Topic 2: Atomic Structures
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
45 Terms
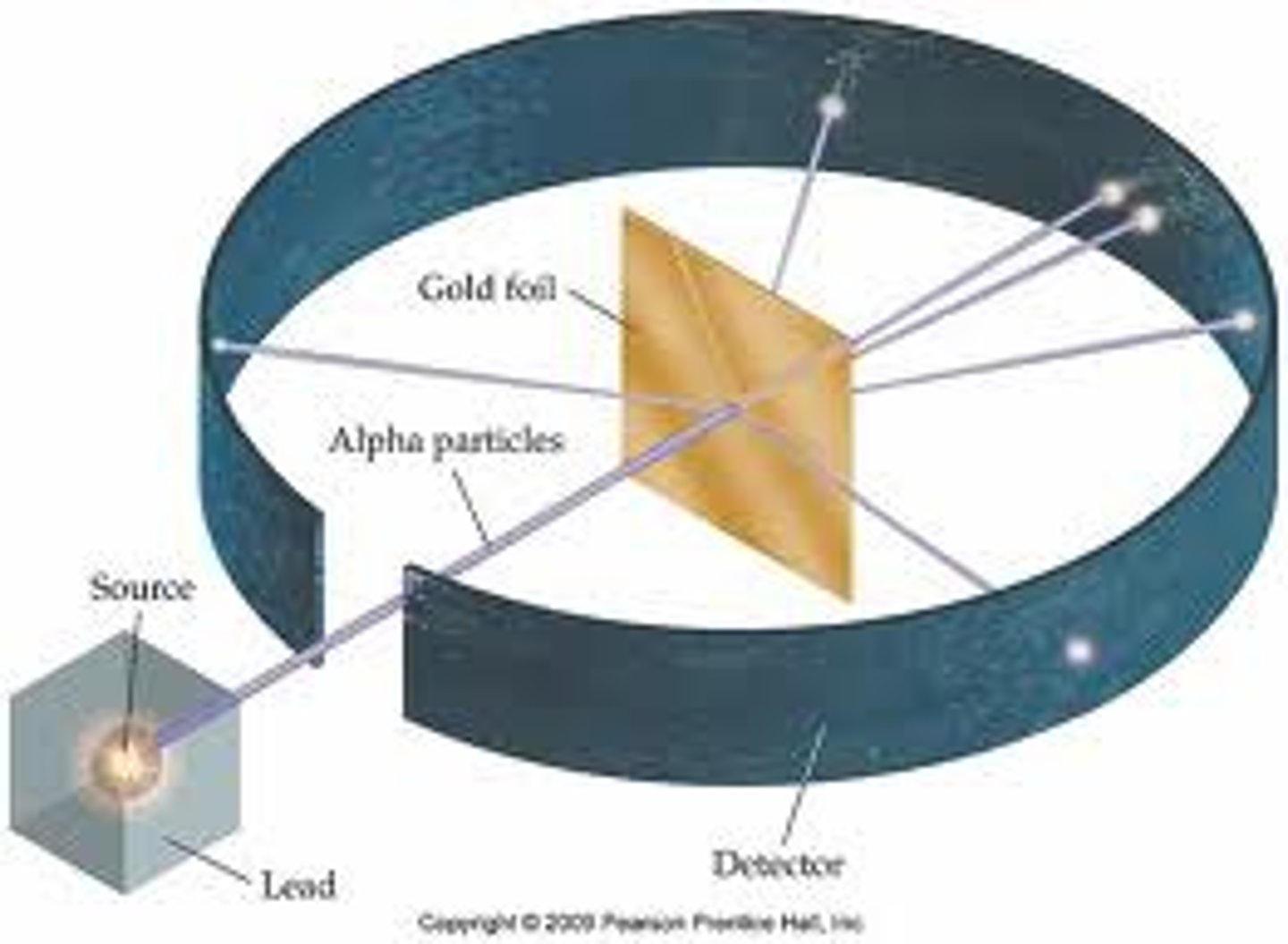
Rutherford's experiment
- Revealed atoms have a nucleus
- He shot alpha particles towards a sheet of gold foil
- Around the foil was a fluorescent screen: flashed when hit with alpha particles
- Predicted: alpha particles go straight through the foil and not get deflected
- However, a small percentage of particles were deflected through angles larger than 90 degrees
- Some particles were scattered back
- Showed:
- atoms are mostly empty space
- atoms had a nucleus
Definitions: Mass Number (A)
Sum of the number of protons and neutrons in the nucleus
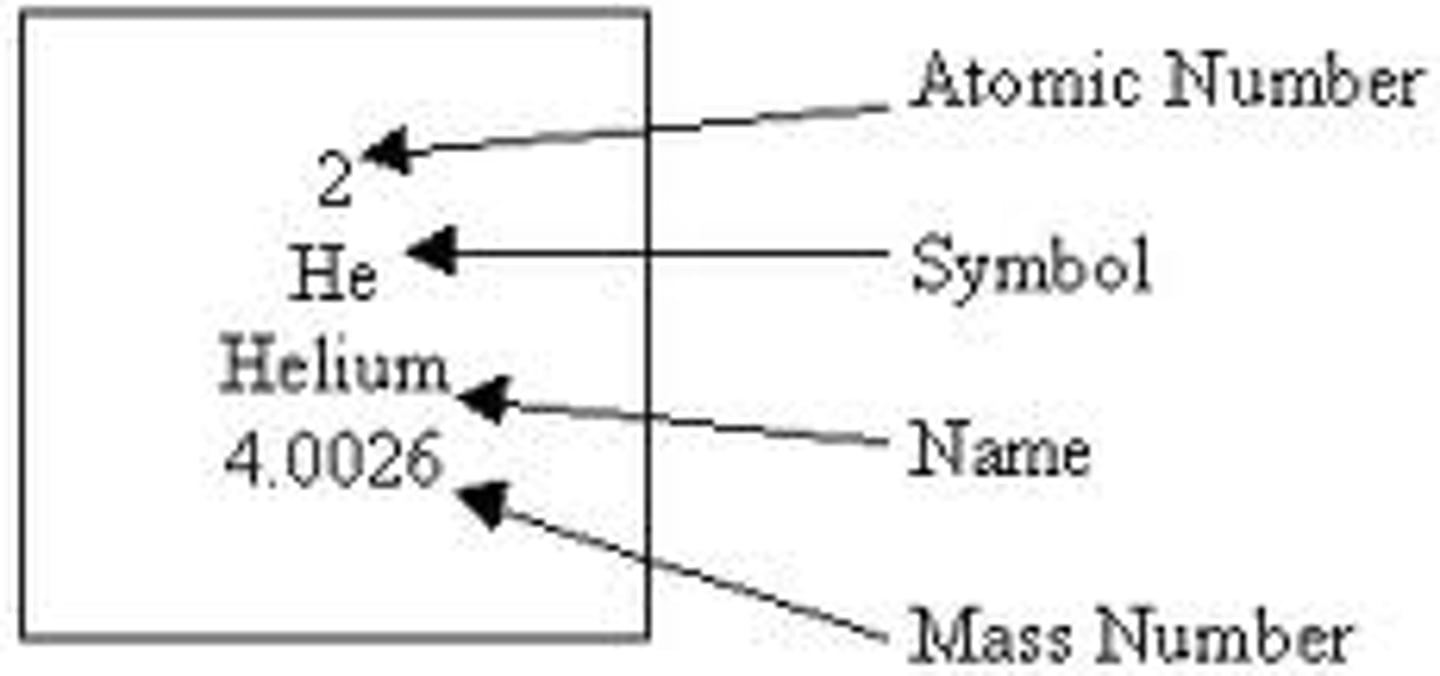
Definitions: Atomic Number (Z)
The number of protons in the nucleus
- since atoms are electrically neutral, the number of protons is equal to the number of electrons
Definitions: Isotope
Atoms of the same element with the same number of protons, but with a different number of neutrons
- Some isotopes may be heavier than other elements despite having a smaller proton count because the element may have a greater proportion of heavier isotopes
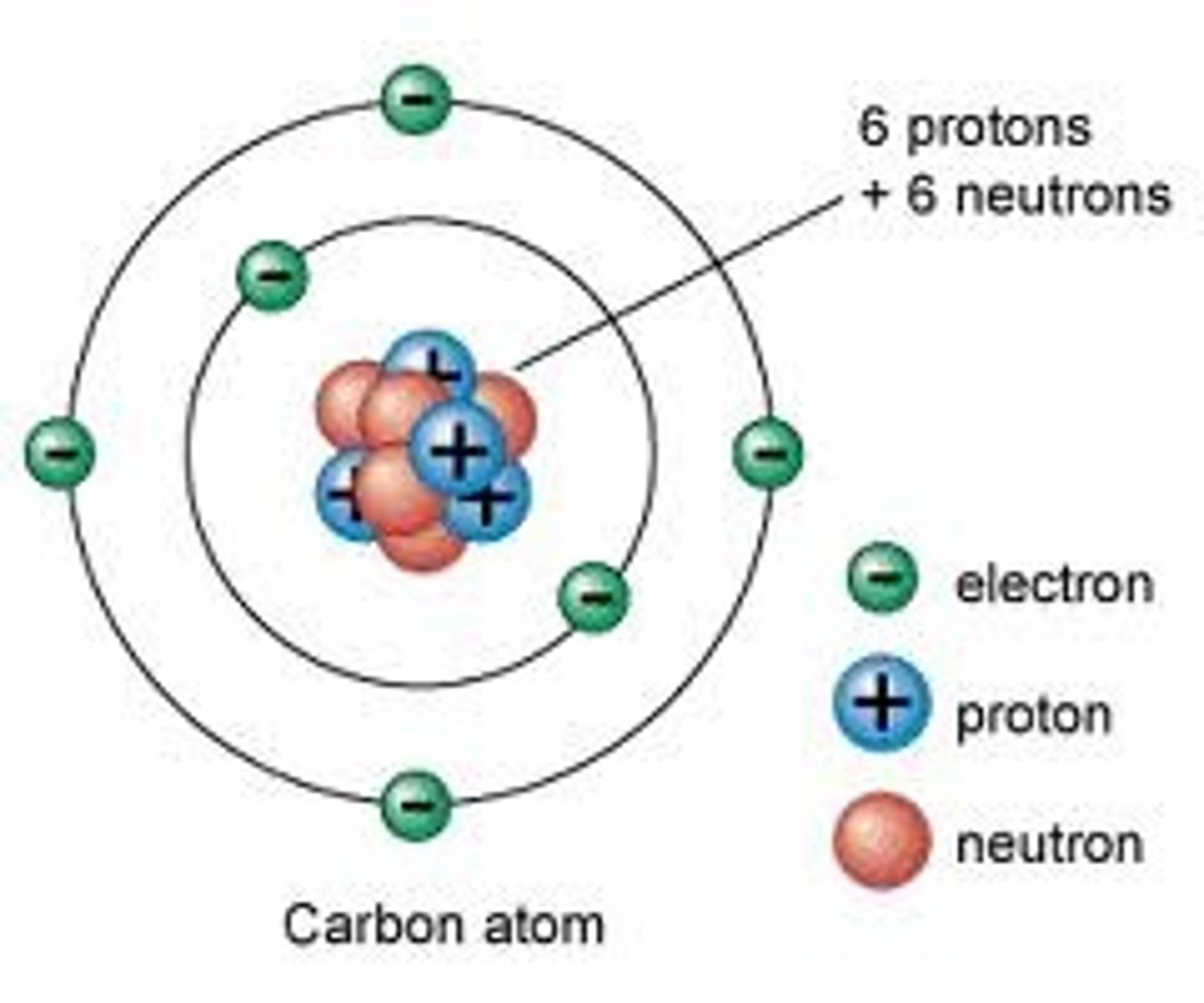
Sub-Atomic Structures
- Atoms are made up of a nucleus containing positively charged protons and neutral neutrons, and negatively charged electrons moving around the nucleus in the valence shells
- Electrons = massless
Sub-Atomic Structures
Sub-Atomic Particle Relative mass Relative charge
- Proton 1 +1
- Neutron 1 0
- Electron 1/1836 -1
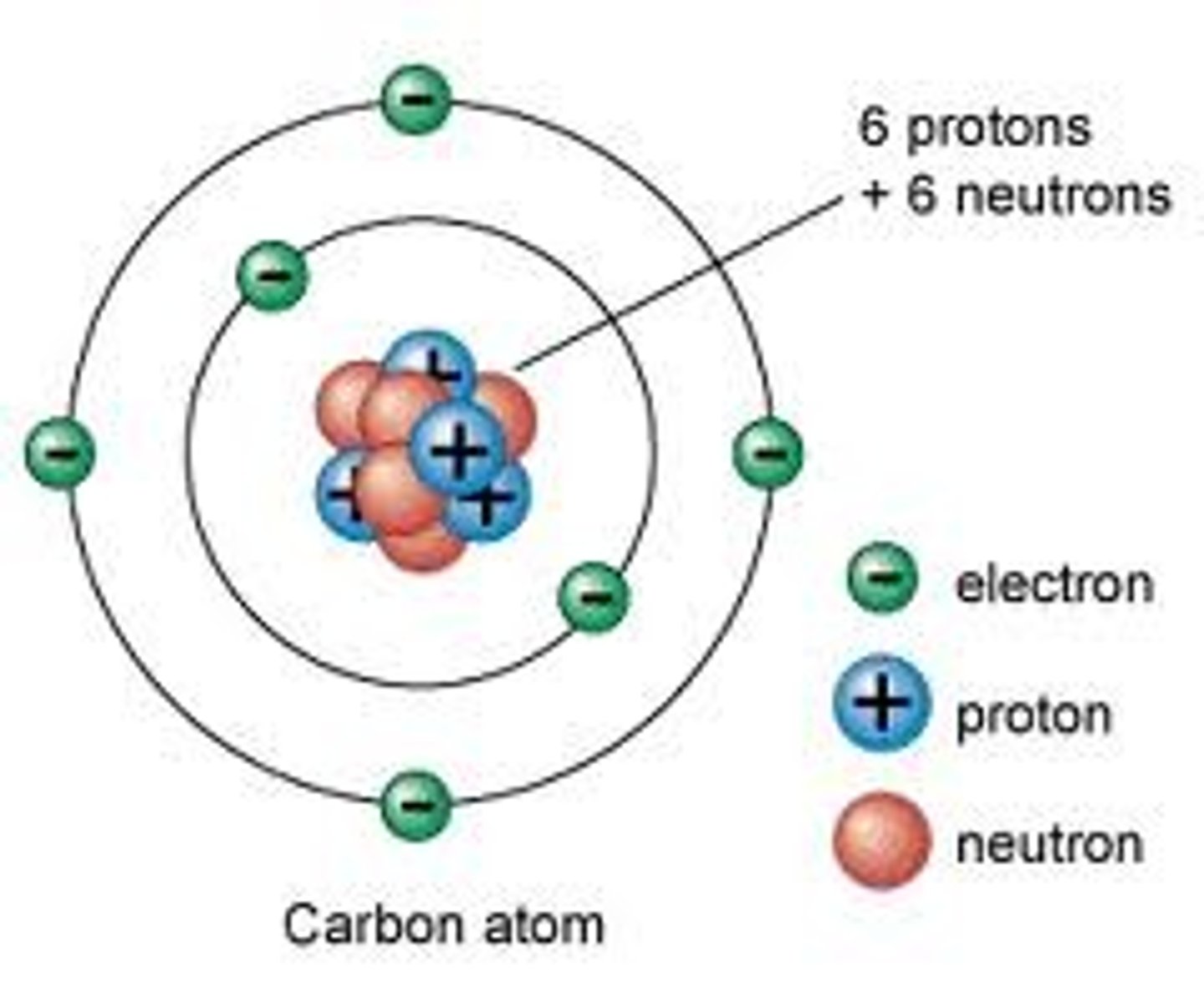
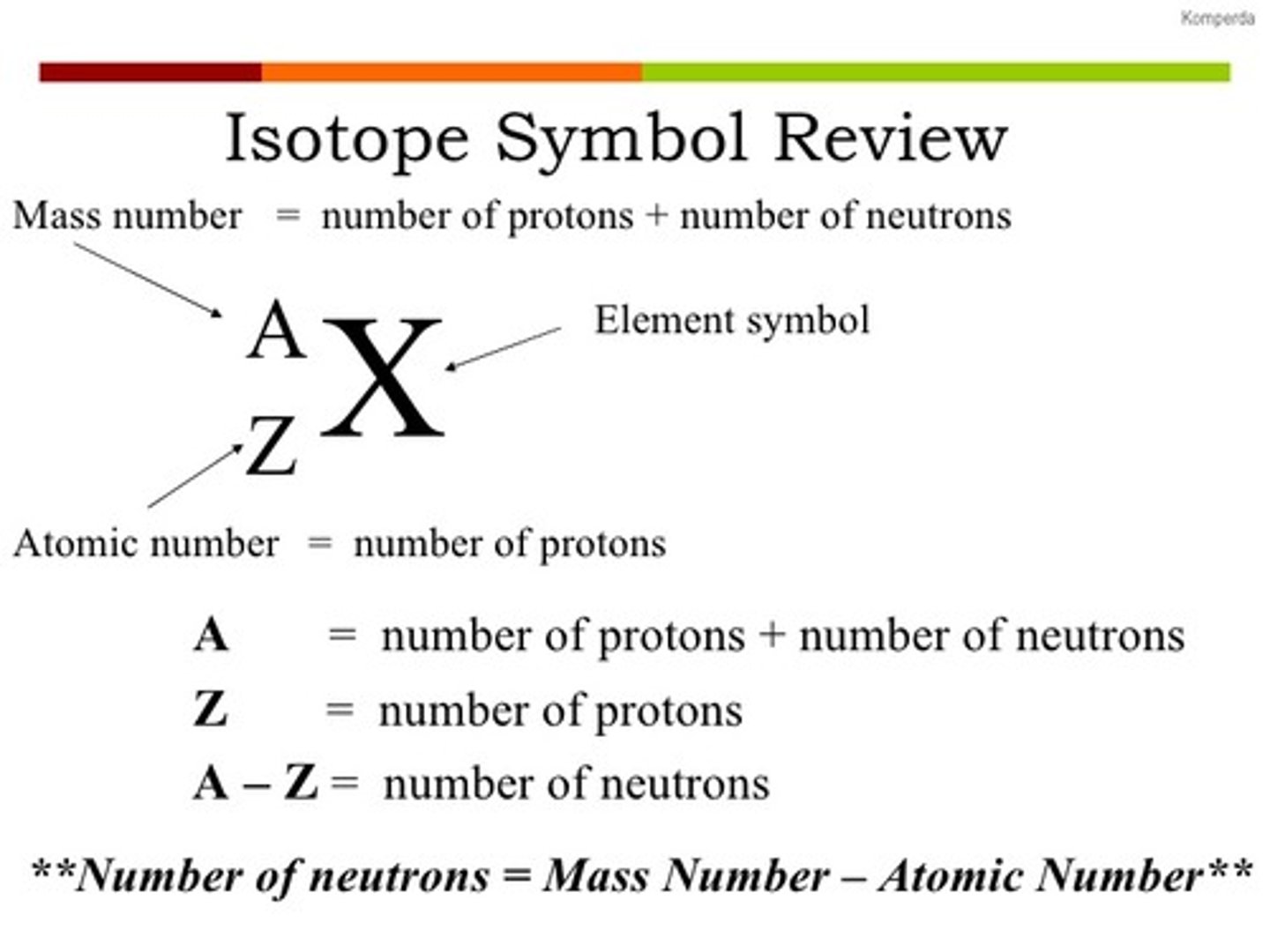
Nuclear Notation
- shows the mass number, atomic number, and element symbol to represent a particular isotope
- Atomic Number: number of protons
- Mass Number: number of protons and neutrons (n = mass number - atomic number)
- Number of Electrons: atomic number

Properties of Isotopes
- Chemical properties depend on the number of outer shell electrons
- Isotopes have the same number of electrons and protons
- Different number of neutrons = different mass number
- Same number of protons
- Physical properties depend on the isotopes' nuclei
- Changes:
- number of neutrons
- density
- rate of diffusion
- melting/boiling point
- mass
Radio Active Isotopes
- The nuclei are more prone to breaking down spontaneously
- Radiation is emitted
- Can occur naturally or man-made
Uses of Radioisotopes: Carbon-14
- Estimates the age of organisms
- Process= Radiocarbon dating
- Penetrating
- Used to treat cancerous cells
Uses of Radioisotopes: Cobolt-60
- Powerful gamma emitter
- Useful for cancer treatments
- Used to stop immune response to transported organs
- Used for leveling devised to sterilize food and spices
Uses of Radioisotopes: Iodine-131/Iodine-125
- Iodine-131
- Releases both gamma and beta radiation
- Used for treating thyroid cancer and functionality
- Iodine-125
- Gamma emitters
- Used to treat prostate cancer and brain tumors
Mass Spectrometry
- Measure the individual masses of atoms
- Separates individual isotopes from a sample of atoms and determines the mass of each isotope
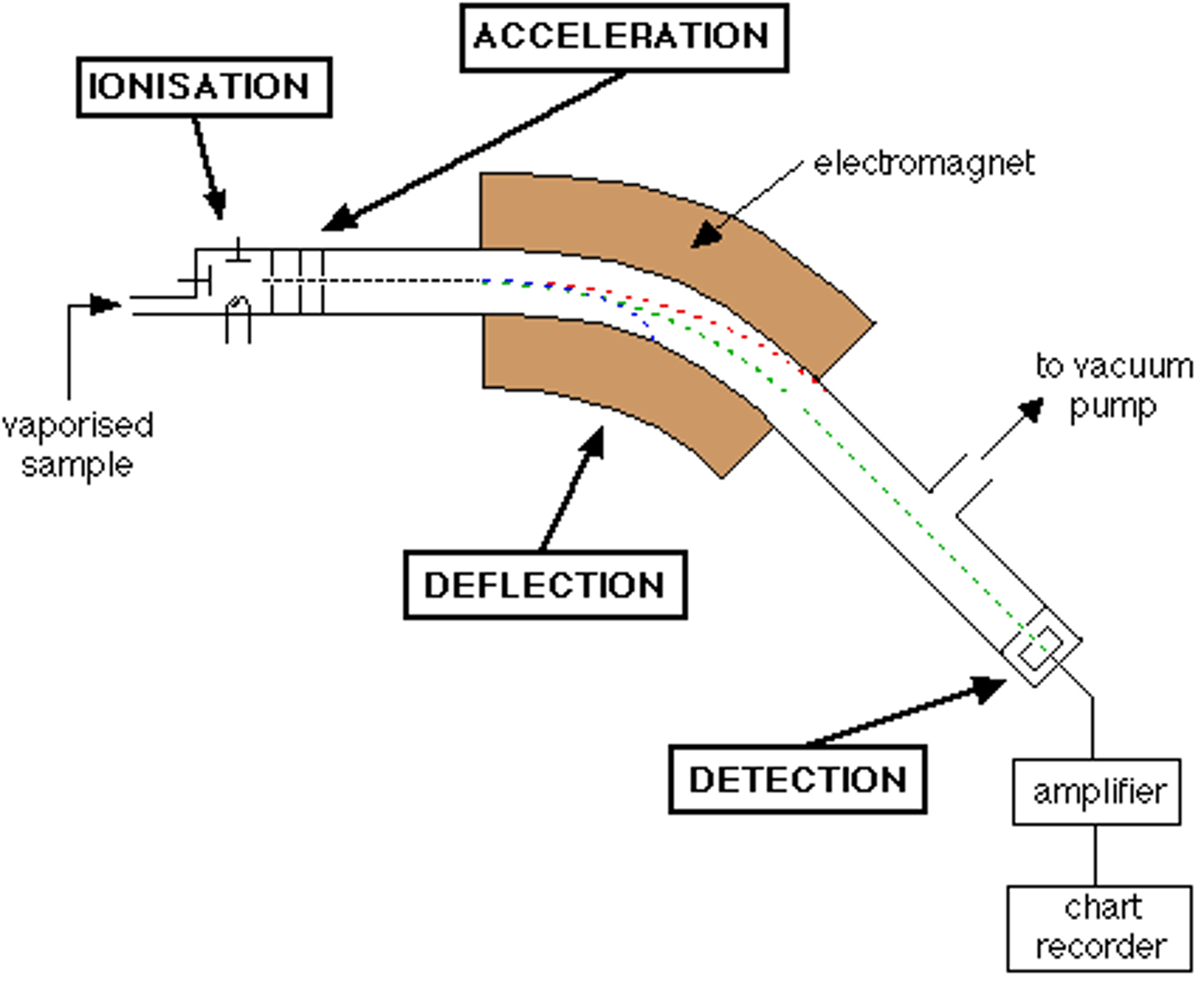
Stages 1 of Mass Spectrometry: Vaporization
- Heated
- Vaporized
- Passed through into an evacuated tube
- This process separates the particles
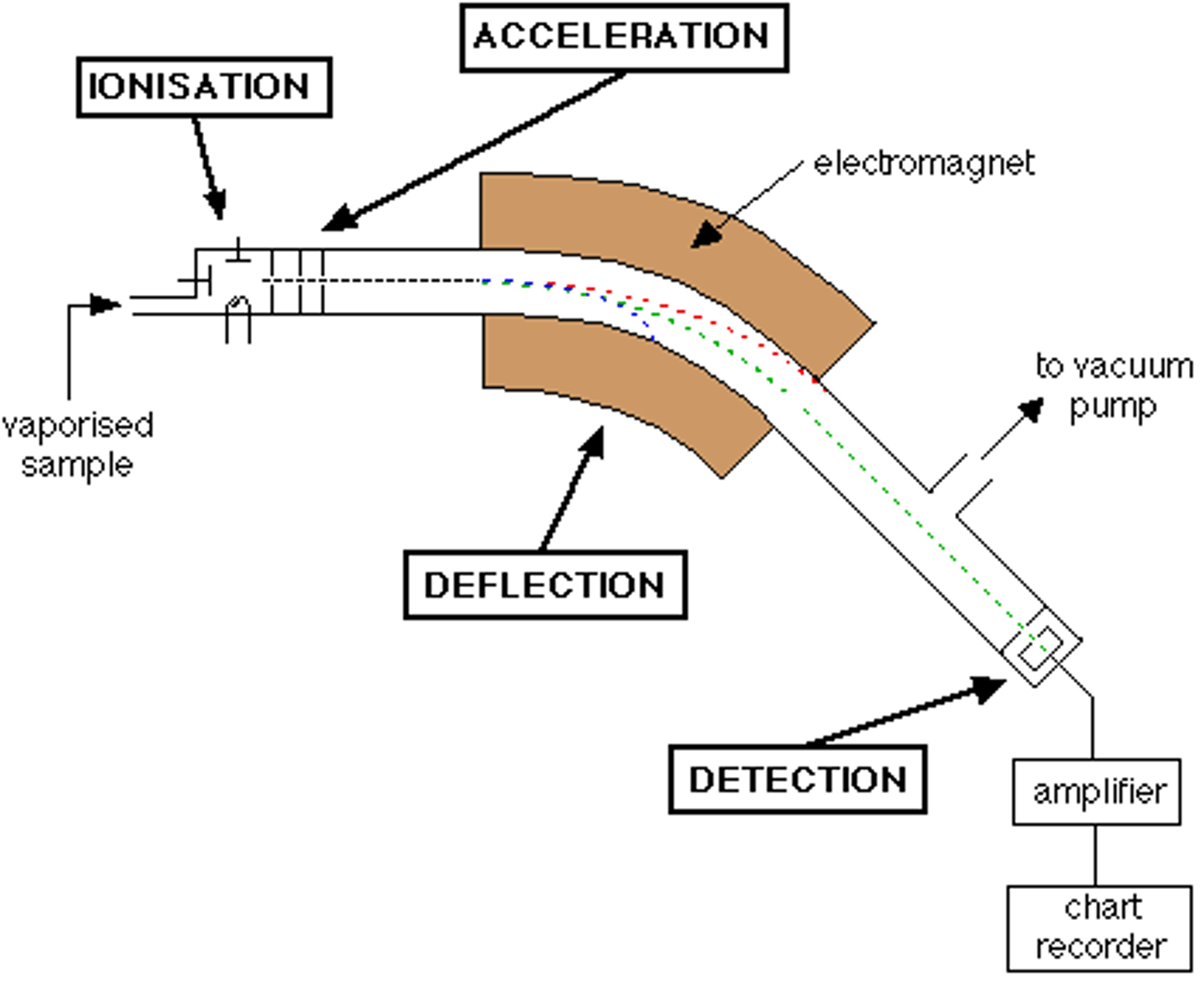
Stages 2 of Mass Spectrometry: Ionization
- Atoms/molecules are bombarded by a stream of high energy electrons
- Knocking electrons off the particles
- Results in ions with 1+ charge
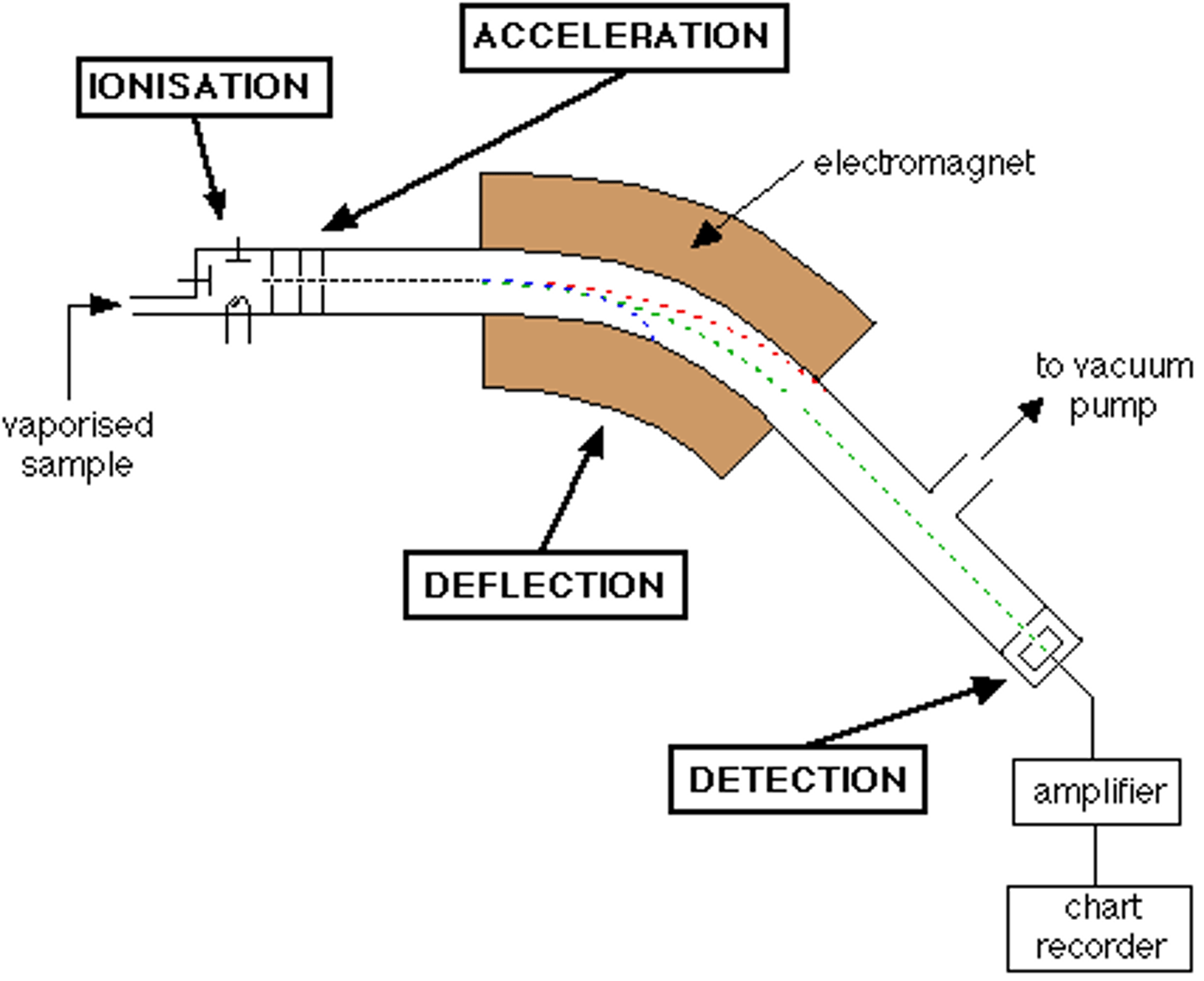
Stages 3 of Mass Spectrometry: Acceleration
- Positively charged ions are accelerated along the evacuated tube by the means of attraction towards negatively charged plates
- Ions pass through slits
- Controls the direction and velocity of their motion
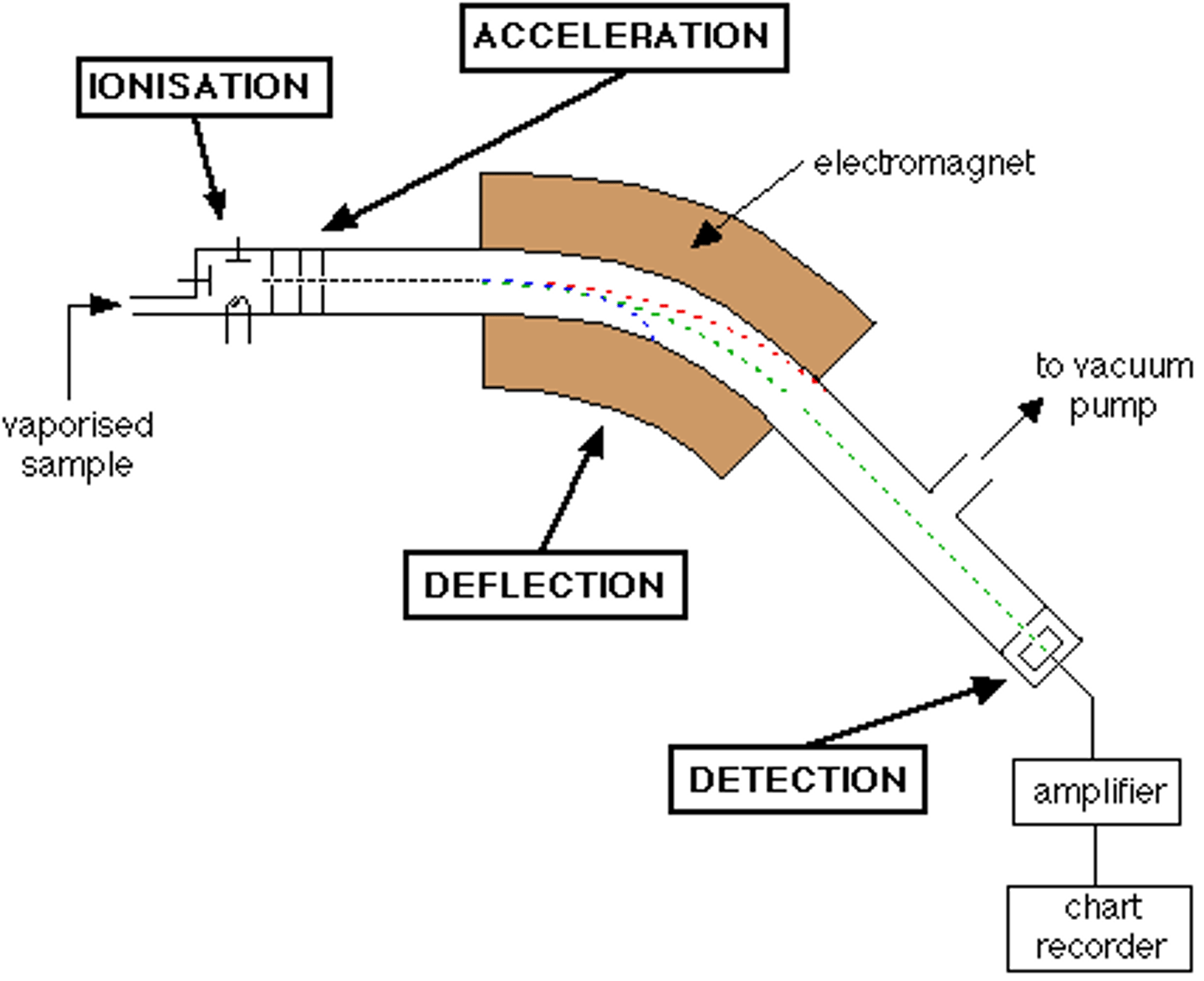
Stages 4 of Mass Spectrometry: Deflection
- Ions are passed into a very strong magnetic field
- Ions are deflected in a curved path
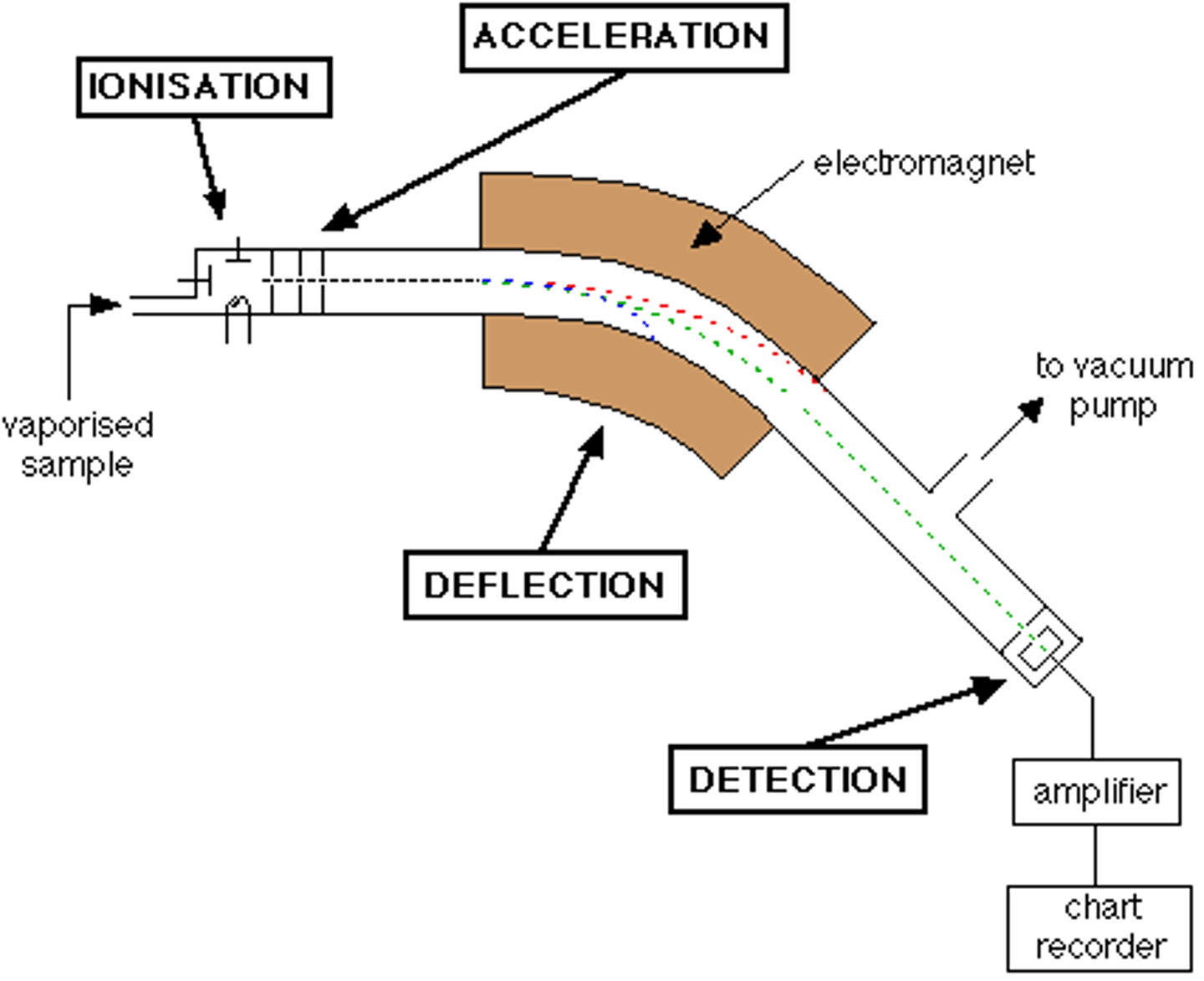
Stages 5 of Mass Spectrometry: Detection
- Ions are electronically detected
- the device used measures the location and the number of particles of an ion
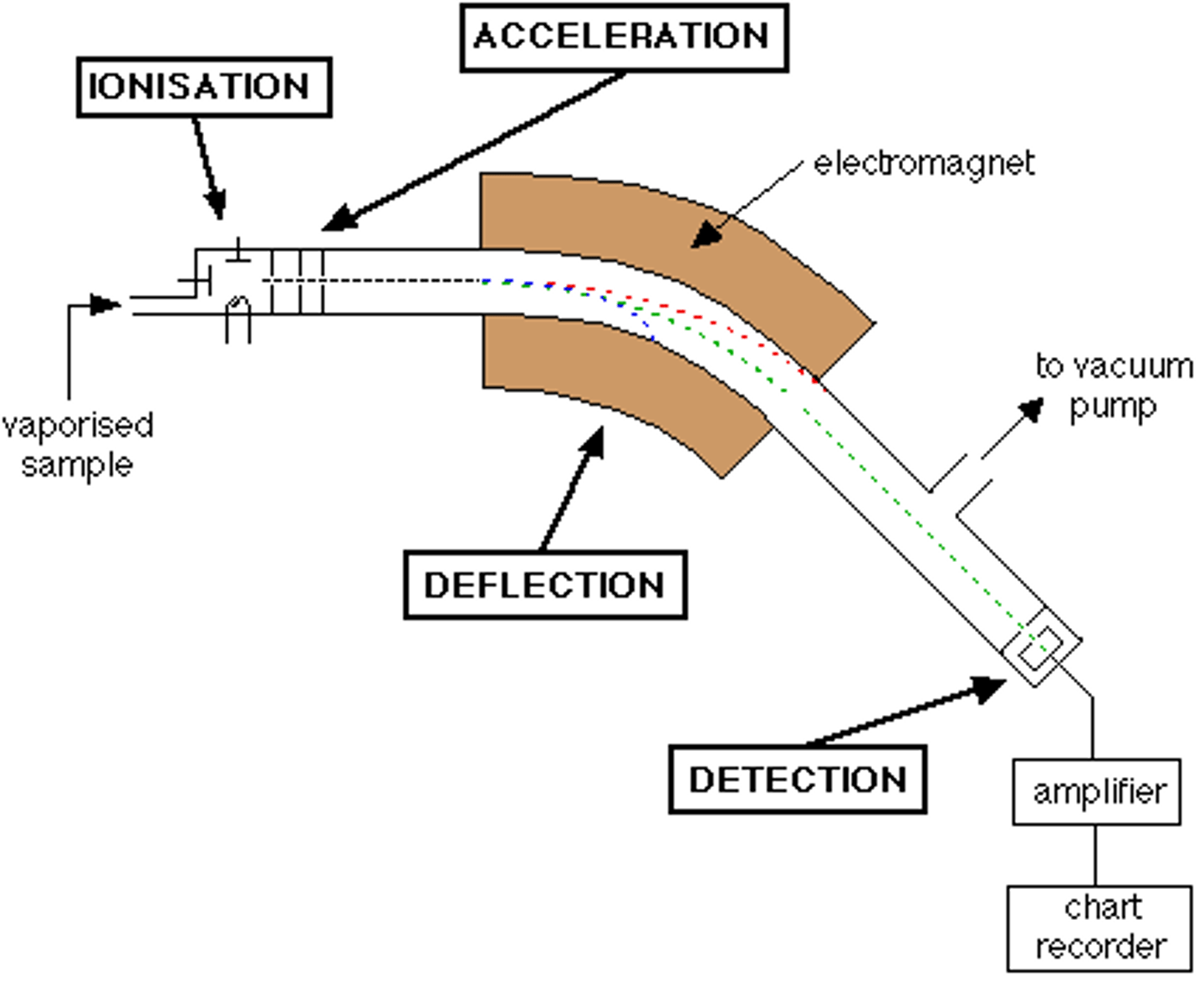
Stages 6 of Mass Spectrometry: The Deflection or path of an ion in a mass spectrometer
- Absolute mass of an ion
- Charge of an ion
- Strength of the magnetic field
- Velocity (speed) of an ion
Mass Spectrum
- Shows the number of isotopes of an element
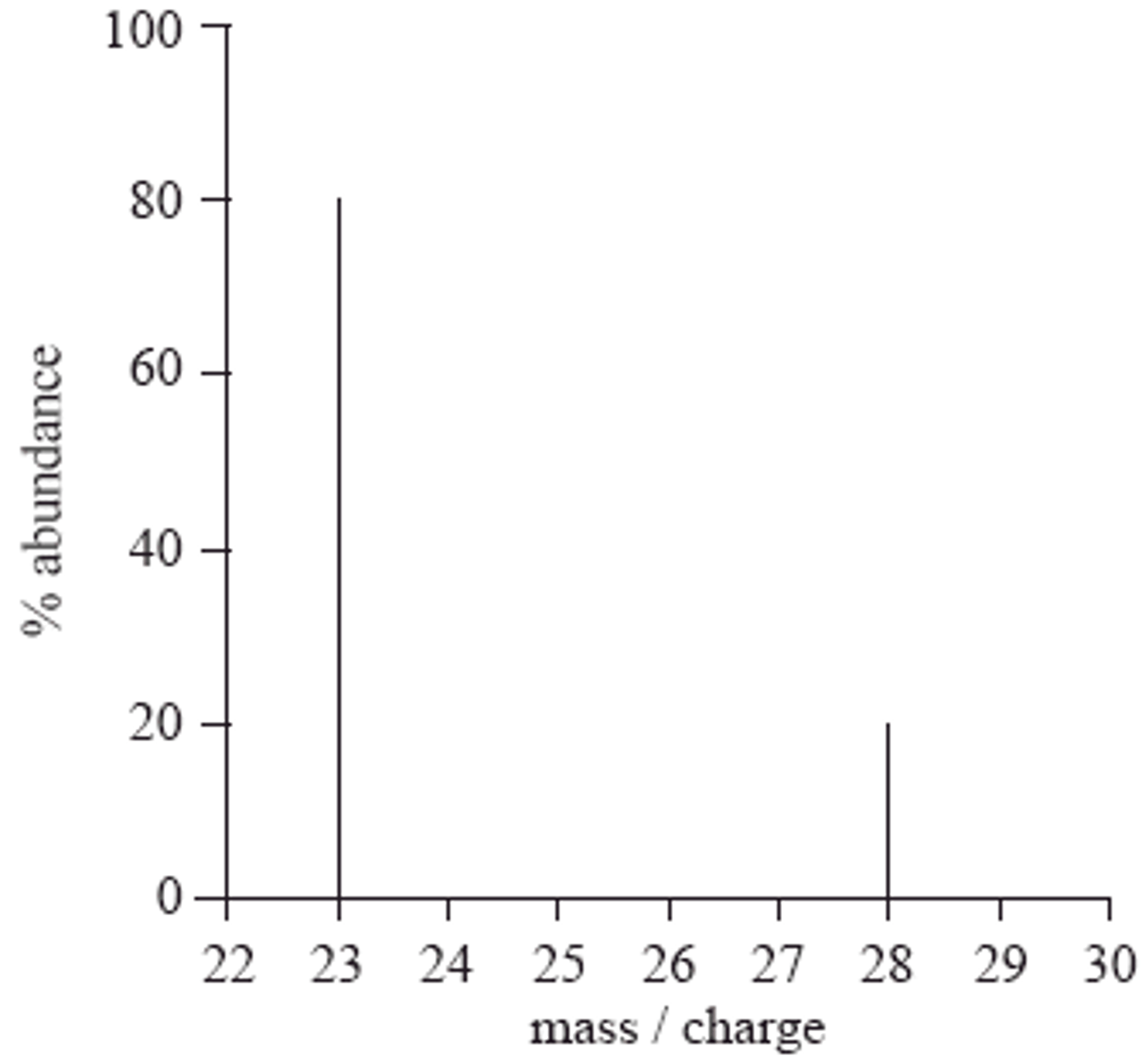
How to read the Mass Spectrum
- The number of peaks indicates the number of isotopes
- The position of each peak in the horizontal axis indicates the relative isotopic mass
- The relative heights of the peaks correspond to the relative abundance of the isotopes
Calculations: Atomic Mass
Ar = (relative atomic mass 1 x % abundance 1) + (relative atomic mass 2 x % abundance 2)
Ar= The answer to above / 100
Calculation Example: Atomic Mass
Chlorine has two isotopes. 35Cl and 37Cl. Cl has a relative atomic mass of 35.5. What are the abundances?
- x = abundance of 35Cl
- Ar = (relative atomic mass 1 x % abundance 2) + (relative atomic mass 2 x % abundance 2)
- Ar= The answer to above / 100
- 35.5 = x x 35 + (100 - x) x 37
- 35.3 x 100 = 35x + 3700 - 37x
- 2x = 150
- both sides divided by 2
- x = 75%
- the other abundance is 25%
Electron Configuration: Bohr's Model
The term energy levels to describe orbitals of different energy
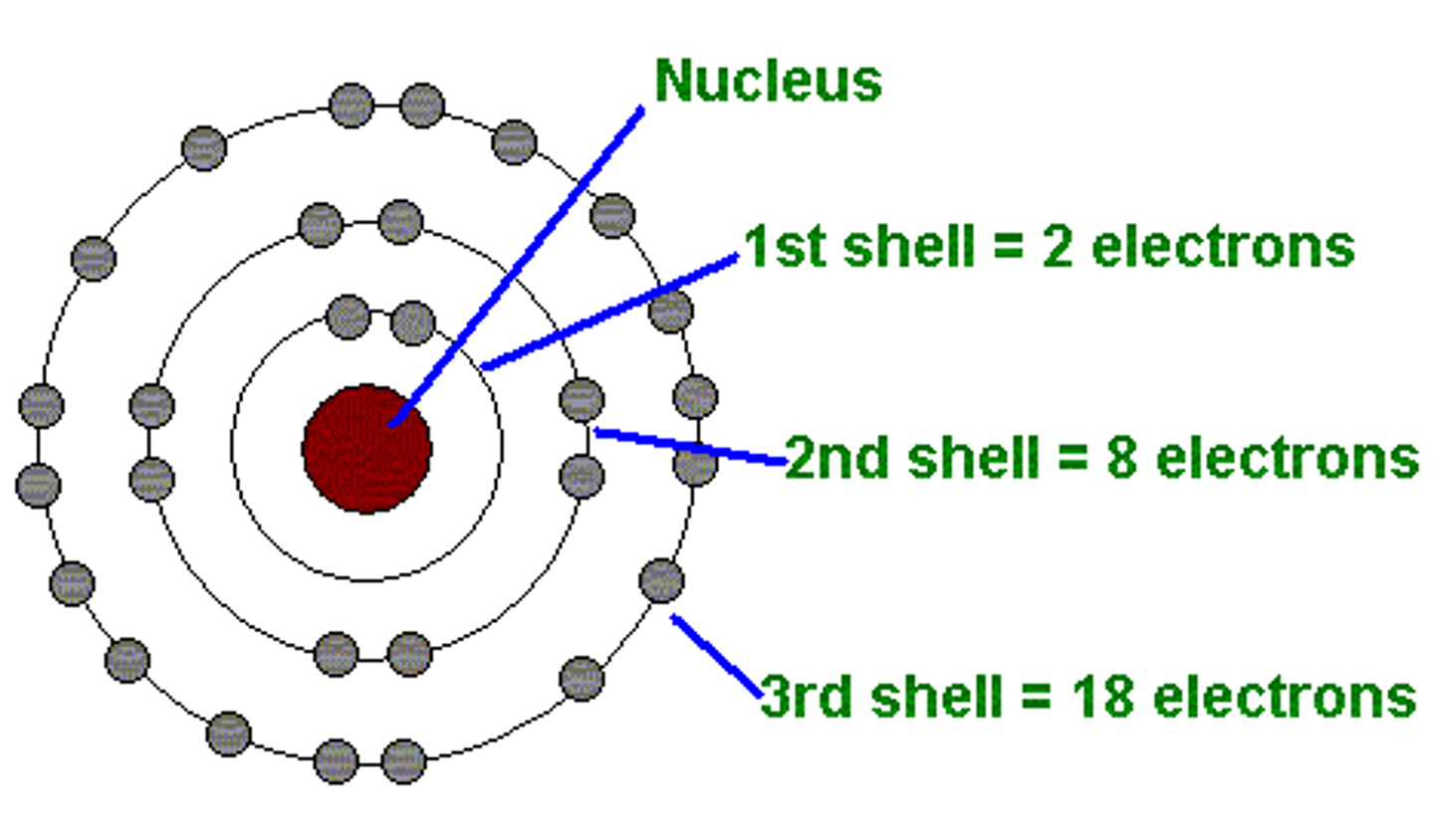
Bohr's Model Four Main Ideas
- Electrons orbit the nucleus in ring-like paths at fixed energy levels
- the higher the energy level the farther away the electron is from the nucleus = higher energy
- Orbits further from the nucleus exist at higher energy levels
- Electrons are quantized
- Electrons can only occur in one energy level not outside of it
- Electrons can only move from one orbital to another orbital at one time
- When electrons return to a lower energy level they emit energy
- These emissions are not always visible
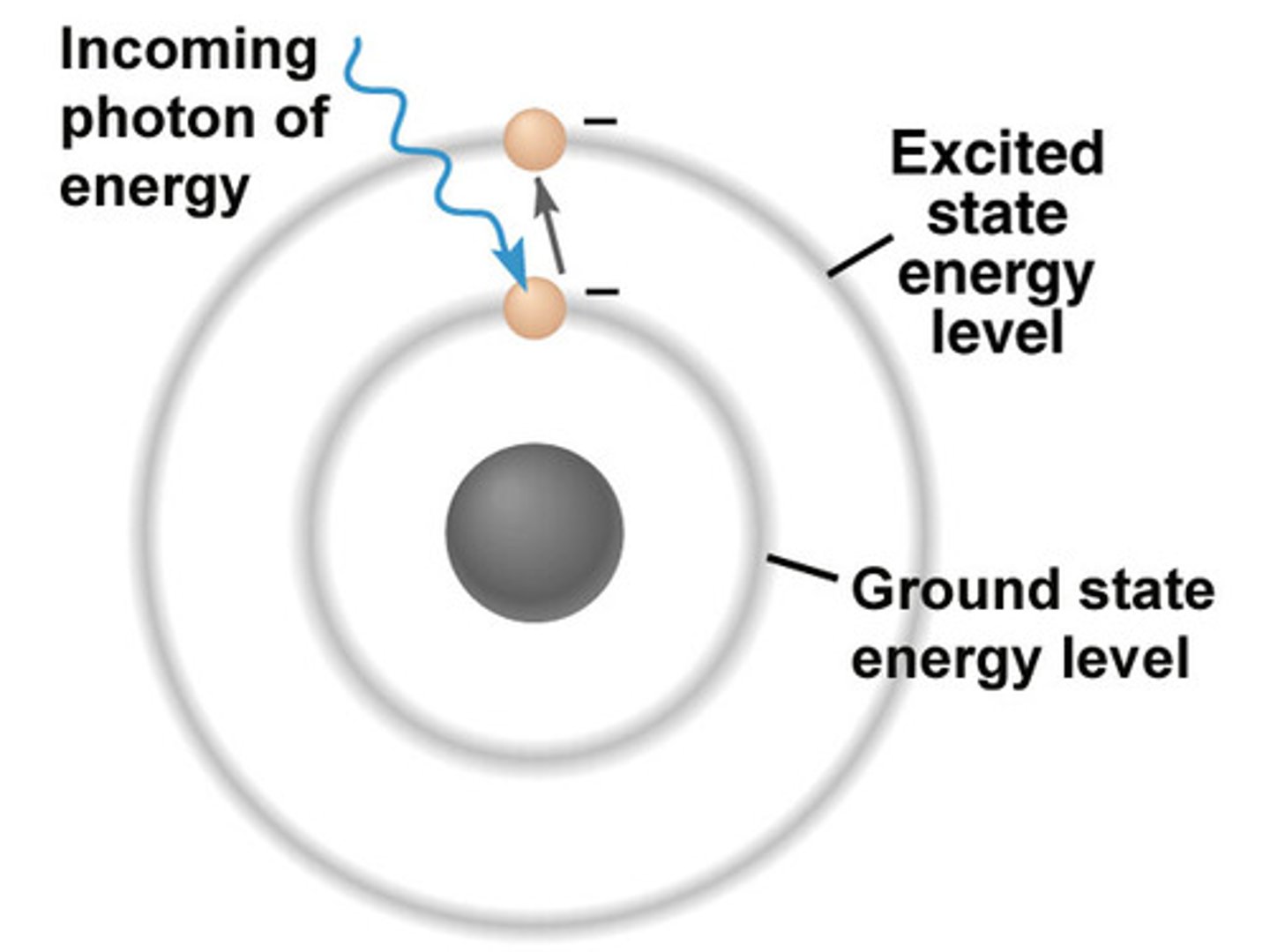
Definitions: Ground State
- The energy level an electron normally occupies
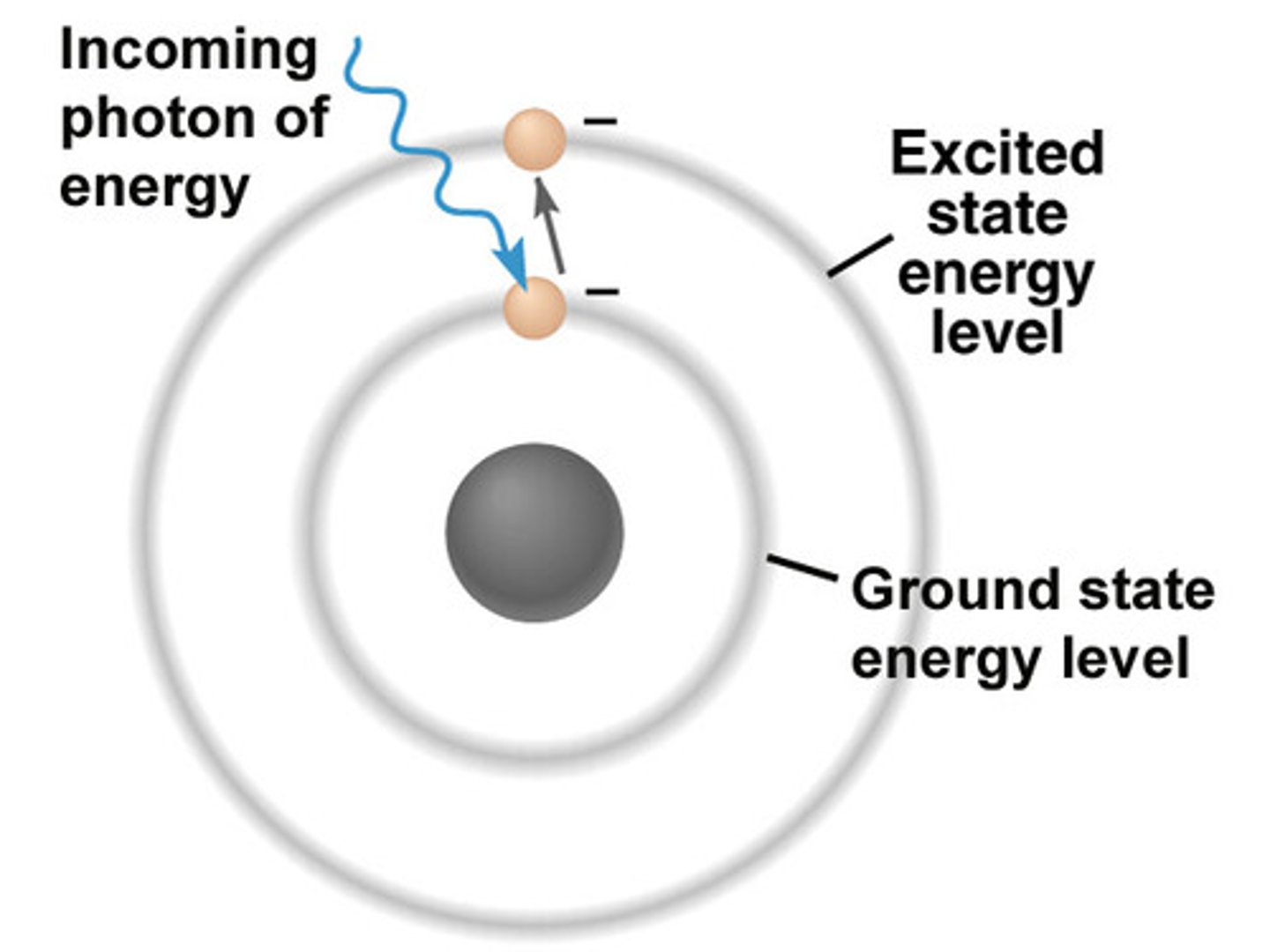
Definitions: Excited State
- The electrons that absorb energy as they move up to a higher energy level
Emission Spectrum
- Produced when photons are emitted from atoms as excited electrons return to a lower energy level
- Each electron transitions from a higher energy level to a lower energy level
- Corresponds with a specific frequency/wavelength
- Only contains specific colours (frequencies/wavelengths) of visible light
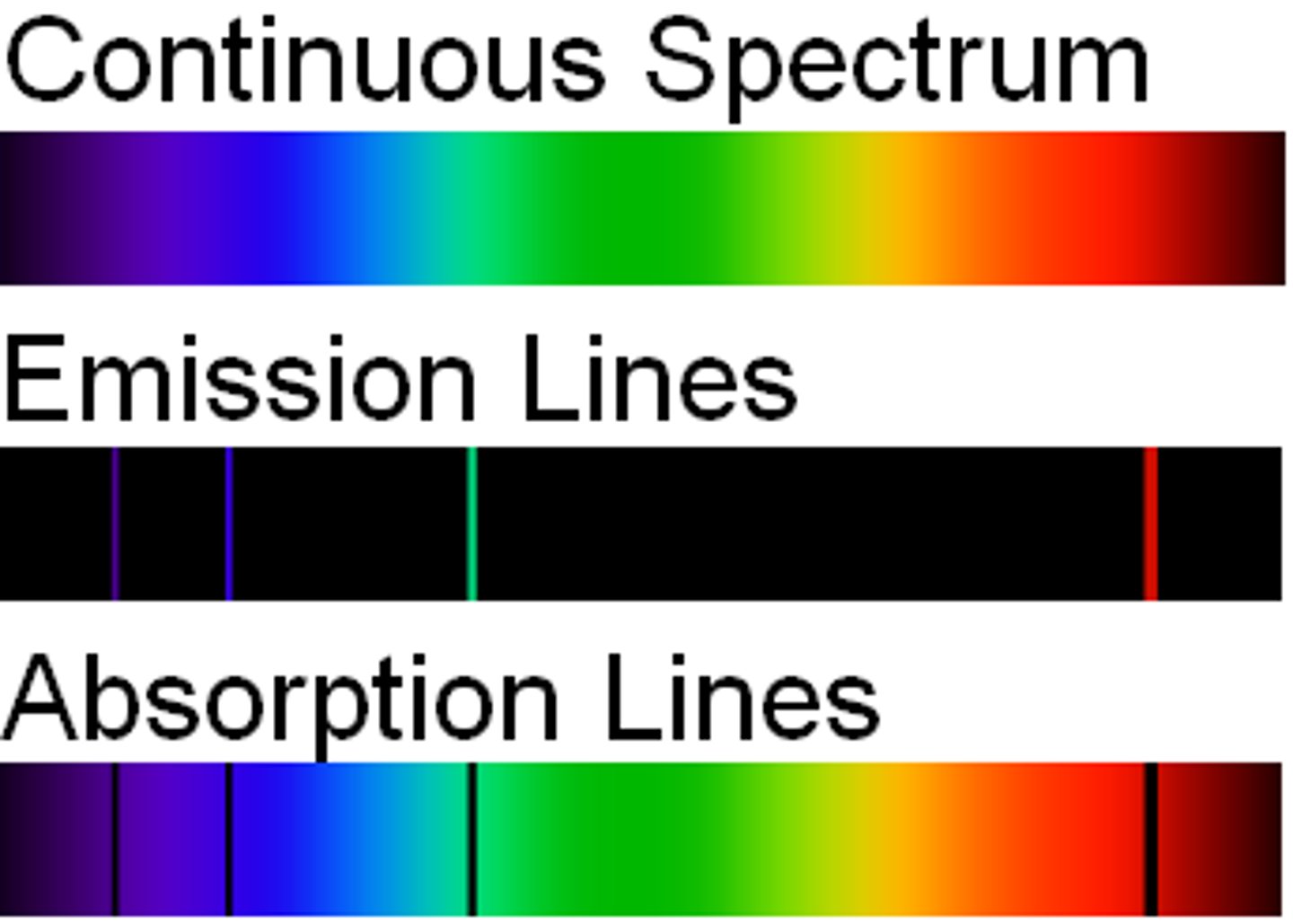
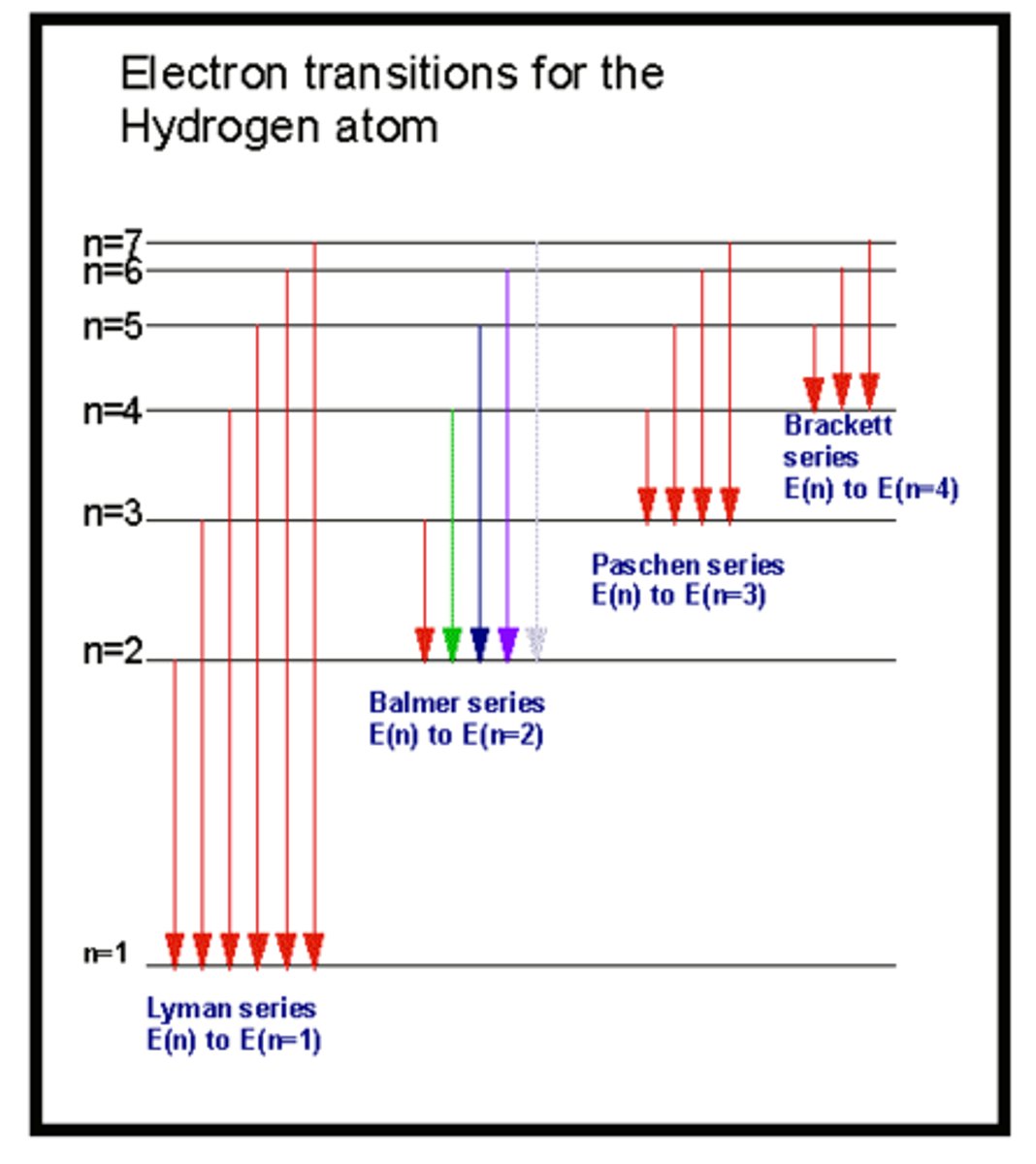
Emission Spectrum Example: Hydrogen
- The energy lines on the emission spectrum correspond to a difference in energies between energy levels
- Hydrogen provides evidence for the existence of electrons in discrete energy levels
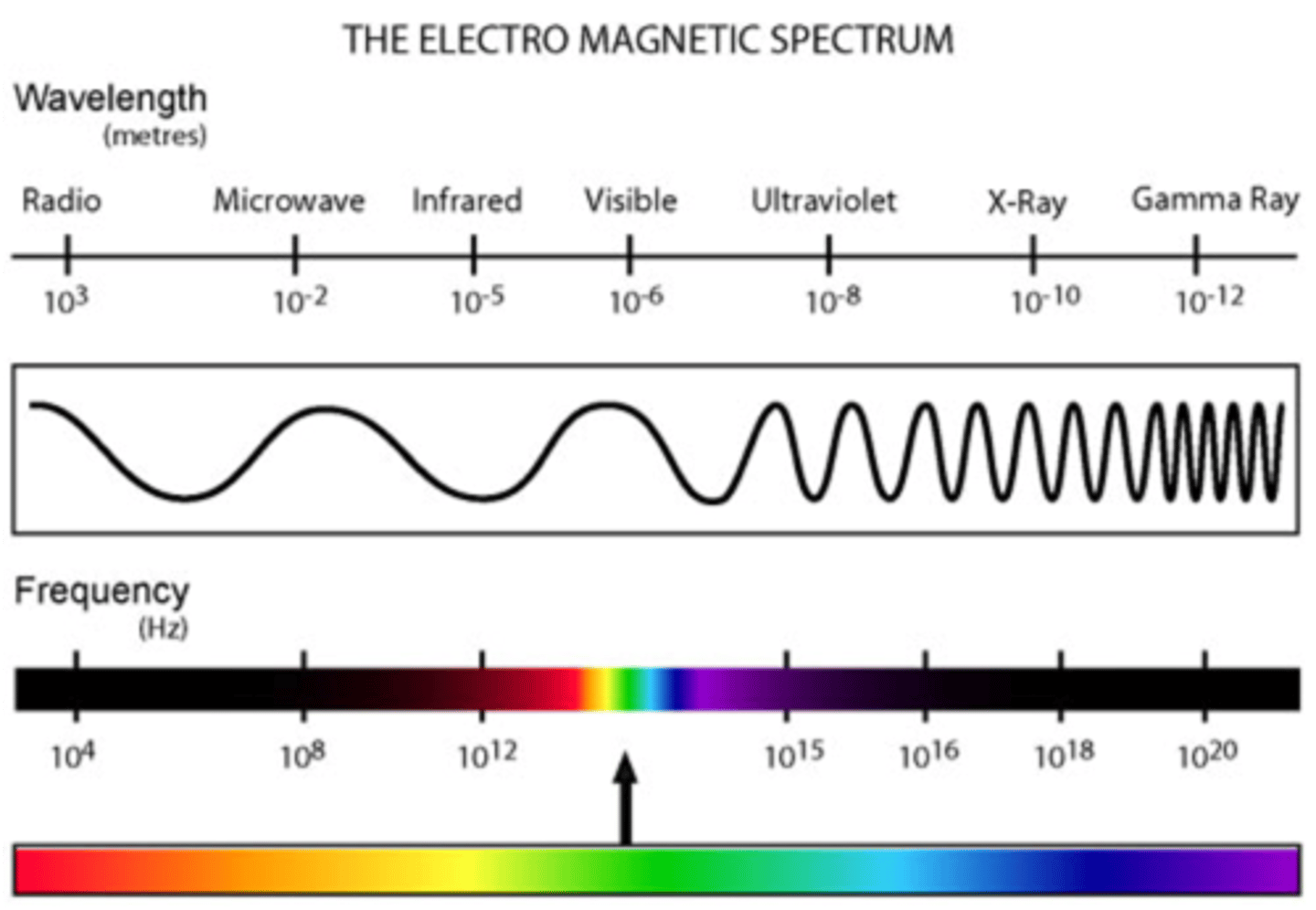
Electromagnetic Spectrum
- Electromagnetic waves travel at the same speed
- Shows the range of all possible frequencies of electromagnetic radiation
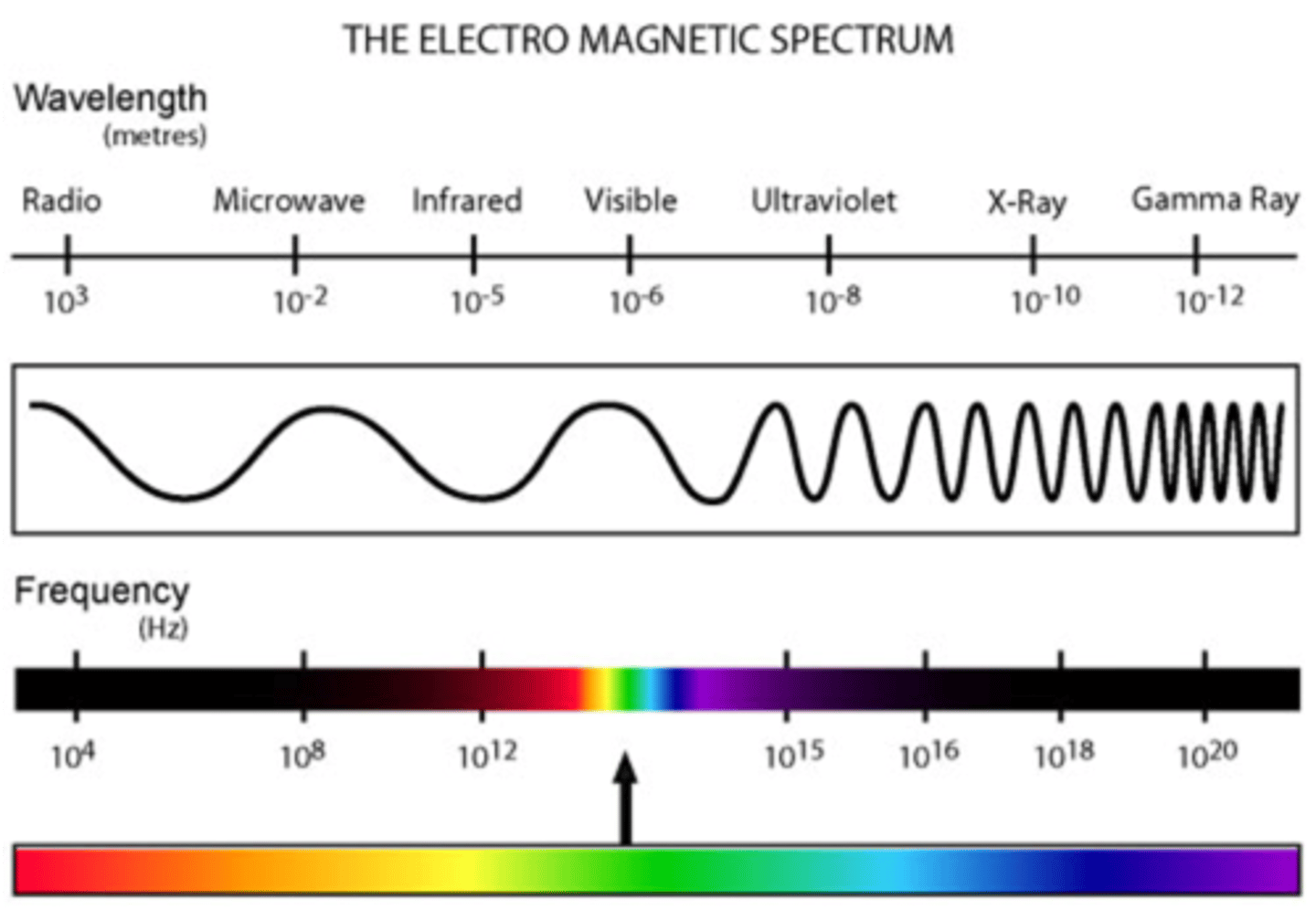
Electromagnetic Spectrum: Order of Spectrum
- Rabbits = Radio waves
- Mate = Microwaves
- In = Infrared rays
- Very = Visible light rays
- Unusual = Ultraviolet rays
- eXpensive = X-rays
- Gardens = Gamma Rays
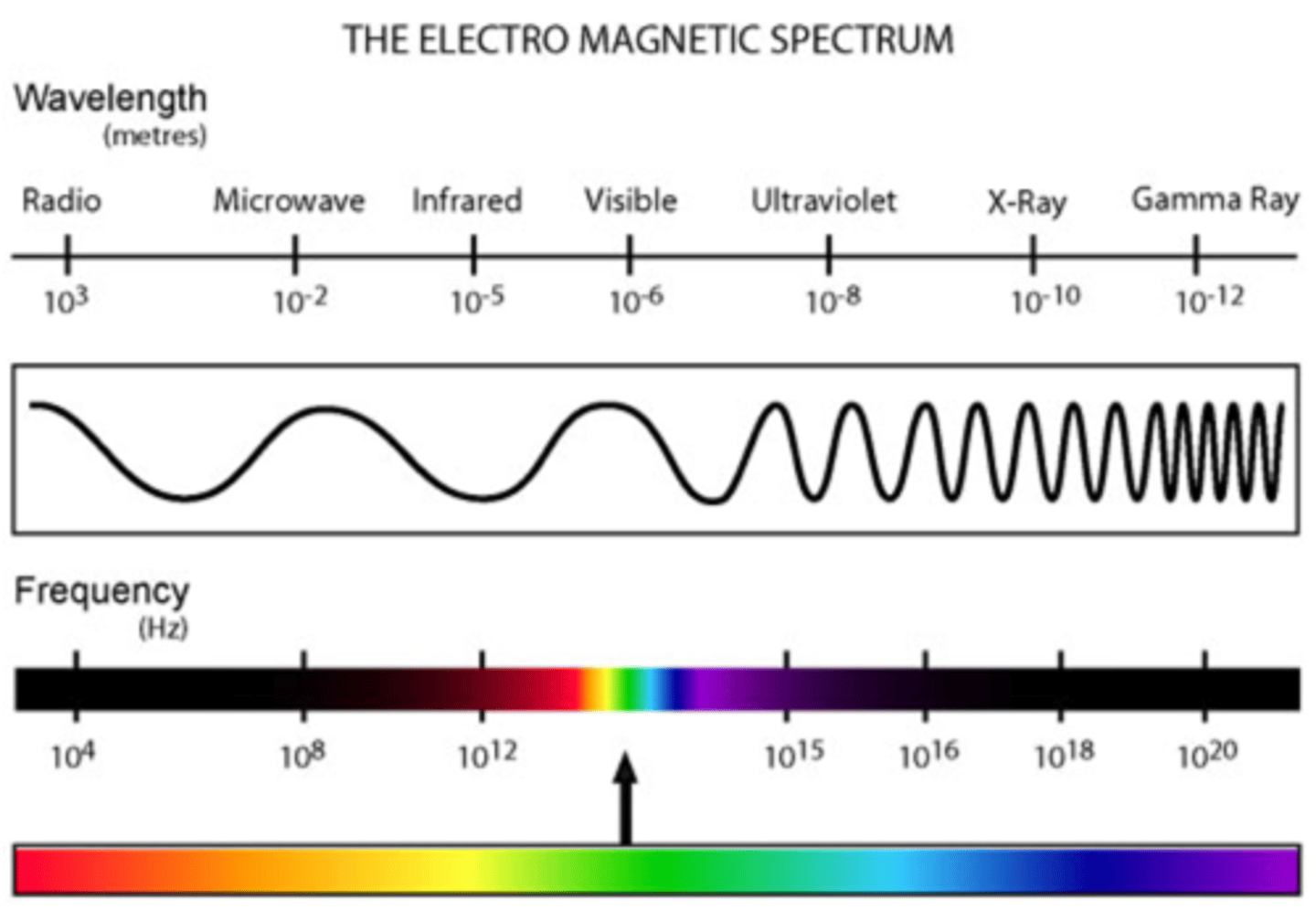
Electromagnetic Spectrum: Wavelengths
- The distance between two successive crests
- A smaller/shorter wavelength = Higher frequency = more energy
- A larger/longer wavelength = Lower frequency = less energy
- As energy increases so does frequency
- Red light = low energy
- Violet energy = high energy
-
Orbitals
- A region of space where the probability of finding an electron is greater than or equal to 95%
- Each orbit can hold a maximum of two electrons with opposite spins
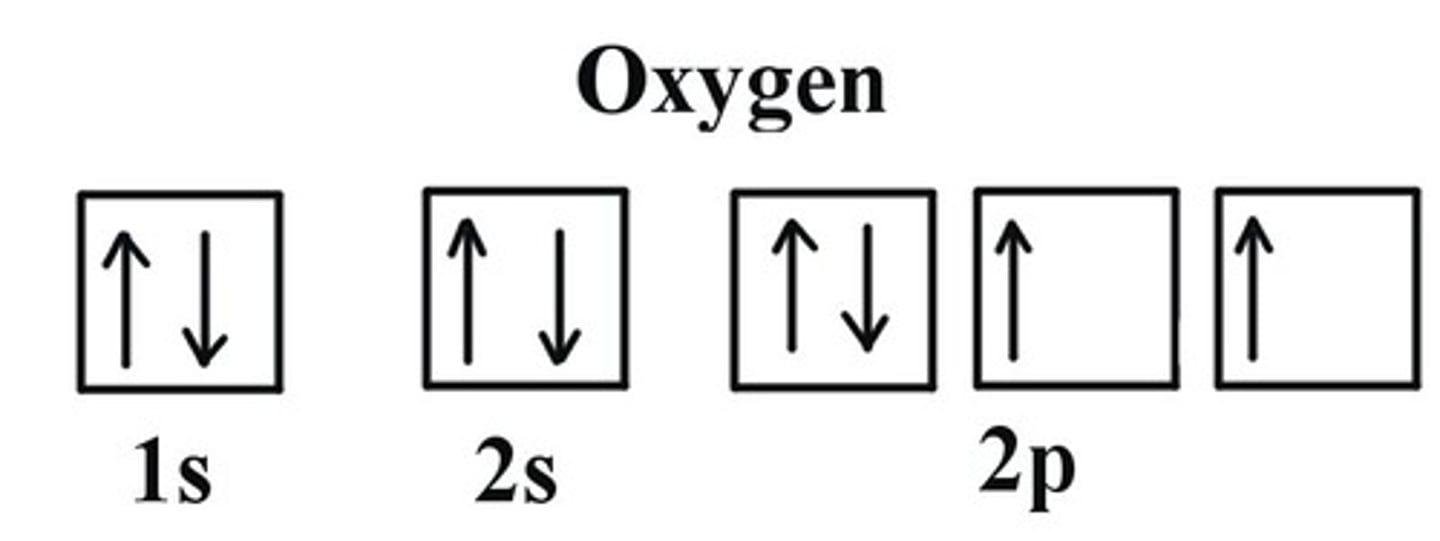
Orbits can be represented as boxes with electrons depicted with arrows
- Up and down arrows imitate the spin
- all orbits must have an up arrow before having a down arrow
Orbitals: Heisenberg Uncertainty Principle
- Impossible to define the exact position of an electron
Orbitals: Pauli's Exclusion Principle
- Controls how electrons fill atomic orbitals
- No more than two electrons per each orbital
- if two electrons are in one orbital they must spin in opposite direction (up and down arrow)
Orbitals: Aufbau Principle
- Controls how electrons fill atomic orbitals
- Electrons are placed into orbitals of the lowest energy first
Orbitals: Hunds' Third Rule
- Controls how electrons fill atomic orbitals
- Orbitals of the same sublevel are filled single and then doubly (all filled with an up arrow then finished with down arrows)
- If more than one orbital in a sub-level is available, electrons occupy different orbitals with parallel spins (the extra orbitals fill with up arrows)
S/P/D/F Sub-levels
- A group of orbitals with particular properties: shape and angular momentum
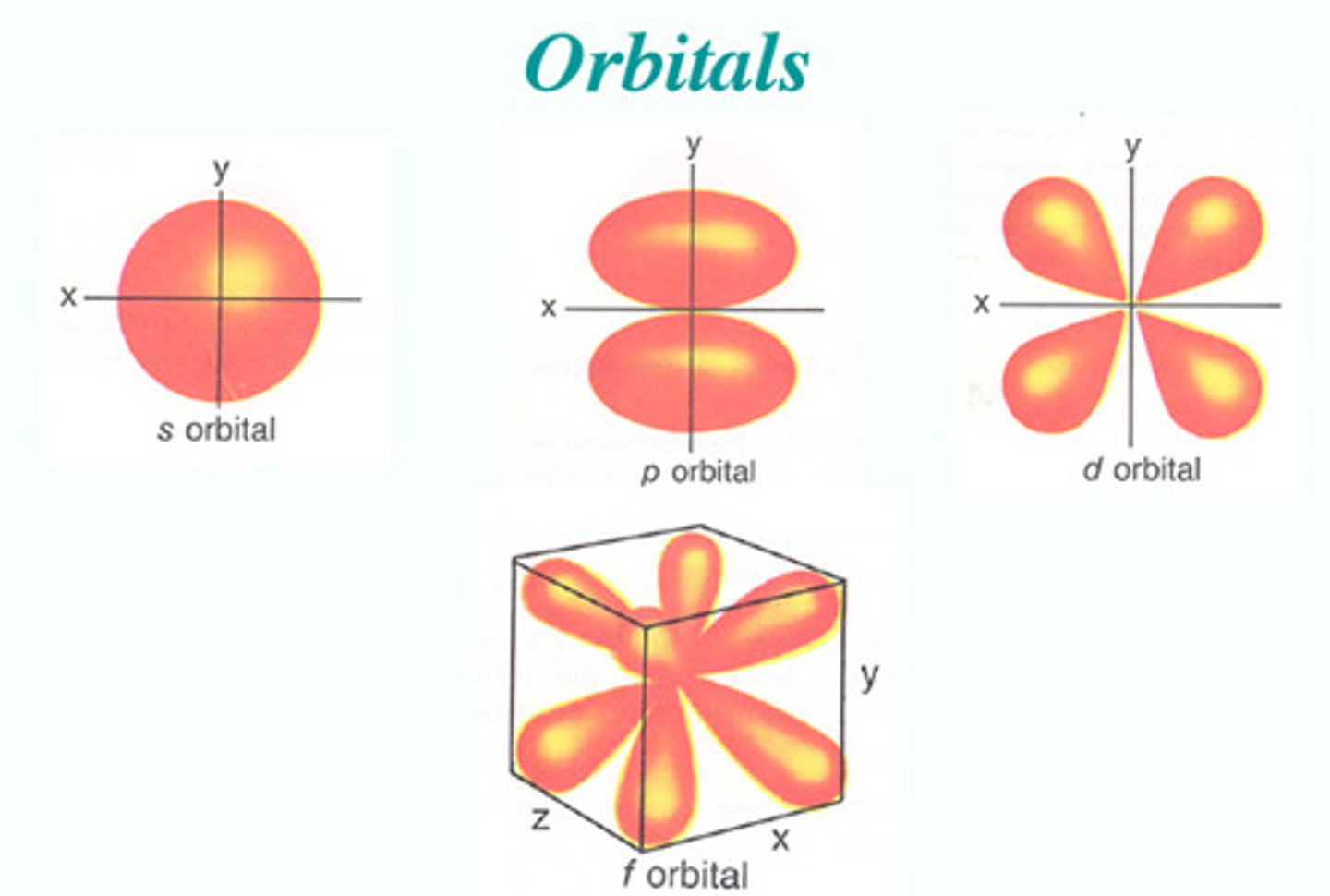
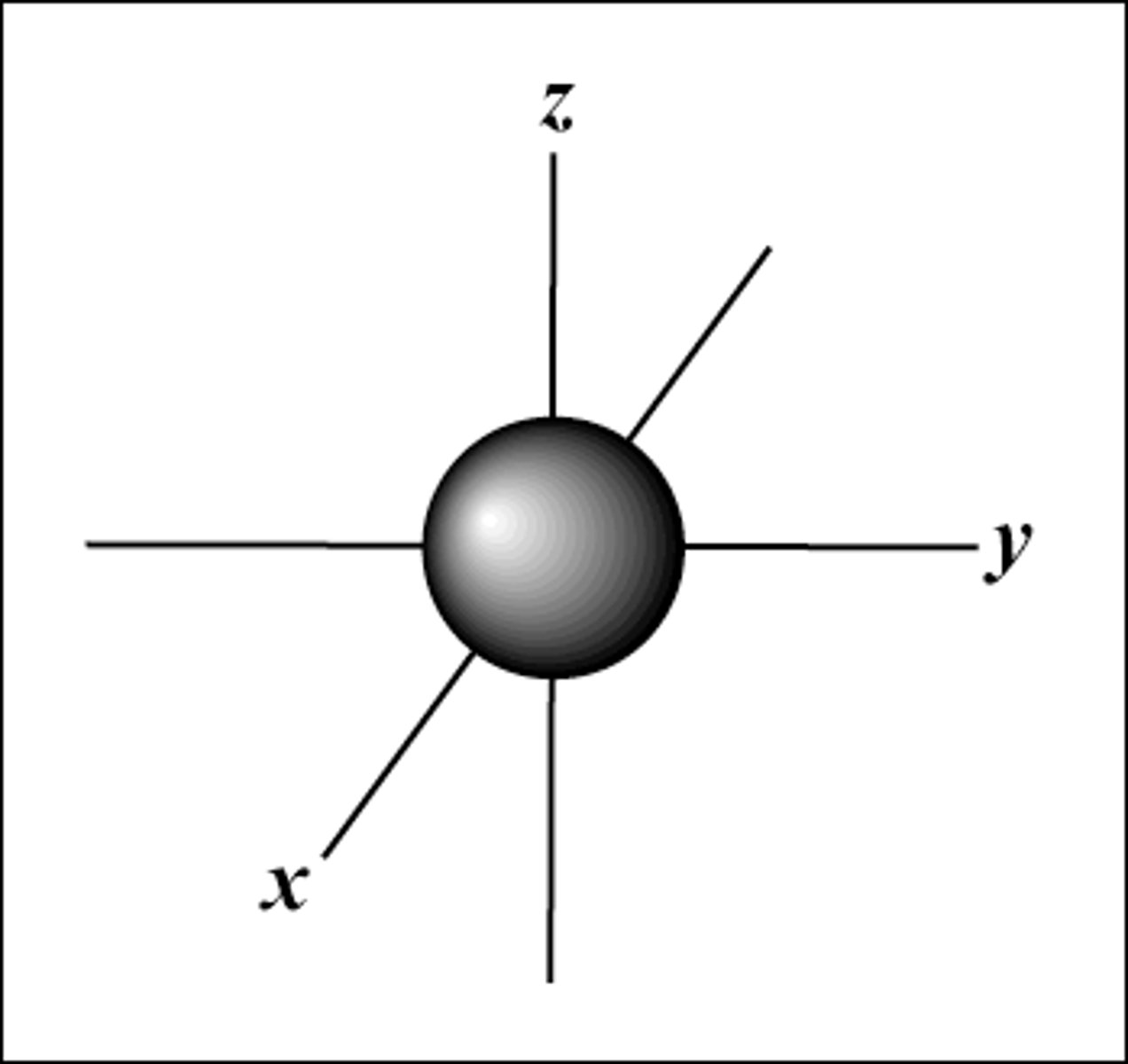
S-Orbital
- Spherical shape
- 1s = 1 orbital
- 2 electrons
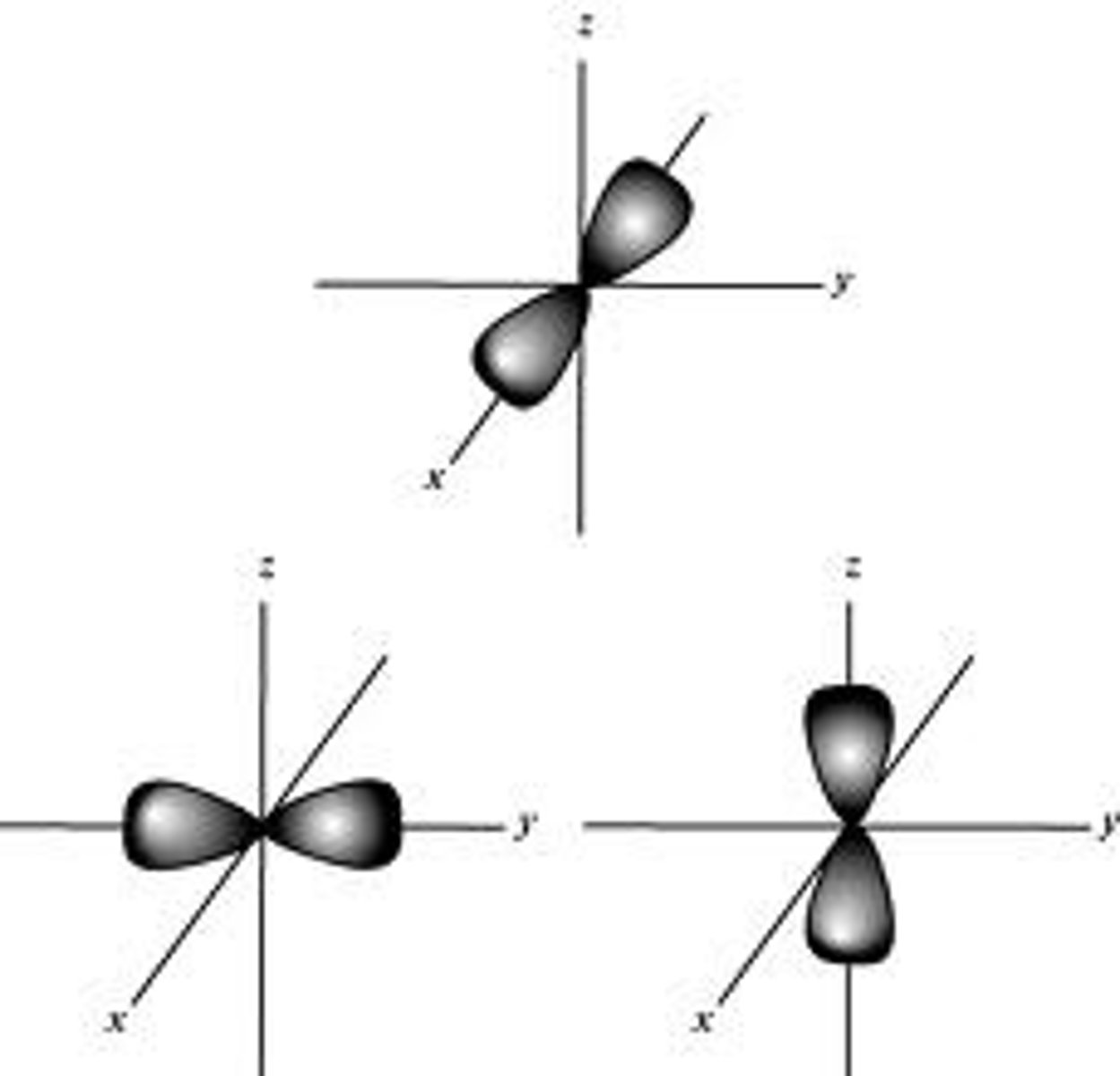
P-Orbital
- Dumbbells' shape
- 3p = 3 orbitals
- 6 electrons
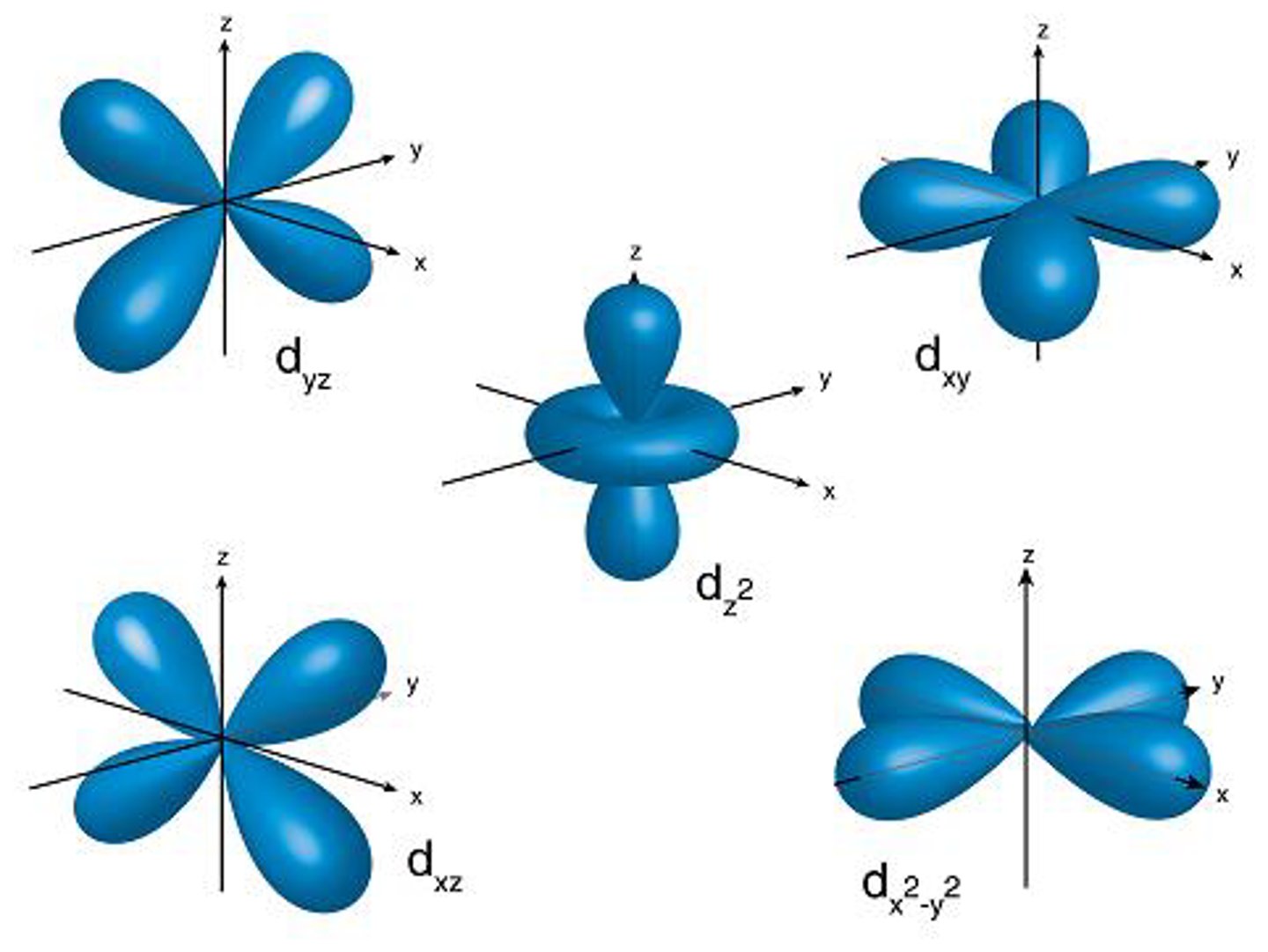
D-Orbital
- 5d = 5 orbitals
- 10 electrons
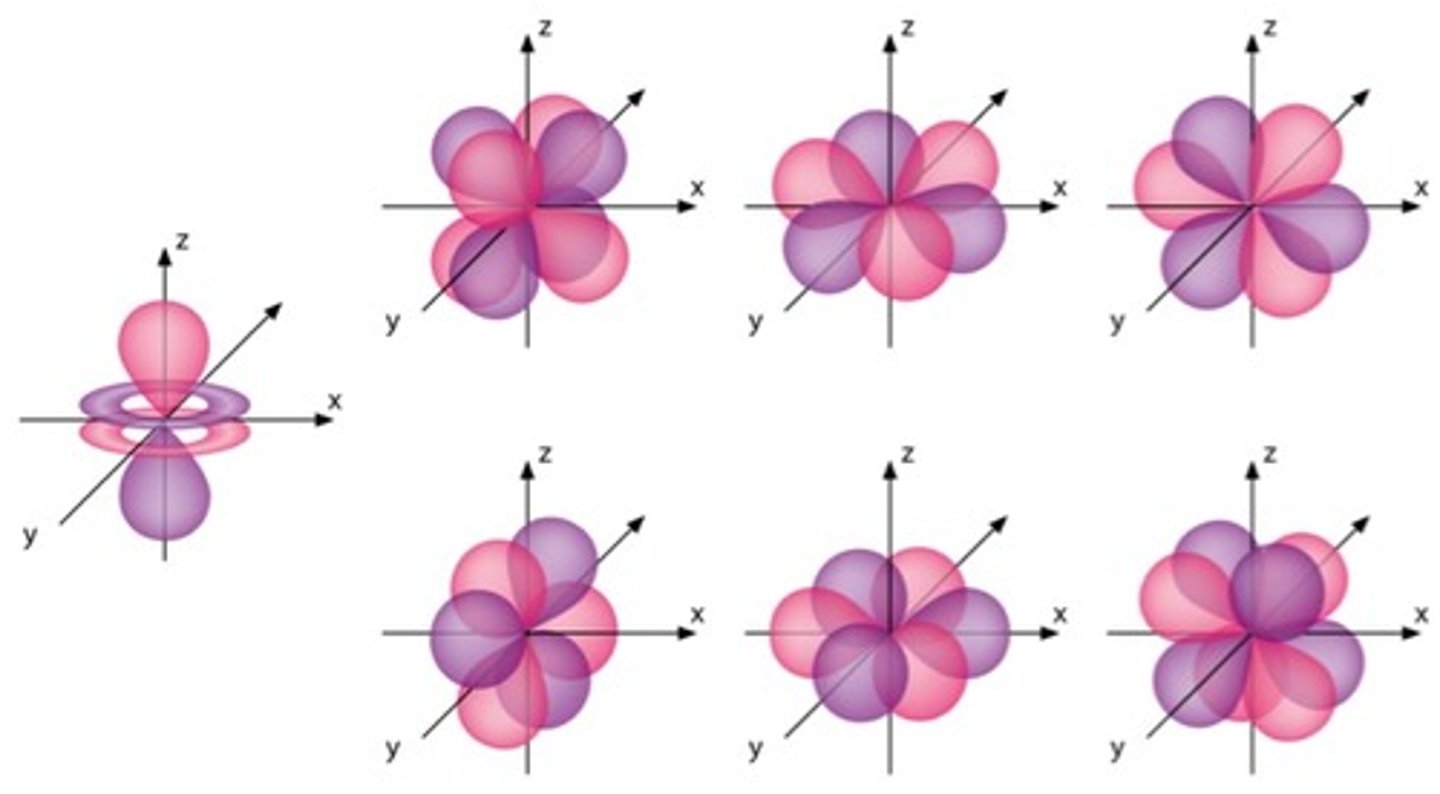
F-Orbital
- 7f = 7 orbitals
- 14 electrons
Main Energy level can hold a maximum of 2n^2 electrons
- Refer to Chart

Writing Electron Configurations/Arrangements
- Determine the total number of electrons
- Fill the lowest subshells first then higher energy levels until all electrons are used (Use chart)
- Note: 4s orbital is filled before 3d
- Example: Ti = 1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^2

Writing Electron Configurations/Arrangements Condensed
- Write the symbol in square brackets [ ] for the nearest smallest noble gas
- Write the following electron configuration after the noble gas configuration
- Example: Abbreviated electron configuration of Ca
- Ar = 1s^2 2s^2 2p^6 3s^2 3p^6
- Ca [Ar] 4s^2