Mitochondria
1/98
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
Similarities between eukaryotic and prokaryotic cells
both have cell membrane, cytoplasm, and DNA
Differences between eukaryotic and prokaryotic cells
-Prokaryotes don’t have mitochondria, nucleus, chloroplasts
-Eukaryotes are more advances and have organelles
Mitochondria and Chloroplasts similarities
both involved in converting energy from one form to another
-both only present in eukaryotic cells
-both have multiple layers of membranes filled with enzymes
Similarities between mitochondria/chloroplasts to bacteria
-Contain their own DNA, have the ability to replicate and make RNA/protein
-Their DNAS are circular and its sequence resembles that of bacteria
-The DNA lacks histones/ribosomes (like in bacteria)
-Mitochondria and Chloroplasts undergo fission (which is how bacteria divide)
How is mitochondria inherited
Maternally inherited
What does mitochondria do
cellular respiration (produces chemical energy (ATP) from food)
What is mitochondria comprised of
outer membrane, inner membrane (has lots of folding), matrix, intermembrane space
How does mitochondria adapt to meet energy demand
-Size depends on the cell and its energy demand
-Mostly located in the cytosol where energy demand is highest
-Number of mitochondria increase with increasing energy demand
Additional functions of mitochondria
Calcium regulation, cell death, antiviral actions, regulation of stem cells
What’s mitochrondria fission
The process by which mitochondria divides (similar to bacteria)
What is mitochondrial fusion
Mitochondria fuse together so that damaged and healthy mitochrondria and mix
What are cristae and what do they do
They are inner membrane folds that increase surface area which allows for more ATP to be produced by the ETC
What do porins do and where are they found
Found in the outer membrane and they allow passage of water, pyruvate, and other small molecules into the intermembrane space
Does inner membrane have porins and why
No, because it has proteins of ETC, ATP Synthase, and pumps to move soluted in and out of the matrix
Is the inner membrane permeable and why
IMpermeable because of its unique lipid composition
What does the matrix contain a lot of and what are they used for
Contains lots of proteins making it viscous and they are used in the citric acid cycle to generate ATP
What does matrix contain in addition to proteins
DNA and ribosomes (MT has about 37 genes but around 1000 proteins that are synthesized by nuclear DNA)
What lipids are a part of mitochondria
Phosphatidylcholine and Cardiolipin
Where does mitochondria get its lipids
the ER
What is outer membrane lipid composition similar to
Plasma membrane
What happens to some of the imported lipids in mitochondria
It gets converted to cardiolipin
Whats cardiolipin
A phospholipid with 4 hydrophobic tails which helps make inner membrane impermeable
How much cardiolipin in inner and outer membranes
Makes up 20% of lipid in the inner membrane, None in outer membrane
What is mitochondrial fussion stimulated by
energy demand/stress
What does mitochondial fission do
generates new organelles and facilitates quality control
Mitochondrial encephalomytopathy with lactic acidosis and stroke-like episodes (MELAS)
caused by mutations in several mitochondrial genes
affects brain and muscles
Myoclonic epilepsy with ragged-red fibers (MERRF)
caused by mutations in several mitochondrial genes
affects brain and muscles
Leigh syndrome
caused by mutations in several mitochondrial genes
affects brain and spinal cord
Kearns Sayre syndrome
caused by deletions/rearrangements in mtDNA
Affects brain, spinal cord, eyes, and muscles
Alpers syndrome
caused by mutations in the POLG gene (polymerase gamma), which functions to replicate/maintain mtDNA
affects brain, spinal cord, and muscles
Aging related diseases like ALS, Alzheimer’s, cancer metabolic disorders, loss of muscle function
caused by epigenetic modifications (mutations/methylation changes) that alter expression of MT genes, ATP production, and calcium storage
What do high-energy electrons from NADH and FADH2 do
drive oxidative phosphorylation of ADP → ATP
Inner mitochondria membrane role in oxidative phosphorylation
Acts as an energy device that converts the high energy of electrons stored in NADH and FADH into phosphate bond of ATP
What does oxidative phosphorylation involve
consumption of O2 and additions of a phosphate group to ADP to form ATP
Where does ETC take place
inner membrane
How do NADH and FADH2 transfer electrons to ETC
through oxidizing other molecules
What do electrons pass through in the ETC and what happens when they pass
They pass through a series of electron acceptor/donor molecules that form the chain.
Electrons fall to successively lower energy states as they pass through the chain
What is the energy released from electrons passing through the chain used for
It drives H+ ions (protons) across the membrane into the intermembrane space from the matrix
What does driving protons across the membrane do
generates a transmembrane gradient of protons that serve as a source of energy for phosphorylation of ADP to generate ATP
What happens at the end of the electron transport chain
electrons are added to O2 molecules, which combines with H+ to produce H2O
How does ETC begin
the remove of a hydride ion (a hydrogen atom w/extra electron) from NADH or FADH2
What happens to hydride ion when it is removed from NADH or FADH2
Enters ETC and the NADH dehydrogenase complex where it is converted into a proton and 2 high-energy electrons
What does removal of high-energy electrons from NADH and FADH2 do
Creates NAD+ and FAD which can be reduced again in the citric acid cycle
What does NADH dehydrogenase complex do
catalyzes the reaction of hydride ion (H-) into a proton and 2 electrons
Ubiquionone
a small hydrophobic molecule in the bilayer that carries electrons/donates them (doesn’t generate protons)
Cytochrome C
carry electrons/donates them (doesn’t generate protons)
What are the 3 complexes in ETC (in order)
(1) NADH dehydrogenase complex (2) Cytochrome c reductase complex (3) cytochrome c oxidase complex)
What do all 3 complexes in ETC have
Each possesses metal ions and chemical groups that form a pathway for electrons through the enzyme
LEO goes GER
Lose electron → oxidation
Gain electron → reduction
What does oxidized ubiquinone do
Receives electrons from the NADH dehydrogenase complex (or directly from FADH2) and then picks up protons from the aqueous enviornment
What does reduced ubiquinone do
Passes electrons and protons to the cytochrome c reductase complex
What does cytochrome c reductase complex do
pumps protons into the intermembrane space and passes on the electrons to cytochrome c
Why might certain cells have abundant inner membranes
Because they are more active and may need more energy (like muscle cells)
What’s the total electrochemical gradient of H+ across the IMM
-large force due to membrane potential
-small force due to H+ concentration gradient
what’s proton-motive force
the role of membrane potential + pH gradient that adds to the driving force pulling H+ across the membrane (low pH to high pH) = electrochemical gradient
Essential requirements for harnessing ATP energy
(1) membrane containing a series of electron carriers (ETC), a pump protein, and ATP synthase
(2) pump harnesses energy of electron trasnfer to pump protons and creates proton gradient across membrane
(3) source of high-energy electrons (e-) derived from the oxidation of food
What does the proton gradient do
Serves as an energy store and is used to drive the synthesis of ATP by the ATP synthase
Stage 1 of chemiosmosis
High-energy electron passing through proton pump drives proton across IMM AGAISNT its gradient
Concentration of proton gradient created by proton pumping in the intermembrane versus matrix
High concentration of protons in the intermembrane space (pH = 7.2) than the matrix (pH = 7.9)
Stage 2 of chemiosmosis
Electrochemical H+ gradient drives protons through ATP synthase which phosphorylates ADP to ATP
Whats Chemiosmotic coupling
ATP production (chemi) is brought about by transport across a membrane (osmosis)
Batteries and chemical reactions
Chemiosmotic mechanisms allow cell to harness energy of electron transfer in the same way energy stored in batteries can be harnessed to do smth
Energy of electrons from NADH
enough to pump protons across IMM at 3 places
Energy of electrons from FADH2
donated to ubiquinone and have enough energy to pump protons across IMM at 2 places
How does proton gradient lead to generation of ATP
ATP synthase forms pathway for protons across the IMM and the protons pumped through ATP synthase by the electrochemical proton gradient provide energy to phosphorylate ADP to ATP
What is ATP synthase composed of
head portion (F1 ATPase) and transmembrane H+ carrier (F0 Rotor) → both are multi-subunit complexes
How much ATP can ATP synthase produce per second
100 (3 molecules of ATP per revolution)
What does it mean if ATP synthase is a reversible coupling device
ATP can convert energy of electrochemical proton gradient into chemical-bond energy, and vice versa meaning that it can (1) synthesize ATP w/H+ gradient or (2) pump protons against electrochemical gradient by hydrolyzing ATP to make ADP depending on which direction the rotation goes
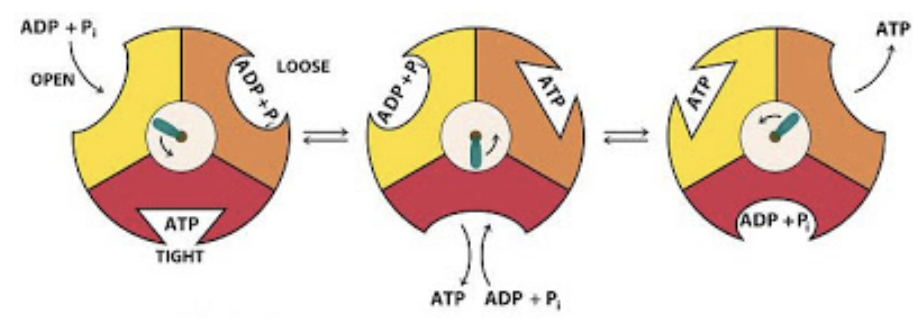
Steps in which ATP synthase converts mechanical energy to chemical energy
(1) Proton flow to the stroma turns the rotor
(2) ATP synthase head has 3 subunits that each consist of an alpha and beta subunit, where ADP + Pi bind to an active site
(3) Rotation of the central stalk changes the conformation of the proteins in the head → phosphorylates ADP to ATP
(4) ATP is released, and the process is repeated
If IMM is impermeable, how are molecules transported in/out of the matrix
pumps and transporters
How does pyruvate/protons enter matrix
Symport
How do phosphate and protons enter matrix
symport
How does ADP enter the matrix and ATP exit
antiport
What drives the import of pyruvate and Pi
electrochemical gradient of H+
What does pump out of ATP and pump in of ADP (ADP-ATP exchange) depend on
voltage gradient across membrane (membrane potential)
The ADP-ATP exhcange moves on negative charge out of the mitochondrion
Can you separate proton generation from ATP production
Yes, with uncoupling agents which are H+ carriers that can insert into the IMM
How does uncoupling occur naturally in some specialized fat cells (brown fat cells)
Most of the energy from oxidation is dissipated as heat rather than converted to ATP
What’s special of IMM of brown fat cells
contain special transport protein that allows protons to move across (important in protecting sensitive areas of newborns from the cold)
What do uncoupling agents do
prevent ATP from being made
what’s 2, 4-dinitrophenol (DNP)
an uncoupling agent that causes the inner membrane to be permeable to protons, allowing H+ flow into mitochondrion without passing thru ATP synthase → ATP is not made
Use of DNP for weight loss
Highly toxic, small amounts were given, effective in melting away pounds (especially loss of fat reserves), patients had elevated temps and sweated a lot during treatment
With each of the 3 respiratory enzyme complexes, where do electrons mainly move
between metal atoms that are tightly bound to the proteins
What carries electrons between different complexes
electron carriers which are molecules that diffuse along the lipid bilayer
Quinones
only electron carriers in ETC that function without being tightly bound to a protein
How does long hydrophobic tail affect ubiquinone
It confines ubiquinone to the membrane
Where is cytochrome c held
Held loosely on the outer face of the inner membrane by ionic interaction
How many amino acids does cytochrome c contain
just over 100
What makes up cytochrome c
a iron atom in the bound heme group can carry a single electron
What does heme group in hemoglobin do
reversibly binds O2 rather than electron
How can you demonstrate proton gradients power ATP production
When bacteriorhodopsin is added to artificial vesicle, protein generates proton gradient in response to light
What happens in artificial vesicles containing both bacteriorhodopsin and ATP synthase
proton gradient drives formation of ATP
What happens if theres either only bacteriorhodopsin or only ATP synthase
No ATP generated
What happens in uncoupling agent is also there with bacteriorhodopsin and ATP synthase
uncoupling agent abolishes gradient and stops ATP synthesis
How does proton go from one side to another
NADH dehydrogenase complex picks up proton from one side of membrane when it accepts eelctron from NADH, and then it releases the proton to the other side of the membrane when it donates the electron to ubiquinone
What is cytochrome oxidase
protein complex that receives electrons from cytochrome c and donates it to O2
What is cytochrome oxidase composed of
Subunits I and II
What is present at active site of cytochrome oxidase where O2 is bound i
Cytochrome oxidase contains complex of a heme iron atom juxtaposed with tightly bound copper atom
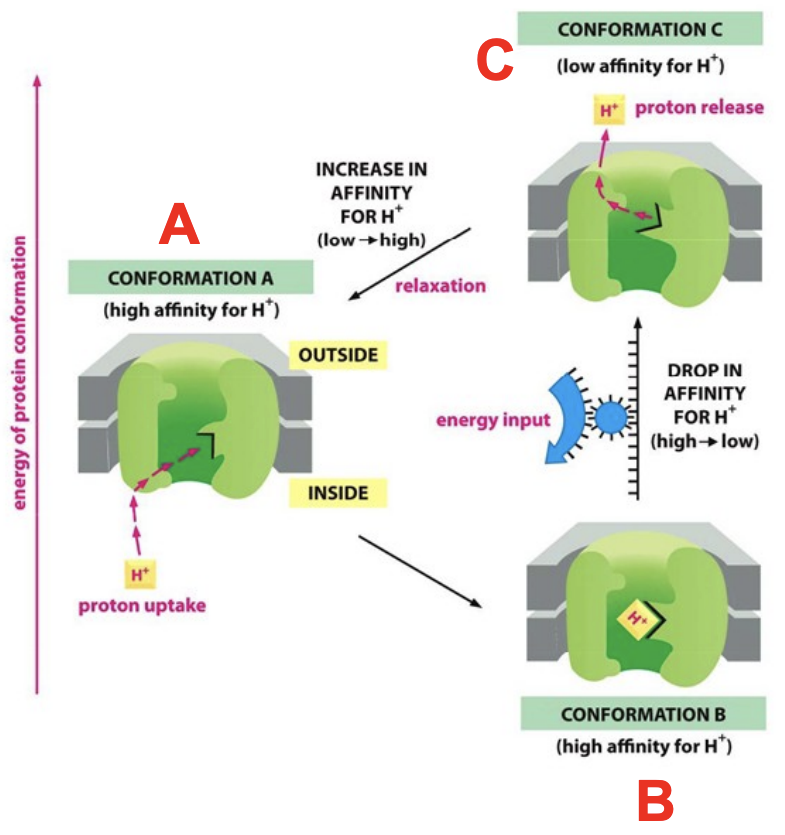
How can conformational change in a protein pump driven by an energetically favorable reaction cause H+ pumping
Proteins, like NADH dehydrogenase or cytochrome oxidase, are driven through a cycle of 3 conformations (A, B, C)
A and B have a high affinity for H+, so it picks H+ up
C has low affinity for H+, so it releases H+
The transition from B to C is energetically unfavorable and needs to be coupled to a favorable reaction (aka electron transport)