a2 chemistry- chemical energetics
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
56 Terms
ionic bond strength is determined by
lattice energy
covalent bond strength is determined by
bond energy
lattice formation enthalpy
is always exothermic
"energy released when 1 mole of an ionic solid is formed from its gaseous cation and anion under standard condition"
lattice dissociation enthalpy
endothermic.
"energy absorbed when 1 mole of an ionic solid is converted into gaseous cation and anion under standard condition"
lattice energy depends on
ionic charge and ionic radius- charge density
lattice energy is directly proportional to
charge of ions
lattice energy is inversely proportional to
ionic radius
when is lattice energy highly exothermic
if the charge of the ion is higher, size of the ion is smaller, charge density increases, electrostatic attraction (b/w cation and anion) will become stronger so more energy is released
which one has higher value of lattic energy
anion same--->explanation on basis of cation
charge same--->ionic radius
priority given to the
charge of the ion
born haber cycle is an application of
hess's law
born haber cycle is only applicable for
pure ionic crystals (only exhibit ionic bonding, consists of only 2 elements)
impure ionic crystals
consists of more than three elements
consists of both ionic and covalent bonding
standard enthalpy change of formation
enthalpy change that occurs when 1 mole of a compound is formed from its constituent elements in their standard states under standard condition- can be exo/endo
standard enthalpy change of atomisation (for metals)
1. enthalpy change occurs when 1 mole of gaseous atom is formed from its constituent elements in their standard states under standard condition
2. is always endothermic
energy values need to be
multiplied with number of moles
standard enthalpy change of atomisation (for non-metals)
1. decomposition
2. is always endothermic
3. if it's not given, then half the bond energy value
4. do not multiply the enthalpy change of vap/sub with no. of moles
ionisation energy of metals
1. don't use ionisation energy of non metals
2. amount of energy required to remove one mole of an electron from one mole of neutral gaseous atom to give a monopositive ion
3. is always endothermic
4. charge of the ion decide the ionisation energy, not the moles of electron
electron affinity of non-metals
1. only in non metals
2. energy released when one mole of an electron is added to one mole of neutral gaseous atom to give an anion with a charge of 1-
3. it is a measure of the force of attraction between incoming electron and nucleus
4. stronger the force of attraction, more energy released and vice versa
5.exo/endo
why the first electron affinity is exo while the second and third are endo?
both anion and electron are negatively charged, experience repulsion, to overcome this, absorption of energy takes place.
electron affinity depends on
1. atomic size
2. shielding effect
3. nuclear charge
electron affinity (down the group)
atomic size increases, number of shells increase and so does the shielding effect, nucleus to electron attraction becomes weak so electron affinity becomes less exo
elctron affinity (across the period)
nuclear charge increases, nucleus to electron attraction increases, more energy is released so electron affinity becomes more exo
dissolving ionic solids in water
1. standard enthalpy change of solution
2. lattice dissociation enthalpy
3. hydration enthalpy
standard enthalpy change of solution
enthalpy change occus when 1 mole of compound is dissolved in large amount of water to give an infinite dilute solution under standard condition
can be exo/endo
exo if soluble in water
endo if partially soluble/insoluble
lattice dissociation enthalpy-2
amount of energy required to break 1 mole of ionic solid into gaseous cation and anion
is always endo
hydration enthalpy
enthalpy change occurs when 1 mole of gaseous cation and anion are dissolved in large amount of water to give an infinite dilute solution
is always exo bec electrostatic attraction is formed b/w ion and water molecule
hydration enthalpy can be highly exo or less exo
stronger the electrostatic attraction, more energy is released.
hydration enthalpy--->highly exo
hydration enthalpy depends on
charge of cation
size of cation
charge density
hydration enthalpy (overview with example)
magnesium ion has high magnitude of charge, smaller ionic radii, high charge density. strong electrostatic attraction exists b/w ion and water so more energy is released. if more energy is released, then hydration enthalpy is highly exo
dissolving ionic solids equation
sol=-le+hyd
lattice enthalpy equation
h1+le-hf=0
solubility can be explained on the basis of
lattice energy and hydration enthalpy
both lattice energy and hydration enthalpy depend on
charge density
also, both are exo
bigger ionic radius of cO3(2-) and so(2-) ions
electrostatic attraction b/w ions is not effective, hence lattice energy will not play a key role
le<hyd
more exo, sol is exo, compound is exo
le>hyd
more endo, sol is endo, compound is insoluble or partially soluble
solubility of group ii sulfates/carbonates
down the group, ionic radius increases so charge density decreases, both hydration and lattice enthalpy decrease, hydration enthalpy decreases more significantly than lattice energy, sol is endothermic, solubility decreases
mgso4
soluble
baso4
insoluble
solubility of group ii hydroxides
due to smaller ionic radii of oh- ion, electrostatic attraction b/w ions exist, so lattice energy will play a key role
solubility of group ii hydroxides (trend)
down the group ionic radius increases, charge density decreases, both hyd and le decreases, lattice energy decreases more significantly than hydration, sol is more exo, solubility increases down the group
thermal stability of group ii carbonates and nitrates
MCO3----->MO+CO2 (heat)
2M(NO3)2---->2MO+4NO2+O2 (heat)
experiment for thermal stability
place small amount of hydrated Mg(No3)2.6H2O in a boiling tube, heat the boiling tube gently.
Mg(No3)2.6H20---->Mg(No3)2+6h2o
water vapours are condensed onto the mouth of boiling tube
heat the boiling tube strongly
2Mg(No3)2---->2MgO+4No2+O2
NO2
dark brown gas
O2
colorless gas which relights a glowing splint
NO2 (structure)
double bond, dative bond
free radical
thermal stability depends on
charge density and polarisation
polarisation
distortion of electrons of anion
when does decomposition takes place at low temp
higher the charge density, higher the polarisation, easier the decomposition
thermal stability trend down group ii
down the group, ionic radius increases
charge density and polarisatio decrease
cation cannot distort the electrons of CO32- easily
for thermal decomposition, more energy is needed
decomposition occurs at a high temp
thermal stability increases down the group
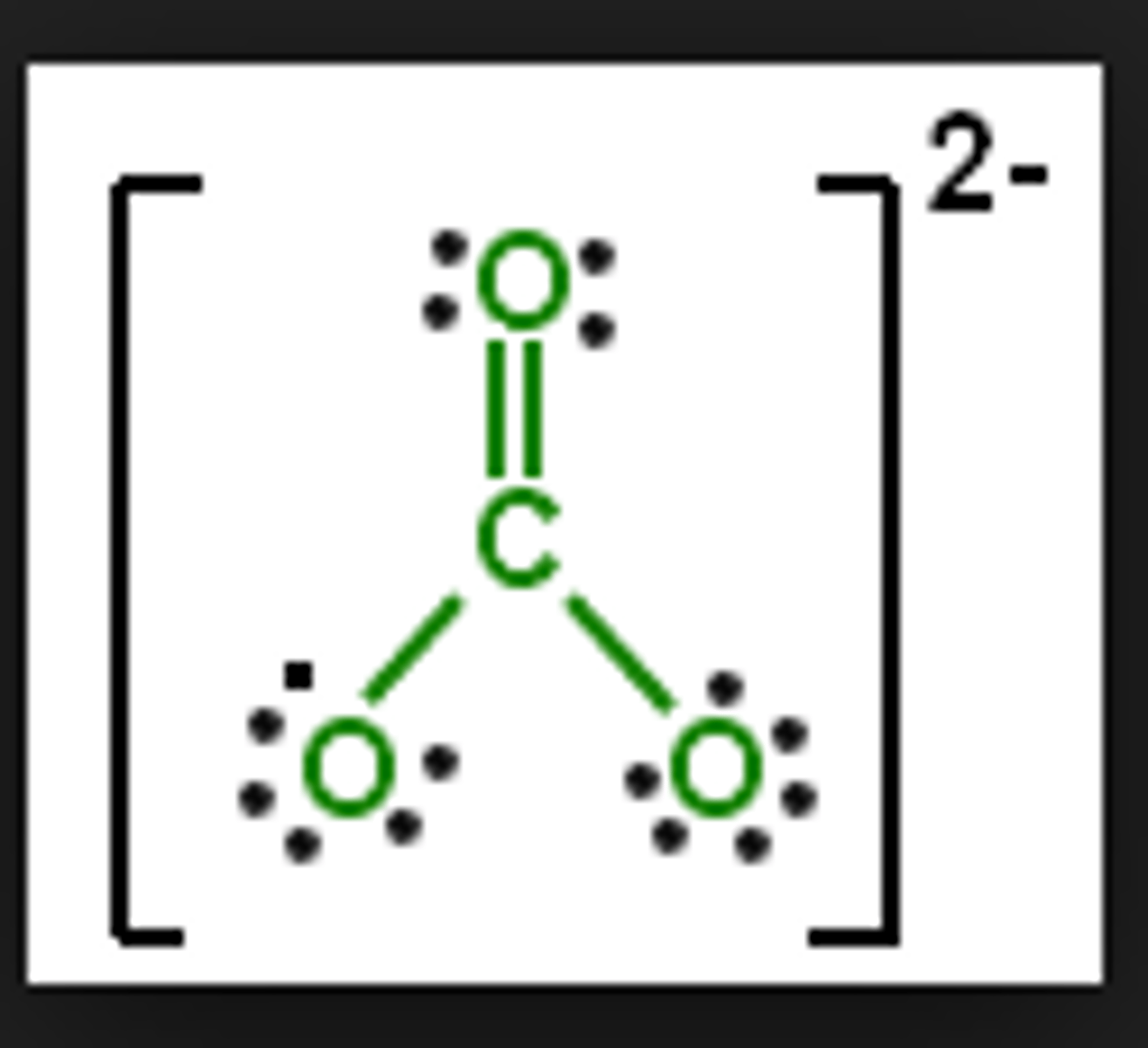
carbonate ion
negative charge on the side oxygens
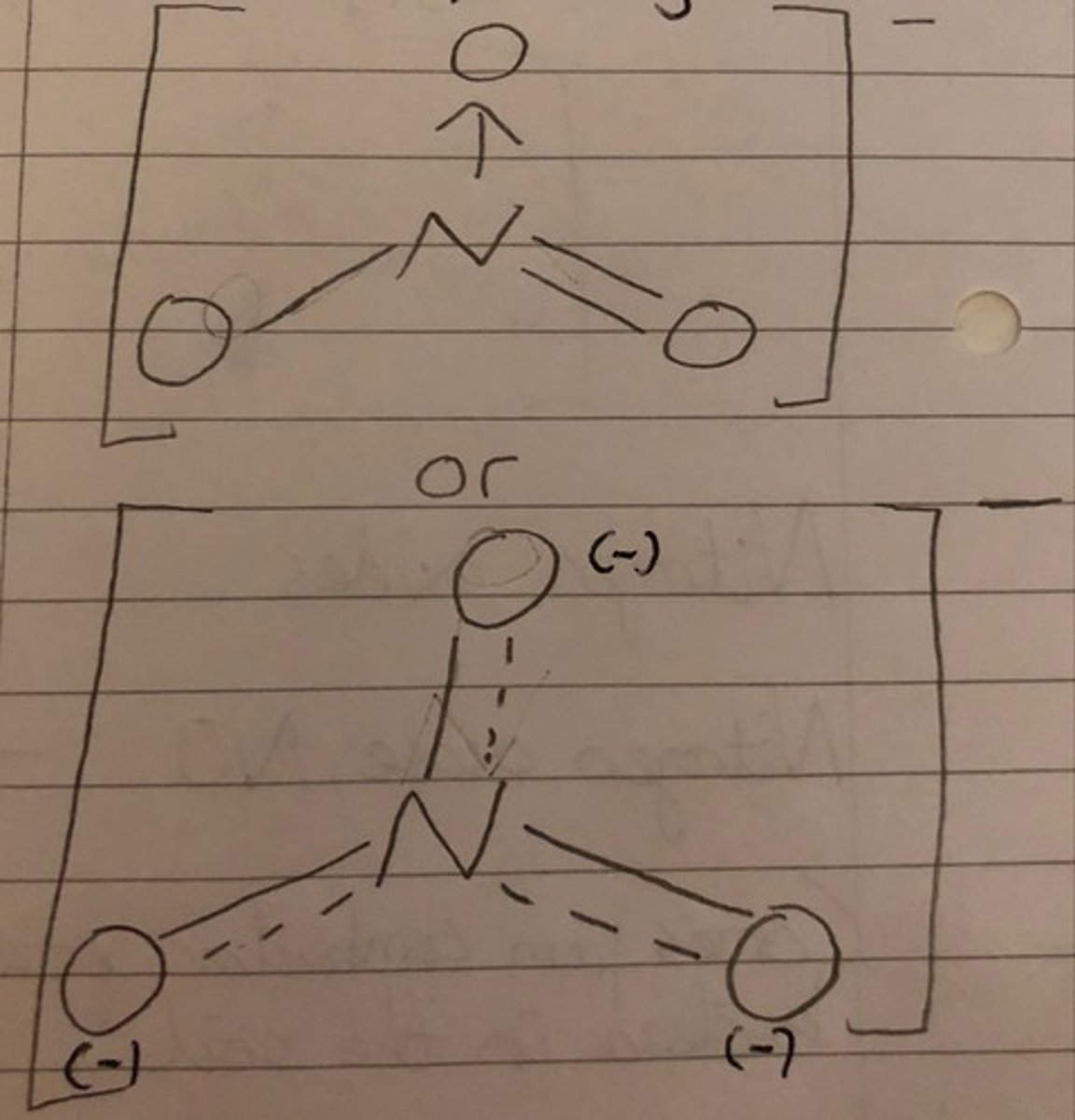
nitrate ion structure
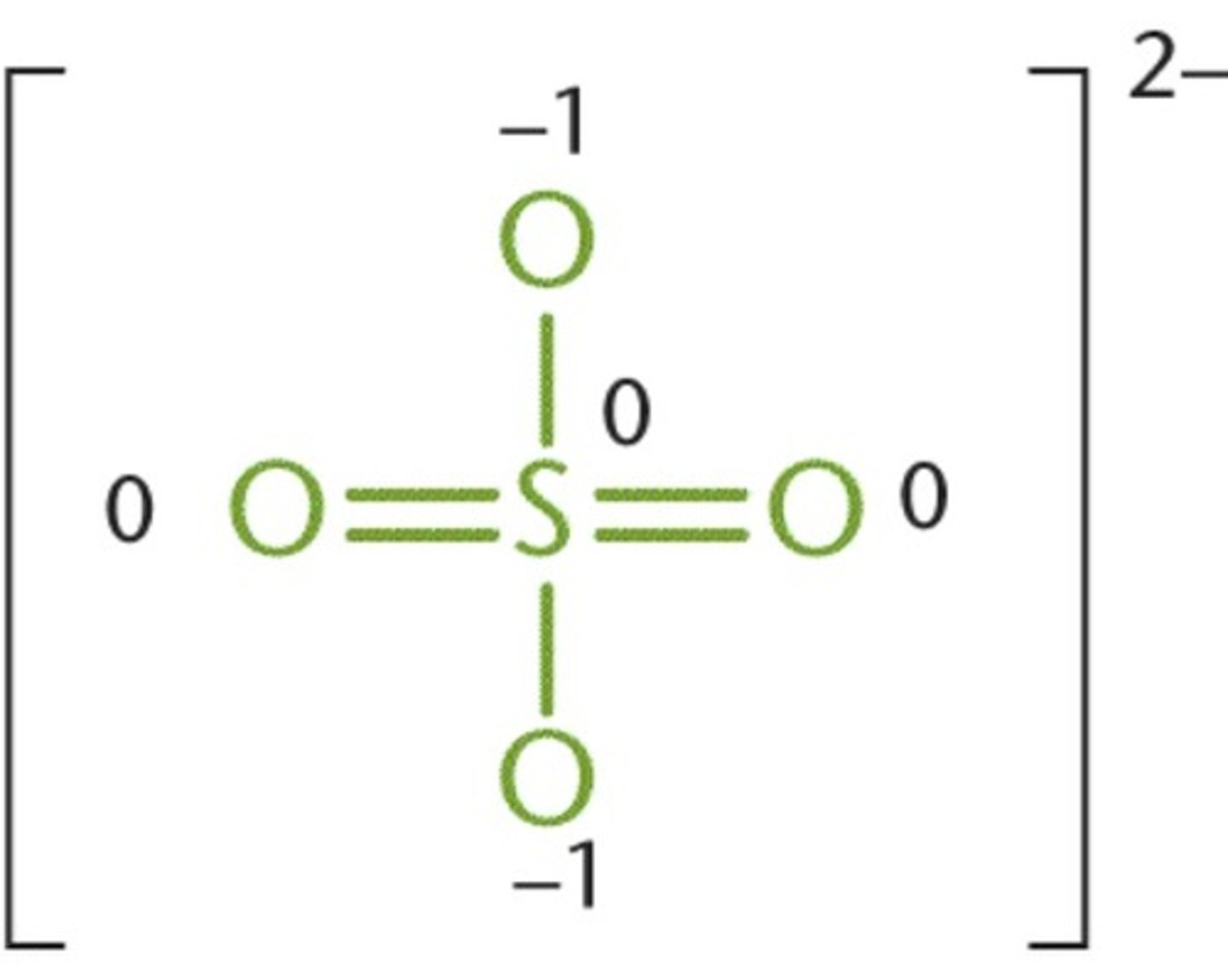
sulfate ion structure
azide ion structure
dative bond and - sign on side N
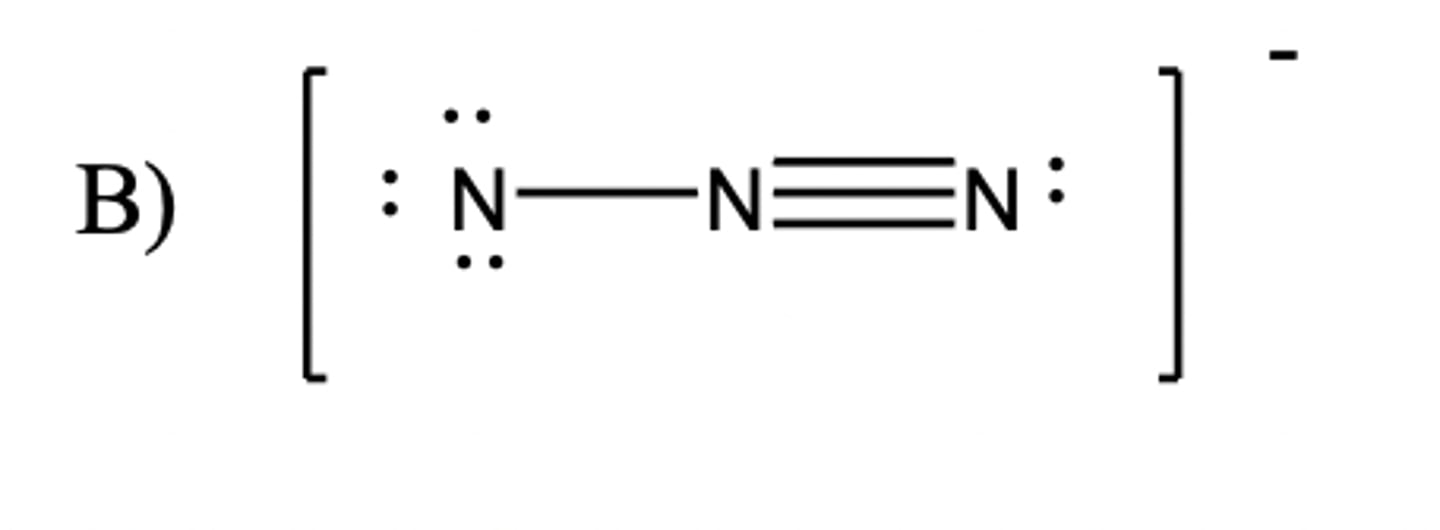
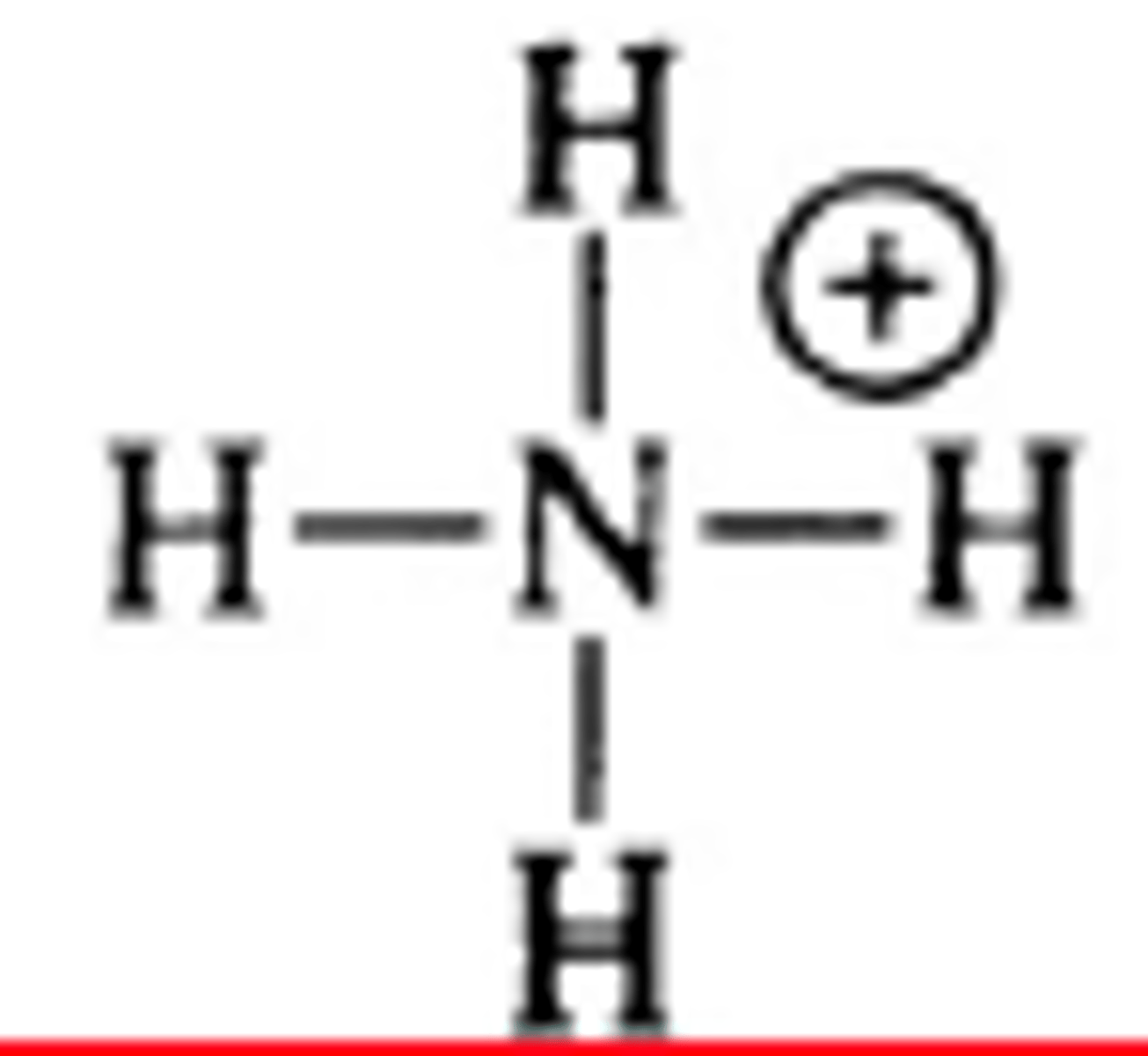
ammonium ion structure