Addition polymers (3.3.4.3)
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
What are addition polymers produced from?
Addition polymers are produced from alkenes when the pi bond is broken to form a repeating unit.
How do you draw a repeating units and unit of an addition polymer from its corresponding alkene?
1) Break the carbon-carbon double bond
2) Add extended bonds either side
3) Add square brackets so the bonds go through it
4) Add an n before the alkene and to the bottom right of the square brackets
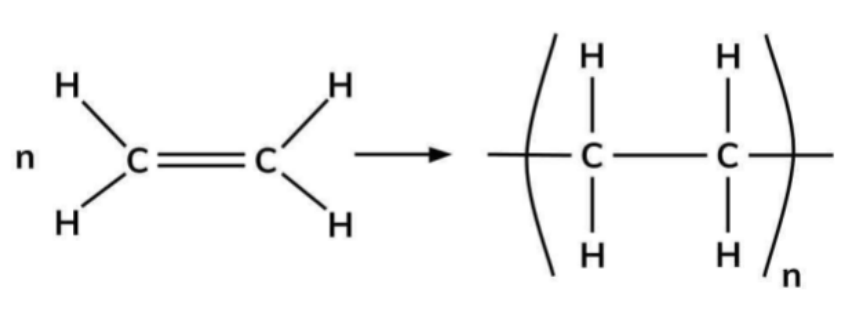
To draw a single repeat unit, remove the brackets and the n
How do you name addition polymers?
Put brackets around the alkene name and put poly before it. e.g. ethene → poly(ethene)
What are the uses of addition polymers and why are these the uses?
They are used to manufacture everyday plastic products like shopping bags.
This is because they are unreactive hydrocarbon chains which consist of many non-polar covalent bonds
This also means they aren’t biodegradable.
How does changing the conditions of the reaction influence the properties of addition polymers?
A high pressure and temperature - branched polymers with relatively weak intermolecular forces - pack less densely so are more flexible
A low pressure and temperature - straight chain polymers with relatively strong intermolecular forces - pack more closely together so are strong and rigid
What are plasticisers and how do they work?
Plasticisers can be used to make straight chain polymers more flexible.
They do this by sticking themselves between the molecules and keep the polymers further apart, weakening the LDFs and let the chains slide more easily
What are the uses of PVC and how can its properties be modified?
PVC (poly(vinylchloride)) now known as poly(chloroethene) is used for rigid plastics like drain pipes.
If you add a plasticiser you can make a more flexible PVC material that can be used in flooring tiles