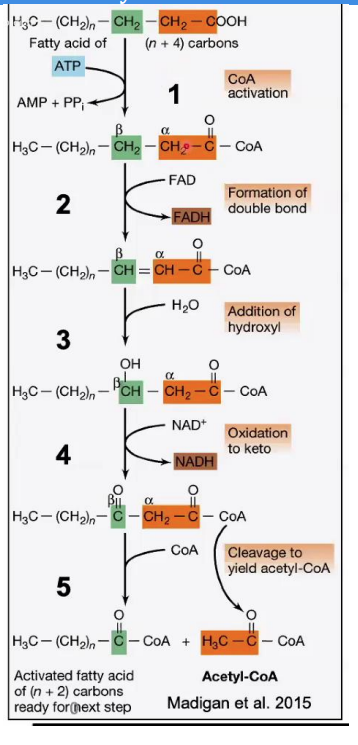
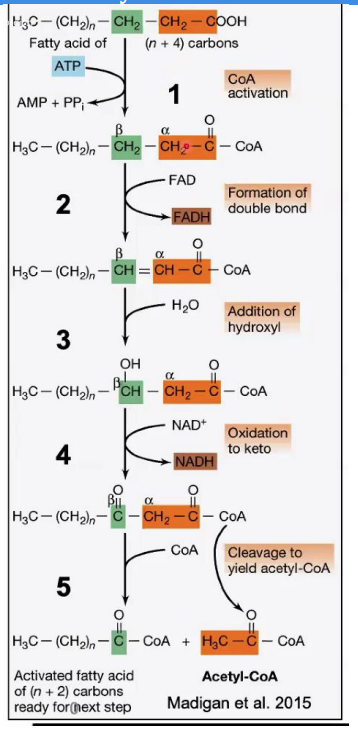
Oxidation of Hydroxyl Group
______________________
Reaction: The hydroxyl group (-OH) on the β-carbon is oxidized to a keto group (C=O), producing β-ketoacyl-CoA.
Enzyme: β-hydroxyacyl-CoA dehydrogenase.
Key Input/Output: NAD⁺ is reduced to NADH
Steps in β-Oxidation.
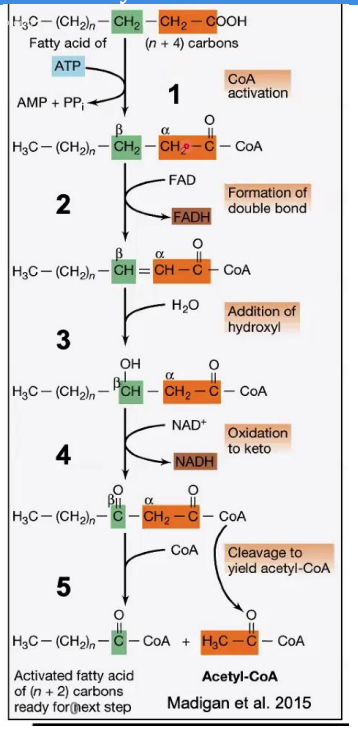
β-hydroxyacyl-CoA dehydrogenase.
Oxidation of Hydroxyl Group
Reaction: The hydroxyl group (-OH) on the β-carbon is oxidized to a keto group (C=O), producing β-ketoacyl-CoA.
Enzyme: __________________
Key Input/Output: NAD⁺ is reduced to NADH
Steps in β-Oxidation.
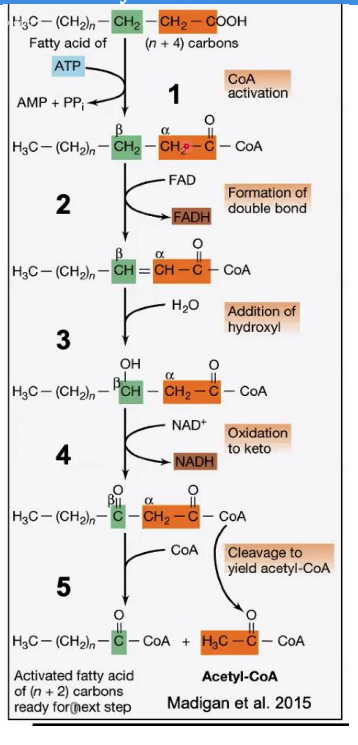
Cleavage (Thiolysis)
____________________
Reaction: The bond between the α and β carbons is broken, releasing acetyl-CoA and a shortened fatty acyl-CoA (n-2 carbons).
Enzyme: β-ketothiolase.
Key Input/Output: A new CoA molecule is attached to the shortened chain.
Steps in β-Oxidation.
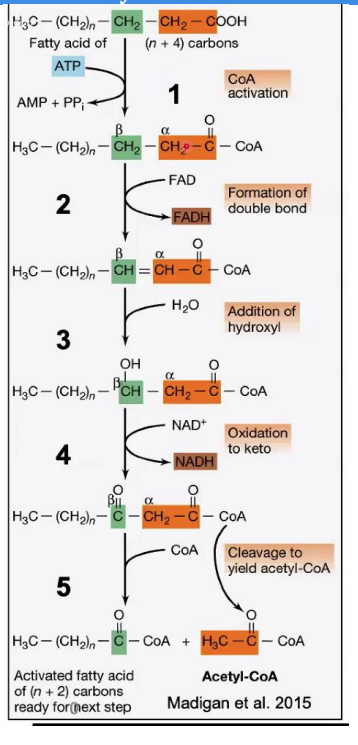
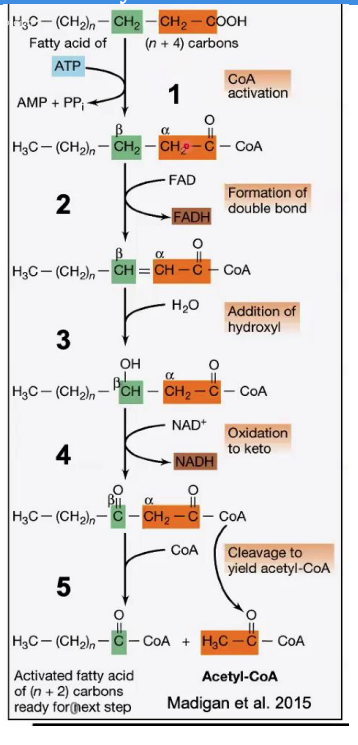
β-ketothiolase
Cleavage (Thiolysis)
Reaction: The bond between the α and β carbons is broken, releasing acetyl-CoA and a shortened fatty acyl-CoA (n-2 carbons).
Enzyme: _________________.
Key Input/Output: A new CoA molecule is attached to the shortened chain.
Steps in β-Oxidation.
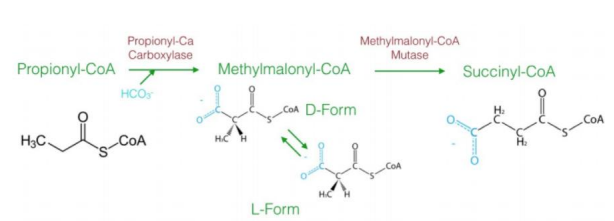
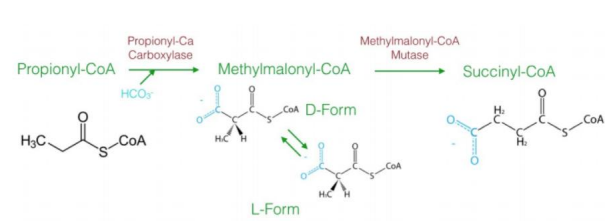
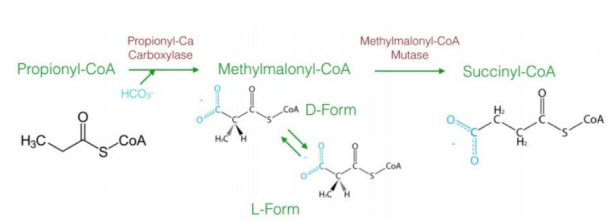
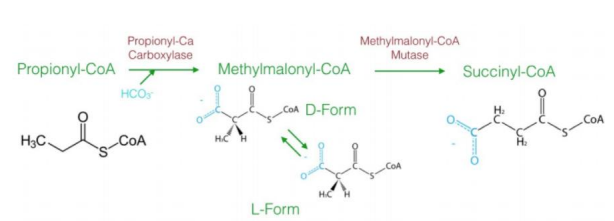
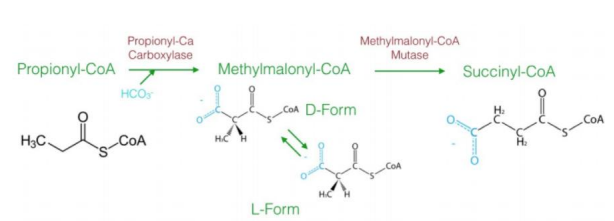
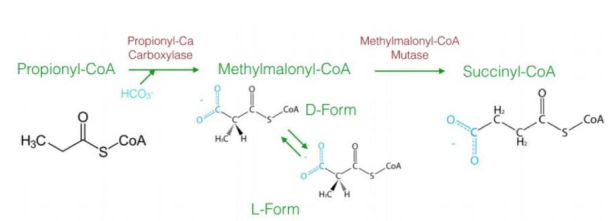
a three-carbon compound (propionyl-CoA)
The diagram explains the oxidation of odd-chain fatty acids, which differs from even-chain fatty acid oxidation. Odd-chain fatty acids produce ________________ during their final cycle of β-oxidation. This requires a unique pathway to convert propionyl-CoA into a metabolizable intermediate.
Steps in Oxidation of Odd-Chain Fatty Acids1. Carboxylation of Propionyl-CoA to D-Methylmalonyl-CoA
Reaction: Propionyl-CoA is carboxylated to form D-methylmalonyl-CoA.
Enzyme: Propionyl-CoA carboxylase.
Cofactor: Biotin (vitamin B7) is required.
Input/Output: ATP is used in the carboxylation reaction.
Significance: Adds a carboxyl group to the molecule, forming a four-carbon compound.
2. Isomerization to L-Methylmalonyl-CoA
Reaction: D-methylmalonyl-CoA is converted to its stereoisomer, L-methylmalonyl-CoA.
Enzyme: Methylmalonyl-CoA epimerase.
This step ensures the molecule has the correct stereochemistry for further processing.
3. Rearrangement to Succinyl-CoA
Reaction: L-methylmalonyl-CoA is rearranged to succinyl-CoA.
Enzyme: Methylmalonyl-CoA mutase.
Cofactor: Vitamin B12 (cobalamin) is required.
Significance: Succinyl-CoA enters the citric acid cycle, linking odd-chain fatty acid oxidation to energy production.
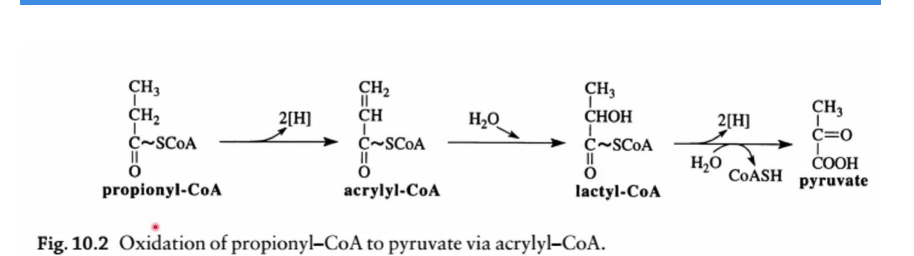
Alternate Pathway via Acryl-CoA
Propionyl-CoA can also be converted to pyruvate via intermediate steps involving acryl-CoA and lactyl-CoA.
Significance:
Pyruvate can enter the TCA cycle for energy generation.
Pyruvate can also participate in gluconeogenesis, providing glucose for anabolic pathways.
Importance
Odd-chain fatty acids are less common than even-chain fatty acids but are found in some dietary fats and bacterial-derived lipids.
This pathway enables complete utilization of odd-chain fatty acids for energy and glucose production.
Vitamin B12 and biotin deficiencies can impair this process, leading to metabolic issues.
Carboxylation of Propionyl-CoA to D-Methylmalonyl-CoA
Steps in Oxidation of Odd-Chain Fatty Acids
1. _____________________________
Reaction: Propionyl-CoA is carboxylated to form D-methylmalonyl-CoA.
Enzyme: Propionyl-CoA carboxylase.
Cofactor: Biotin (vitamin B7) is required.
Input/Output: ATP is used in the carboxylation reaction.
Significance: Adds a carboxyl group to the molecule, forming a four-carbon compound
Propionyl-CoA carboxylase.
Steps in Oxidation of Odd-Chain Fatty Acids
1. Carboxylation of Propionyl-CoA to D-Methylmalonyl-CoA
Reaction: Propionyl-CoA is carboxylated to form D-methylmalonyl-CoA.
Enzyme: _______________________
Cofactor: Biotin (vitamin B7) is required.
Input/Output: ATP is used in the carboxylation reaction.
Significance: Adds a carboxyl group to the molecule, forming a four-carbon compound
Isomerization to L-Methylmalonyl-CoA
Steps in Oxidation of Odd-Chain Fatty Acids
2. __________________________
Reaction: D-methylmalonyl-CoA is converted to its stereoisomer, L-methylmalonyl-CoA.
Enzyme: Methylmalonyl-CoA epimerase.
This step ensures the molecule has the correct stereochemistry for further processing.
Methylmalonyl-CoA epimerase.
Steps in Oxidation of Odd-Chain Fatty Acids
2. Isomerization to L-Methylmalonyl-CoA
Reaction: D-methylmalonyl-CoA is converted to its stereoisomer, L-methylmalonyl-CoA.
Enzyme: _______________________________
This step ensures the molecule has the correct stereochemistry for further processing.
Rearrangement to Succinyl-CoA
Steps in Oxidation of Odd-Chain Fatty Acids
3. ___________________________
Reaction: L-methylmalonyl-CoA is rearranged to succinyl-CoA.
Enzyme: Methylmalonyl-CoA mutase.
Cofactor: Vitamin B12 (cobalamin) is required.
Significance: Succinyl-CoA enters the citric acid cycle, linking odd-chain fatty acid oxidation to energy production.
Methylmalonyl-CoA mutase
Steps in Oxidation of Odd-Chain Fatty Acids
3. Rearrangement to Succinyl-CoA
Reaction: L-methylmalonyl-CoA is rearranged to succinyl-CoA.
Enzyme: _______________________
Cofactor: Vitamin B12 (cobalamin) is required.
Significance: Succinyl-CoA enters the citric acid cycle, linking odd-chain fatty acid oxidation to energy production.
Acryl-CoA
Alternate Pathway via _________________
Propionyl-CoA can also be converted to pyruvate via intermediate steps involving _________________ and lactyl-CoA.
Significance:
Pyruvate can enter the TCA cycle for energy generation.
Pyruvate can also participate in gluconeogenesis, providing glucose for anabolic pathways.
Steps in Oxidation of Odd-Chain Fatty Acids
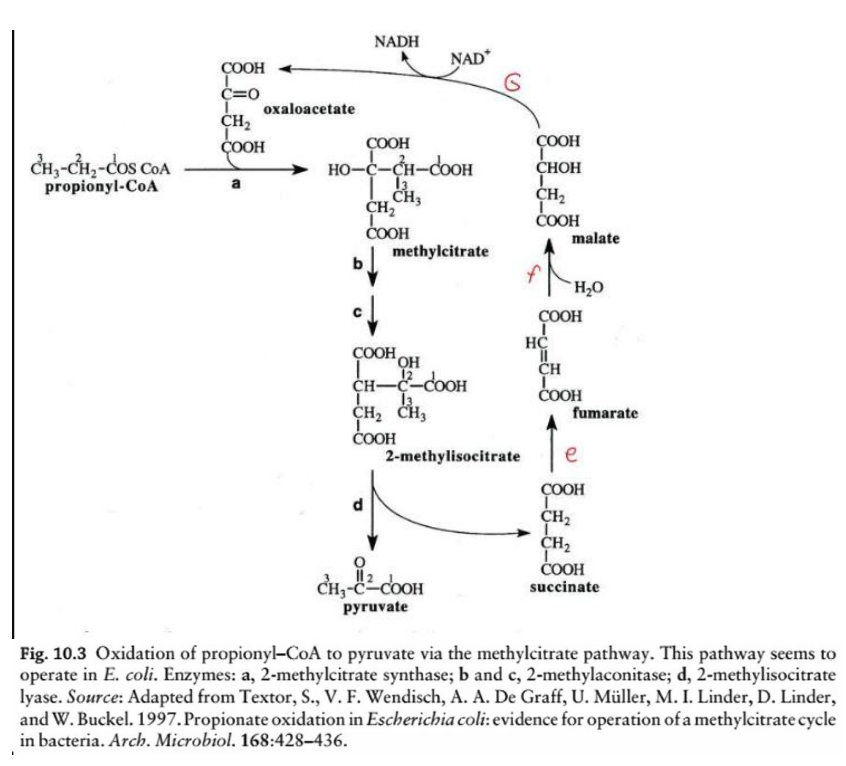
methylcitrate pathway
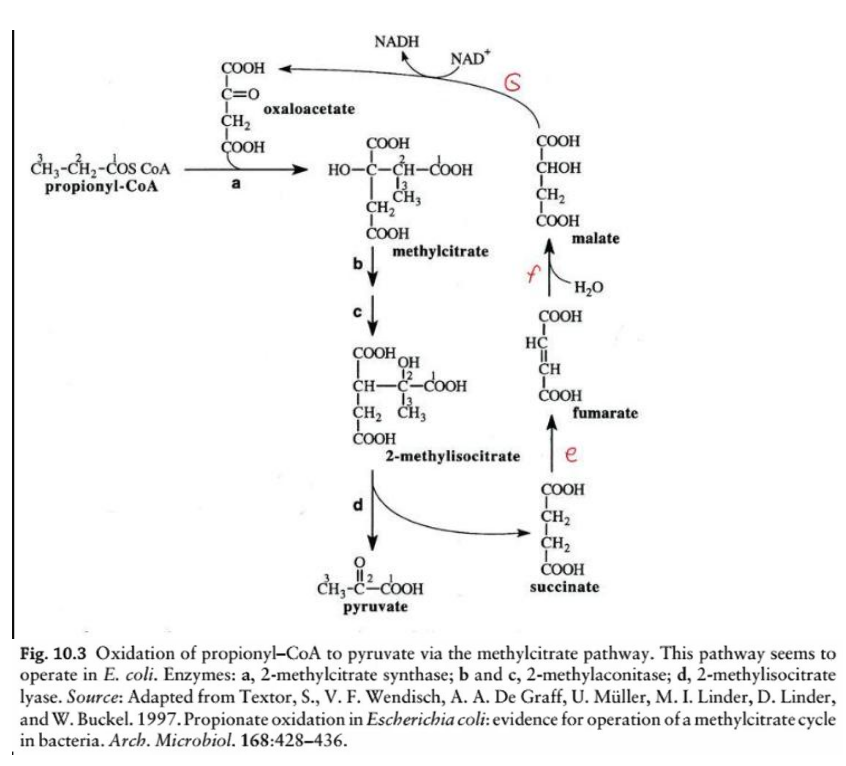
This diagram illustrates the oxidation of odd-chain fatty acids via the __________________, a biochemical process in Escherichia coli for the metabolism of propionyl-CoA. Here’s a step-by-step breakdown:
Overview:
Odd-chain fatty acids yield propionyl-CoA as a final product during beta-oxidation. The methylcitrate pathway converts propionyl-CoA into pyruvate, which can then enter central metabolic pathways like the TCA cycle or gluconeogenesis.
Key Reactions and Enzymes:
Step a: Formation of methylcitrate
Enzyme: 2-methylcitrate synthase
Reaction: Propionyl-CoA condenses with oxaloacetate to form methylcitrate.
Step b & c: Isomerization of methylcitrate to 2-methylisocitrate
Enzyme: 2-methylcitrate dehydratase (aconitase-like activity)
Reaction: Methylcitrate undergoes dehydration and rehydration to form 2-methylisocitrate.
Step d: Cleavage of 2-methylisocitrate
Enzyme: 2-methylisocitrate lyase
Reaction: 2-methylisocitrate is cleaved into pyruvate and succinate.
Steps e & f: TCA cycle intermediates
Succinate is converted into fumarate, malate, and eventually back to oxaloacetate (via TCA cycle enzymes), replenishing the cycle.
NADH Production
During the conversion of malate to oxaloacetate (via malate dehydrogenase), NADH is generated.
Importance:
Odd-chain fatty acid metabolism: This pathway addresses the metabolic challenge posed by propionyl-CoA, which cannot be directly oxidized by the TCA cycle.
Energy production: The conversion of propionyl-CoA to pyruvate and TCA intermediates allows cells to extract energy and integrate odd-chain fatty acids into mainstream metabolic pathways.
Bacterial survival: In organisms like E. coli, this pathway is crucial for utilizing propionyl-CoA derived from external carbon sources.
Formation of methylcitrate
Key Reactions and Enzymes:
Step a: ________________________
Enzyme: 2-methylcitrate synthase
Reaction: Propionyl-CoA condenses with oxaloacetate to form methylcitrate.
methylcitrate pathway
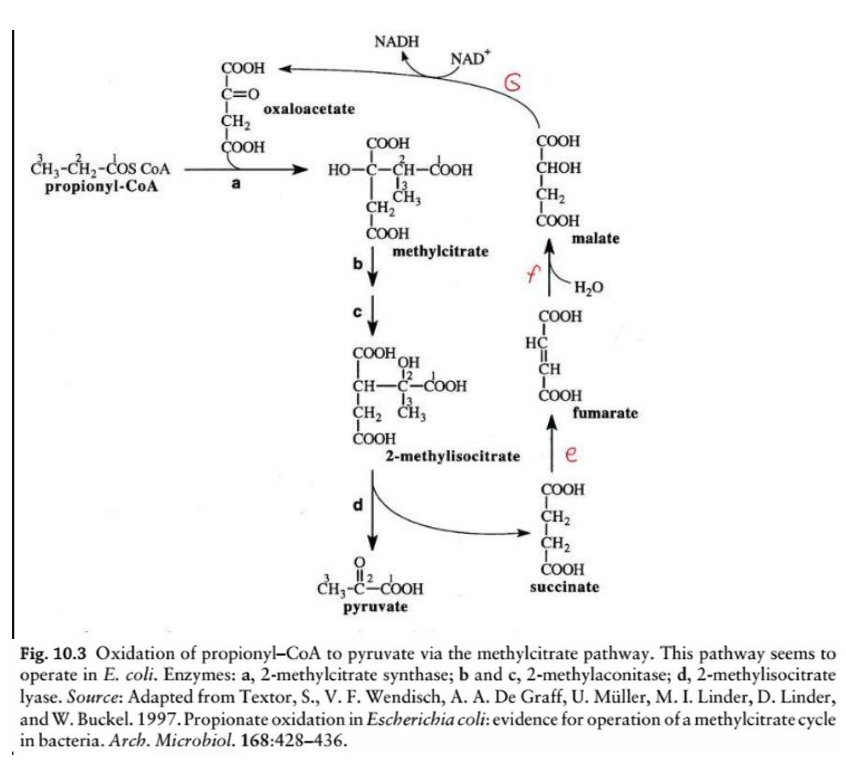
Key Reactions and Enzymes:
Step a: Formation of methylcitrate
Enzyme: __________________________
Reaction: Propionyl-CoA condenses with oxaloacetate to form methylcitrate.
methylcitrate pathway
Isomerization of methylcitrate to 2-methylisocitrate
Step b & c: ________________________
Enzyme: 2-methylcitrate dehydratase (aconitase-like activity)
Reaction: Methylcitrate undergoes dehydration and rehydration to form 2-methylisocitrate.
methylcitrate pathway
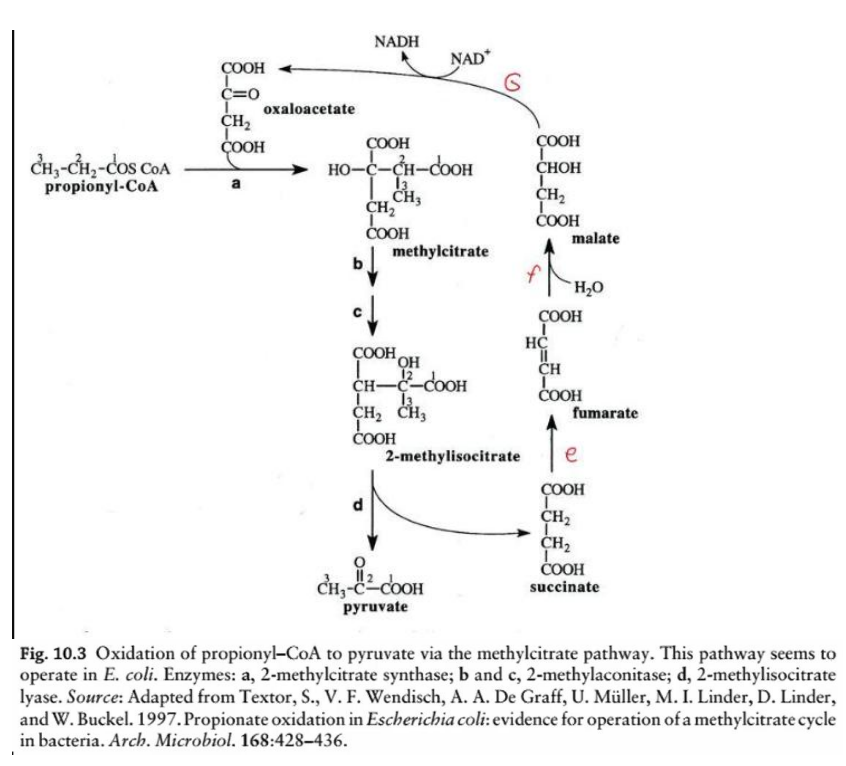
2-methylcitrate dehydratase (aconitase-like activity)
Step b & c: Isomerization of methylcitrate to 2-methylisocitrate
Enzyme: _________________________
Reaction: Methylcitrate undergoes dehydration and rehydration to form 2-methylisocitrate.
methylcitrate pathway
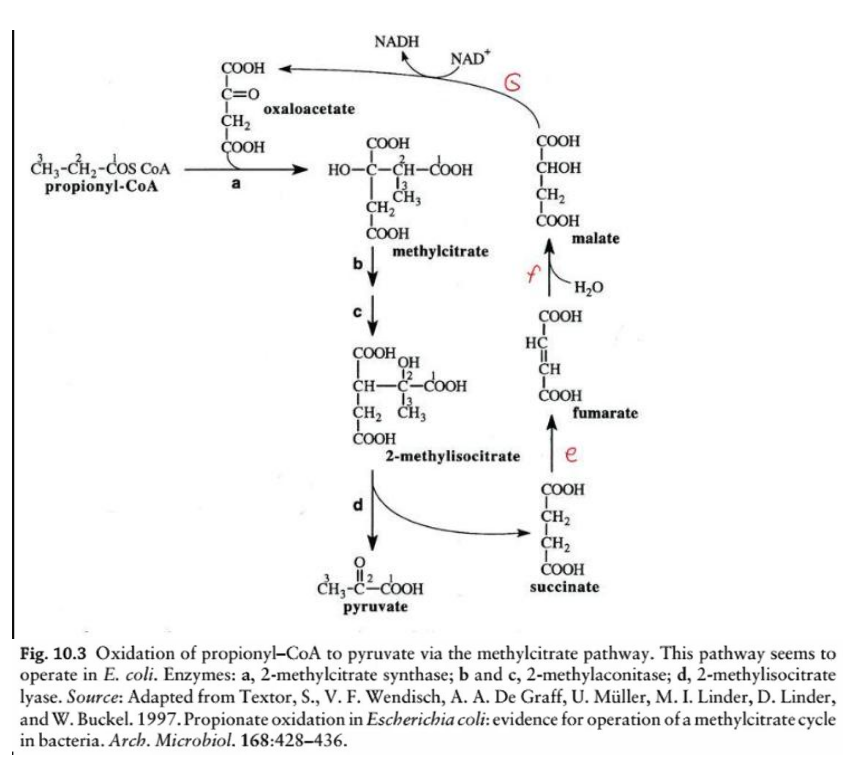
Cleavage of 2-methylisocitrate
Step d: _______________________
Enzyme: 2-methylisocitrate lyase
Reaction: 2-methylisocitrate is cleaved into pyruvate and succinate.
methylcitrate pathway
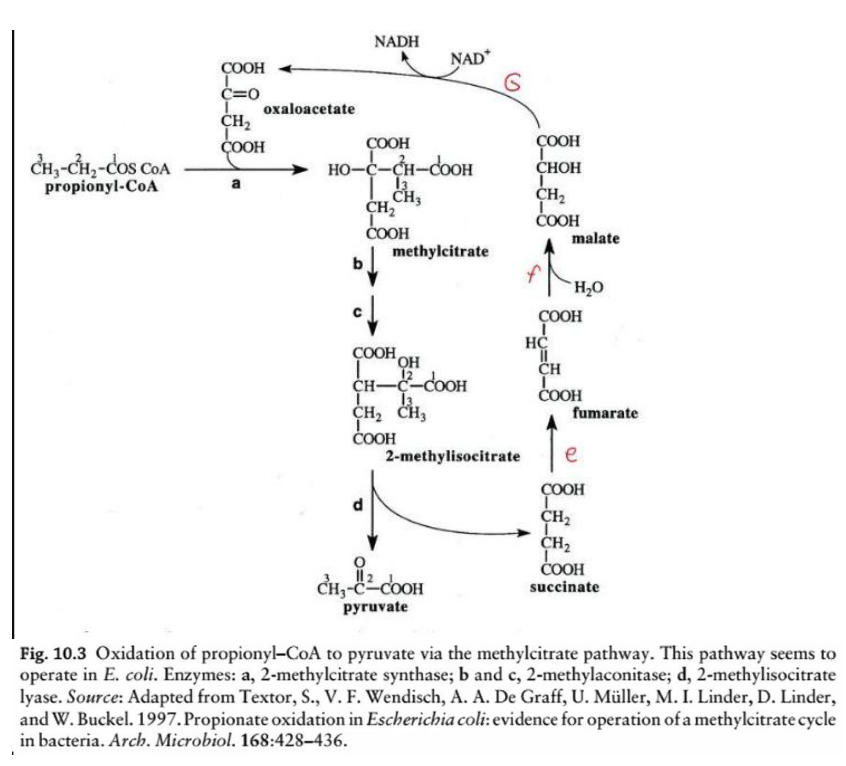
2-methylisocitrate lyase
Step d: Cleavage of 2-methylisocitrate
Enzyme: ______________________
Reaction: 2-methylisocitrate is cleaved into pyruvate and succinate.
methylcitrate pathway
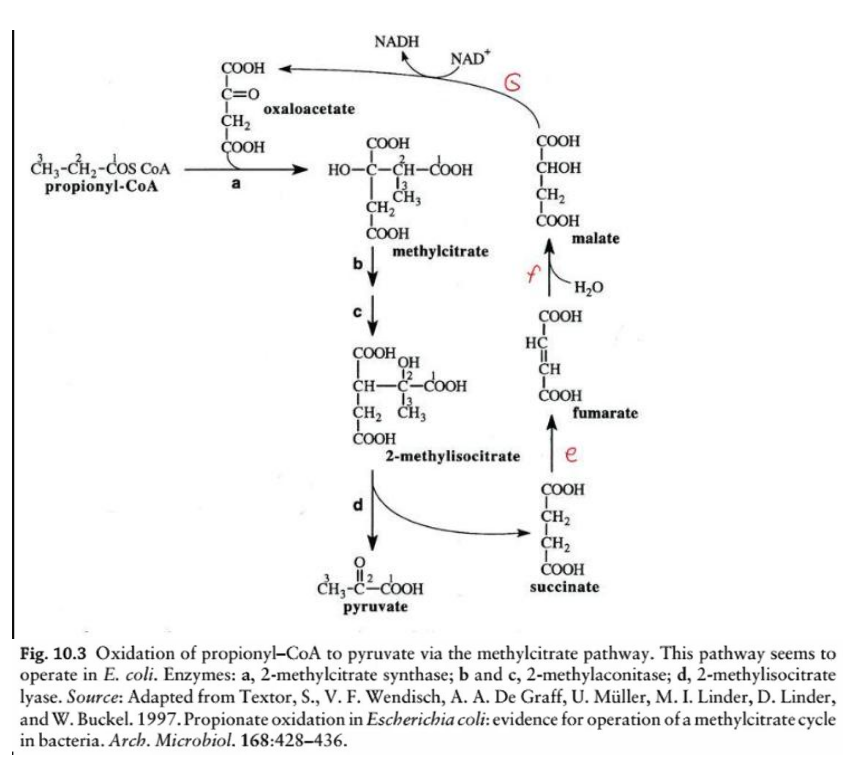
TCA cycle intermediates
Steps e & f: _______________________
Succinate is converted into fumarate, malate, and eventually back to oxaloacetate (via TCA cycle enzymes), replenishing the cycle.
methylcitrate pathway
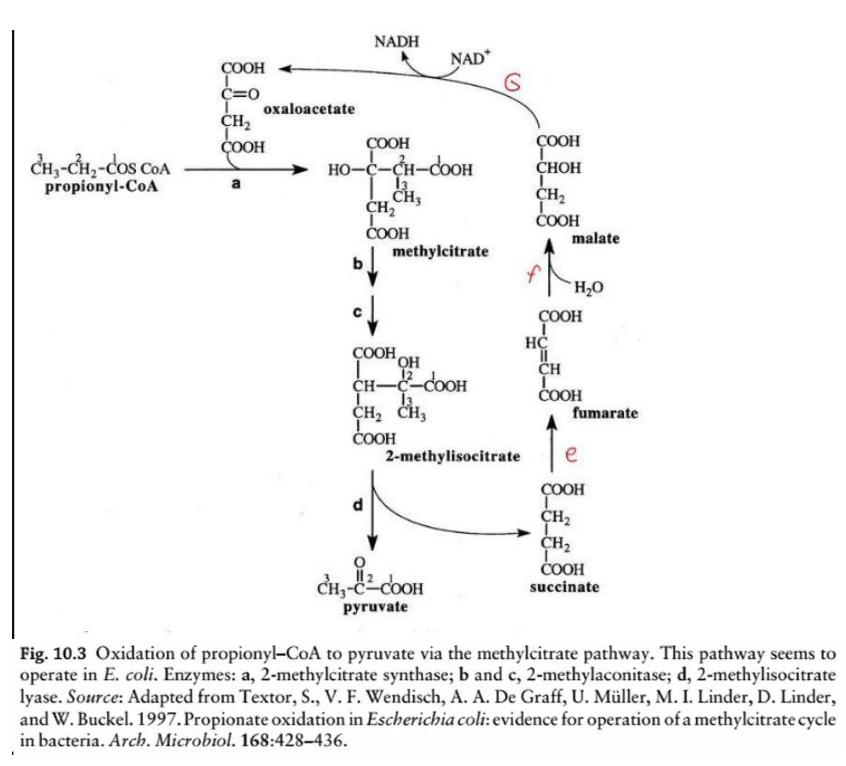
TCA cycle enzymes
Steps e & f: TCA cycle intermediates
Succinate is converted into fumarate, malate, and eventually back to oxaloacetate (via _____________), replenishing the cycle.
methylcitrate pathway
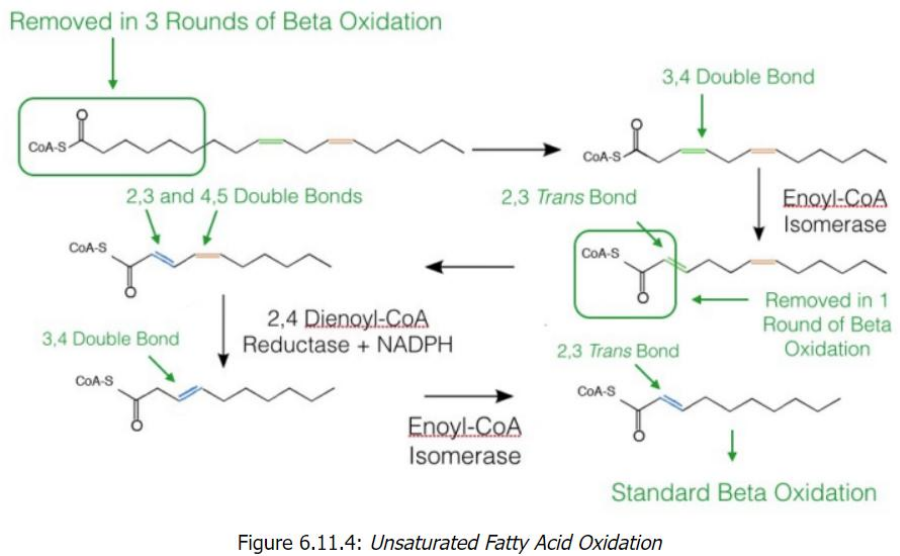
2,4-Dienoyl-CoA reductase
Unsaturated Fatty Acid Oxidation Overview
Unsaturated fatty acids differ from saturated ones because they contain double bonds in their carbon chains.
The presence of double bonds requires additional enzymatic steps for oxidation to proceed via the standard beta-oxidation pathway.
Two key enzymes handle these double bonds:
Enoyl-CoA isomerase: Works on odd-numbered double bonds.
_____________________: Handles even-numbered double bonds (in conjunction with enoyl-CoA isomerase).
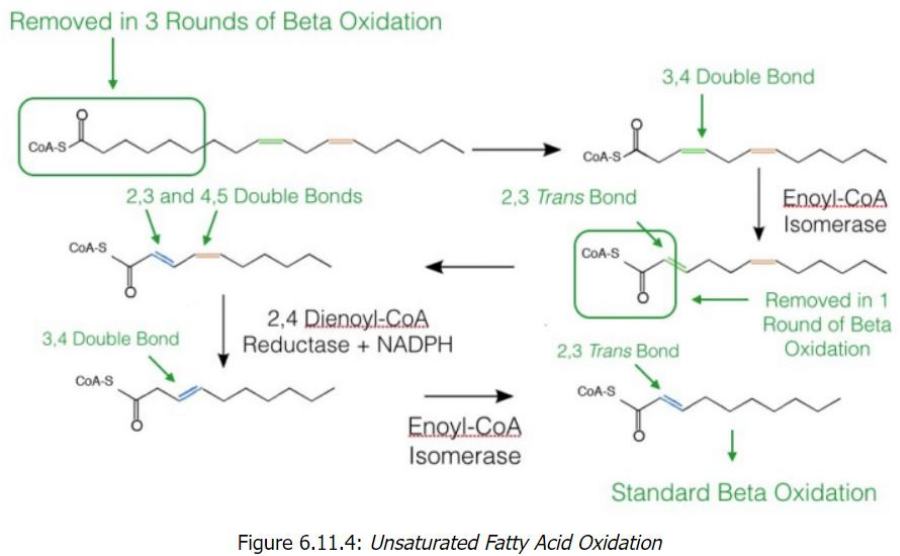
Enoyl-CoA isomerase
Unsaturated Fatty Acid Oxidation Overview
Unsaturated fatty acids differ from saturated ones because they contain double bonds in their carbon chains.
The presence of double bonds requires additional enzymatic steps for oxidation to proceed via the standard beta-oxidation pathway.
Two key enzymes handle these double bonds:
______________: Works on odd-numbered double bonds.
2,4-Dienoyl-CoA reductase: Handles even-numbered double bonds (in conjunction with enoyl-CoA isomerase).
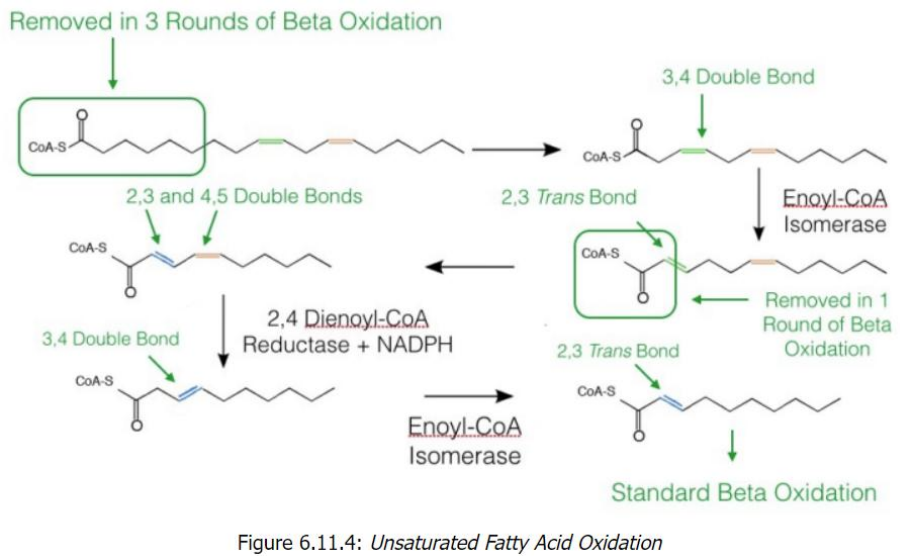
trans-Δ2 configuration
Key Enzymes and Steps1. Odd-Numbered Double Bonds
Double bonds at odd positions (e.g., cis-Δ3) cannot be directly processed by beta-oxidation.
Enzyme: Enoyl-CoA isomerase converts the cis-Δ3 double bond into a _______________, which is a substrate for beta-oxidation.
Once converted, the fatty acid resumes standard beta-oxidation.
2. Even-Numbered Double Bonds
Even-numbered double bonds (e.g., cis-Δ4) require an extra step because they cannot be processed directly either.
Step 1: 2,4-Dienoyl-CoA reductase reduces the 2,4-diene (two double bonds) into a single trans-Δ3 double bond, using NADPH.
Step 2: Enoyl-CoA isomerase converts this trans-Δ3 double bond into a trans-Δ2 configuration, allowing beta-oxidation to proceed.
Enoyl-CoA isomerase
Key Enzymes and Steps
1. Odd-Numbered Double Bonds
Double bonds at odd positions (e.g., cis-Δ3) cannot be directly processed by beta-oxidation.
Enzyme: _______________ converts the cis-Δ3 double bond into a trans-Δ2 configuration, which is a substrate for beta-oxidation.
Once converted, the fatty acid resumes standard beta-oxidation.
2. Even-Numbered Double Bonds
Even-numbered double bonds (e.g., cis-Δ4) require an extra step because they cannot be processed directly either.
Step 1: 2,4-Dienoyl-CoA reductase reduces the 2,4-diene (two double bonds) into a single trans-Δ3 double bond, using NADPH.
Step 2: Enoyl-CoA isomerase converts this trans-Δ3 double bond into a trans-Δ2 configuration, allowing beta-oxidation to proceed.
2,4-Dienoyl-CoA reductase
Key Enzymes and Steps
1. Odd-Numbered Double Bonds
Double bonds at odd positions (e.g., cis-Δ3) cannot be directly processed by beta-oxidation.
Enzyme: Enoyl-CoA isomerase converts the cis-Δ3 double bond into a trans-Δ2 configuration, which is a substrate for beta-oxidation.
Once converted, the fatty acid resumes standard beta-oxidation.
2. Even-Numbered Double Bonds
Even-numbered double bonds (e.g., cis-Δ4) require an extra step because they cannot be processed directly either.
Step 1: ________________ reduces the 2,4-diene (two double bonds) into a single trans-Δ3 double bond, using NADPH.
Step 2: Enoyl-CoA isomerase converts this trans-Δ3 double bond into a trans-Δ2 configuration, allowing beta-oxidation to proceed.
trans-Δ3
Key Enzymes and Steps
1. Odd-Numbered Double Bonds
Double bonds at odd positions (e.g., cis-Δ3) cannot be directly processed by beta-oxidation.
Enzyme: Enoyl-CoA isomerase converts the cis-Δ3 double bond into a trans-Δ2 configuration, which is a substrate for beta-oxidation.
Once converted, the fatty acid resumes standard beta-oxidation.
2. Even-Numbered Double Bonds
Even-numbered double bonds (e.g., cis-Δ4) require an extra step because they cannot be processed directly either.
Step 1: 2,4-Dienoyl-CoA reductase reduces the 2,4-diene (two double bonds) into a single ________________ double bond, using NADPH.
Step 2: Enoyl-CoA isomerase converts this trans-Δ3 double bond into a trans-Δ2 configuration, allowing beta-oxidation to proceed.
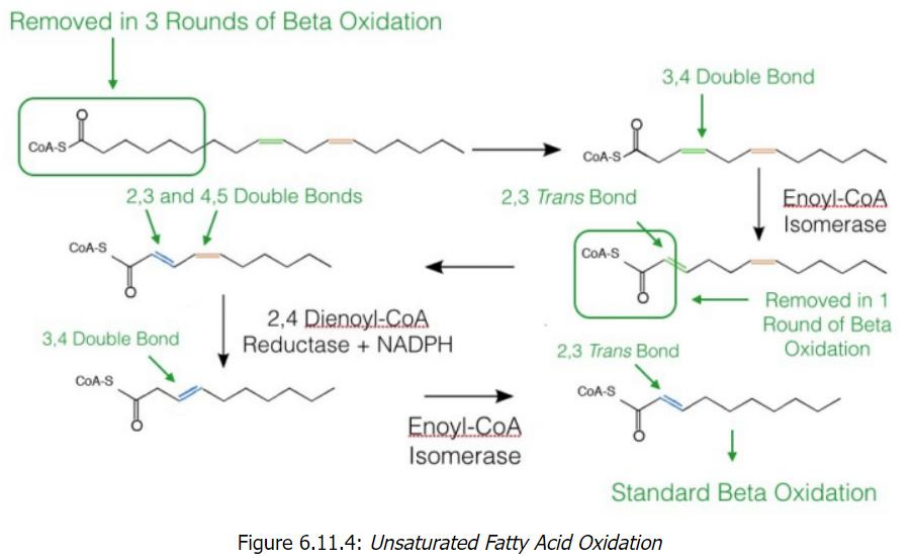
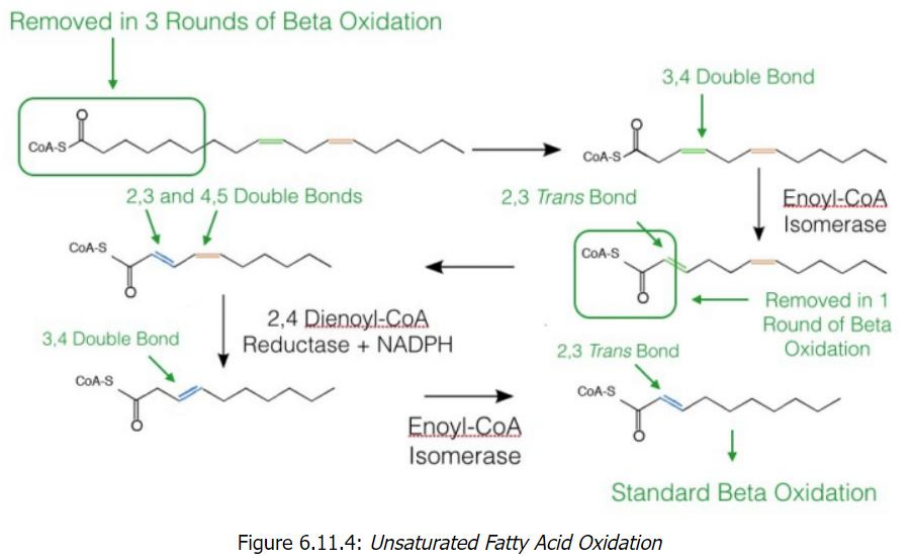
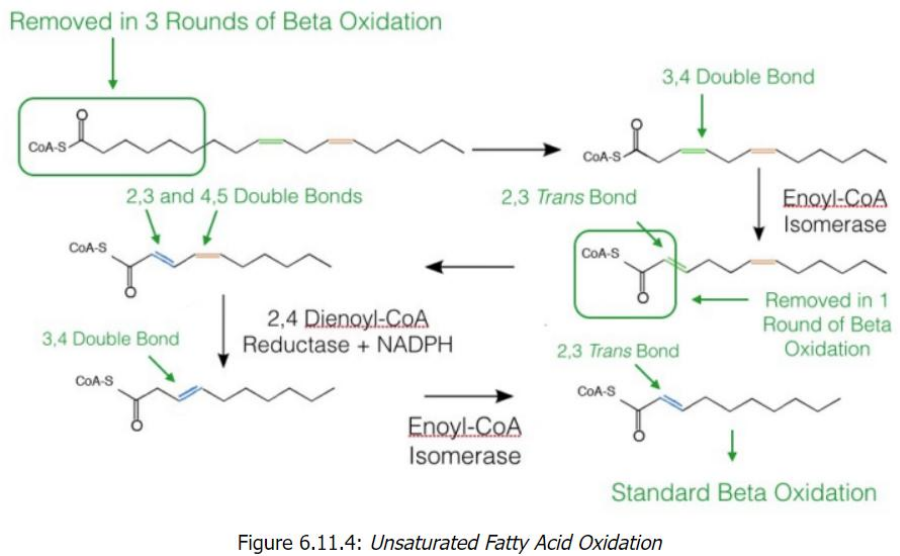
Unsaturated Fatty Acid Oxidation
Start with Linoleoyl-CoA (18 carbons, cis-Δ9, cis-Δ12)
After 6 cycles of beta-oxidation, 3 acetyl-CoA molecules are removed, leaving a fatty acid with a cis-Δ3 bond.
Processing the cis-Δ3 Double Bond
Enoyl-CoA isomerase converts cis-Δ3 to trans-Δ2.
Beta-oxidation resumes, producing another acetyl-CoA.
Encounter with a cis-Δ4 Bond
2,4-Dienoyl-CoA reductase reduces the diene structure (cis-Δ4) to a single trans-Δ3 bond.
Enoyl-CoA isomerase converts this to trans-Δ2, allowing beta-oxidation to proceed.
Final Oxidation Cycles
Standard beta-oxidation continues until all carbons are converted into acetyl-CoA (5 total in the example).
_____________________________ Overview
Key Points
Energy Efficiency: Unsaturated fatty acid oxidation yields slightly less ATP compared to saturated fatty acids due to the involvement of NADPH in the reductase step.
Adaptability: This process allows cells to metabolize a wide variety of fatty acids, including those with double bonds, making it crucial for energy production in diverse diets.
12 ATP
This breakdown calculates the ATP yield from fatty acid oxidation for both even- and odd-numbered fatty acids, taking into account the energy contributions from NADH, FADH₂, and acetyl-CoA during beta-oxidation. Here’s an explanation of each calculation:
Key Terms
FADH₂: Each FADH₂ contributes 2 ATP during electron transport.
NADH: Each NADH contributes 3 ATP during electron transport.
Acetyl-CoA: Each acetyl-CoA generates __________________ through the TCA cycle.
Even-Numbered Saturated Fatty Acids ATP generated
____________________________
For a fatty acid with n carbons:
Number of oxidations: n/2−1
Beta-oxidation breaks down the chain into 2-carbon acetyl-CoA units. For even chains, this produces n/2, and the number of beta-oxidation cycles is one less than the number of acetyl-CoA molecules.
ATP Yield from Each Oxidation Cycle:
Each cycle generates:
1 FADH₂ → 2 ATP
1 NADH → 3 ATP
Total per cycle: 2+3=52 + 3 = 52+3=5 ATP.
ATP from Acetyl-CoA:
Each acetyl-CoA generates 12 ATP via the TCA cycle.
Total acetyl-CoA: n/2 × 12
Correction for Activation Energy:
Fatty acid activation (conversion to acyl-CoA) consumes 2 ATP.
Formula:
Total ATP Yield=(n/2−1) × 5 + n/2 × 12−2
Odd-Numbered Saturated Fatty Acids ATP Generated
____________________
For a fatty acid with n carbons:
Number of oxidations: n / 2 − 1.5
Odd-numbered fatty acids undergo the same beta-oxidation process until a 3-carbon propionyl-CoA remains.
ATP Yield from Propionyl-CoA:
Propionyl-CoA is converted to succinyl-CoA, which enters the TCA cycle. This produces 1 FADH₂ (2 ATP), 1 NADH (3 ATP), and one ATP-equivalent via GTP (total 5 ATP).
ATP Yield Formula:
Beta-oxidation cycles yield:
(n / 2− 1.5) × 5
Acetyl-CoA yield:
(n / 2−0.5) × 12
Add the ATP from propionyl-CoA (+5) and subtract 2 ATP for activation.
Total ATP Yield=(n / 2−1.5) × 5 +(n / 2− 0.5) × 12 + 5 − 2
cytoplasm
Overview of Fatty Acid Synthesis:
Location: This process occurs in the _____________.
Purpose: Fatty acid synthesis creates long-chain fatty acids for energy storage or membrane formation.
Substrate: The process begins with acetyl-CoA, which is derived from carbohydrates, fats, or amino acids.
Key Features Highlighted:
Small Protein Involved:
Fatty acid synthesis requires a small protein called the acyl carrier protein (ACP). It holds and transfers acyl groups during the synthesis process.
This is distinct from CoA, which is used in degradation pathways (e.g., β-oxidation).
Precursor:
Acetyl-CoA serves as the main starting material.
It reacts with malonyl-CoA to build fatty acid chains.
Reductant:
NADPH is the electron donor, providing reducing power for the anabolic process (biosynthesis).
NADH is not used in this pathway, as it is more commonly involved in catabolic processes.
CO₂ Requirement:
The biosynthesis uses a carboxylated derivative of acetyl-CoA, namely malonyl-CoA, which is formed by acetyl-CoA carboxylase (ACC).
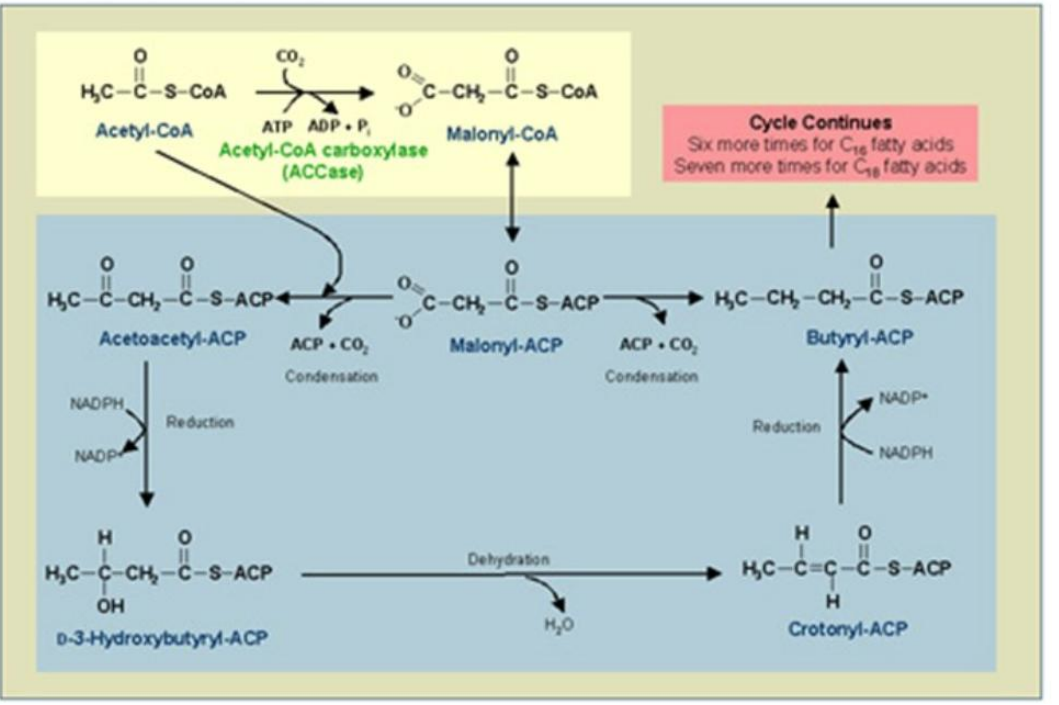
Steps of Fatty Acid Synthesis:
Initiation:
Acetyl-CoA and malonyl-CoA attach to ACP, forming acetyl-ACP and malonyl-ACP.
Enzymes involved:
Acetyl-CoA Carboxylase (ACC): Converts acetyl-CoA into malonyl-CoA.
FabD: Transfers malonyl group to ACP.
Elongation (2 Carbons Added at a Time):
Acetyl-ACP reacts with malonyl-ACP to form a β-keto-acyl-ACP intermediate.
This intermediate is modified through reduction, dehydration, and a second reduction to yield an elongated acyl-ACP.
Key enzymes:
FabH/FabB/FabF: Catalyze the condensation reaction.
FabG: Reduces β-keto-acyl-ACP to β-hydroxy-acyl-ACP.
FabZ/FabA: Catalyze dehydration to form trans-2-enoyl-ACP.
FabI/FabK/FabL: Catalyze the final reduction step to form acyl-ACP.
Termination:
When the fatty acid chain reaches its full length (e.g., 16 carbons for palmitate), it is released as a free fatty acid by thioesterase (TesA).
Distinctions from β-Oxidation:
Fatty Acid Synthesis:
Anabolic process.
Uses NADPH.
Occurs in the cytoplasm.
Involves ACP as a carrier protein.
β-Oxidation:
Catabolic process.
Produces NADH and FADH₂.
Occurs in the mitochondria.
Uses CoA for substrate activation and transport
Summary Points:
Two carbons are added at a time during elongation.
Malonyl-CoA is the critical donor of the two-carbon units.
The process ends when the fatty acid reaches its target length and is hydrolyzed by thioesterase.
Key Points:Energy Requirements: ATP is required for the initial formation of malonyl-CoA, while NADPH provides reducing power during elongation.
ACP's Role: Acts as a dynamic carrier for intermediates, ensuring efficient transfer between enzymes.
Regulation: Acetyl-CoA Carboxylase (ACC) is a critical regulatory enzyme in fatty acid synthesis.
This pathway is tightly regulated and essential for cellular lipid metabolism.
Acyl Carrier Protein (ACP):
________________
EC No.: 6.4.1.2
Role: Carries acyl intermediates in a thioester linkage, acting as a flexible arm during synthesis.
Note: ___________is not an enzyme but a carrier protein critical for the process.
The Fatty Acid Synthase Complex:
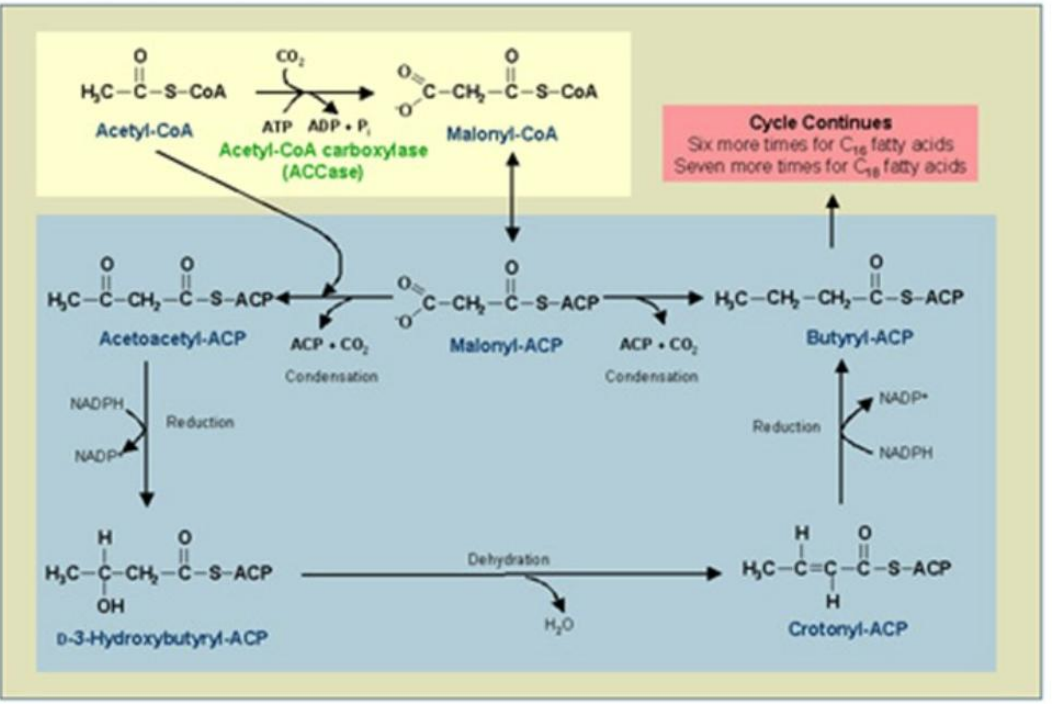
Acyl-CoA–ACP Transacylase (AT)
_________________:
EC No.: 2.3.1.9
Role: Transfers the acyl group from CoA to the cysteine residue of β-ketoacyl synthase (KS).
The Fatty Acid Synthase Complex:
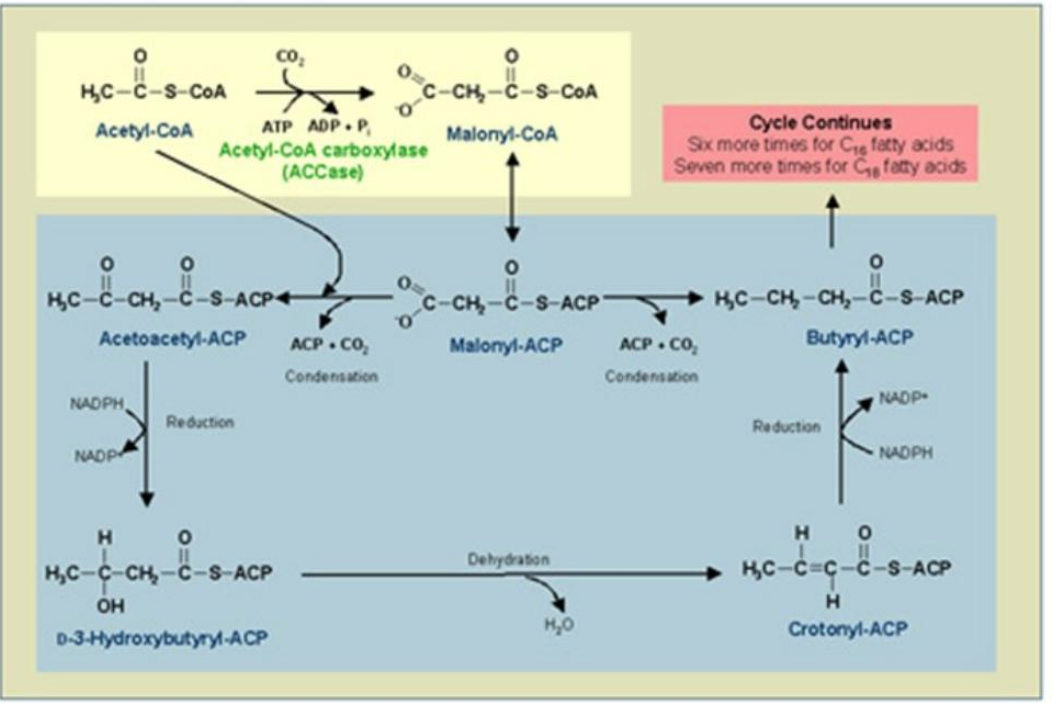
Malonyl-CoA–ACP Transacylase (MT)
____________________:
EC No.: 2.8.3.3
Role: Transfers malonyl groups from CoA to ACP, forming malonyl-ACP.
The Fatty Acid Synthase Complex:
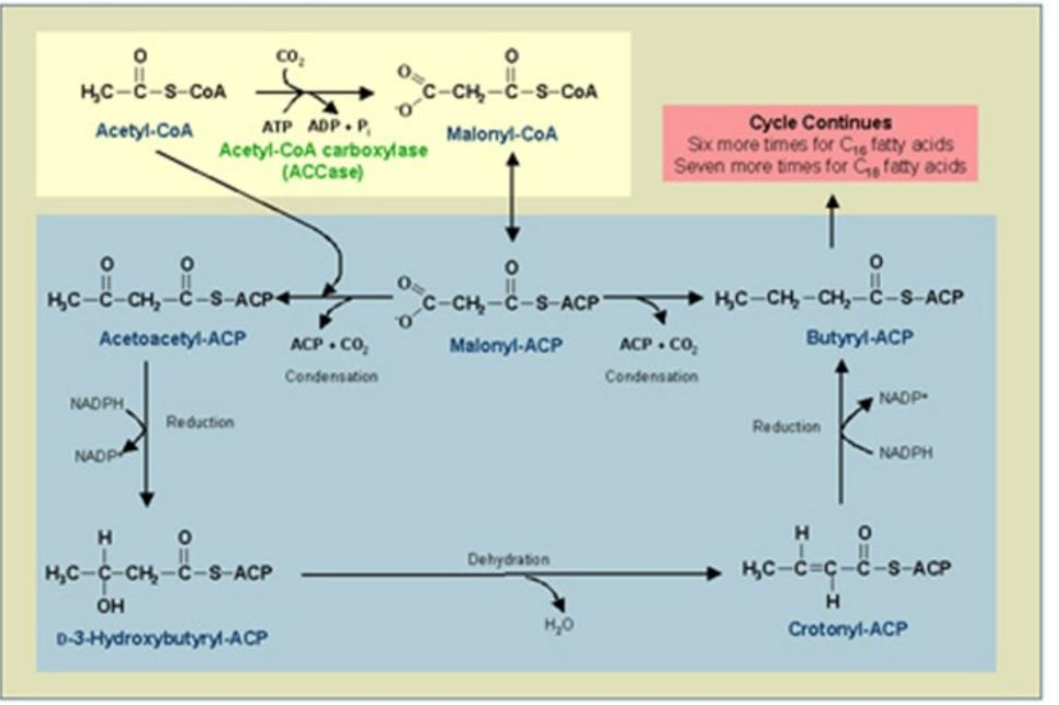
β-Ketoacyl-ACP Synthase (KS)
________________________
EC No.: 2.3.1.16
Role: Catalyzes the condensation reaction between acetyl-ACP and malonyl-ACP, forming β-ketoacyl-ACP.
The Fatty Acid Synthase Complex:
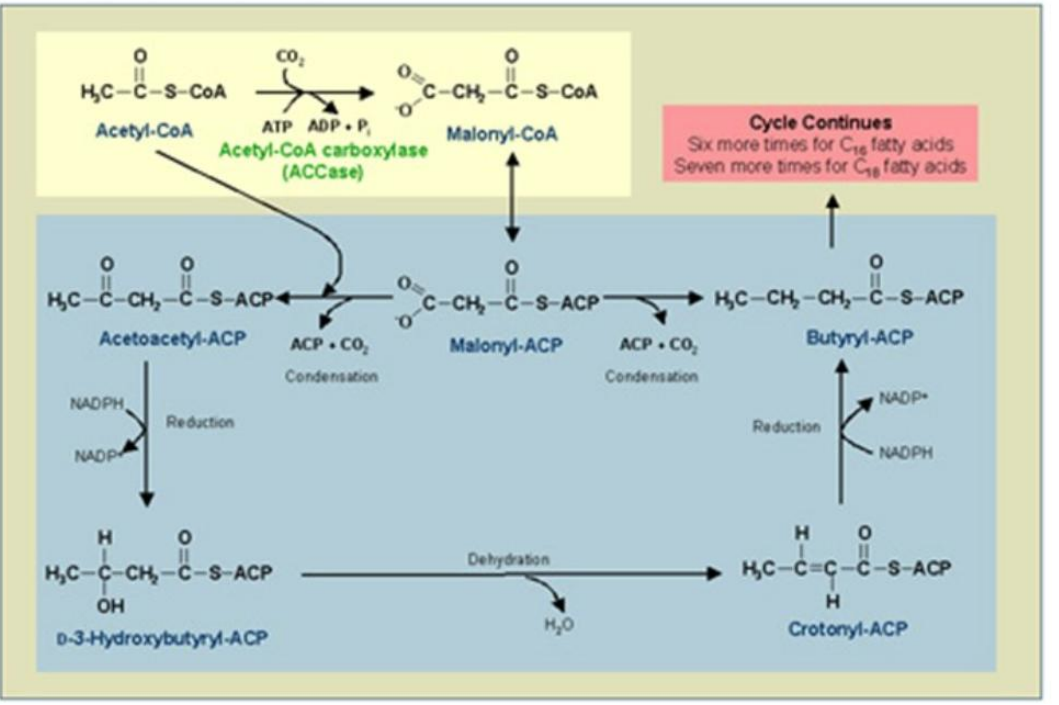
β-Ketoacyl-ACP Reductase (KR)
_________________________:
EC No.: 1.1.1.36
Role: Reduces β-ketoacyl-ACP to β-hydroxyacyl-ACP using NADPH.
The Fatty Acid Synthase Complex:
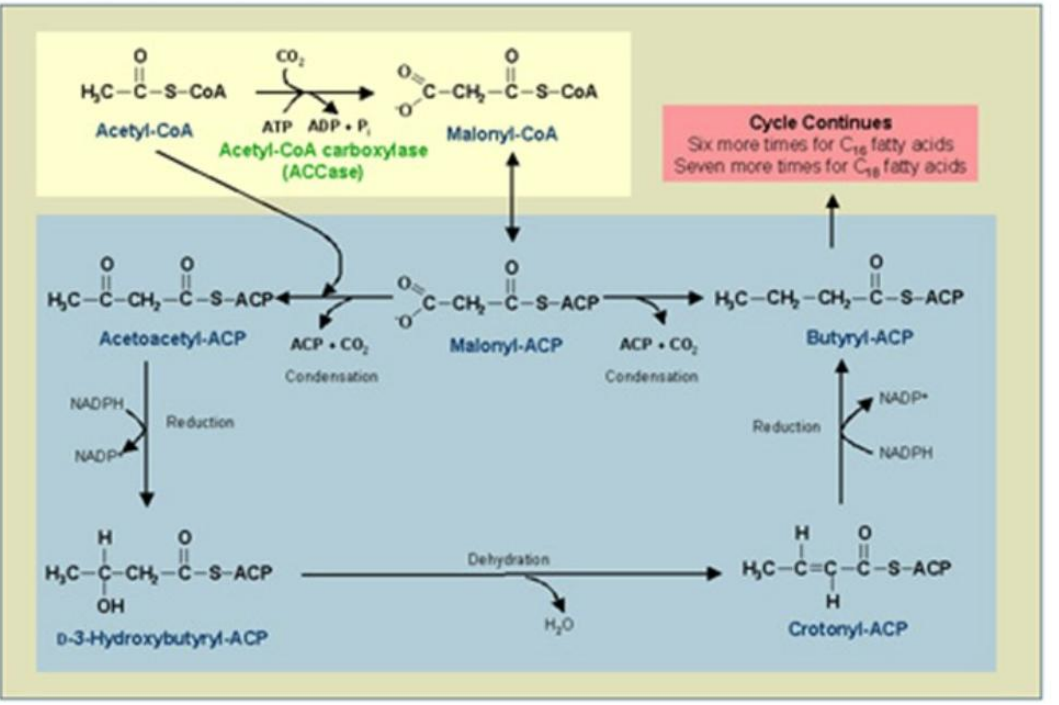
β-Hydroxyacyl-ACP Dehydratase (HD)
________________________:
EC No.: 4.2.1.17
Role: Dehydrates β-hydroxyacyl-ACP, forming enoyl-ACP (with a double bond).
The Fatty Acid Synthase Complex:
Enoyl-ACP Reductase (ER)
____________________:
EC No.: 1.3.99.2
Role: Reduces the double bond in enoyl-ACP, forming a saturated acyl-ACP.
The Fatty Acid Synthase Complex:
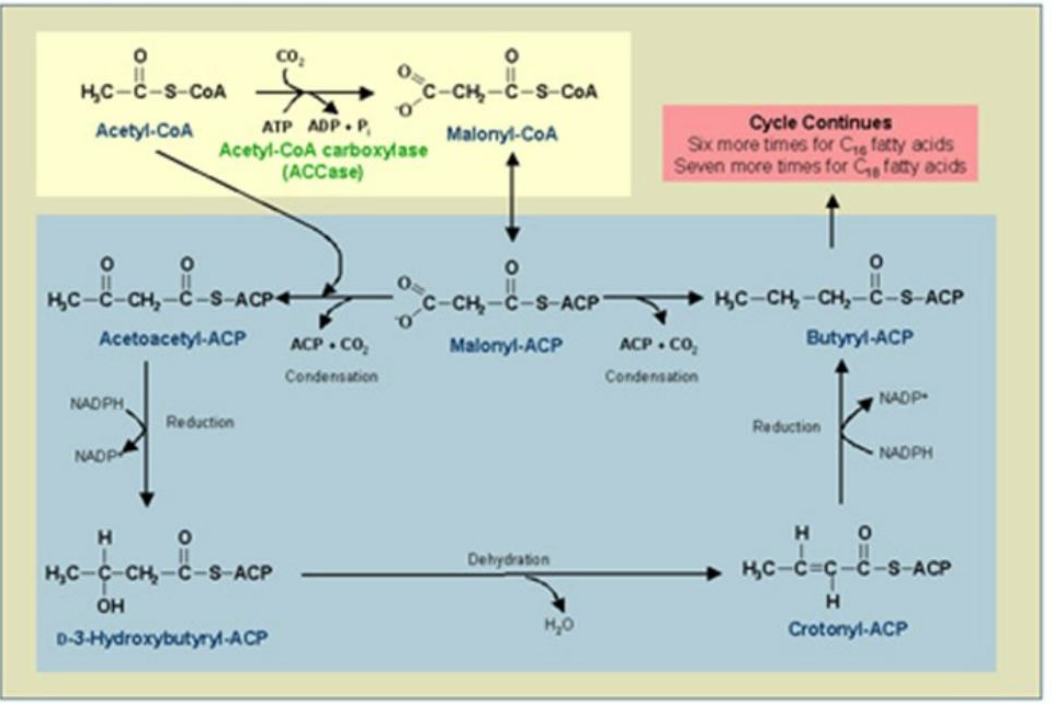
Conversion of Acetyl-CoA to Malonyl-CoA
1. ______________________________:
Reaction: Acetyl-CoA is carboxylated to malonyl-CoA.
Catalyst: Acetyl-CoA Carboxylase (ACC).
Energy Requirement: ATP and bicarbonate (HCO₃⁻).
Synthesis of Fatty Acids
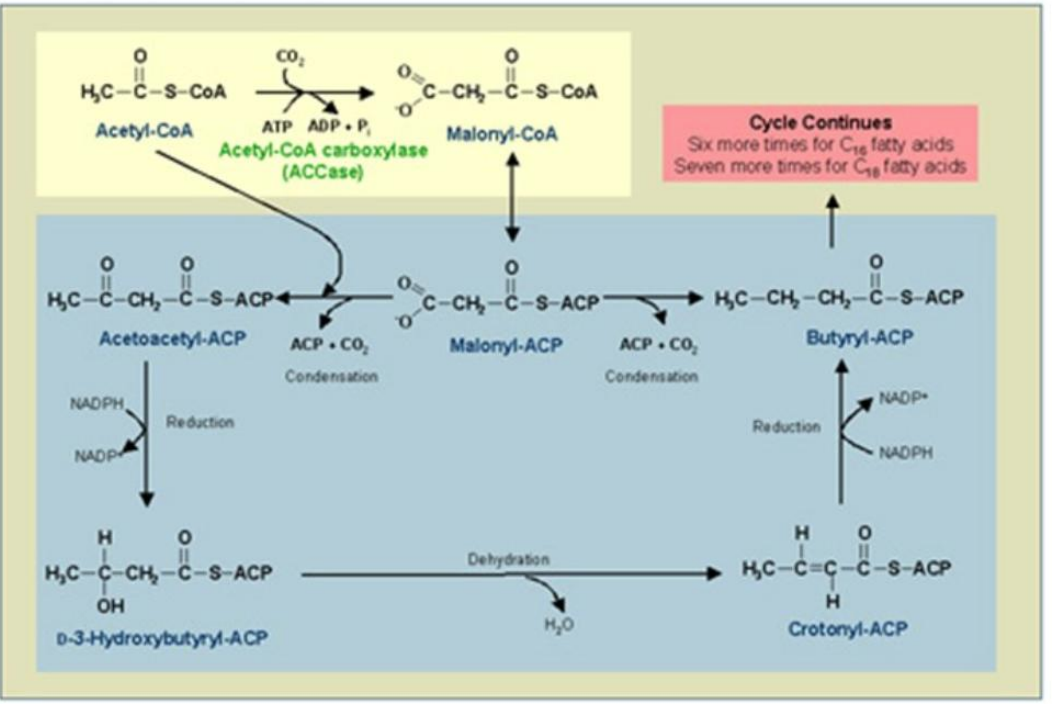
Acetyl-CoA Carboxylase (ACC)
1. Conversion of Acetyl-CoA to Malonyl-CoA:
Reaction: Acetyl-CoA is carboxylated to malonyl-CoA.
Catalyst: ____________________.
Energy Requirement: ATP and bicarbonate (HCO₃⁻).
Synthesis of Fatty Acids
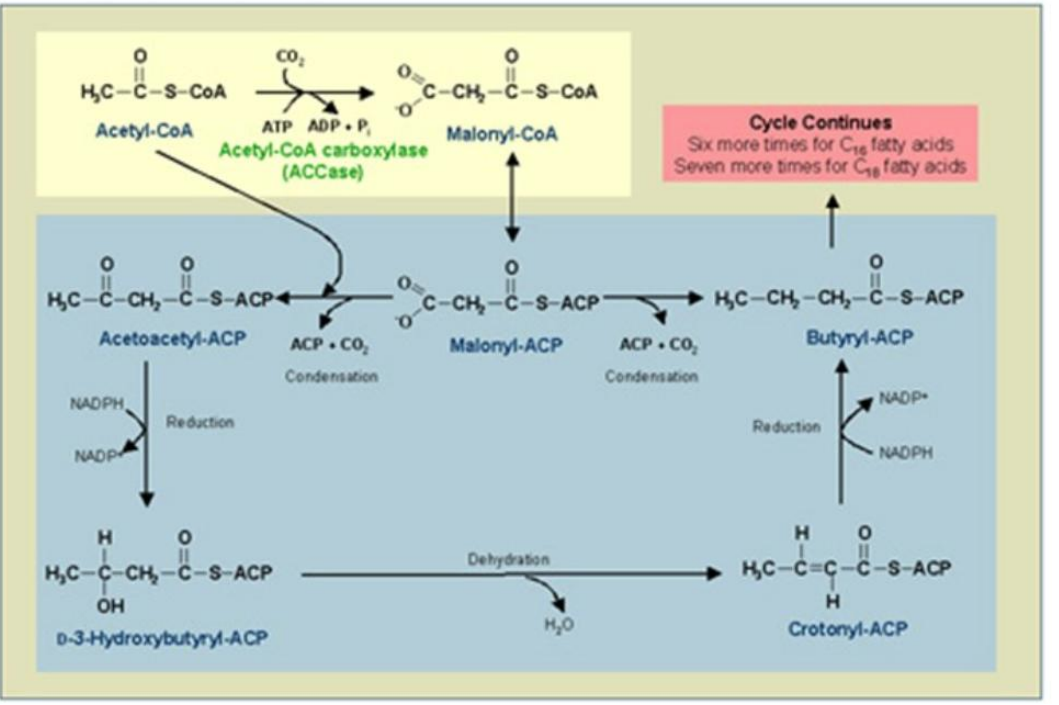
Loading ACP with Acetyl and Malonyl Groups
2. _______________________:
Enzymes Involved:
Acetyl-CoA transacylase.
Malonyl-CoA transacylase.
Synthesis of Fatty Acids
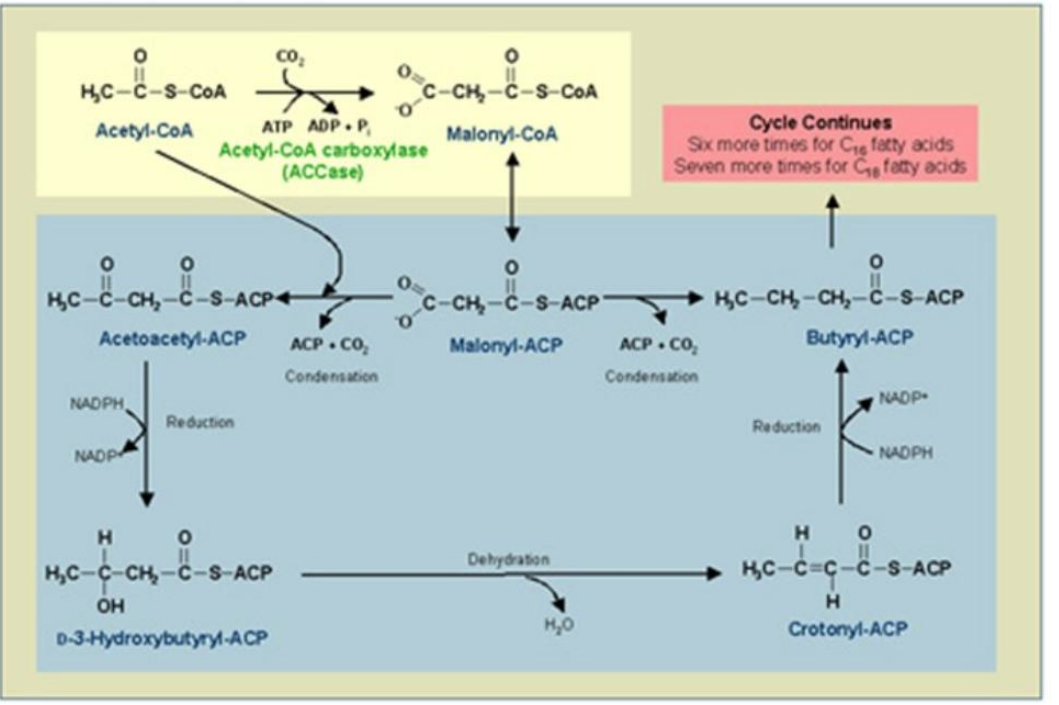
Acetyl-CoA transacylase.
Malonyl-CoA transacylase.
2. Loading ACP with Acetyl and Malonyl Groups:
Enzymes Involved:
____________________
Synthesis of Fatty Acids
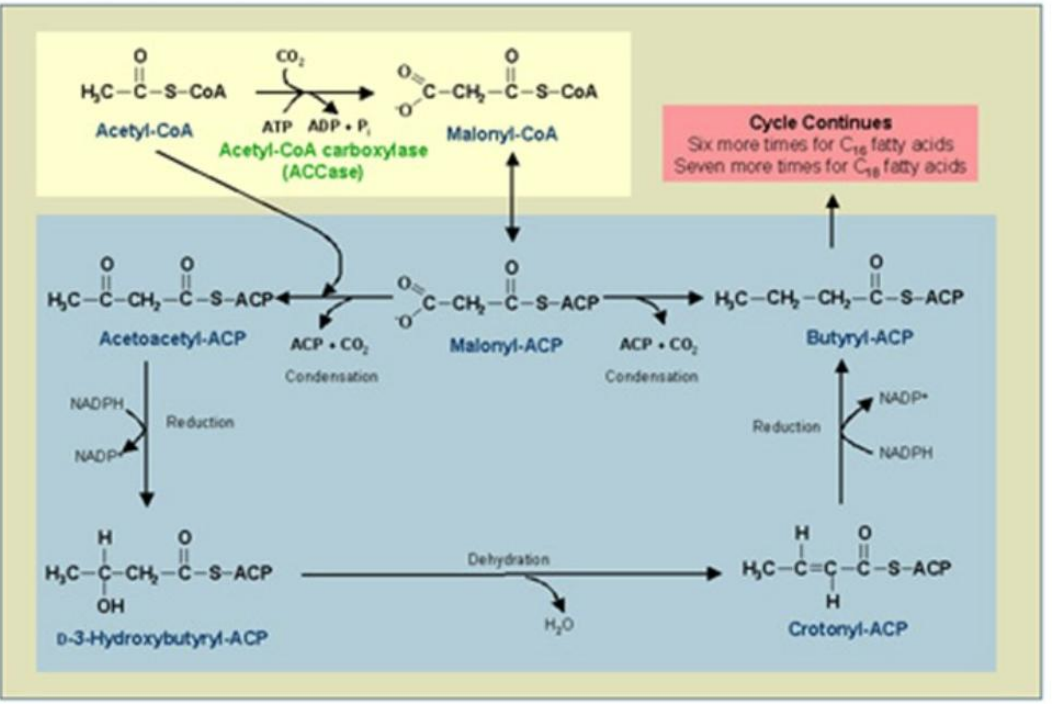
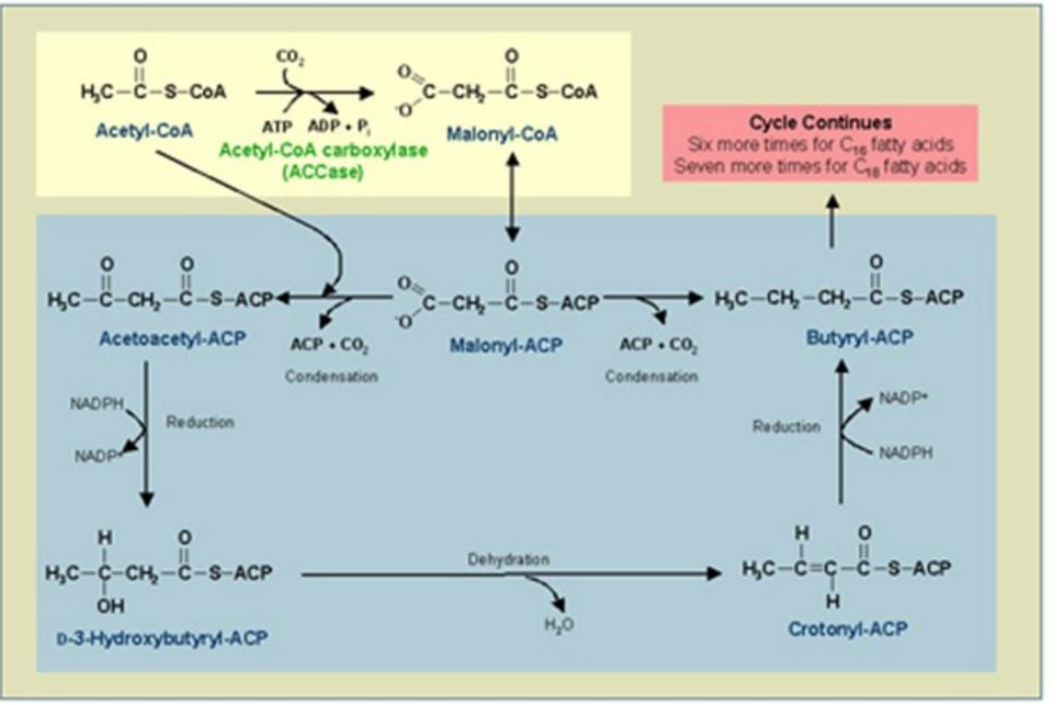
Condensation
_______________:
Acetyl-ACP and malonyl-ACP undergo a condensation reaction, releasing CO₂.
Catalyst: β-Ketoacyl-ACP Synthase (KS).
Product: β-Ketoacyl-ACP.
Synthesis of Fatty Acids
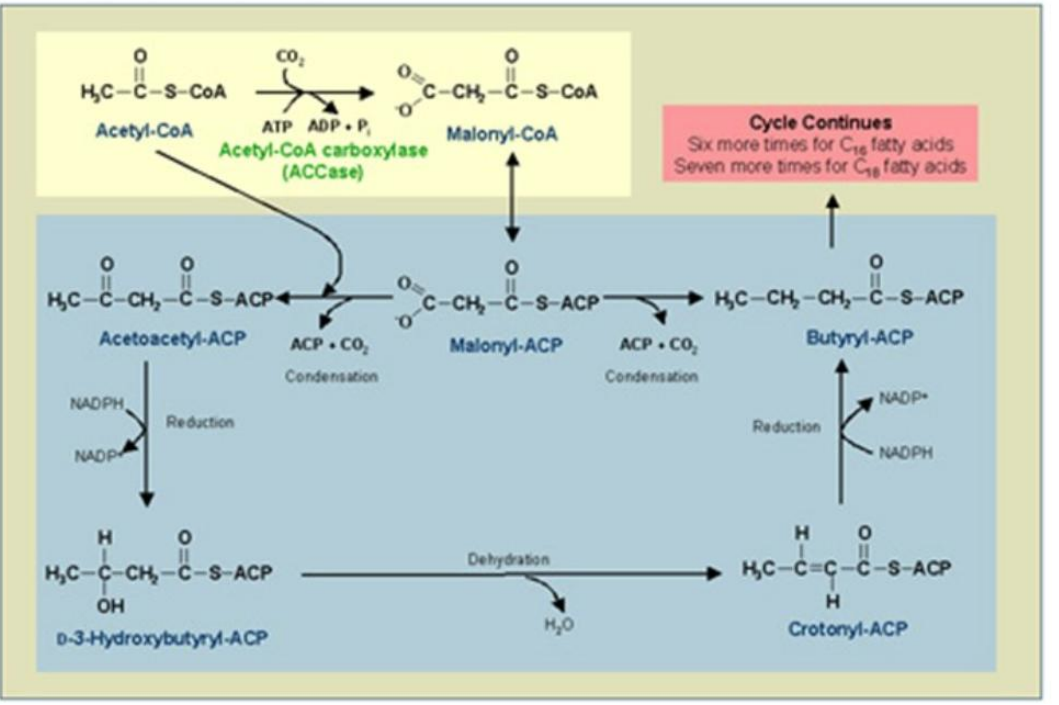
β-Ketoacyl-ACP Synthase (KS)
Condensation:
Acetyl-ACP and malonyl-ACP undergo a condensation reaction, releasing CO₂.
Catalyst: ________________).
Product: β-Ketoacyl-ACP.
Synthesis of Fatty Acids
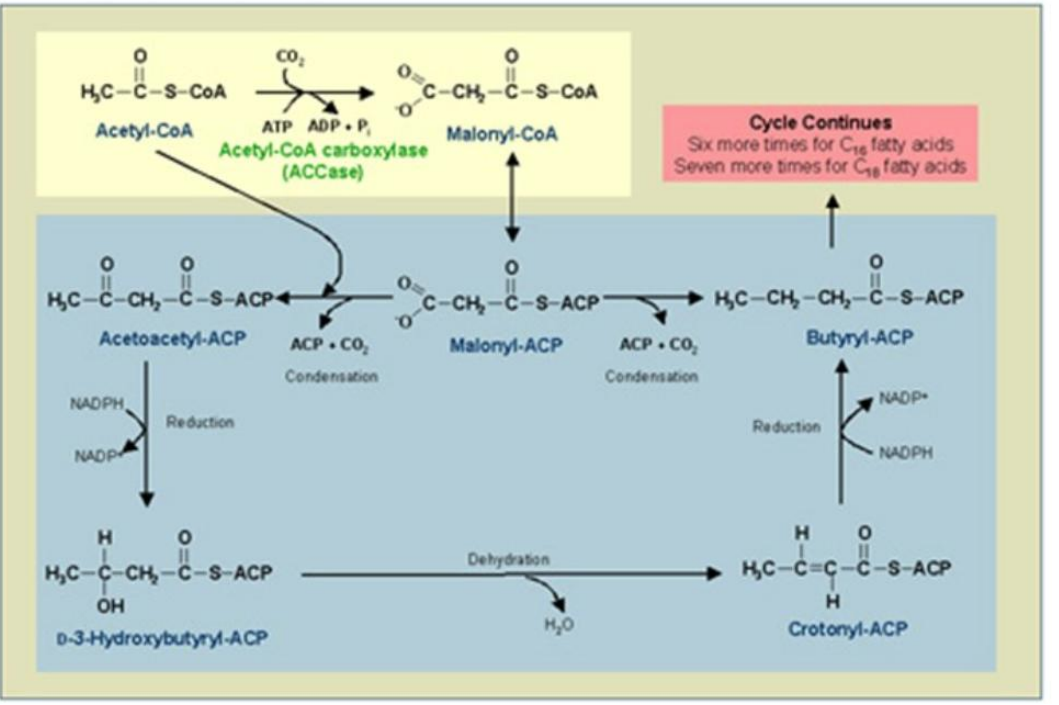
Reduction of β-Ketoacyl-ACP
_________________________:
First Reduction: β-Ketoacyl-ACP is reduced to β-hydroxyacyl-ACP using NADPH.
Catalyst: β-Ketoacyl-ACP Reductase (KR).
Synthesis of Fatty Acids
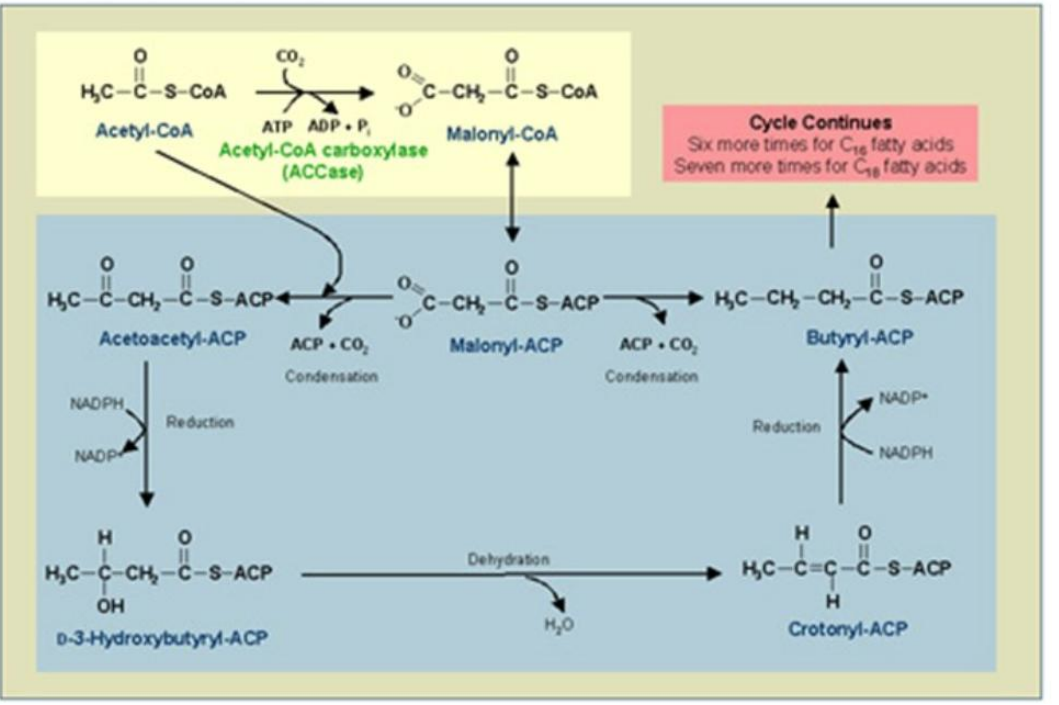
β-Ketoacyl-ACP Reductase (KR)
Reduction of β-Ketoacyl-ACP:
First Reduction: β-Ketoacyl-ACP is reduced to β-hydroxyacyl-ACP using NADPH.
Catalyst: _____________________.
Synthesis of Fatty Acids
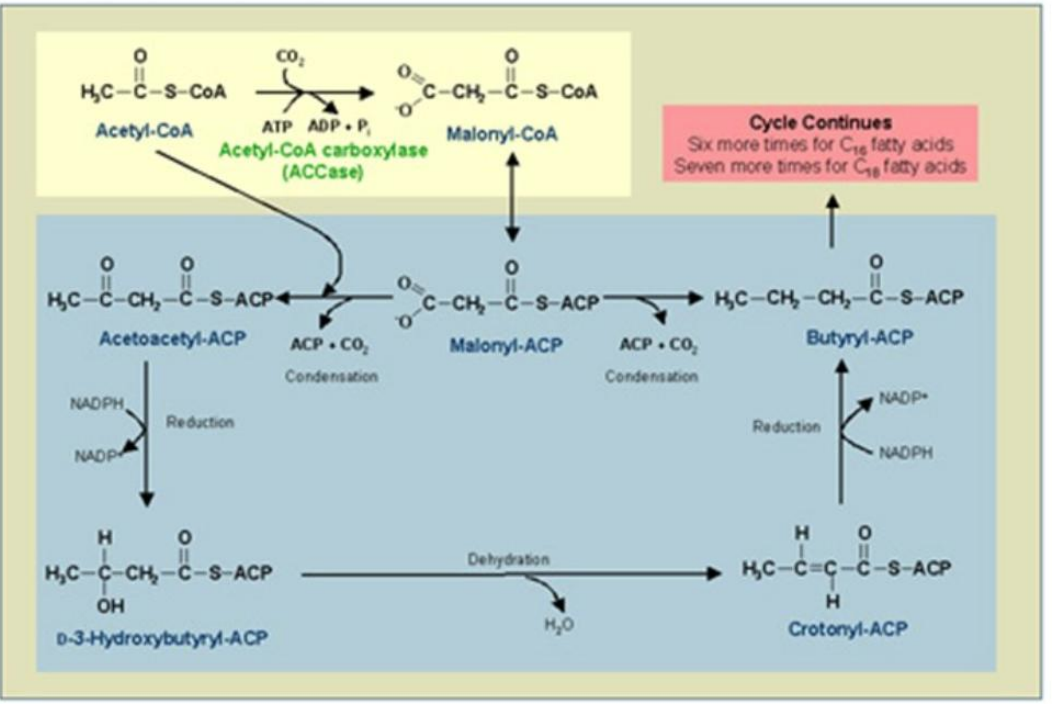
Dehydration of β-Hydroxyacyl-ACP
___________________________:
Water is removed, forming a double bond in enoyl-ACP.
Catalyst: β-Hydroxyacyl-ACP Dehydratase (HD).
Synthesis of Fatty Acids
β-Hydroxyacyl-ACP Dehydratase (HD)
Dehydration of β-Hydroxyacyl-ACP:
Water is removed, forming a double bond in enoyl-ACP.
Catalyst: _______________________.
Synthesis of Fatty Acids
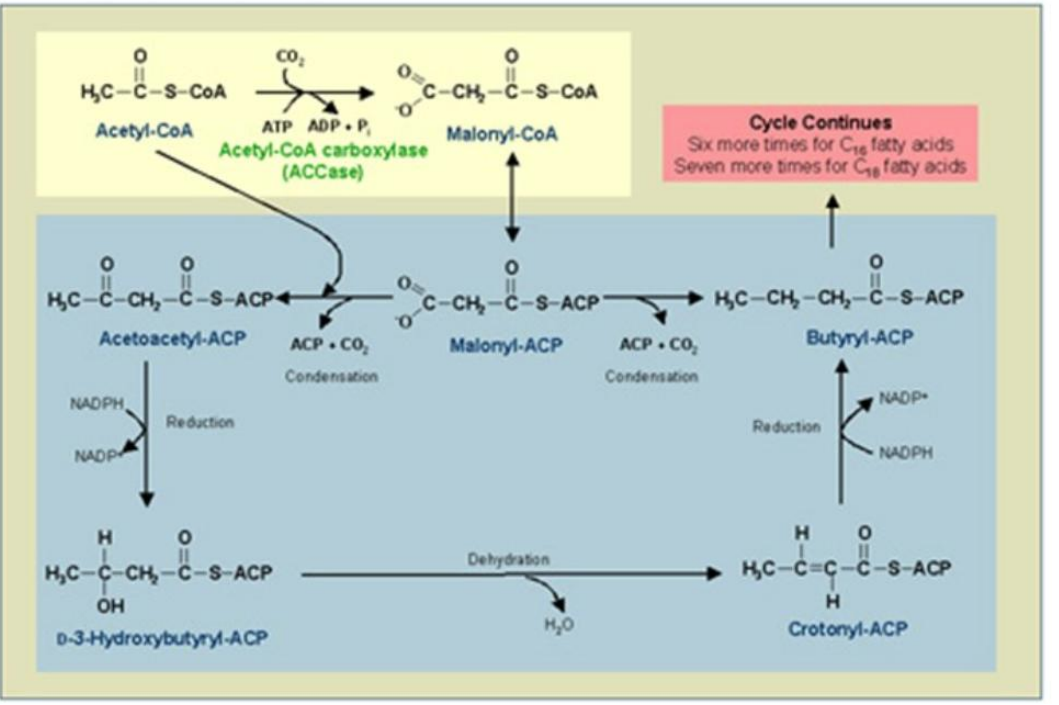
Reduction of Enoyl-ACP:
_____________________
The double bond in enoyl-ACP is reduced, forming a saturated acyl-ACP.
Catalyst: Enoyl-ACP Reductase (ER).
Reductant: NADPH.
Synthesis of Fatty Acids
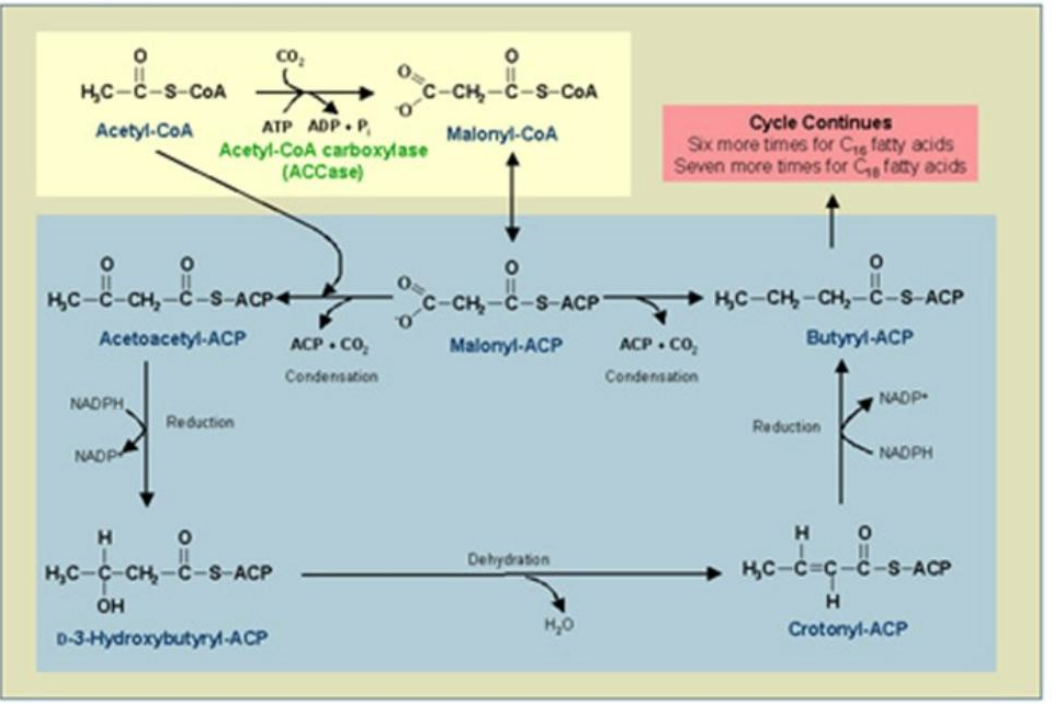
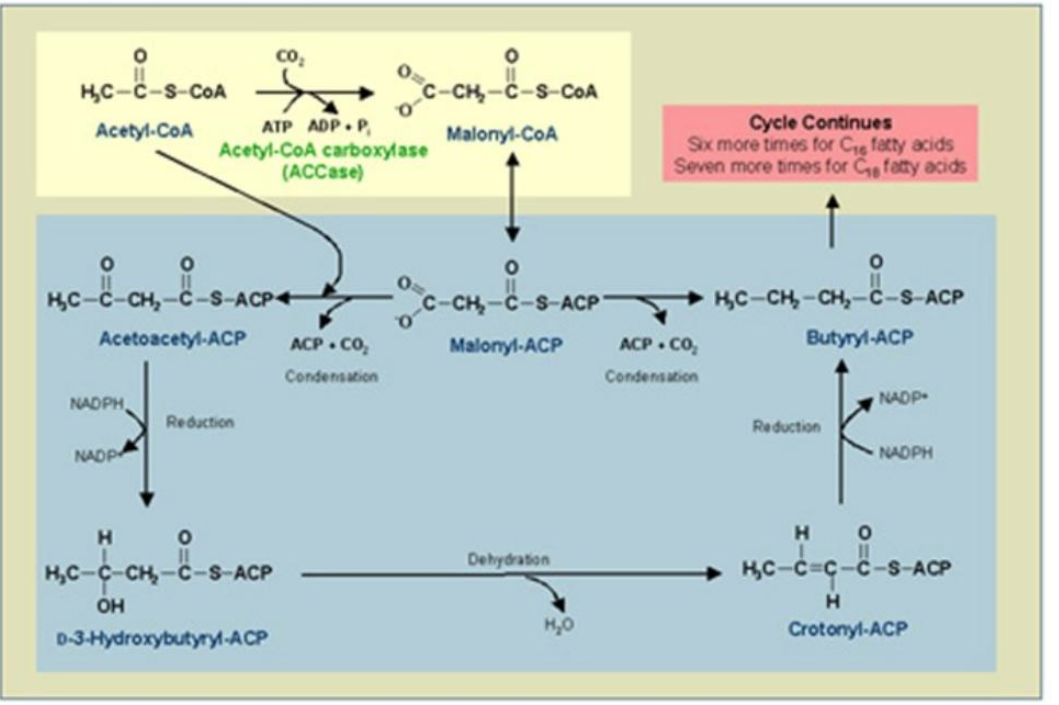
Enoyl-ACP Reductase (ER)
Reduction of Enoyl-ACP:
The double bond in enoyl-ACP is reduced, forming a saturated acyl-ACP.
Catalyst: ___________________.
Reductant: NADPH.
Synthesis of Fatty Acids
Mechanisms for Unsaturated Fatty Acid Synthesis
________________________________
For Aerobic Organisms:
Aerobic organisms utilize molecular oxygen (O₂) and NADPH for introducing double bonds into fatty acid chains.
The process is catalyzed by fatty acyl-CoA desaturase, a membrane-bound enzyme.
Example Reaction:
A saturated fatty acyl-CoA is oxidized using O₂ and electrons donated by NADPH (via cytochrome b5 reductase) to produce a monounsaturated fatty acyl-CoA.
For Anaerobic Organisms:
Anaerobes synthesize unsaturated fatty acids during fatty acid synthesis rather than modifying existing saturated fatty acids.
This is achieved by dehydration of hydroxy fatty acids followed by isomerization:
β-hydroxydecanoyl-ACP → (dehydration) → trans-2-decenoyl-ACP → (isomerization by FabA) → cis-3-decenoyl-ACP.
The unsaturated intermediate undergoes chain elongation to produce unsaturated fatty acids.
Key Points:
Double bonds in fatty acids are introduced via a redox reaction in aerobic pathways.
In anaerobes, double bonds are incorporated enzymatically during the synthesis.
Summary
Aerobic organisms use oxygen-dependent desaturases and electron transfer systems to introduce double bonds.
Anaerobic organisms utilize enzymatic dehydration and isomerization during fatty acid synthesis.
Eukaryotes synthesize polyunsaturated fatty acids using sequential desaturation steps, catalyzed by specific desaturases. These pathways are vital for producing structurally diverse lipids and maintaining cellular functions.
fatty acyl-CoA desaturase
For Aerobic Organisms:
Aerobic organisms utilize molecular oxygen (O₂) and NADPH for introducing double bonds into fatty acid chains.
The process is catalyzed by ____________________, a membrane-bound enzyme.
Example Reaction:
A saturated fatty acyl-CoA is oxidized using O₂ and electrons donated by NADPH (via cytochrome b5 reductase) to produce a monounsaturated fatty acyl-CoA.
Mechanisms for Unsaturated Fatty Acid Synthesis
Summary
Aerobic organisms use oxygen-dependent desaturases and electron transfer systems to introduce double bonds.
Anaerobic organisms utilize enzymatic dehydration and isomerization during fatty acid synthesis.
Eukaryotes synthesize polyunsaturated fatty acids using sequential desaturation steps, catalyzed by specific desaturases. These pathways are vital for producing structurally diverse lipids and maintaining cellular functions.
monounsaturated fatty acyl-CoA
For Aerobic Organisms:
Aerobic organisms utilize molecular oxygen (O₂) and NADPH for introducing double bonds into fatty acid chains.
The process is catalyzed by fatty acyl-CoA desaturase, a membrane-bound enzyme.
Example Reaction:
A saturated fatty acyl-CoA is oxidized using O₂ and electrons donated by NADPH (via cytochrome b5 reductase) to produce a ______________________.
Mechanisms for Unsaturated Fatty Acid Synthesis
Summary
Aerobic organisms use oxygen-dependent desaturases and electron transfer systems to introduce double bonds.
Anaerobic organisms utilize enzymatic dehydration and isomerization during fatty acid synthesis.
Eukaryotes synthesize polyunsaturated fatty acids using sequential desaturation steps, catalyzed by specific desaturases. These pathways are vital for producing structurally diverse lipids and maintaining cellular functions.
dehydration of hydroxy fatty acids
For Anaerobic Organisms:
Anaerobes synthesize unsaturated fatty acids during fatty acid synthesis rather than modifying existing saturated fatty acids.
This is achieved by ______________ followed by isomerization:
β-hydroxydecanoyl-ACP → (dehydration) → trans-2-decenoyl-ACP → (isomerization by FabA) → cis-3-decenoyl-ACP.
The unsaturated intermediate undergoes chain elongation to produce unsaturated fatty acids.
Mechanisms for Unsaturated Fatty Acid Synthesis
Summary
Aerobic organisms use oxygen-dependent desaturases and electron transfer systems to introduce double bonds.
Anaerobic organisms utilize enzymatic dehydration and isomerization during fatty acid synthesis.
Eukaryotes synthesize polyunsaturated fatty acids using sequential desaturation steps, catalyzed by specific desaturases. These pathways are vital for producing structurally diverse lipids and maintaining cellular functions.
Synthesis of Polyunsaturated Fatty Acids (PUFAs)
__________________________
Desaturation Pathways:
Desaturases are enzymes that introduce double bonds into fatty acids.
In eukaryotes, this process often occurs while fatty acids are part of phospholipids, such as phosphatidylcholine.
Examples of Fatty Acid Desaturation:
Oleate (18:1, Δ⁹) → (desaturase) → Linoleate (18:2, Δ⁹,¹²):
A second double bond is introduced between carbons 12 and 13.
Linoleate (18:2, Δ⁹,¹²) → (desaturase) → Linolenate (18:3, Δ⁹,¹²,¹⁵):
A third double bond is added between carbons 15 and 16.
Biological Importance:
These reactions are critical for the synthesis of essential fatty acids, which are precursors for signaling molecules (e.g., prostaglandins and leukotrienes).
Polyunsaturated fatty acids like linoleate and linolenate are integral components of cell membranes, ensuring fluidity and function.
Summary
Aerobic organisms use oxygen-dependent desaturases and electron transfer systems to introduce double bonds.
Anaerobic organisms utilize enzymatic dehydration and isomerization during fatty acid synthesis.
Eukaryotes synthesize polyunsaturated fatty acids using sequential desaturation steps, catalyzed by specific desaturases. These pathways are vital for producing structurally diverse lipids and maintaining cellular functions.
Desaturases
_________________Pathways:
______________ are enzymes that introduce double bonds into fatty acids.
In eukaryotes, this process often occurs while fatty acids are part of phospholipids, such as phosphatidylcholine.
Examples of Fatty Acid Desaturation:
Oleate (18:1, Δ⁹) → (_______________) → Linoleate (18:2, Δ⁹,¹²):
A second double bond is introduced between carbons 12 and 13.
Linoleate (18:2, Δ⁹,¹²) → (desaturase) → Linolenate (18:3, Δ⁹,¹²,¹⁵):
A third double bond is added between carbons 15 and 16.
Biological Importance:
These reactions are critical for the synthesis of essential fatty acids, which are precursors for signaling molecules (e.g., prostaglandins and leukotrienes).
Polyunsaturated fatty acids like linoleate and linolenate are integral components of cell membranes, ensuring fluidity and function.
Synthesis of Polyunsaturated Fatty Acids (PUFAs)
Summary
Aerobic organisms use oxygen-dependent desaturases and electron transfer systems to introduce double bonds.
Anaerobic organisms utilize enzymatic dehydration and isomerization during fatty acid synthesis.
Eukaryotes synthesize polyunsaturated fatty acids using sequential desaturation steps, catalyzed by specific desaturases. These pathways are vital for producing structurally diverse lipids and maintaining cellular functions.
Metabolism of Nucleotides
_________________________
Nucleic Acid Metabolism:
Nucleic acids (DNA and RNA) are composed of nucleotides, which serve as the building blocks.
Roles in Metabolism:
Nucleotide Triphosphates (NTPs):
Provide energy for many metabolic reactions through hydrolysis.
Example: ATP and GTP.
Regulatory Molecules:
Nucleotides play a role in regulating various cellular processes.
RNA/DNA Synthesis:
Nucleotide triphosphates are the precursors used for polymerization into RNA and DNA.
Components of Coenzymes:
Nucleotides form part of important coenzymes like NAD⁺, FAD, and CoA.
Nitrogenous Base
Nucleotide Structure
Basic Components:
____________________:
Purines: Adenine (A) and Guanine (G).
Pyrimidines: Cytosine (C), Thymine (T), and Uracil (U).
Pentose Sugar:
Ribose (in RNA) or deoxyribose (in DNA).
Phosphate Group:
Attached to the 5' carbon of the sugar.
Nucleoside:
Contains only a nitrogenous base and a sugar (no phosphate group).
Nucleotide:
A nucleoside with one or more phosphate groups attached.
Pentose Sugar
Nucleotide Structure
Basic Components:
Nitrogenous Base:
Purines: Adenine (A) and Guanine (G).
Pyrimidines: Cytosine (C), Thymine (T), and Uracil (U).
__________________:
Ribose (in RNA) or deoxyribose (in DNA).
Phosphate Group:
Attached to the 5' carbon of the sugar.
Nucleoside:
Contains only a nitrogenous base and a sugar (no phosphate group).
Nucleotide:
A nucleoside with one or more phosphate groups attached.
Phosphate Group
Nucleotide Structure
Basic Components:
Nitrogenous Base:
Purines: Adenine (A) and Guanine (G).
Pyrimidines: Cytosine (C), Thymine (T), and Uracil (U).
Pentose Sugar:
Ribose (in RNA) or deoxyribose (in DNA).
__________________:
Attached to the 5' carbon of the sugar.
Nucleoside:
Contains only a nitrogenous base and a sugar (no phosphate group).
Nucleotide:
A nucleoside with one or more phosphate groups attached.
Nucleotide
Nucleotide Structure
Basic Components:
Nitrogenous Base:
Purines: Adenine (A) and Guanine (G).
Pyrimidines: Cytosine (C), Thymine (T), and Uracil (U).
Pentose Sugar:
Ribose (in RNA) or deoxyribose (in DNA).
Phosphate Group:
Attached to the 5' carbon of the sugar.
Nucleoside:
Contains only a nitrogenous base and a sugar (no phosphate group).
__________________:
A nucleoside with one or more phosphate groups attached.
Nucleoside
Nucleotide Structure
Basic Components:
Nitrogenous Base:
Purines: Adenine (A) and Guanine (G).
Pyrimidines: Cytosine (C), Thymine (T), and Uracil (U).
Pentose Sugar:
Ribose (in RNA) or deoxyribose (in DNA).
Phosphate Group:
Attached to the 5' carbon of the sugar.
___________________:
Contains only a nitrogenous base and a sugar (no phosphate group).
Nucleotide:
A nucleoside with one or more phosphate groups attached.
6-membered ring | 2 nitrogen atoms
Synthesis of Pyrimidine Nucleotides
Structure of Pyrimidines:
Pyrimidines have a ________________ with ________________.
The ring is synthesized using:
Aspartic acid.
Ammonia (from glutamine).
Carbon dioxide (CO₂).
Key Features:
Pyrimidine rings are built before attaching to ribose-5-phosphate (unlike purines, where the base is built directly on the ribose).
De Novo Pyrimidine Synthesis Pathway
_____________________________
Where It Occurs:
In the cytoplasm.
Does not rely on preexisting nucleotides (hence "de novo" = "from scratch").
Steps:
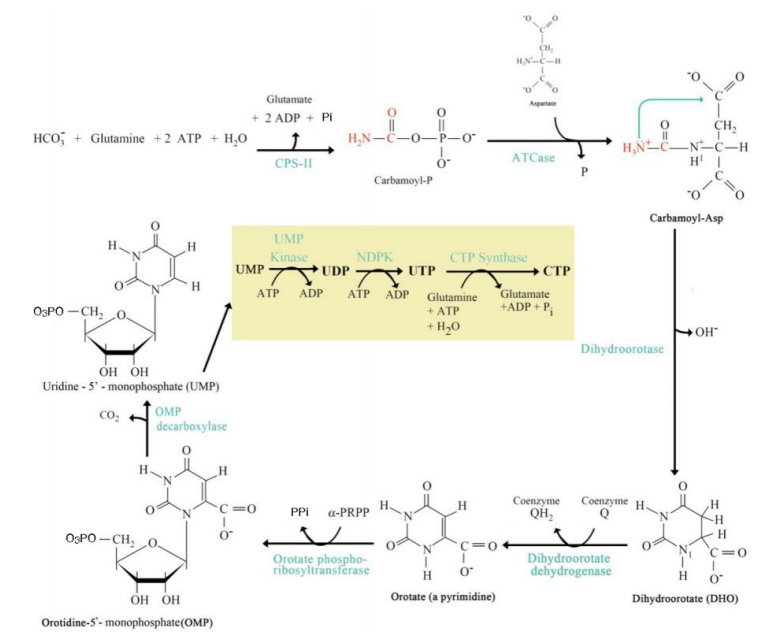
Step 1: Carbamoyl Phosphate Synthesis:
Catalyzed by carbamoyl phosphate synthetase II (CPS-II).
Substrates: HCO₃⁻, glutamine, ATP.
Product: Carbamoyl phosphate.
Step 2: Formation of Carbamoyl Aspartate:
Enzyme: Aspartate transcarbamoylase (ATCase).
Carbamoyl phosphate combines with aspartate.
Step 3: Dihydroorotate Formation:
Cyclization reaction forming dihydroorotate.
Step 4: Orotate Formation:
Dihydroorotate is oxidized to orotate.
Step 5: Addition of Ribose-5-Phosphate:
Orotate reacts with phosphoribosyl pyrophosphate (PRPP) to form orotidine monophosphate (OMP).
Step 6: UMP Formation:
OMP decarboxylated to form uridine monophosphate (UMP).
Step 7: UTP and CTP Formation:
UMP is phosphorylated to form UTP.
UTP is aminated to form CTP.
Significance:
UMP, UTP, and CTP are essential pyrimidine nucleotides used in RNA synthesis.
CTP serves as a precursor for lipid biosynthesis and RNA polymerization.
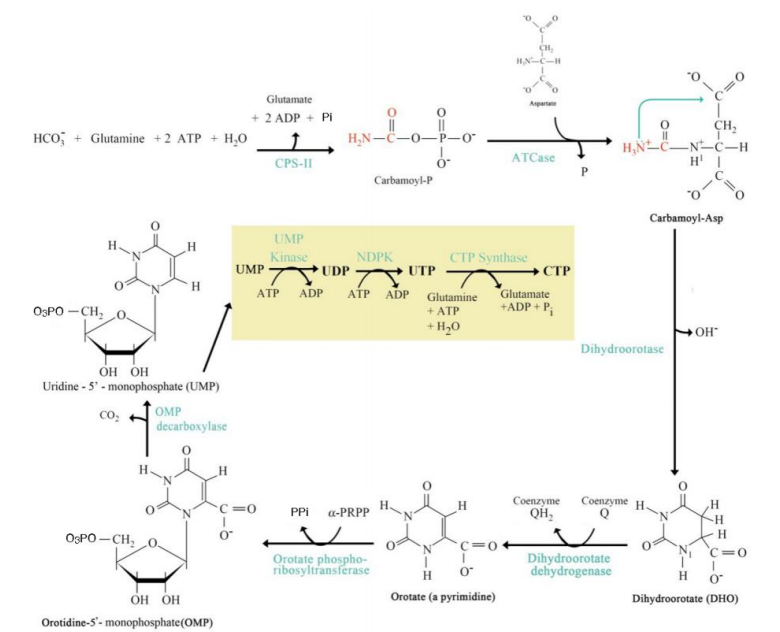
Carbamoyl Phosphate Synthesis
Step 1: _______________________:
Catalyzed by carbamoyl phosphate synthetase II (CPS-II).
Substrates: HCO₃⁻, glutamine, ATP.
Product: Carbamoyl phosphate.
De Novo Pyrimidine Synthesis Pathway
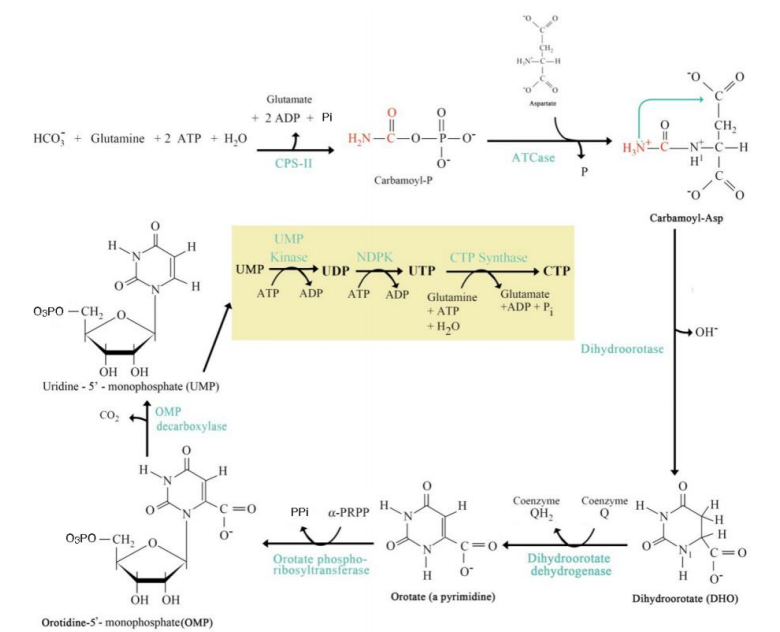
carbamoyl phosphate synthetase II (CPS-II).
Step 1: Carbamoyl Phosphate Synthesis:
Catalyzed by ___________________
Substrates: HCO₃⁻, glutamine, ATP.
Product: Carbamoyl phosphate.
De Novo Pyrimidine Synthesis Pathway
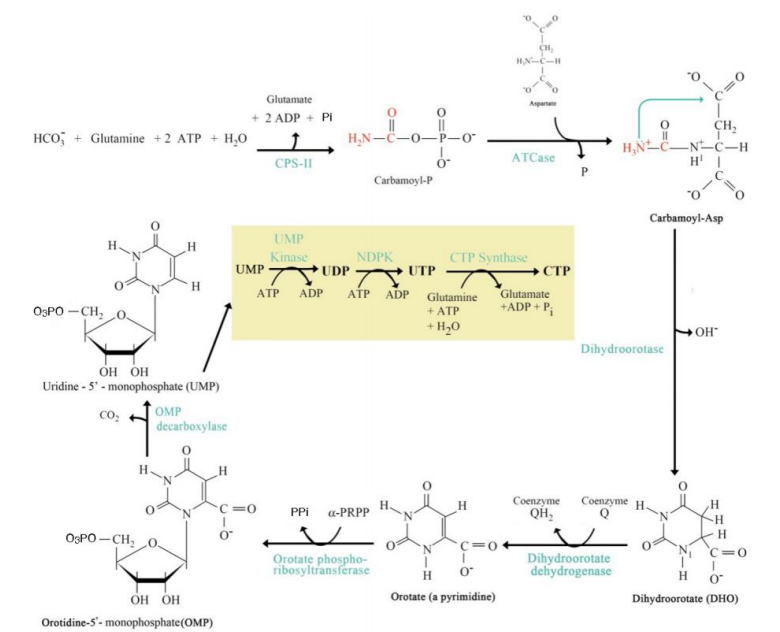
Formation of Carbamoyl Aspartate
Step 2: _________________________:
Enzyme: Aspartate transcarbamoylase (ATCase).
Carbamoyl phosphate combines with aspartate.
De Novo Pyrimidine Synthesis Pathway
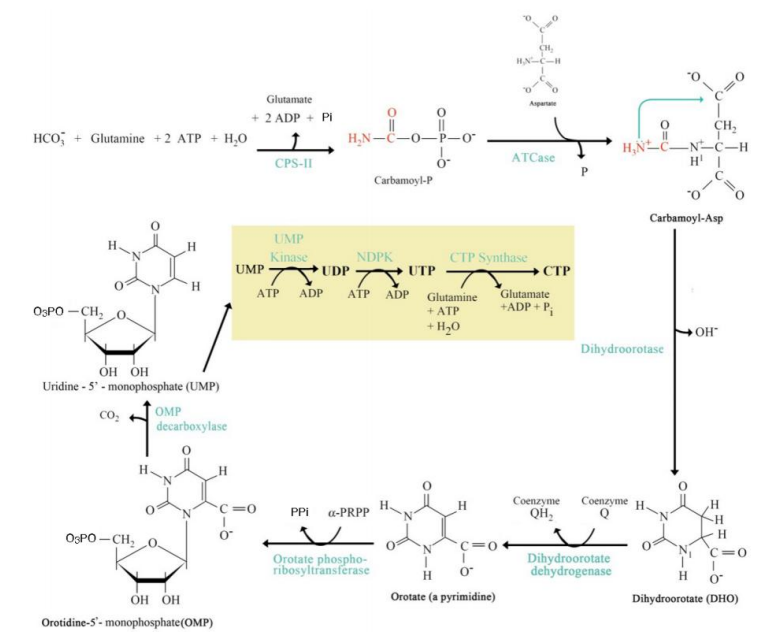
Aspartate transcarbamoylase (ATCase)
Step 2: Formation of Carbamoyl Aspartate:
Enzyme: ___________________.
Carbamoyl phosphate combines with aspartate.
De Novo Pyrimidine Synthesis Pathway
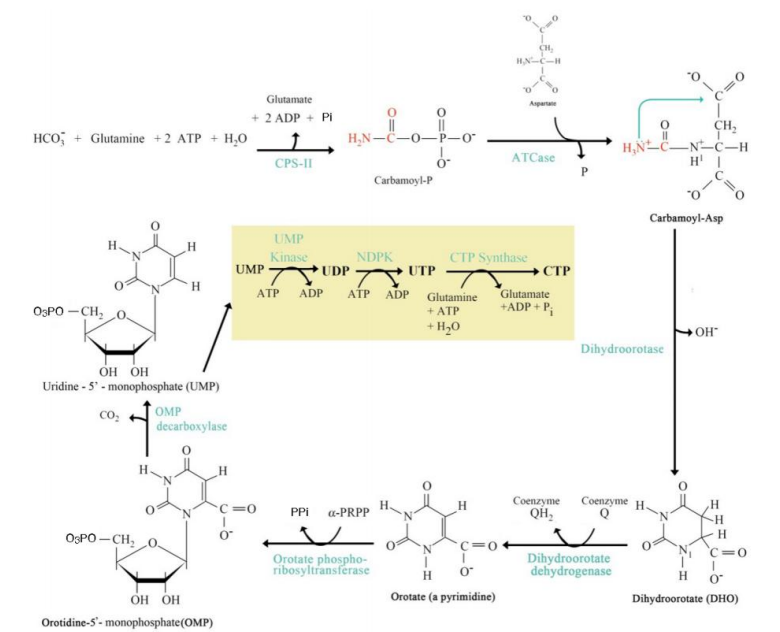
Dihydroorotate Formation
Step 3: ____________________:
Cyclization reaction forming dihydroorotate.
De Novo Pyrimidine Synthesis Pathway
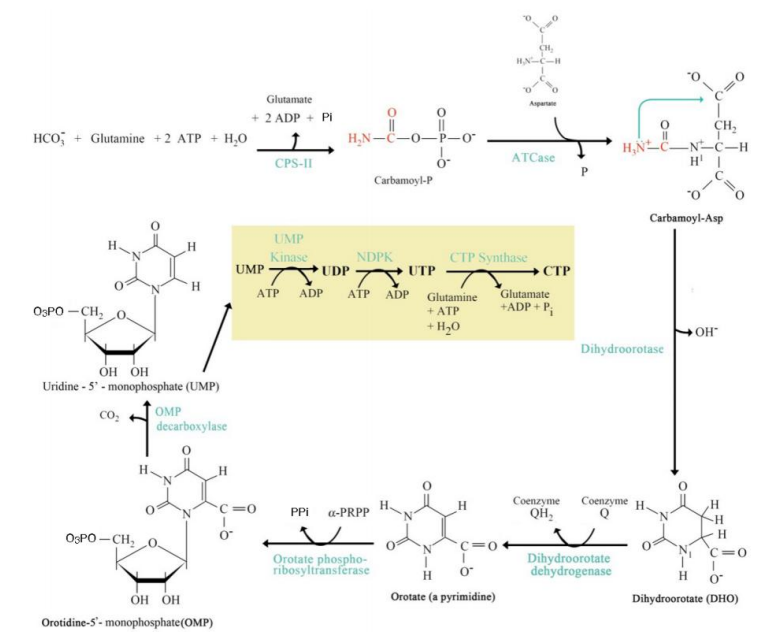
Orotate Formation
Step 4: _______________:
Dihydroorotate is oxidized to orotate.
De Novo Pyrimidine Synthesis Pathway
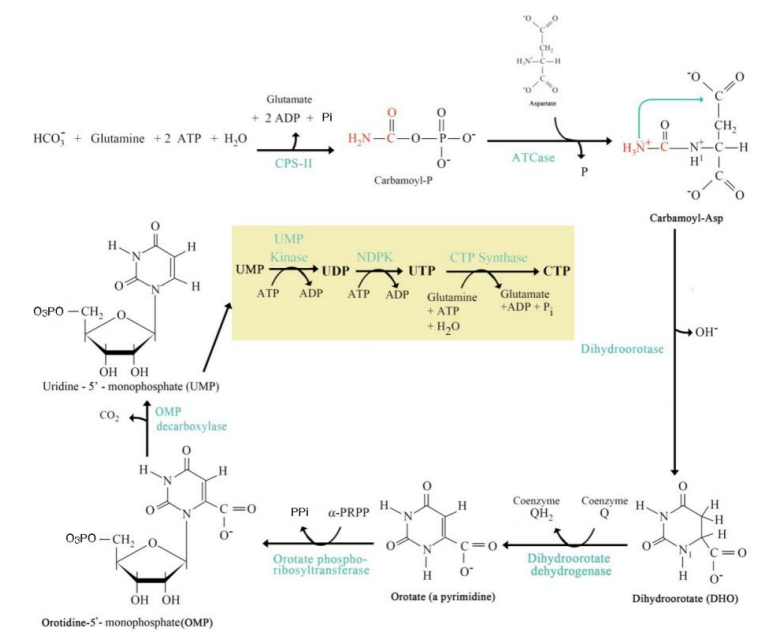
Addition of Ribose-5-Phosphate
Step 5: ______________________:
Orotate reacts with phosphoribosyl pyrophosphate (PRPP) to form orotidine monophosphate (OMP).
De Novo Pyrimidine Synthesis Pathway
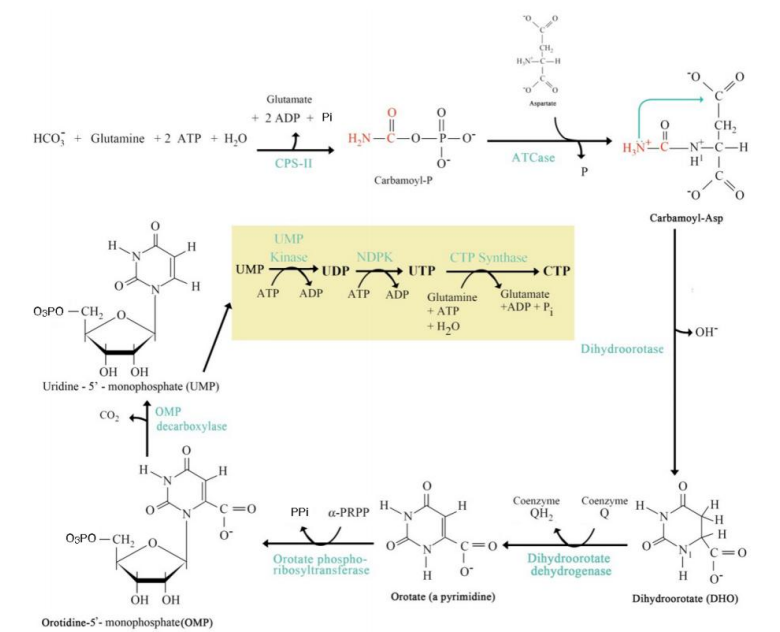
UMP Formation
Step 6: ____________________:
OMP decarboxylated to form uridine monophosphate (UMP).
De Novo Pyrimidine Synthesis Pathway
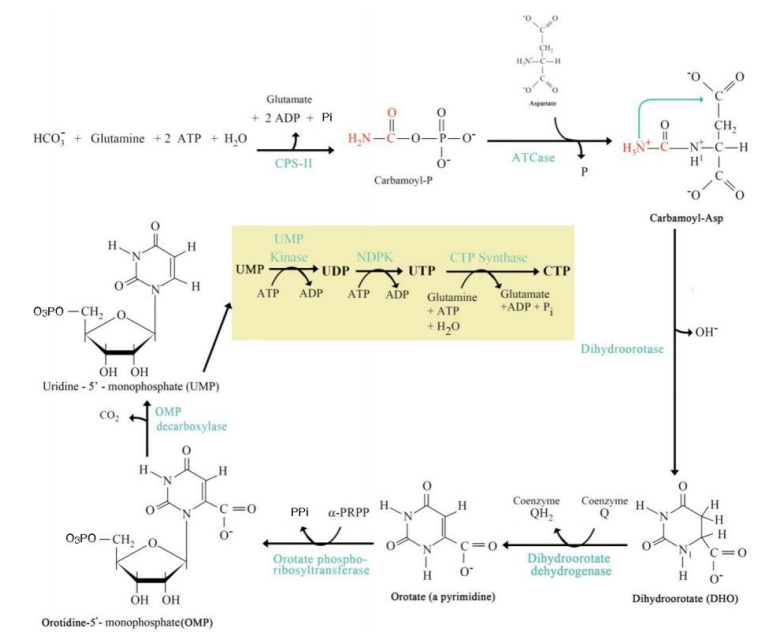
UTP and CTP Formation
Step 7: __________________________:
UMP is phosphorylated to form UTP.
UTP is aminated to form CTP.
De Novo Pyrimidine Synthesis Pathway
salvage pathway of pyrimidine nucleotides
his diagram illustrates the ______________________, a metabolic process that recycles pre-existing pyrimidine bases (e.g., uracil, cytosine) or nucleosides to synthesize nucleotide derivatives (like UTP, CTP) without the need for de novo synthesis.
Key Concepts:
Purpose:
Salvage pathways reuse free bases and nucleosides derived from nucleotide degradation or obtained from the diet.
It is energetically more efficient than de novo synthesis since it avoids starting from scratch.
Pathway Details:
Pyrimidines like uracil, cytosine, and thymine can be converted into their corresponding nucleosides and eventually phosphorylated to nucleotides (e.g., UTP, CTP).
Enzymes facilitate each step in the pathway. The corresponding enzyme commission (EC) numbers and genes associated with each reaction are shown in the diagram.
Steps:
Uridine and cytidine: Converted to uridine monophosphate (UMP) or cytidine monophosphate (CMP) via nucleoside kinases.
Phosphorylation: UMP is phosphorylated sequentially to UDP and UTP by nucleoside monophosphate kinases.
CTP Formation: UTP is aminated by CTP synthetase to form CTP.
Key Enzymes:
Cytosolic enzymes:
Nucleoside kinases (phosphorylation of nucleosides).
UMP-CMP kinase and NDP kinases (for UTP formation).
CTP synthetase: Converts UTP to CTP.
Recycling:
Pyrimidine bases (uracil, cytosine) can directly re-enter the pathway using enzymes like uridine phosphorylase or cytidine deaminase to form nucleosides.
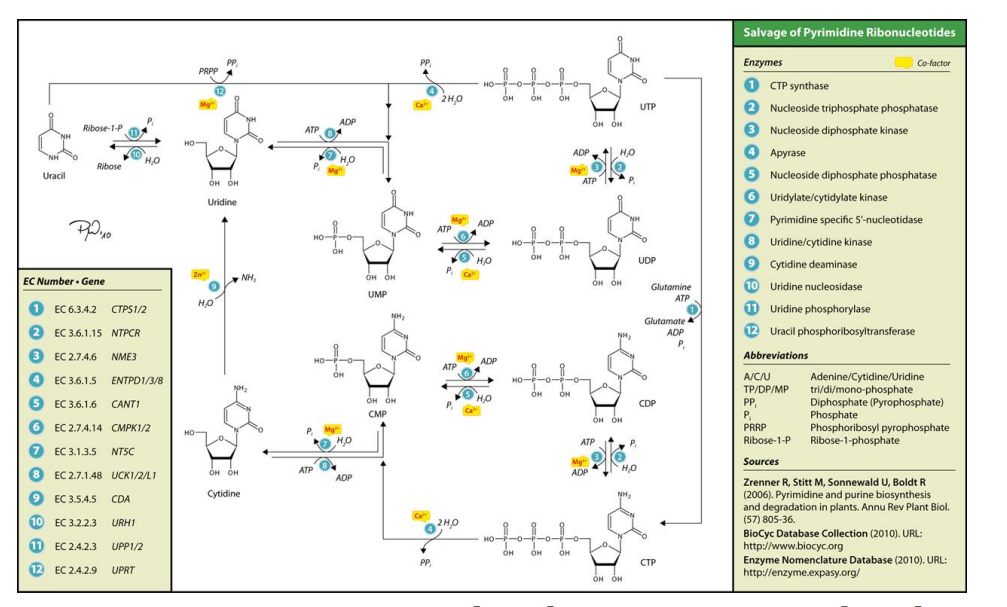
nucleoside monophosphate kinases
Uridine and cytidine: Converted to uridine monophosphate (UMP) or cytidine monophosphate (CMP) via nucleoside kinases.
Phosphorylation: UMP is phosphorylated sequentially to UDP and UTP by _____________________________.
CTP Formation: UTP is aminated by CTP synthetase to form CTP.
salvage pathway of pyrimidine nucleotides steps