Classification of Matter
1/9
Earn XP
Description and Tags
Chem Test 2/20
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
gas
particles are independent, very far apart, with no attractive forces between them. Fill total volume of container. Has most kinetic energy
solid
strong attractive force between particles lock them into a fixed arrangements.

liquid
particles make & break temporary attractions with each other. Particles slip & slide over one another & take shape of container

matter
anything that has mass & takes up space
Pure substance
All particles are the same throughout a sample — composition is fixed or definite. Fixed intrinsic physical properties. CANNOT be separated physically.
elements
PURE SUBSTANCE: cannot be broken down into simpler substances by physical or chemical methods; smallest particle is called a atom.
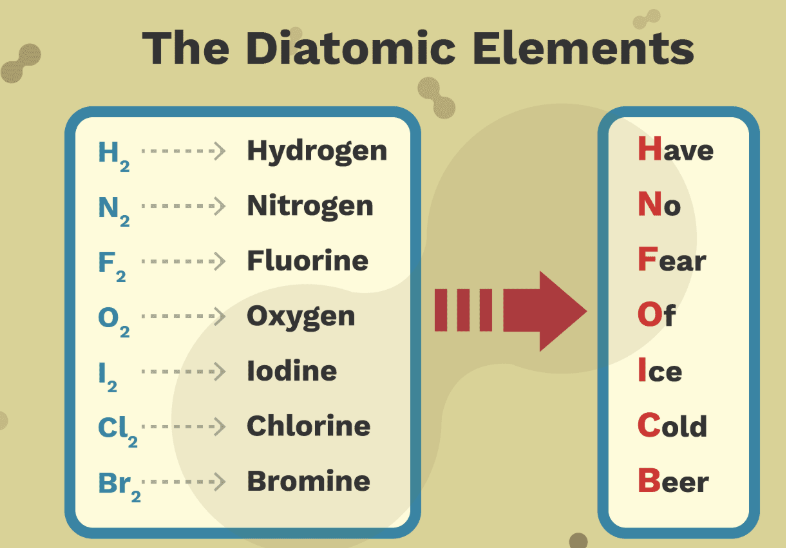
compounds
PURE SUBSTANCE: substances with a definite composition & mass ratio that can be broken down into simpler substances by chemical means

mixture
Composition is variable. Physical properties vary depending on exact composition. CAN BE PHYSICALLY SEPERATED
homogenous MIXTURE
a mixture with one phase throughout the sample, different types of particles are not able to be seen

heterogenous MIXTURE
a mixture with more than one phase throughout the sample, different types of particles are able to be seen
