WIP Temperature dependent Kinetics and complex reactions
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Give the arrhenius equation with units

Linearise the arrhenius equation, when can we do this

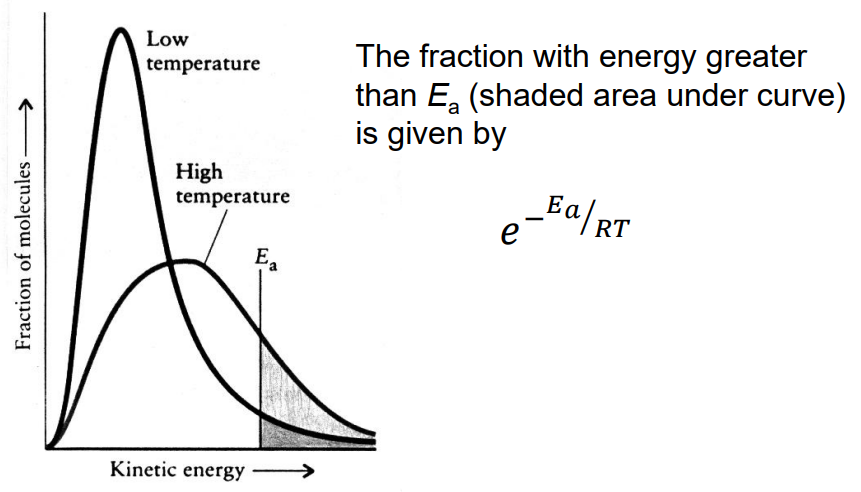
Draw out a boltzmann graph of high and low temperature, state the equation for the fraction of molecules with energy greater than Ea
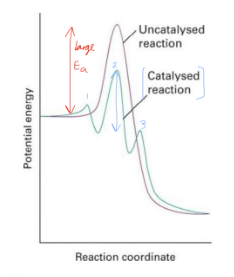
state two ways an energy barrier can be reached
Can overcome the barrier with heat
Need high T experiments
Can lower the barrier with catalysis (or enzymes)
Complex reactions (Barrier is not elementary)
what makes a reaction arrheinious like and what does this mean
Having to break strong bonds means a reaction is arrhenious like
this will mean that there will be a strong dependance on temperature
What is a complex reaction
Multi step reactions where the rate equation doesnt always reflect stoichiometry
Describe the Isolation method
Create a great excess of all reactants but one making them all essentially constant. As such we can analyse the rate equation in respect to just one of the reactants.
from here we can find what order that reactant is.
In sequential reactions what should you look to identify first
The slowest step of the reaction
What is the rate determining step
This is the slowest step
It will not matter how fast subsequent steps are in the reaction sequence, only the RDS and the steps prior to it will effect the obsereved rate.
Use a three step reaction to help describe how RDS effects order
A → B → C → D
Just [A] is first order if A→B is RDS
Just [A] and [B] are second order if B→C is RDS
[A] [B] and [C] are third order if C→D is RDS
Write out a catalysed version of A→B
A + cat → A-cat → B-cat → B + cat
What happens when the critical conversion step at the catalyst is also the RDS
it will be zeroth order meaning rate is independant of concentration
This is because few of the active sites are not consumed at this point aswell as the fact that reagent A is in large excess
Draw a reaction graph of catalysed vs non-catalysed
State what an elementary reaction is
Occur at atomic level, observed order = molecularity, straightforward kinetics
State what a bulk reaction is
Represents the overall stoichiometry. occur via many elementary reaction steps and can have a misleading order. May have very complicated kinetics.
What does steady state approximation mean, and how do we use it
Analysis of complex sequential reactions from its ‘steady state’, where rate of formation = rate of loss of the intermediate
As such the intermediate is not changing with time
As such we have the key assumption of d[intermediate] / dt = 0
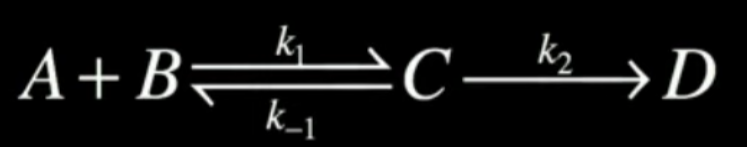
Complete a Steady State approxiamtion of this simple complex reaction

for gas phase reactions what assumptions do we make and when are they good/bad
assume that : gases behave statistically and uses ideal gas behaviours
This works well for Gases like helium that have weak forces and are hard monoatomics, Works poorly for larger molecules with great many forces
When are 2nd order reactions bimolecular
when they are elementary
Give K units for gas phase
1st order , K is s-1
Pseudo 1st order , Keff is s-1
2nd order , K is conc-1time-1 or cm³molecule-1seconds-1
Give the collision rate coefficient equation, stating what each part means as well as what it implies

How can we estiamte the rate of gas phase radical equations
We look at collision theory to estimate K.
In general at T=298K, Kcoll = ~2×10-10 cm3molecule-1s-1
However sterics and orientation can alter these values
True K is =< Kcoll
Describe Discharge flow for gas-phase
Inititation by mixing, the DF will mix within ms
Time resolution dependant on distance from the detector
Monitors radical A sensitively, in steady state at the end of the tube, as such no fast detectors are needed. (UV-Vis, Flouresence, Mass-Spec)
Monitoring unchanging [B] accurately (dilution, GC, NMR, FTIR)
give equation for rection time when using fast flow tubes
reacton time , t (s) = distance to detector (m) / gas flow velocity (m/s)
why requires more energy bond dissociation or or activation energy
BDE »»» Ea
What does boltzmann graphs display and give the equation for the fraction of molecules capable of reacting
Boltzmann distributions statistically describe how molecules of a gas all exist at different energies
The fraction of molecules with energy greater than the Ea is e-Ea/RT
what happens to arrhenious plot over large range of T
It becomes curved
What is the significance of A in bimolecular reactions
A is related to the collision rate, kcoll
give equation for kcoll
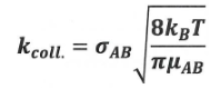
what other factors does A relate too
A = kcoll x P
it relates to a steric factor (P) aswell
It also as we see via its relation to kcoll depends on T
Why was transition state theory developed
As a way of using quasi-equilibrium to understand massive reaction mechanisms like octane and diesel
Providing a more detailed