Lecture 08 - Protein Purification
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
What is specific activity?
units of activity per mg of isolated protein
specific activity = total activity / total protein
What is the workflow for protein purification?
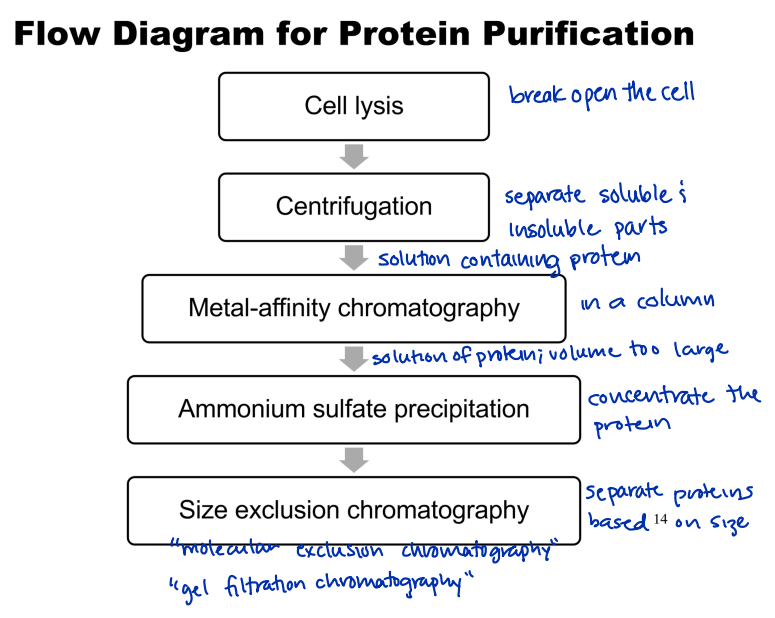
What is ammonium sulfate precipitation?
separates proteins based on their solubility in salt
When should ammonium sulfate precipitation be used to purify a protein?
to concentrate dilute protein samples, remove unwanted proteins, or when working with large samples
What is metal-affinity chromatography?
separates proteins in a column on their ability to bind metal ions (ie: Ni)
When should metal-affinity chromatography be used to purify a protein?
When the protein has His-tag. Histidine (or C or W) can bind immobilized metal ions without denaturing). Release the protein with imidazole or by lowering pH
What is gel filtration (size exclusion) chromatography?
separates molecules based on size when they pass through a column with carbohydrate polymer beads
When should gel filtration chromatography be used to purify a protein?
gentle, non‑binding purification to remove contaminants
How can protein purity be determined using SDS-PAGE?
SDS denatures the protein and gives it a negative charge, 2-mercaptoethanol is added to break disulfide bonds, electrophoresis moves the protein based on size
What is a protein purification table?
summary table tracking protein through each purification step, shows how purity, yield, and specific activity change
How to determine yield in a purification table?
yield = total activity (2) / total activity (1). multiply by 100 (%)
How to determine purification (fold) in a purification table?
purification = specific activity (interest) / specific activity @ 100 yield