7.2 Non-ferrous Metal Alloy
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
Properties of pure aluminium and aluminium alloy
low density
low MP (limited high temp capabilities)
high electrical and thermal conductivity
corrosion resistant →produce thin layer of alumina
only can weld in inert atmosphere (have to remove oxide layer for metal to weld together)
difficult to extract as only exist in mineral form
low strength(pure), comparable strength to iron(alloy)
but high ductility (pure), lower ductility (alloy)
Two main type of aluminium alloy
cast (eutectic) → casted then straight to service
wrought (formed)
→ further categorised into heat treatable or not
Advantages of having a eutectic composition for casting processes
no intermediate process between liquid and solid (convert fully from liquid to solid in one temeprature)
→ easier for no defects to form during casting
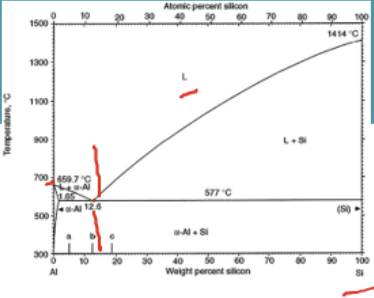
Main alloying addition to aluminium
Cu & Mg→ solid solution strengthening + precipitation hardening
Si→ for eutectic composition(~10%Si)
Changes in property with addition of Cu to Al alloy
solid solution strengthening →minor improvement in strength
Increasing Cu promote intermetallic CuAl2 formation (θ) under equilibrium cooling → hard but poor tensile strength
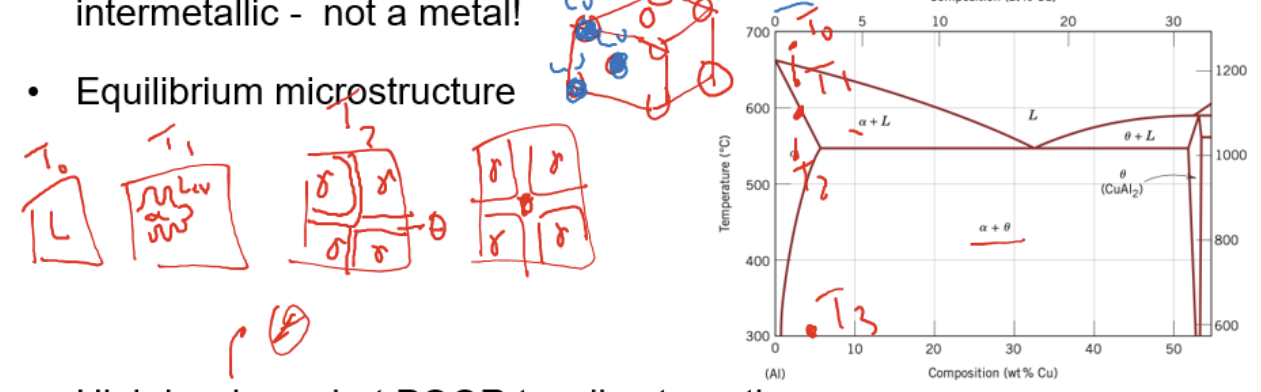
Process of age hardening (form of precipitation hardening)
solution treat alloy to be within single phase super saturated solid solution region(α= low temp phase)
Quench to prevent formation of 2nd phase
reheat/age at 2 phase region for a certain period of time so that hard theta forms in small precipitate
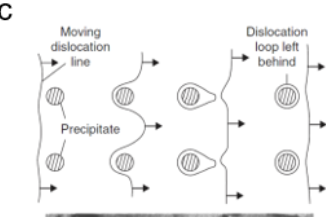
How does precipitate hardening increase strength
precipitate acts as obstacles which require more stress for dislocation line to pass through precipitate
Relationship of precipitate spacing and strength of material
σ inversely propertional to spacing btw precipitate λ
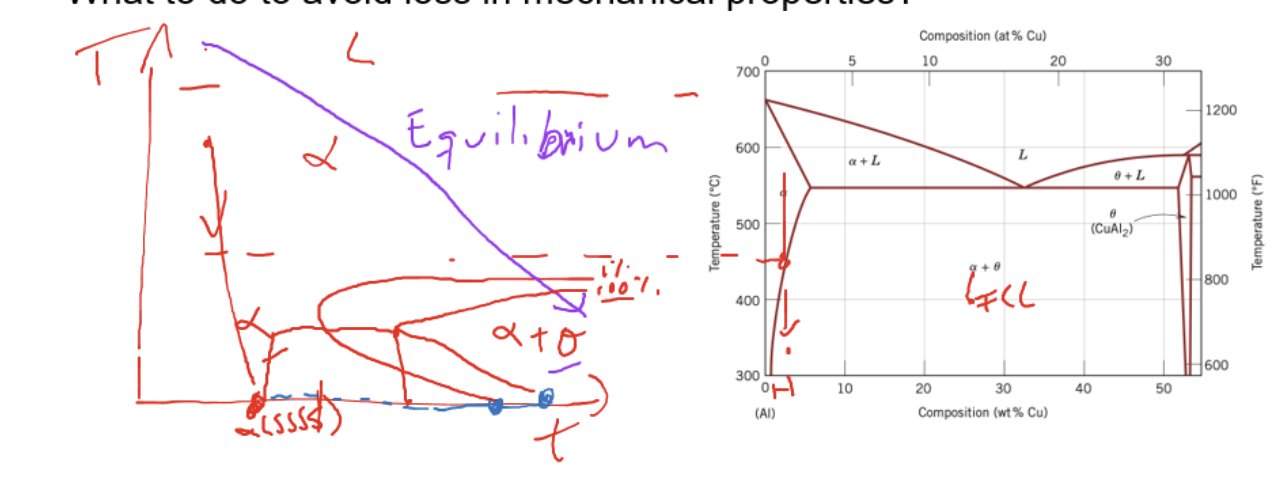
Requirement for an effective strengthening effect
precipitate or second phase is harder than base material
require suitable phase transition (can cool to a single phase first before aging it to two phases)
Quenchable alloy (able to avoid 2 phase transformation initally)
Relationship of mechanical property with aging time
reach peak mechanical property
over aging: longer time for aging increase size of precipitate -. become less effective in preventing dislocation
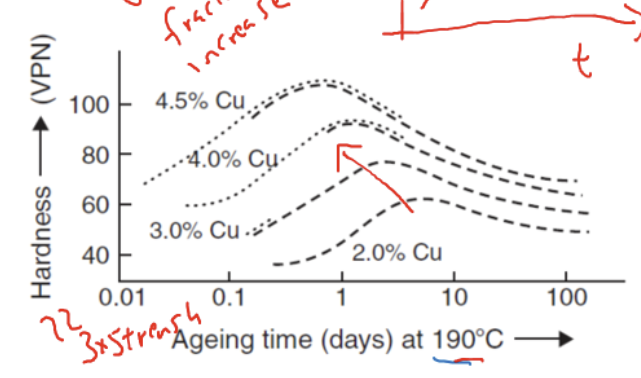
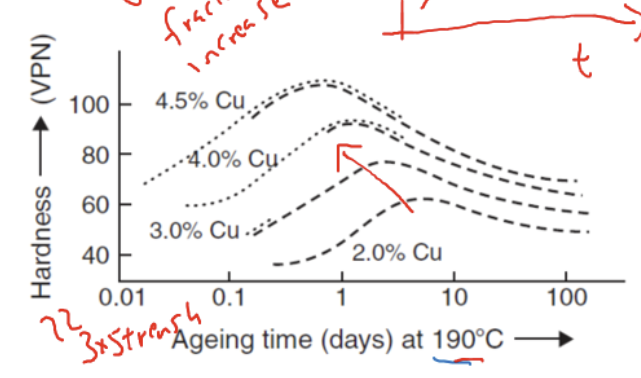
Relationship between composition of alloying addition and mechanical properties
higher Cu→ better mechanical properties + reach peak strength quicker in aging time
Relationship of temperature with peak hardness and aging time
Higher temp → lower time to reach peak mechanical property
higher temp→ lower peak hardness value

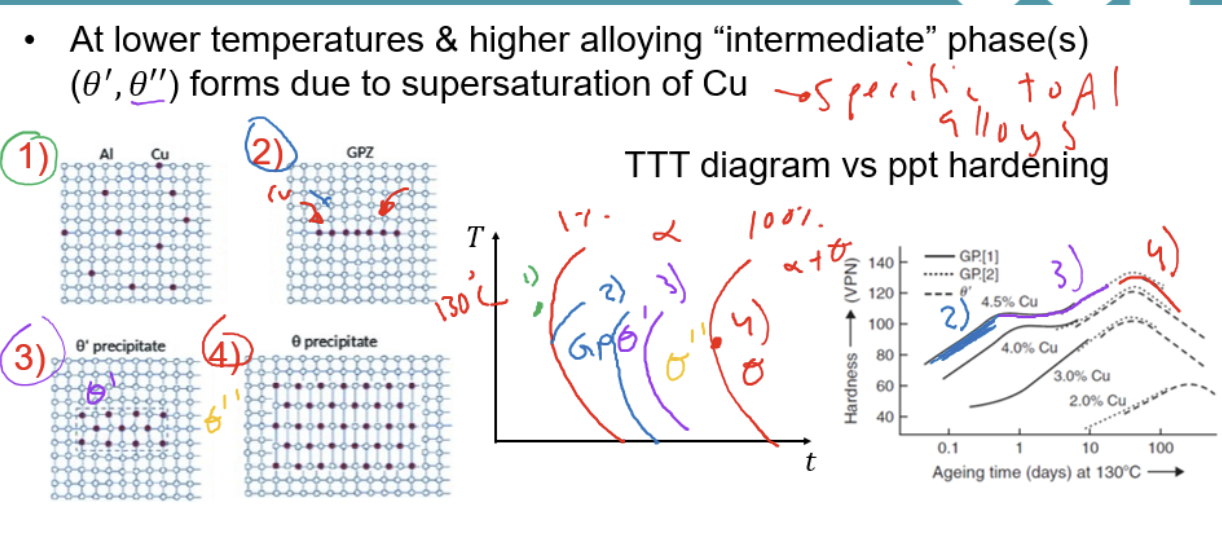
Condition for alloy to go through Guinier-Preston (GP) zones
at lower aging temperature for higher composition
Al alloy only
Process of aging for materials going through GP phase
Before aging
in GP zone, copper gather to form large needle shape during intermediate phase
As more copper joins, transition to more intermediate phase denoted as θ’,θ’’,…
Reaches equilibrium and precipitate size increase to critical
Limitations of aged Al alloy
Limited temperature capabilites
precipitate dissolve at high temperatures
OR lead to overage which deprove mechanical properties
Welding difficulties
when locally heating metal, lose precipitate due to overheating, over aging occur as well
Alloying addition of Al for aerospace
Li and Be
→ reduce weight
Properties of pure copper and copper alloy
good ductility/toughness as they are FCC
good heat and electric conductivity
good corrosion resistance (Cu oxide/sulphide forms on surface)
Main copper alloys types
Bronze (Cu-Sn (12%)): used for marinetime, musical instruments, low friction components
Brass (Cu-Zn)
properties improve by solid solution strengthening
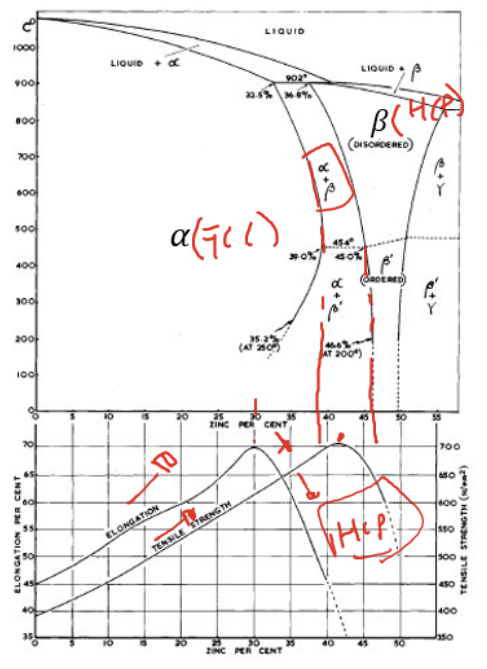
Categories of brass
α brass (max 40% Zn)
FCC single phase for solid solution strengthening
α-β brass (40-45% Zn)
HCP (hexagonal closed packed) + FCC phase → can be heat treated to form various mechanical properties
Generally: ductility and strength is high in α-β copper composition
Property of Nickel alloy
Good ductility/toughness (FCC)
excellent corrosion resistance (Ni-Cr alloy)
very good oxidation resistance (from Cr and Al)
3 types of Ni alloy
Nichrome (20%Cr): high temp application and harsh chemical condition
Monel (28-34%): expensive, marintine ,chemical industry
Ni-based superalloy