AQA A level Chemistry 3.1.8 Thermodynamics
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
What is the standard molar enthalpy of atomisation? (2)
- The enthalpy change for the formation of one mole of gaseous atoms
- From the element in its standard state under standard conditions.
Example reaction for atomisation? (1)
½ Cl₂(g) → Cl(g)
What is the first ionisation energy? (1)
The minimum amount of energy required to remove one mole of electrons from one mole of atoms in the gaseous state
Example reaction for first ionisation energy? (1)
Na(g) → Na⁺(g) + e⁻
Why is the second ionisation energy greater than the first? (1)
It is more difficult to remove an electron from a more positively charged ion.
What is the first electron affinity? (2)
The enthalpy change when a mole of gaseous atoms is converted to a mole of gaseous negative ions
Example reaction for the first electron affinity? (1)
Cl(g) + e⁻ → Cl⁻(g)
Why is the first electron affinity exothermic? (2)
Due to the attraction between the electron and the nucleus
What is the second electron affinity? (2)
- The enthalpy change when a mole of electrons is added to a mole of gaseous negative ions
- Forming ions with a doubly negative charge
Example reaction for the second electron affinity? (1)
O⁻(g) + e⁻ → O²⁻(g)
Why is the second electron affinity always endothermic? (2)
Energy is required to overcome the repulsion between the negative electron and the negative ion
What is lattice formation enthalpy? (1)
The enthalpy change when one mole of a solid ionic compound is formed from its gaseous ions
Example reaction for lattice formation enthalpy? (1)
Na⁺(g) + Cl⁻(g) → NaCl(s)
What is lattice dissociation enthalpy? (2)
The enthalpy change to separate one mole of an ionic substance into its gaseous ions.
Example reaction for lattice dissociation enthalpy? (1)
NaCl(s) → Na⁺(g) + Cl⁻(g)
How are lattice formation and dissociation enthalpies related? (1)
They have the same value but opposite signs.
What is enthalpy of hydration? (2)
The enthalpy change when one mole of gaseous ions becomes aqueous ions
Example reactions for hydration enthalpy? (2)
- Na⁺(g) → Na⁺(aq)
- Cl⁻(g) → Cl⁻(aq)
Why is enthalpy of hydration exothermic? (2)
Due to the attraction between:
- Positive ions and δ⁻ O of water. (Na+)
- Negative ions and δ⁺ H of water. (Cl-)
What is enthalpy of solution? (2)
The enthalpy change when one mole of solid dissolves in water to form aqueous ions.
Example reaction for enthalpy of solution? (1)
NaCl(s) → Na⁺(aq) + Cl⁻(aq)
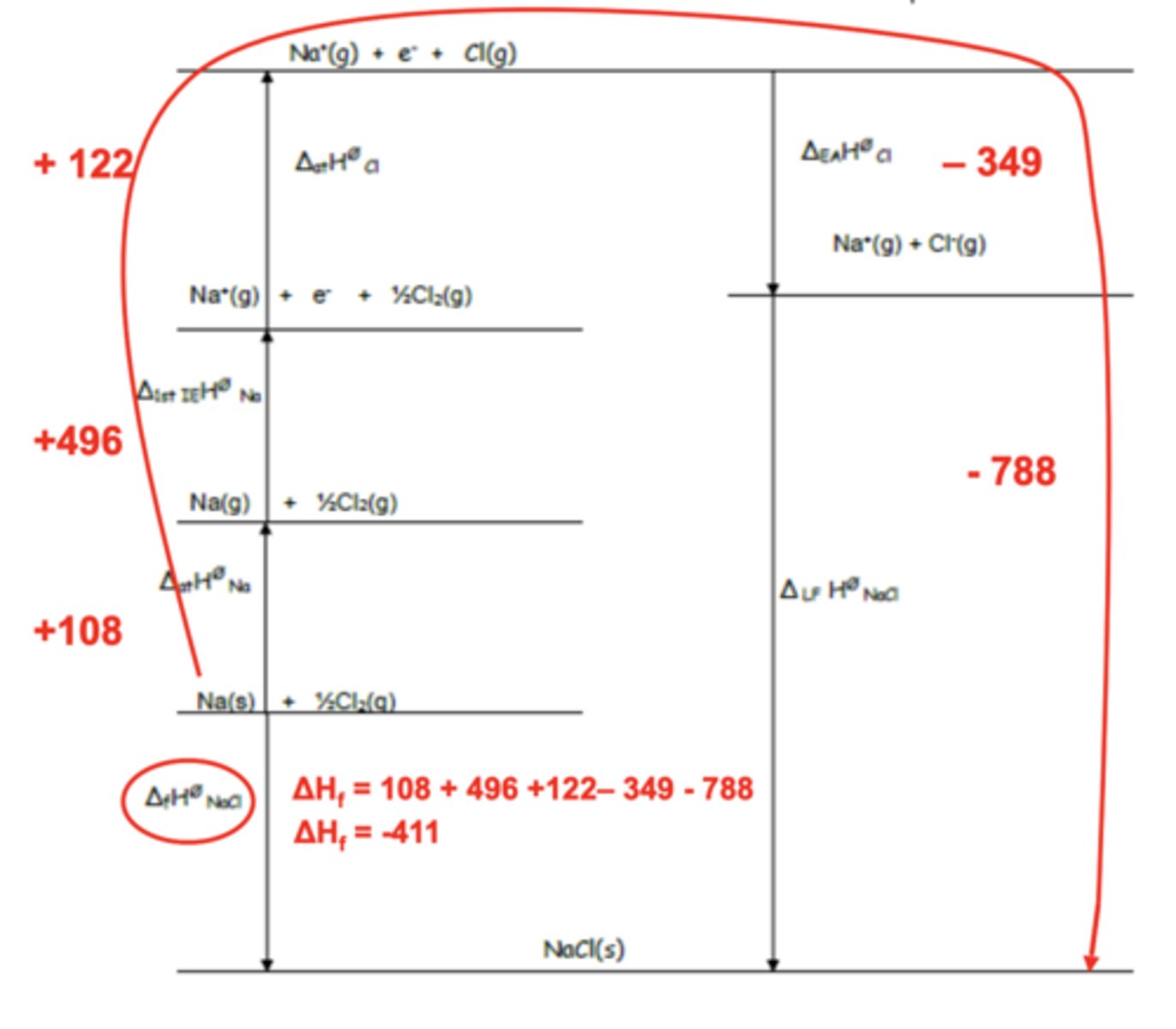
Draw born-haber cycle for NaCl(s) (6)
(ignore numbers)
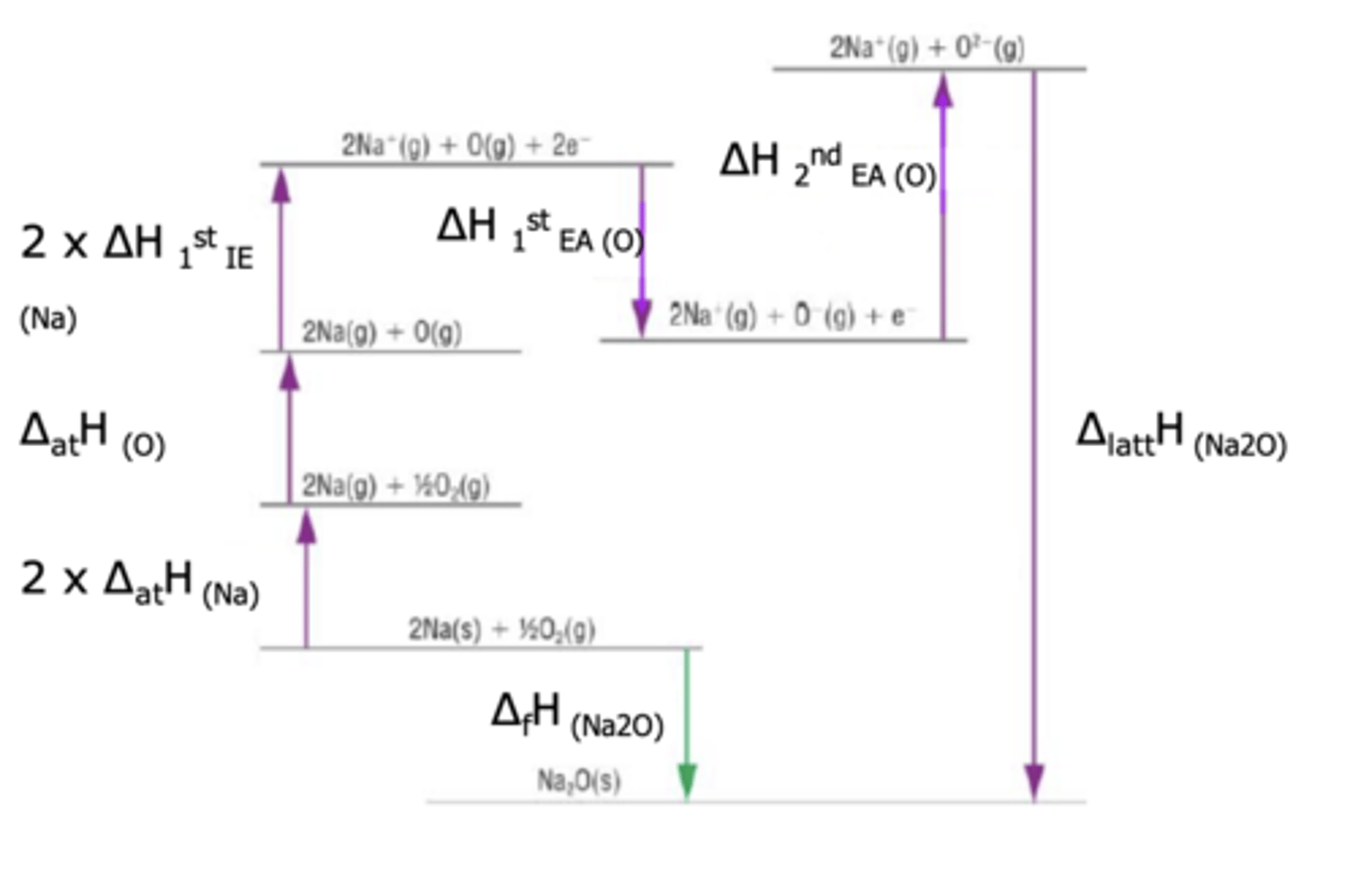
Draw born-haber cycle for Na2O(s) (6)
(ignore numbers)
What two factors affect lattice enthalpy? (2)
- Charge on ions (larger = greater enthalpy)
- Ionic radius (Smaller = greater enthalpy)
Describe and explain trend in lattice formation / dissociation enthalpies across a group (2)
- Lattice dissociation / formation enthalpy decreases
- Size of ion increases
- Larger ions have a weaker electrostatic attraction between oppositely charged ions
What does the perfect ionic model predict? (1)
- All ions are perfect spheres with purely ionic attraction
- Used by theoretical (data-book) values
What does it mean if experimental lattice enthalpy is different from theoretical value?
Due to presence of covalent character
Is it possible for experimental born-haber value to be less than the theoretical?
- No
- It is either the same (purely ionic) or greater than (due to covalent character)
What are two factors that contribute towards the presence of covalent character? (2)
- Size of the ion
- Charge on the ion
What is theoretical lattice enthalpy? (2)
- Calculated using the Perfect Ionic Model.
- Assumes that ions are perfect spheres
What is experimental lattice enthalpy? (2)
- Calculated from a Born-Haber Cycle.
- Shows that covalent character makes the bond stronger.
What is the equation for enthalpy of solution (ΔHₛₒₗ)? (1)
(Enthalpy of lattice dissociation) + (sum of enthalpy of hydration)
What is entropy? (1)
A measure of disorder in a system
What is a reaction considered to be if there is an increase in disorder? (1)
Feasible / spontaneous
What are the two features that occur in a reaction that affect entropy? (2)
- Change of state from reactants to products
- Increase in moles from reactants to products
What is the sign of entropy if an increase in moles from reactants to products happens? (1)
Positive
What is the sign of entropy if an decrease in moles from reactants to products happens? (1)
Negative
What does a more positive value of entropy (ΔS) indicate? (2)
- The system is more random or disordered.
- The reaction is more feasible.
What conditions are entropy values measured at? (3)
- Standard conditions
- 298K
- 100 KPa
What is the formula and units for entropy change? (2)

What is the formula and units for Gibbs free energy?
T = Temperature (K)

What must be done to the units of ΔS for Gibbs free energy calculations? (1)
Convert ΔS from J K⁻¹ mol⁻¹ to kJ K⁻¹ mol⁻¹ by dividing by 1000.
When is a reaction feasible according to ΔG°? (1)
A reaction is feasible when ΔG° is less than or equal to 0
What does a more negative ΔG° value indicate? (1)
The reaction is more feasible
When asked to work out temperature at which a reaction:
- Becomes feasible
- Is no longer feasible
- Is at equilibrium
What do you do? (3)
- Make ∆G = 0
- Rearrange for T: ΔH / ΔS
What determines if a reaction is feasible based on ΔG? (2)
- If TΔS>ΔH, ΔG is negative, so the reaction is feasible.
- If TΔS<ΔH, ΔG is positive, so the reaction is not feasible