Biochem 1B Structural Characterisatin
1/88
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
89 Terms
what is elemental analysis
EA determines the percentage of carbon, hydrogen, nitrogen, and sometimes sulfur in a compound by combusting it and analyzing the gases produced.
Provides the empirical formula of the compound.
what is empirical formula
The empirical formula gives the ratio of the elements in the structure
what is molecular formula
The molecular formula gives the actual number of atoms in each molecule
How to calculate empirical formula
● Divide the amount of each element by its molar mass
● Divide the answers by the smallest value obtained
● If there is a decimal, divide by a suitable number to make it into a whole number
double bond equivalence (Also known as the Index of Hydrogen Deficiency (IHD)
DBE helps determine the number of double bonds or rings in a molecule.
DBE = 1: one double bond or ring
DBE = 2: two double bonds or rings(or one triple bond)
how to calculate DBE
Hreference is the number of hydrogens required to make CnH2n+2 i.e. the saturated alkane.
Hmolecule is the number of hydrogen atoms present
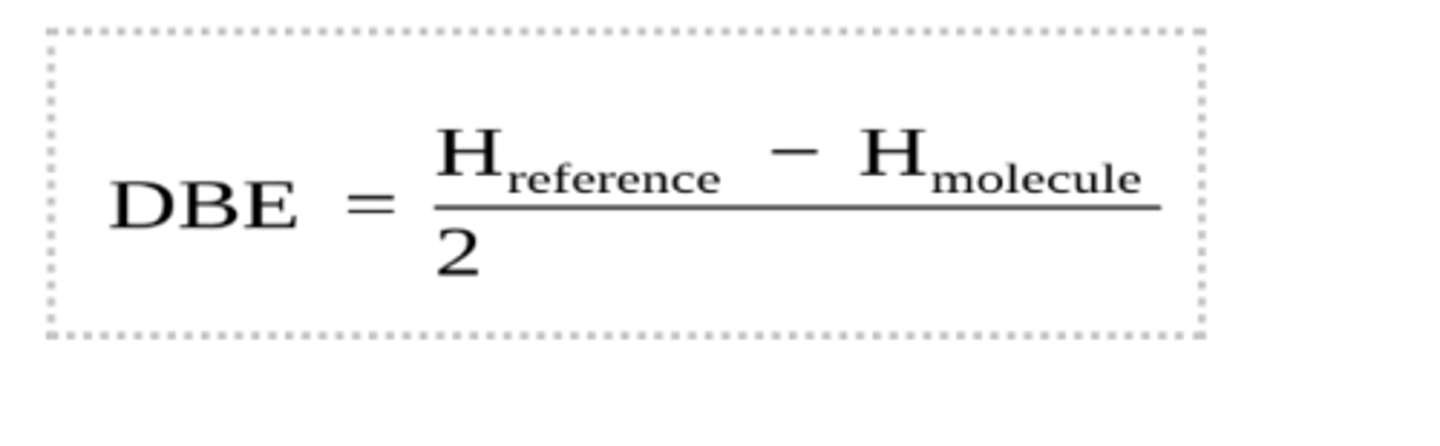
how to calculate DBE for molecules with heteroatoms
Oxygen can be ignored
Subtract one H from Href for every halogen
Add one H to Href for every nitrogen
what does mass spectrometry provide
Mass spectrometry provides the mass of a molecule, distinguishing between empirical and molecular formulas.
e.g. peak at 44 tells us the molecule has a mass of 44 a.m.u
key processes in mass spec
Ionization of molecules, acceleration, and magnetic separation are key processes in mass spectrometry.
mass spectrum of pentane
m/z is mass over charge, charge is usually 1
consider what fragments mass are at 43
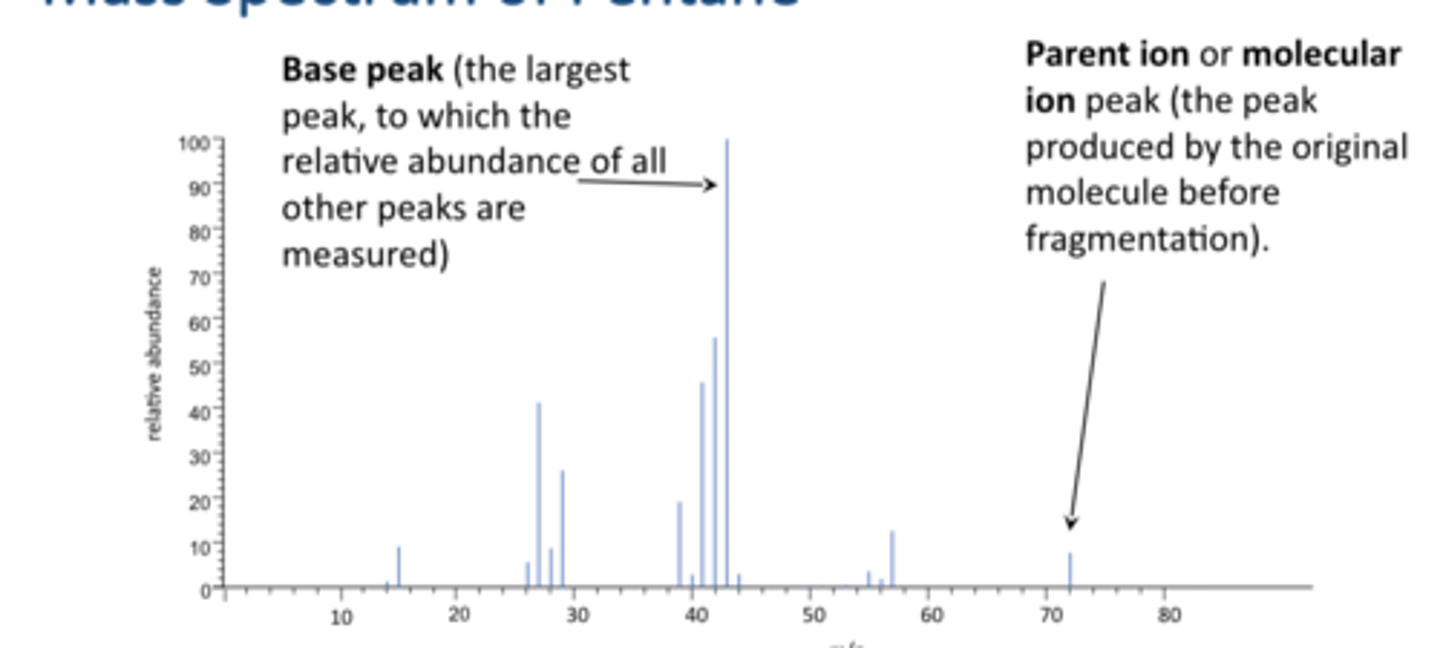
what is the base peak
Base peak (the largest peak, to which the relative abundance of all other peaks are measured)
exact mass determination
Exact mass measurement to four decimal places aids in distinguishing between molecules with the same relative atomic mass (r.a.m).
Accurate mass calculation involves considering the most common isotope of each element.
what is the parent ion peak
Parent ion or molecular ion peak (the peak produced by the original molecule before fragmentation)
can tell us what the mass our molecule was to starat with
how to calculate exact mass
using the mass of the most common isotope
adding the exact masses of the most abundant isotopes of the constituent elements
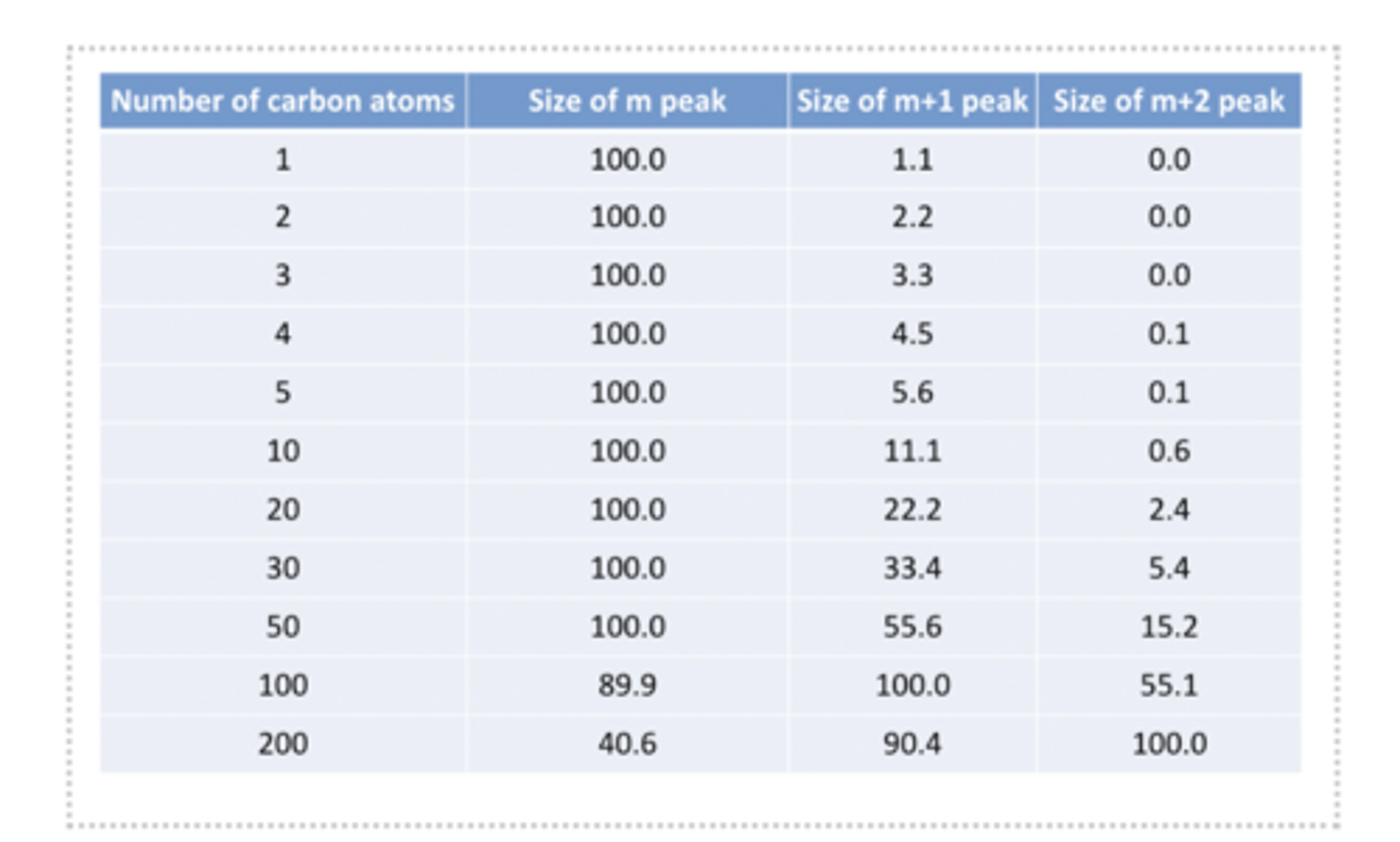
what is the isotope effect
Ratio of peaks in mass spectrometry helps determine the number of carbon atoms present in a molecule.
Can observe the size of the m peaks to tell us this
what does fragmentation provide
fragments can tell you about what you've got in your molecule.
Protein Mass Spectrometry
Involves analyzing amino acid chains, such as alanine attached to cysteine, and identifying fragments between specific functional groups.
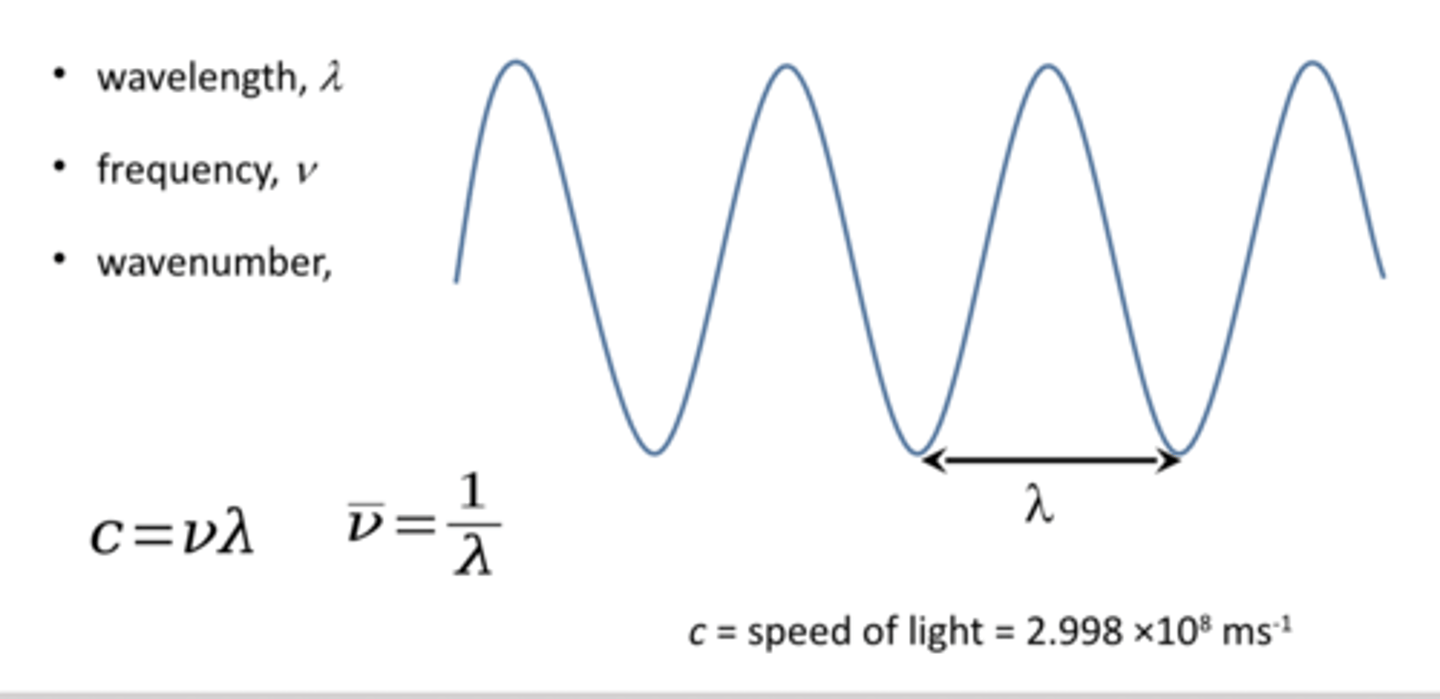
what is electromagnetic radiation
Electromagnetic radiation is a wave consisting of orthogonal electric and magnetic fields that can travel through space
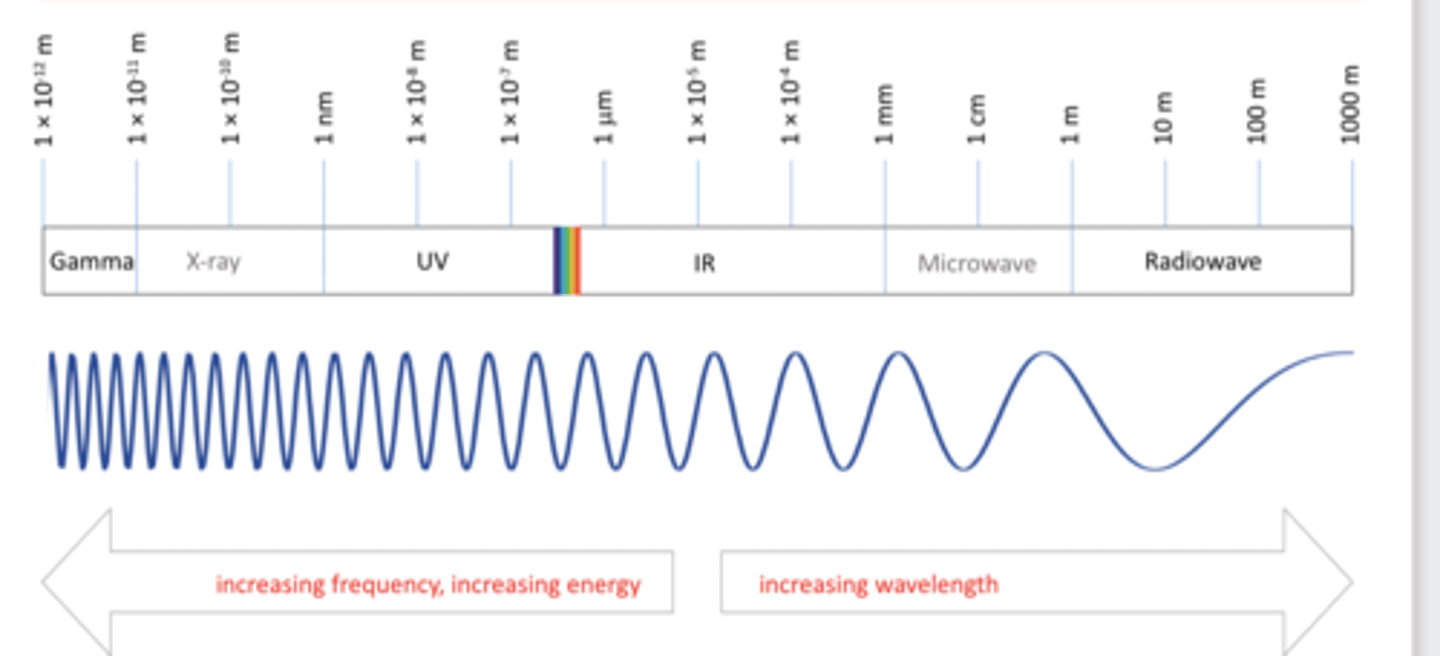
electromagnetic spectrum
defining waves
wave-particle diality
Electromagnetic radiation can be viewed as both a wave and a small packet of energy.
The energy of a photon is directly related to its frequency.
Energy, frequency, and wavenumber are directly proportional to each other.
how can we Convert energies of photons into wavelengths, wave numbers and Hertz
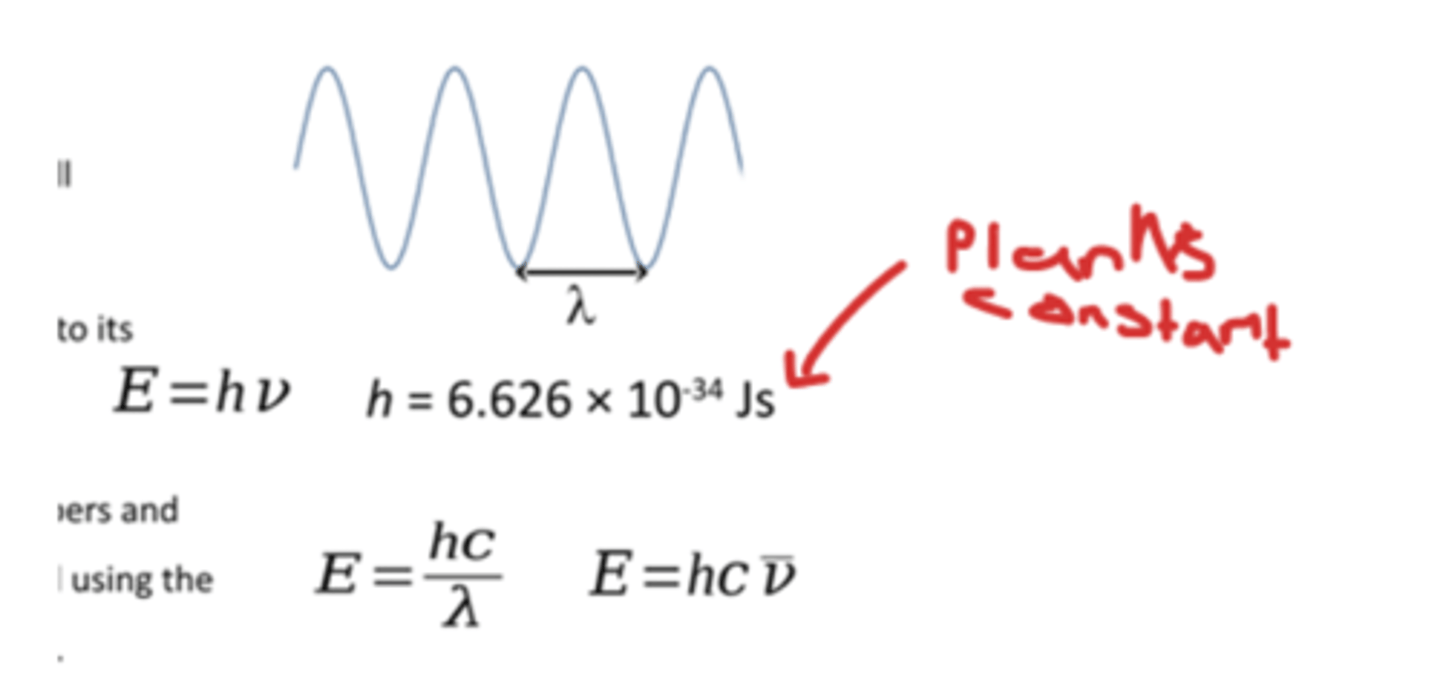
quantisation of energy levels
A molecule has a particular energy associated with it but its not just the energy of that molecule itself, there are electronic, vibrational and rotational energy levels and all of these are quantised.
Electronic, vibrational and rotational energy levels in molecules are quantised
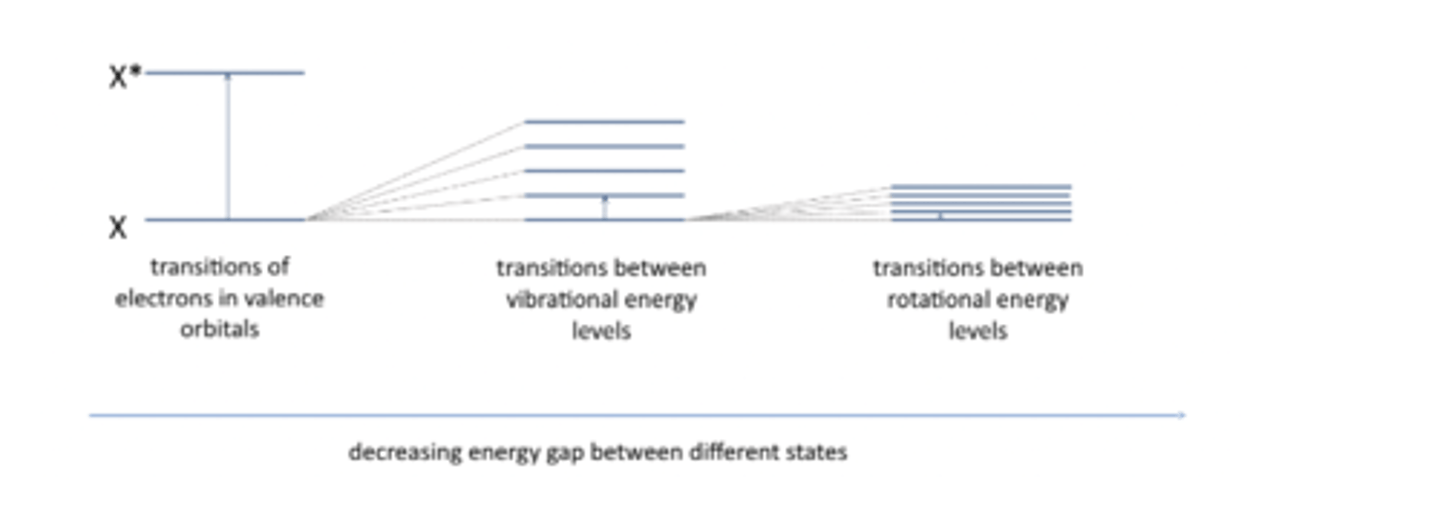
spectroscopy
Spectroscopy is all about the absorption of light
what do spectrometers measure
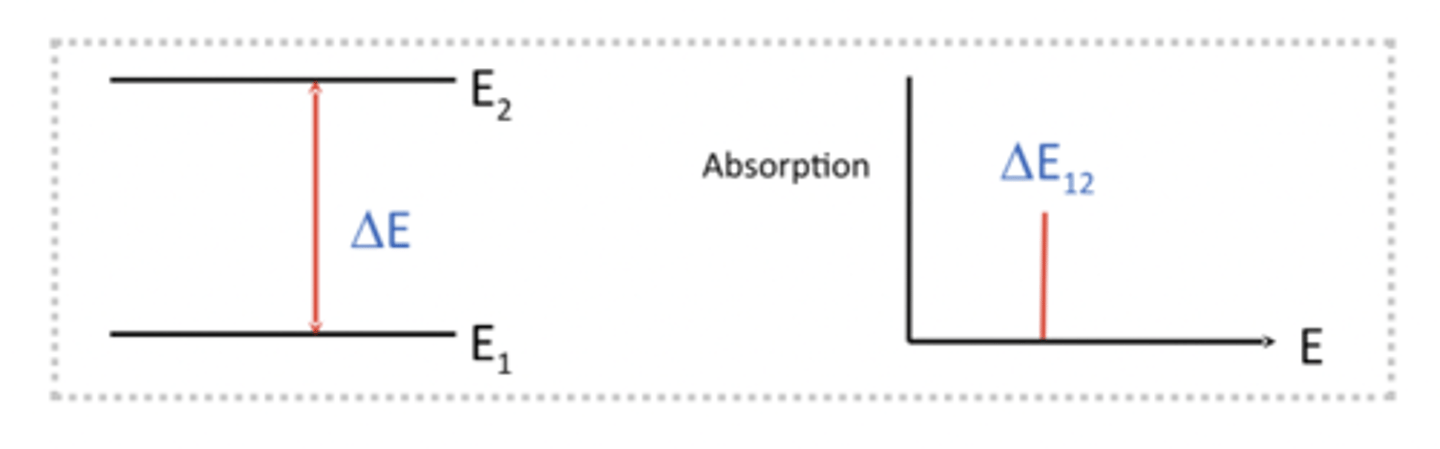
Spetrometer measure the difference in energy levels between two particular states (energy gap)
Spectrometers measure the absorption (or transmission) as a function of the frequency
what does selective absorption result in
Selective absorption results in line spectra
Only photons with the exact energy as the gap will be absorbed
So if we fire lots of photons with diff wavelengths at a sample/moelcule only specific ones with specific frequencies, energies, wavenumbers, etc will be absorbed and the rest will not interact with the molecule whatsoever. Means we will see a peak in our spectrum as the moelcule absorbs that. Known as selective absorption
spectroscopic techniques
Gamma, X-ray, UV-Vis, IR, Microwave and NMR spectroscopy
Gamma spectroscopy
Gamma spectroscopy provides information about nucle
X-ray spectroscopy
X-ray spectroscopy identifies atoms in molecules.
UV-Vis spectroscopy
UV-Vis spectroscopy involves transitions in valence orbitals.
Can promote electrons from one orbital to another. Energy difference in UV visible radiation
IR spectroscopy
IR spectroscopy corresponds to vibrational energy levels.
Microwave spectroscopy
Microwave spectroscopy deals with rotational energy levels.
NMR spectroscopy
NMR spectroscopy focuses on energy differences in nuclear spin states.
how can light interact with molecules
Light can interact
with molecules in different ways, exciting electrons, vibrations or nuclear spin.
what happens i UV-Vis when p orbitals overlap
Overlap of p orbitals in molecules results in sigma (head on) and pi (side on) bonds.
We also getπ antibonding orbitals (don't overlap)
Energy levels in molecules with more pi bonds are more likely to absorb UV/visible radiation.
what happens when a photon is the UV or visible range is absorbed
Promotion of electrons from bonding to antibonding orbitals occurs in UV or visible range
The electrons are those associated with double bonds, which are p-bonds.
We say the electron has undergone a π to π*
transition (π* means antibody orbital).
energy gaps between sigma and sigma* orbitals
Energy gap between sigma and sigma* orbitals is not in the UV/visible region.The gap ΔE is too big and the associated wavelength is less than 200 nm
energy gaps in π orbitals
So if we have lots of π orbitals then this gap between the filled π orbitals and the empty π orbitals(which our electrons are going to get promoted into when it absorbs radiation) is going to narrow down
Molecules with more pi orbitals tend to be colored due to lower energy gaps.
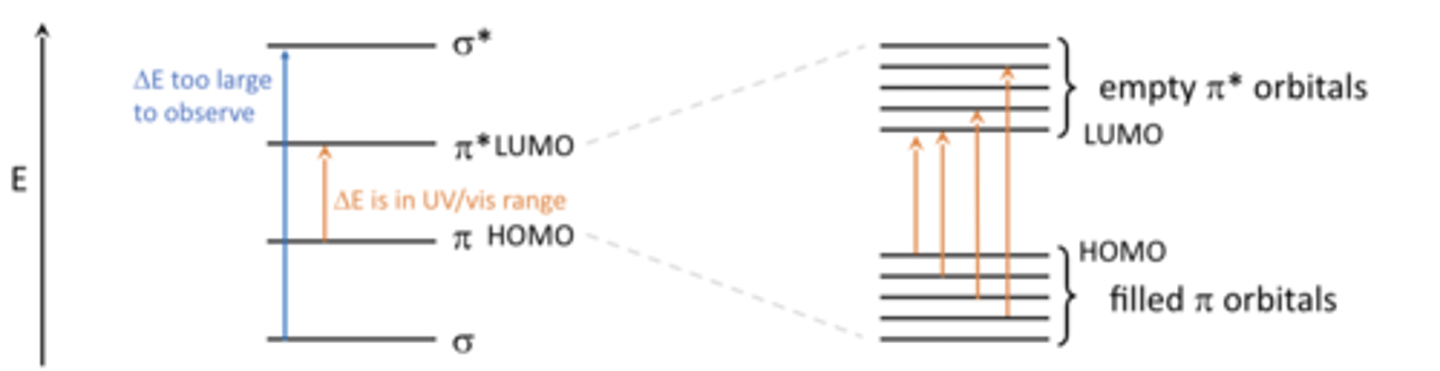
how do you know if a molecule has a π orbital
Molecules that contain double bonds have π / π * molecular orbitals
Single bonds have sigma molecules
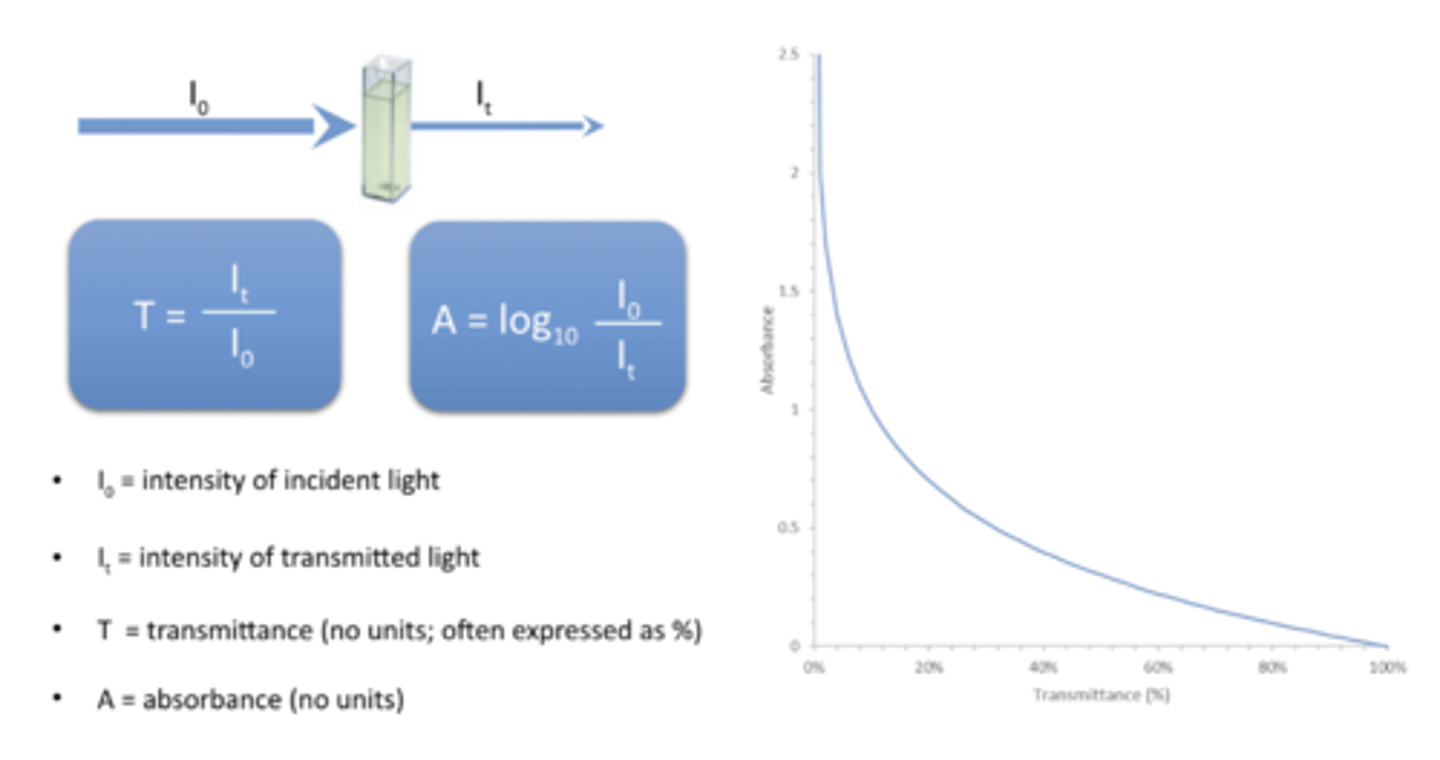
absorbance proportional to
Absorbance is directly proportional to concentration.
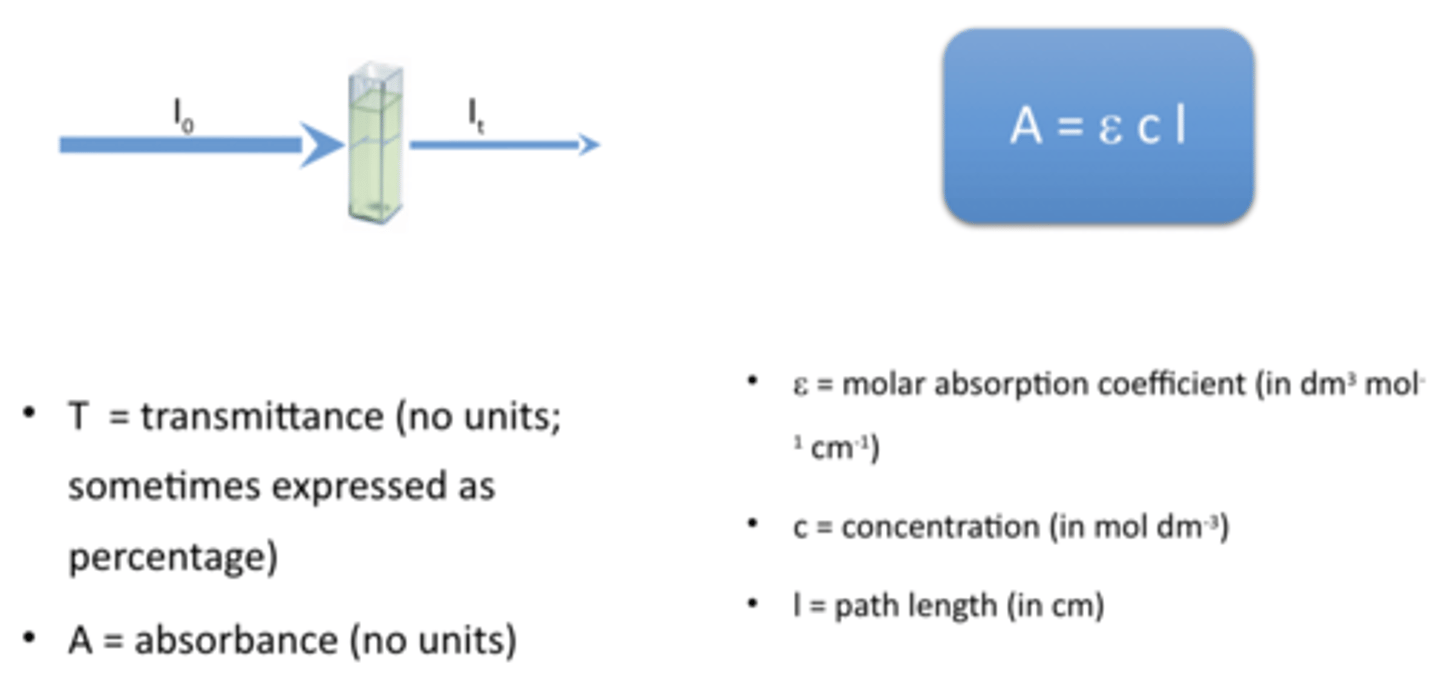
what does the beer lambert law do / calculation
Beer Lambert Law relates absorbance to concentration.
what else affects the energy gap
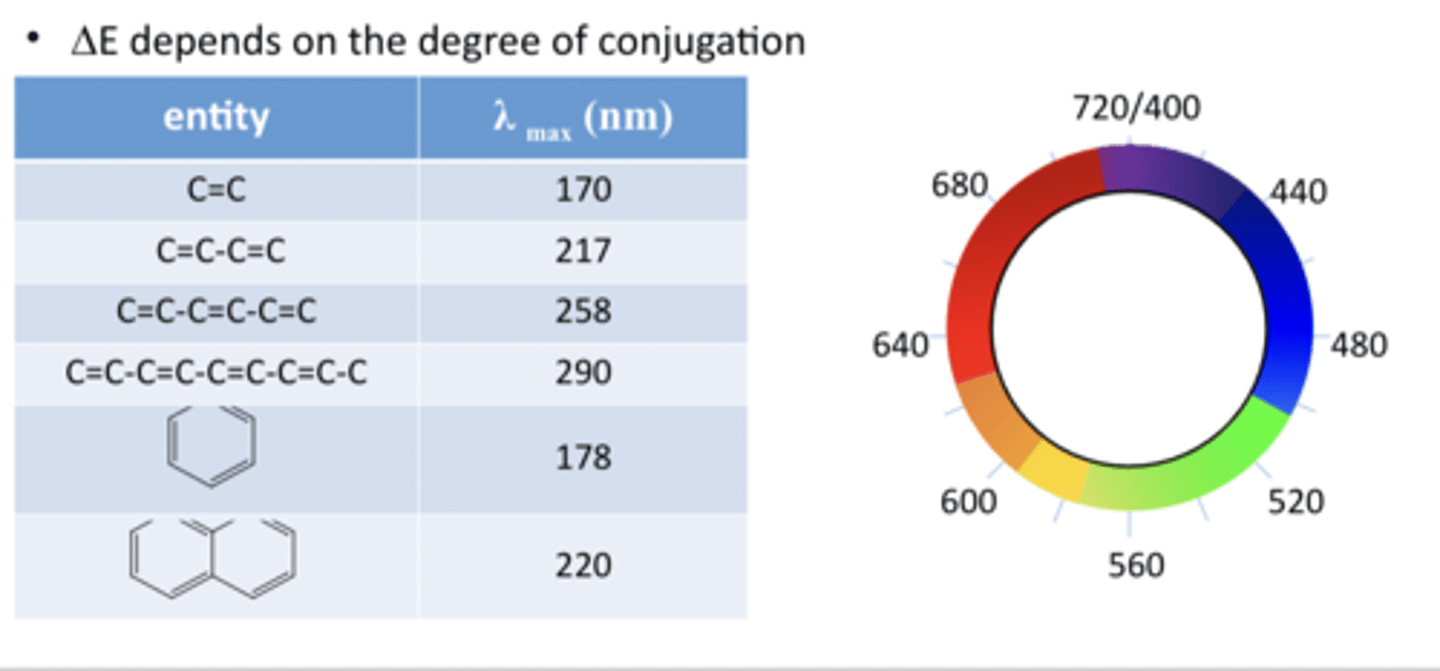
Degree of conjugation affects the energy gap.
More conjugation shifts absorption towards the visible spectrum.
Higher conjugation results in absorption of longer wavelengths
IR
when does a dipole movement occur
A dipole moment occurs when there is a separation of charge. Happens when an atom is bonded to another atom that has a diff electronegativity to it.
when is the dipole moment larger
The dipole moment is larger when:
- the difference in electronegativity increases
- the separation of the atoms decreases
what must happen for IR activity
To be IR active, the dipole moment must change during the vibration. If it doesnt change we dont see an IR peak in the spectrum
what is IR spectroscopy used for
IR spectroscopy is used to identify the presence or absence of functional groups in amolecule
what do IR photons do
Photons in the IR spectrum have lower energy compared to UV and visible light.
IR photons excite vibrations in molecules, causing larger amplitude movements instead of electronic transitions.
Molecular vibrations occur at fixed frequencies based on atom weight and bond strength.
what is the frequency of vibrational absorption affected by
frequency of the vibrational absorption is affected by the mass of the atoms and the stiffness of the bond
atoms relationship with vibrational frequency
As atoms get bigger your value for reduced mass will increase so will get a small stretching frequency. So the largest stretching frequencies will be seen in things with very small masses
Light atoms will give us the highest vibrational frequency ,m eg. Hydrogen bonded to first row of periodic table
common peaks of IR spectrca
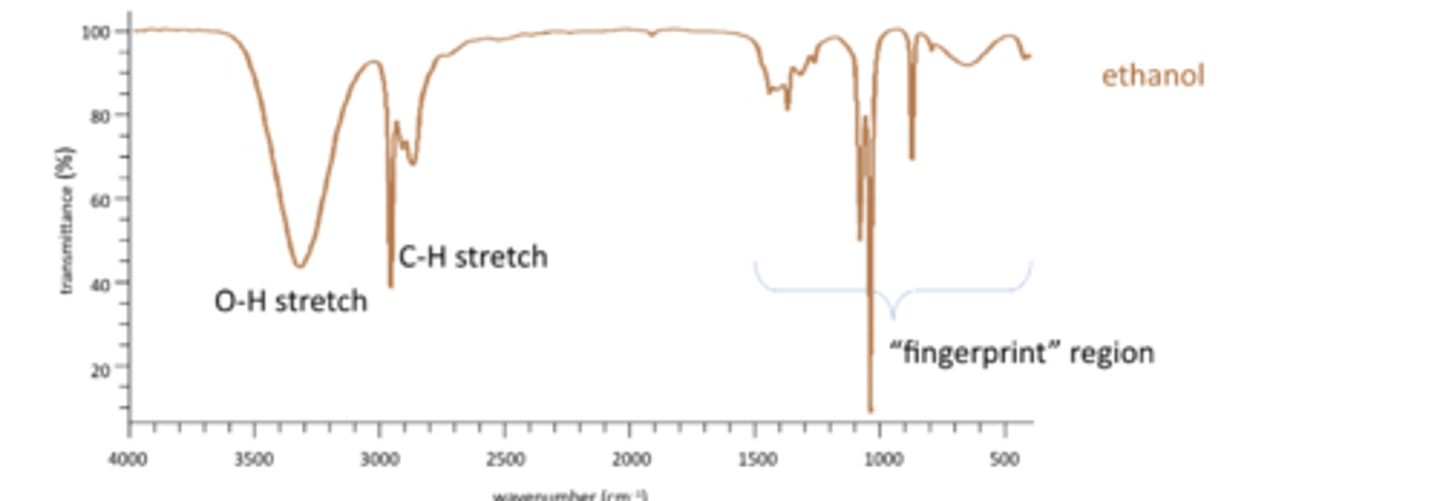
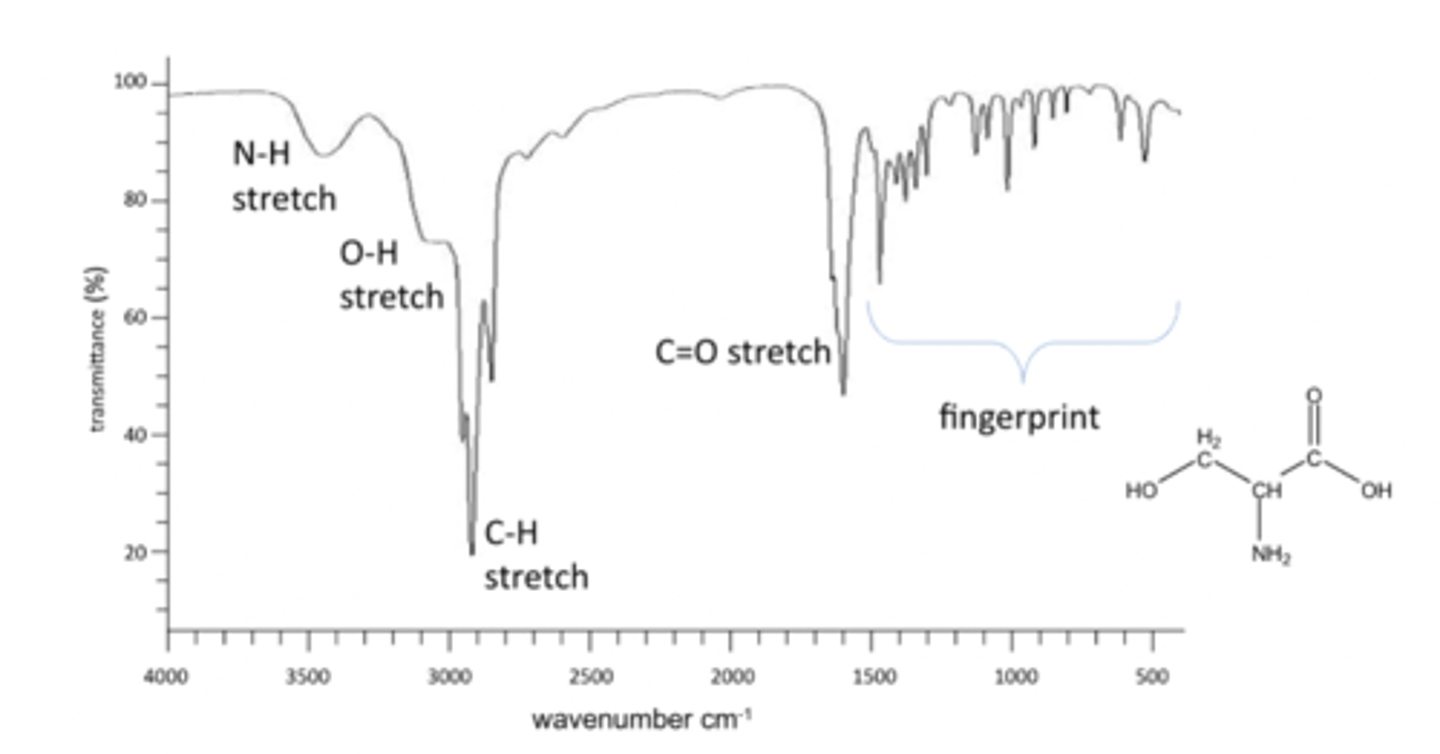
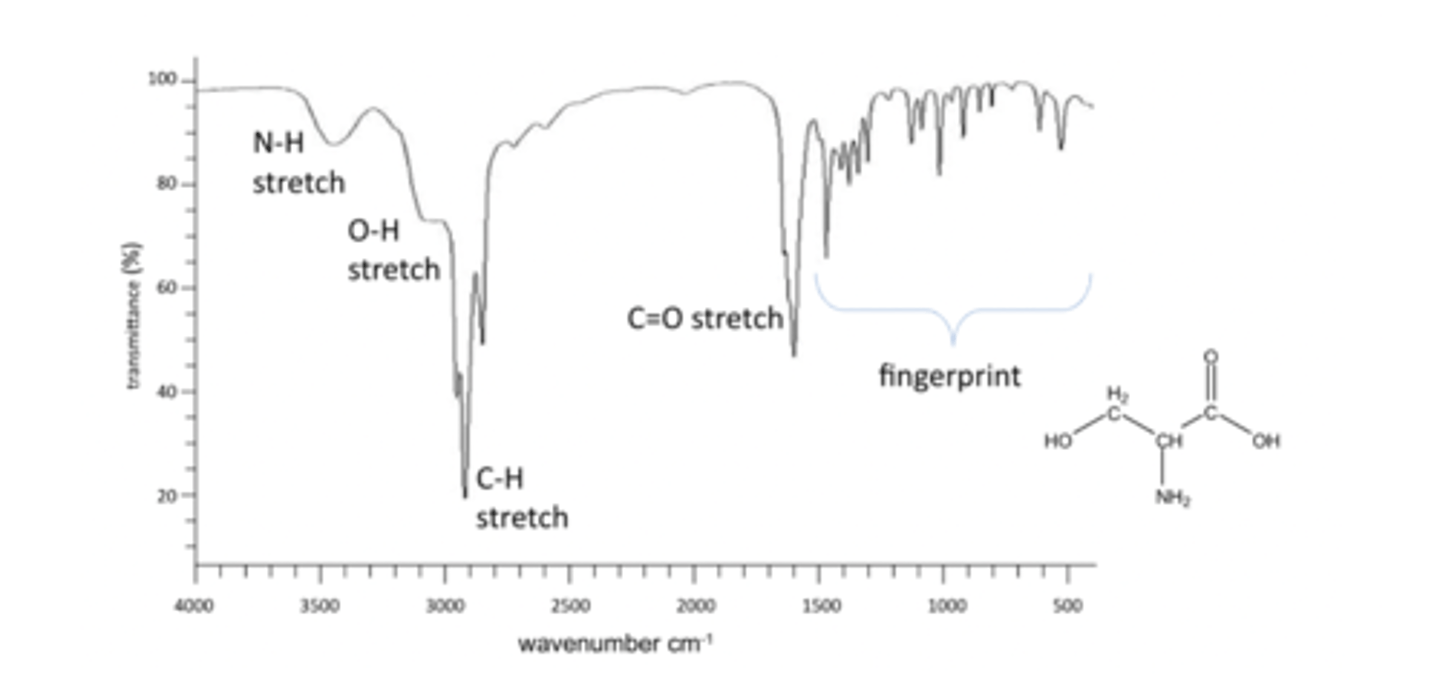
Common peaks include C-H stretch, O-H stretch, N-H bend, C=O stretch, N-H sretchand 'fingerprint' region.
reading an IR spectra
IR spectra are always plotted as % transmittance vs. Frequency (wavenumber) in cm-1
Highest energy are on the LHS, lowest energy on RHS
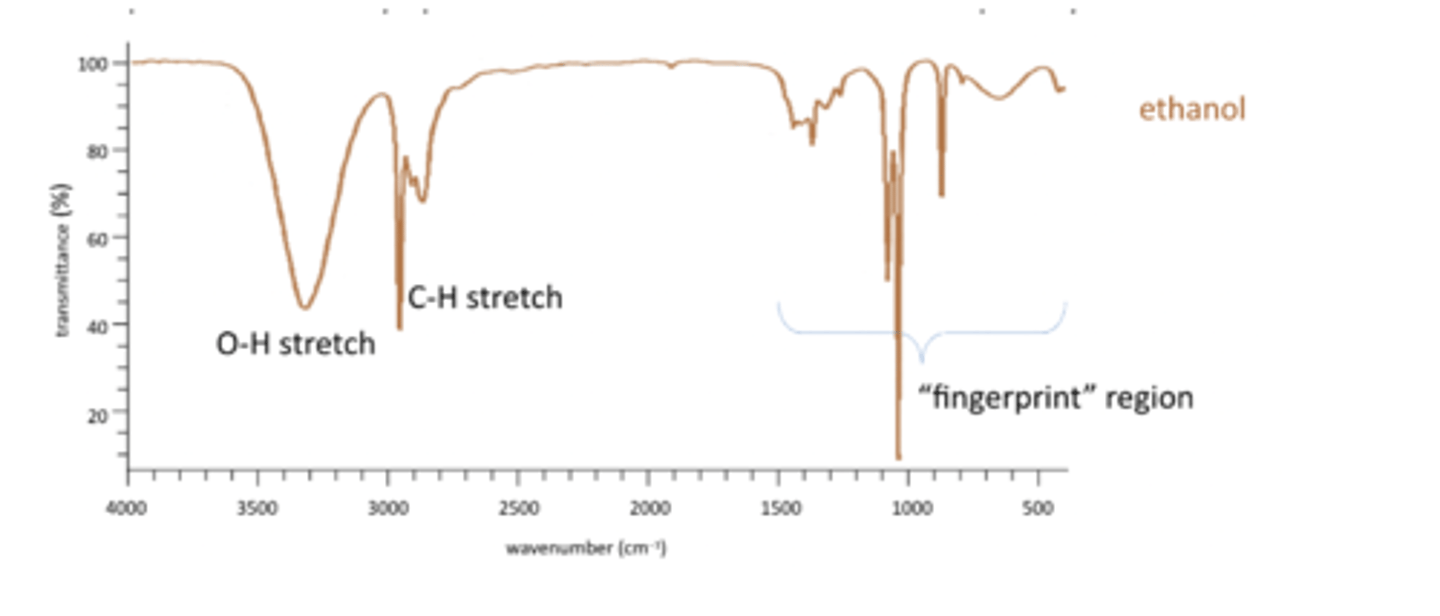
O-H stretch and C-H stretch
Has an O-H stretch, remember bonds to hydrogen produce the highest frequencies. SO O-H and C-H stretches are on the left. Broad peak for O-H as it is involved in hydrogen bonding.
O-H 3500-2300 range
C-H 3300-2700 range
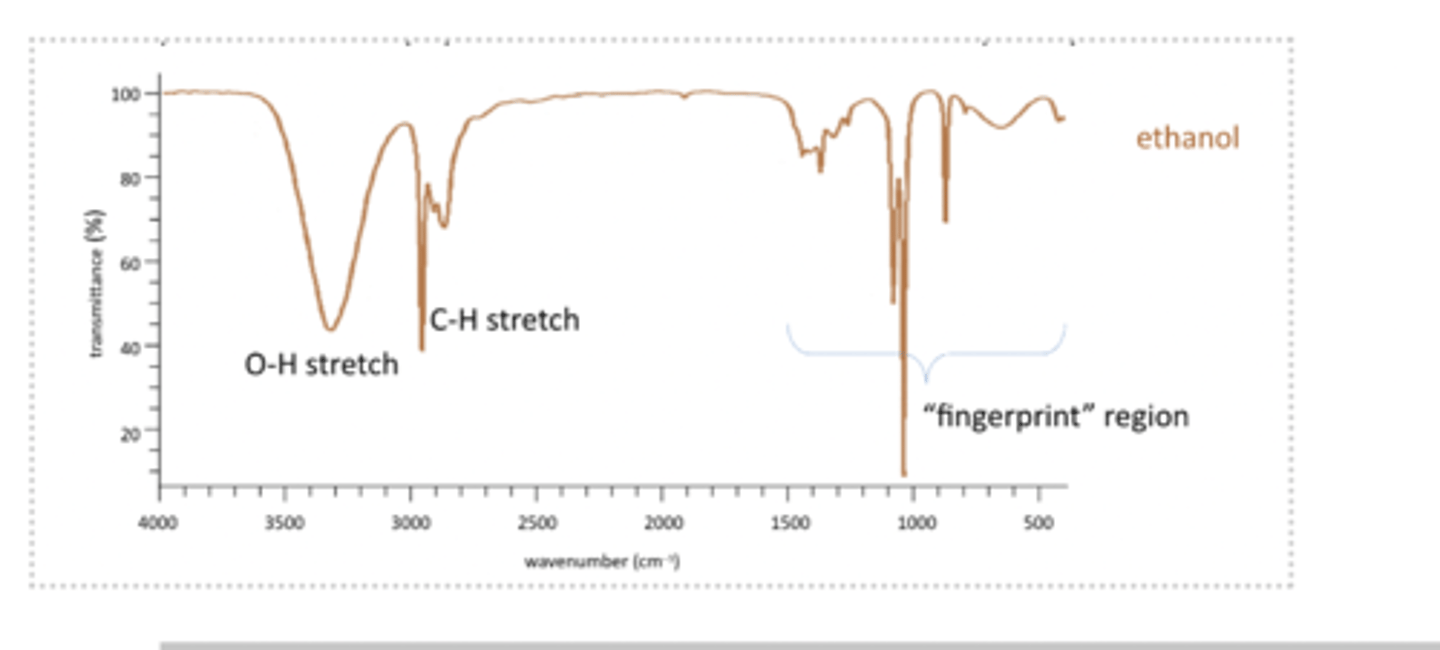
fingerprint region
Fingerprint region peaks are caused by skeletal vibrations. Cant use it for distinguishing
what about a C=O stretch
1800-1650
C=O has a double bond, a C-O single bond would be in the fingerprint region but this is C=O is a distinctive peak.x4
N-H Bend
3400-3200
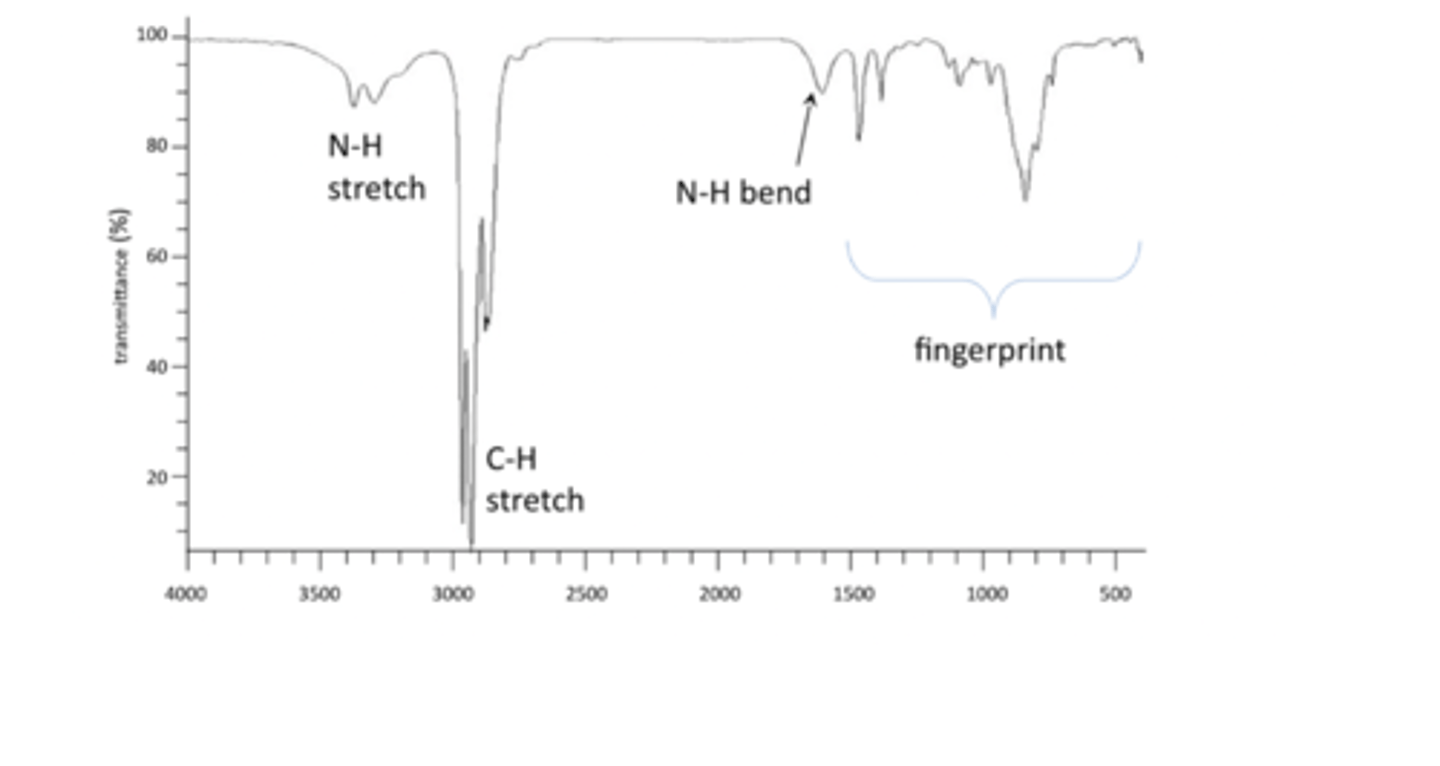
IR of an amine
IR of L-serine
NMR
what is NMR used for
NMR spectroscopy is often used to obtain information on the structure of the carbon-hydrogen framework of the molecules. It also can be used to identifyfunctional groups
Radio waves are utilized in NMR spectroscopy.
what does NMR spectroscopy use
NMR spectroscopy uses
radio waves and signals arise from
transitions between spins states of
nuclei
what gives rise to NMR spectra
Nuclei with either an odd mass number or an odd atomic number have the property ofnuclear spin
1H and 13C both give rise to NMRspectra as they both have a nuclear spin of 1/2
nuclei in a magnetic fiekd
Nuclei align either parallel or anti-parallel to the magnetic field (Bo).
Alignment with the field results in slightly lower energy
increasing the magnetic field
Increasing the magnetic field leads to a larger energy gap between parallel and anti-parallel alignments.
The energy gap (∆E) increases with a larger magnetic field
𝜈 = E/h
what is an NMR spectromerter
An NMR spectrometer is simply a big magnet with a radiofrequency generator, a radiofrequency detector, a computer and a sample tube.
∆𝐸 Dependency
strength of the magnetic field, B0
magnetogyric ratio (or gyromagnetic ratio) - specific to a particular typeof nucleus, e.g. same for all 1H nuclei
degree of shielding
what is degree of shielding
Electrons within atoms have the property of spin and therefore they alsoact as mini-magnets. This local magnetic field opposes the external applied field and therefore:
Bexperienced < Bo
The more electrons you have, the smaller the Bexperienced will be as your nuclei is more shielded.
what does shielding allow us t do
Allows us to distinguish between nuclei in different environments.
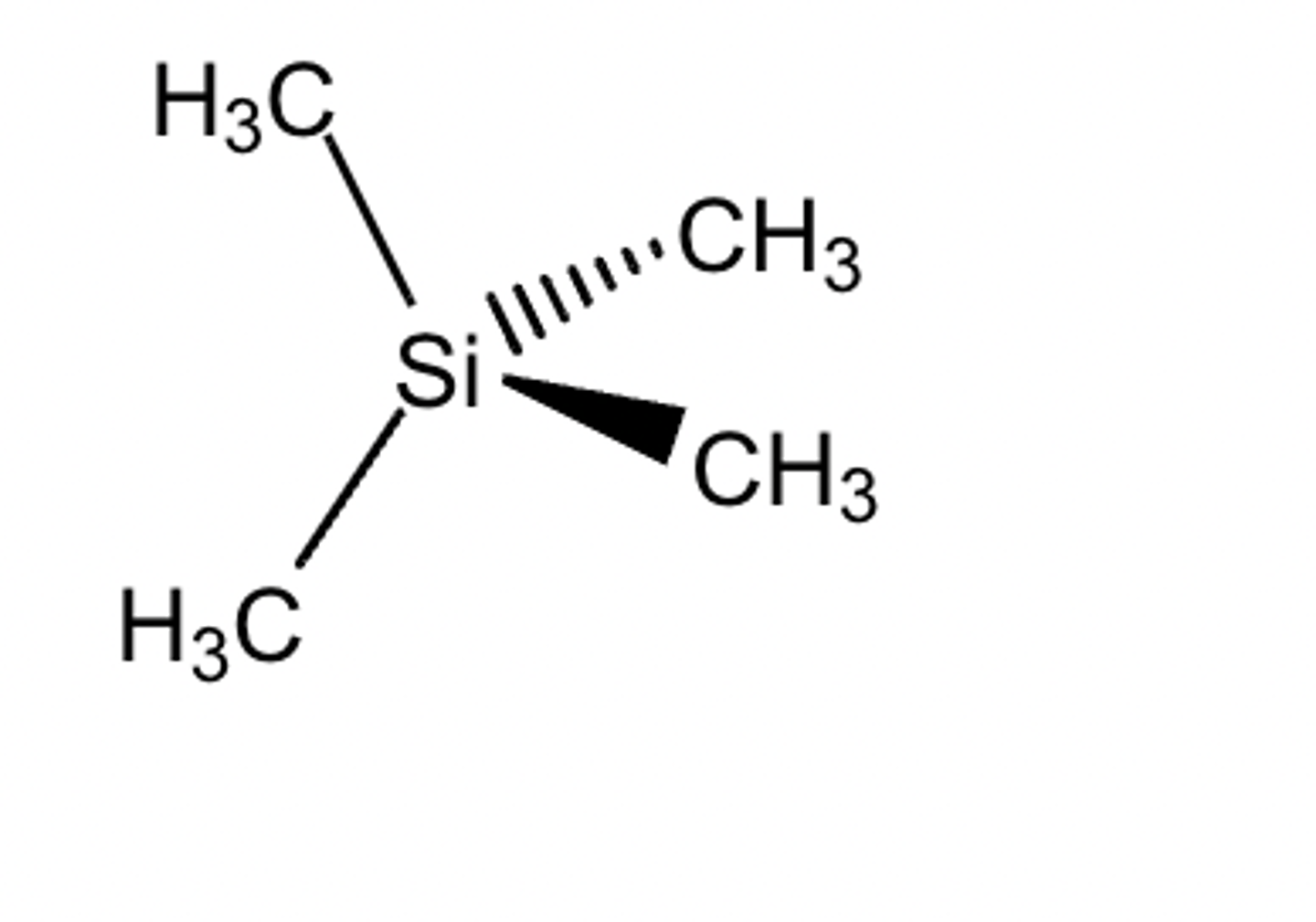
reference signal for 13C and 1H NMR
The reference material for proton and carbon-13 NMR is tetramethylsilane(TMS). The signal for TMS is fixed at 0 ppm
what is chemical shift
The difference in resonance frequency (Dn) is small but measurable and isusually quoted as the chemical shift (d)
remember to tun MHz into Hz by 10^6

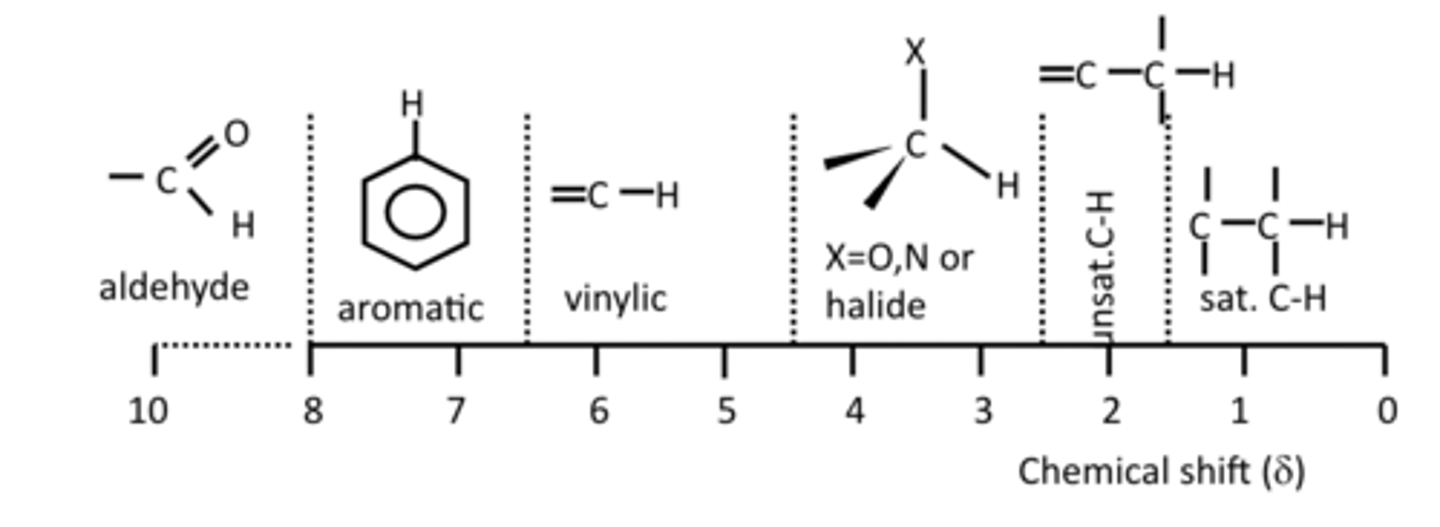
Characteristic 1H chemical shifts
More Deshielded will resonate at a higher frequency
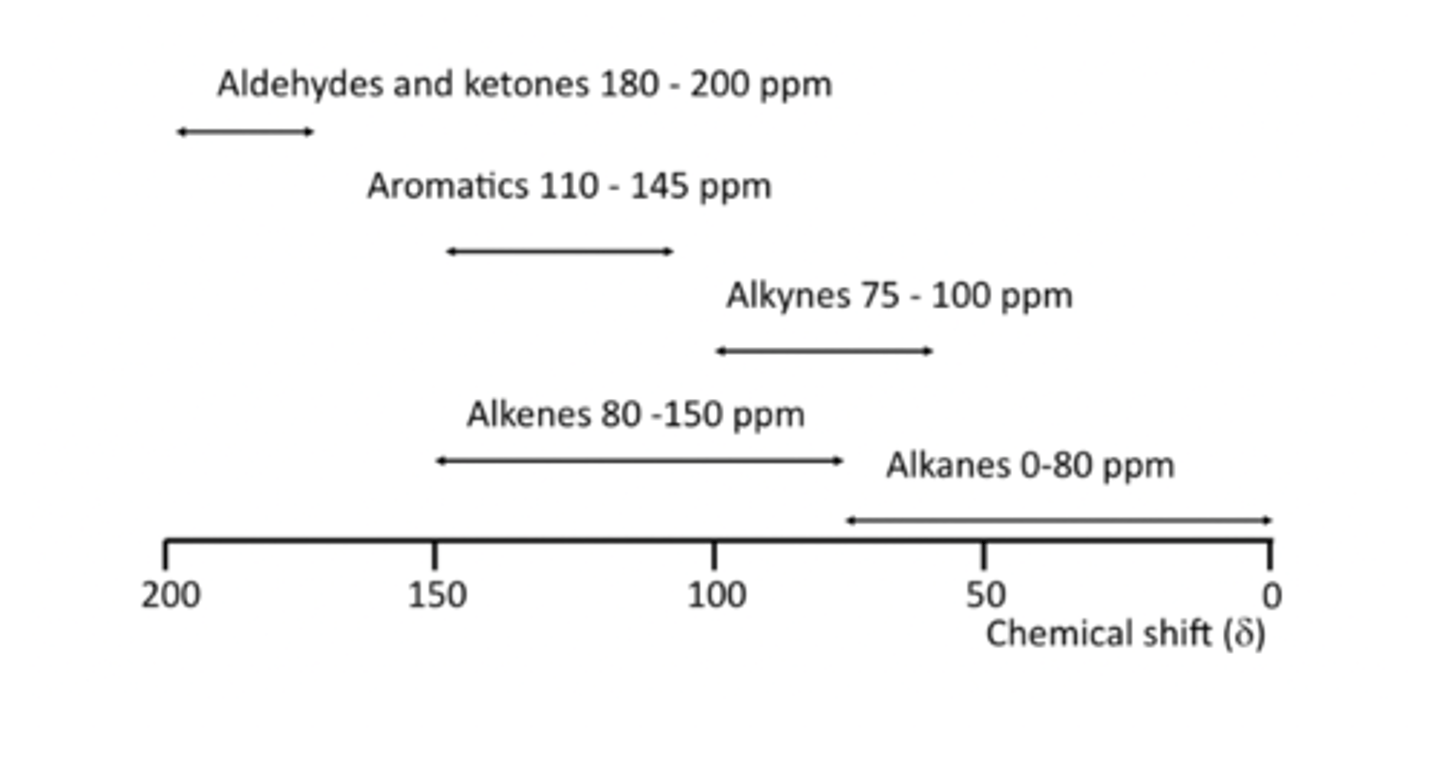
Characteristic 13C chemical shifts
what does NMR tell us using chemical shifts
Protons in different environments exhibit different chemical shifts.
Number of unique peaks in NMR spectra indicates different chemical environments for H or C.
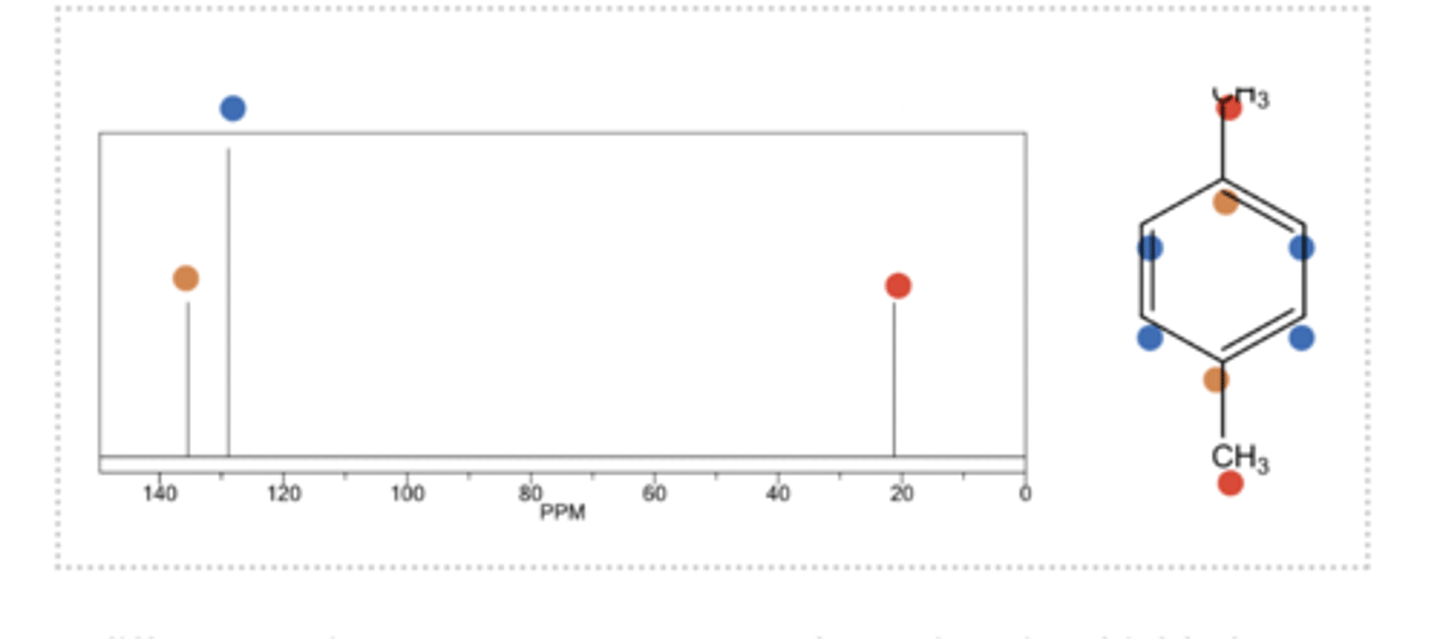
example: 13C NMR of para-xylene
3 different environments so we get 3 peaks. 20 is quite shielded so we can assume it is CH3, the other two are in aromatic environment but one is twice as site (likely to have twice the number of carbons)
what if two peaks in NMR are extremely close
they are similiar but different environemtn
what is a unique environment
an atom that is bonded to different atoms or groups compared to other atoms in the molecule
what does a proton have
va proton has a
magnetic spin field
what is the signal intensity (integral) proportional to
the number of protons.
signal splitting
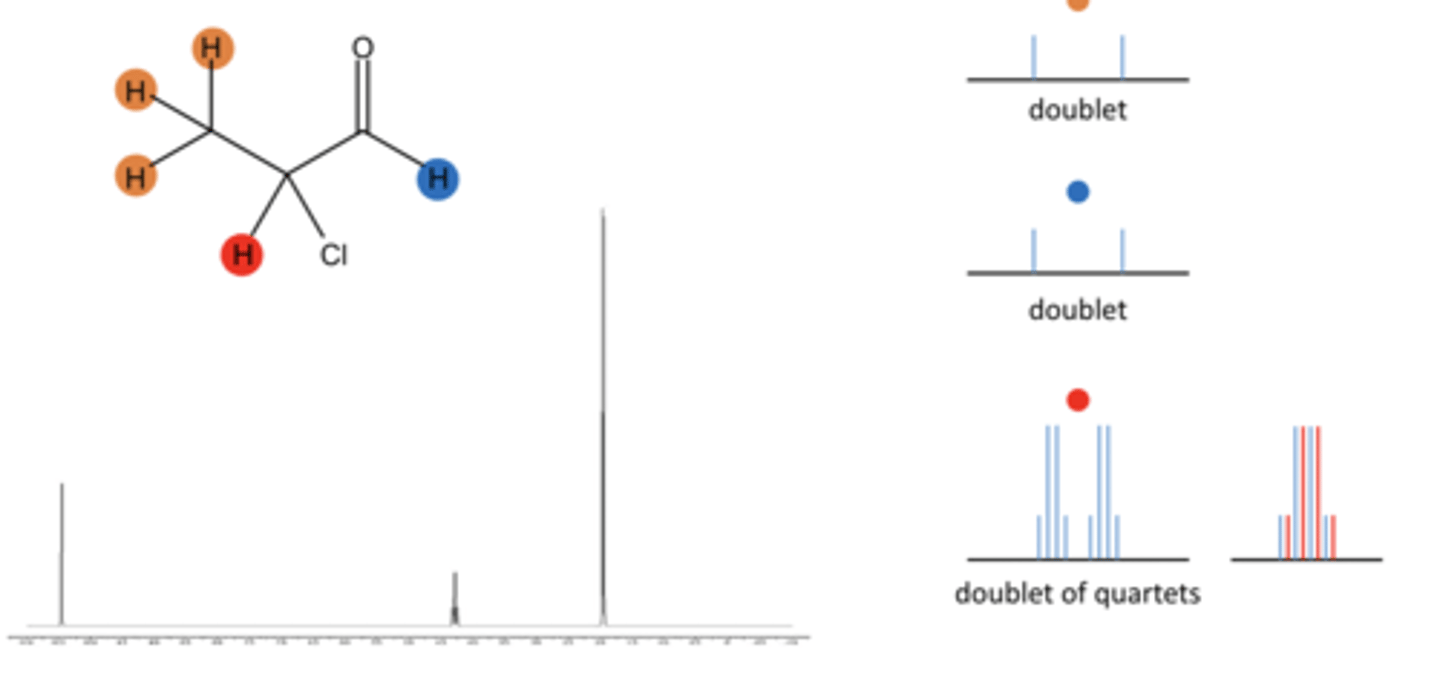
Signal splitting can occur when H atoms on adjacent carbon atoms affect each other's magnetic fields.
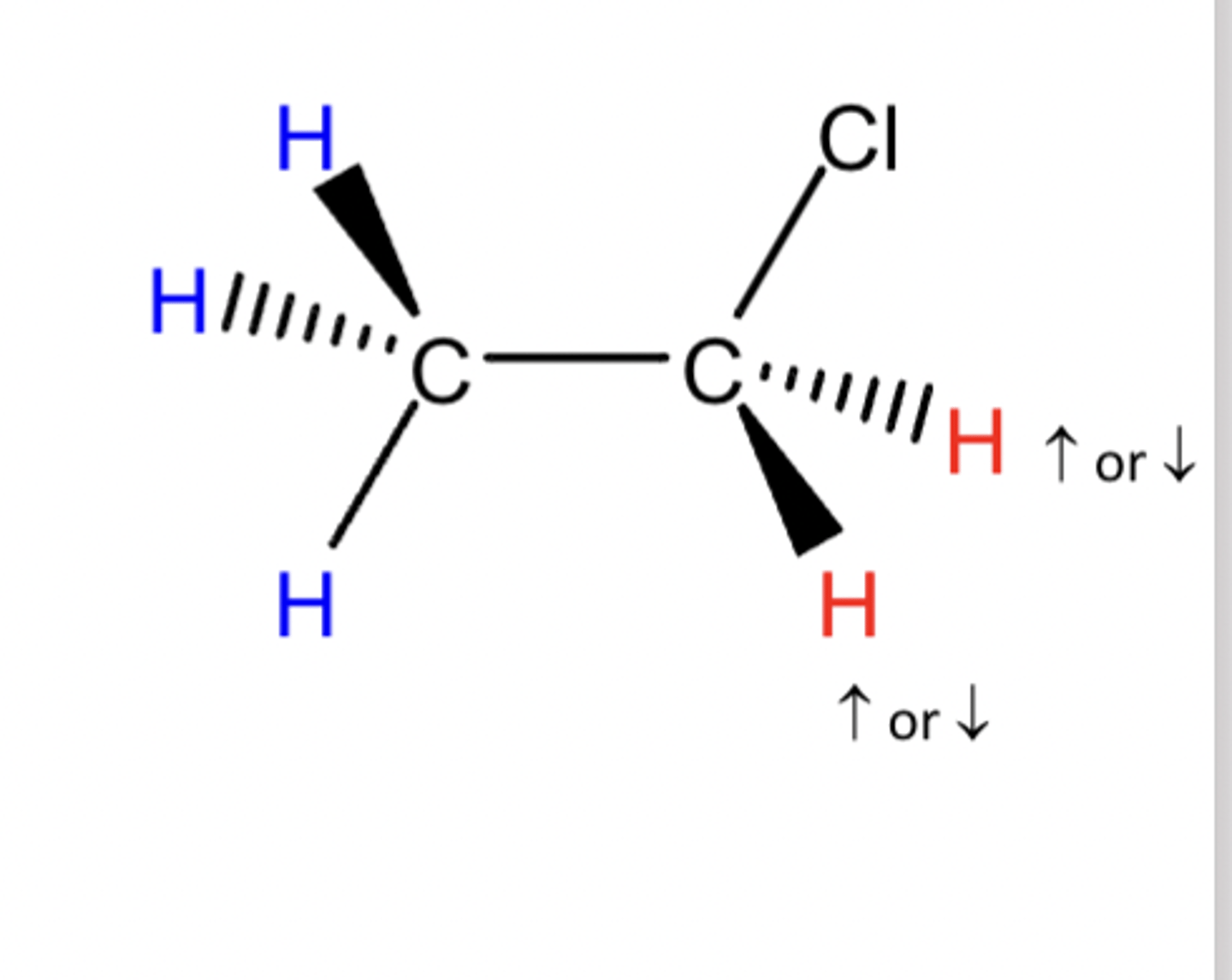
what does each H atom feel
Each H atom feels Bexperienced plus the magnetic fields of the H nuclei.
The H can either align with or against B0
There are three possible magnetic scenarios
three possible magnetic scenarios
Therefore the H atoms see three different magnetic fields– the signal splits into 3
both with B0, one with one against B0, both against B0.

what does the magnetic scenarios do
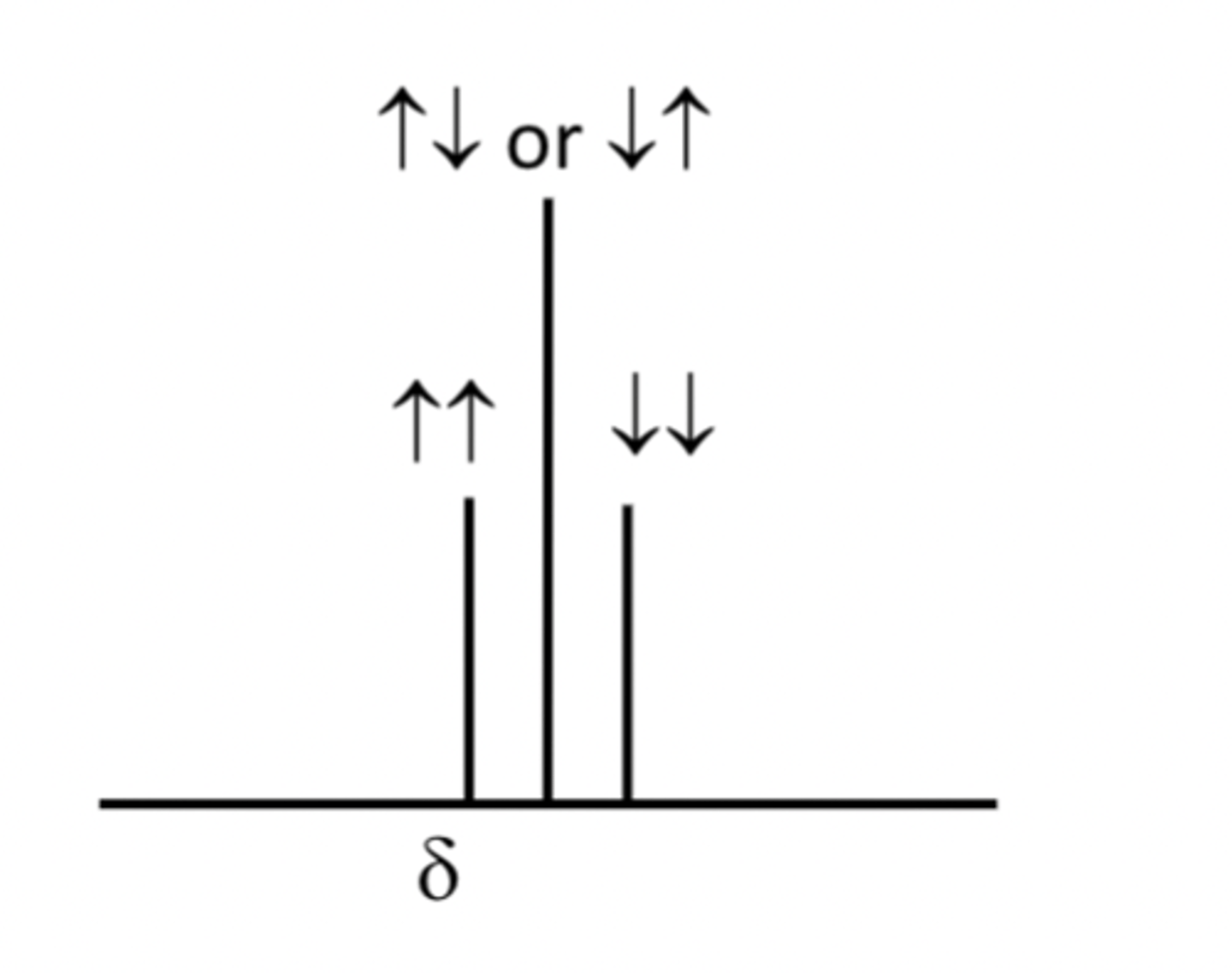
The H atoms in the CH3 group thus feel three slightly different magneticfields and the signal due to these hydrogens is split into a 1:2:1 triplet
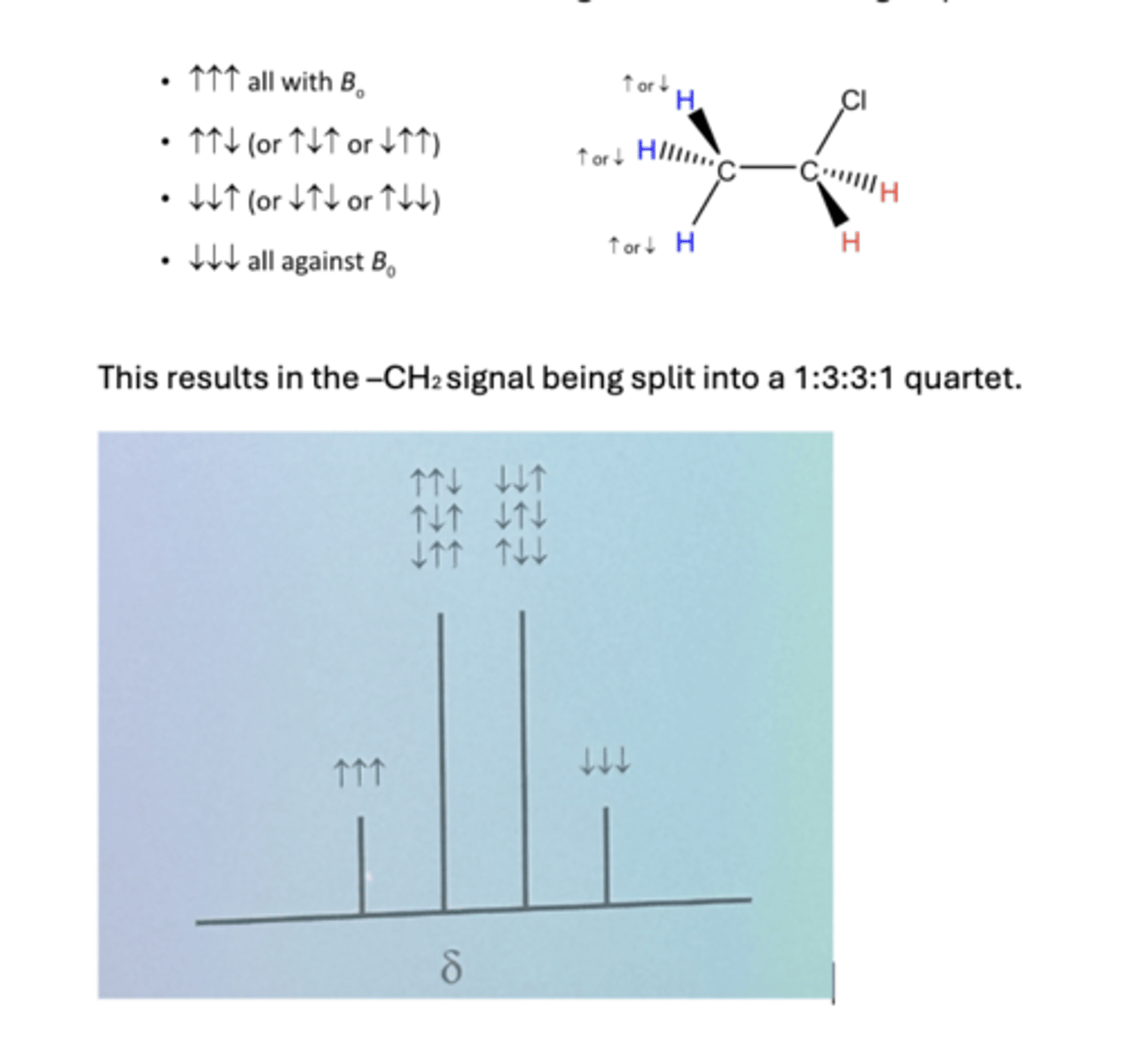
Similarly the H atoms in the -CH2 group will experience 4 different possible scenarios from their H neighbours in the –CH3 group
four possible magnetic scenarios
his results in the -CH2 signal being split into a 1:3:3:1 quartet.
what protons dont affect the signal, where is coupling observed
Protons more than 3 bonds away do not affect the signal; focus is on protons on neighboring carbon atoms.
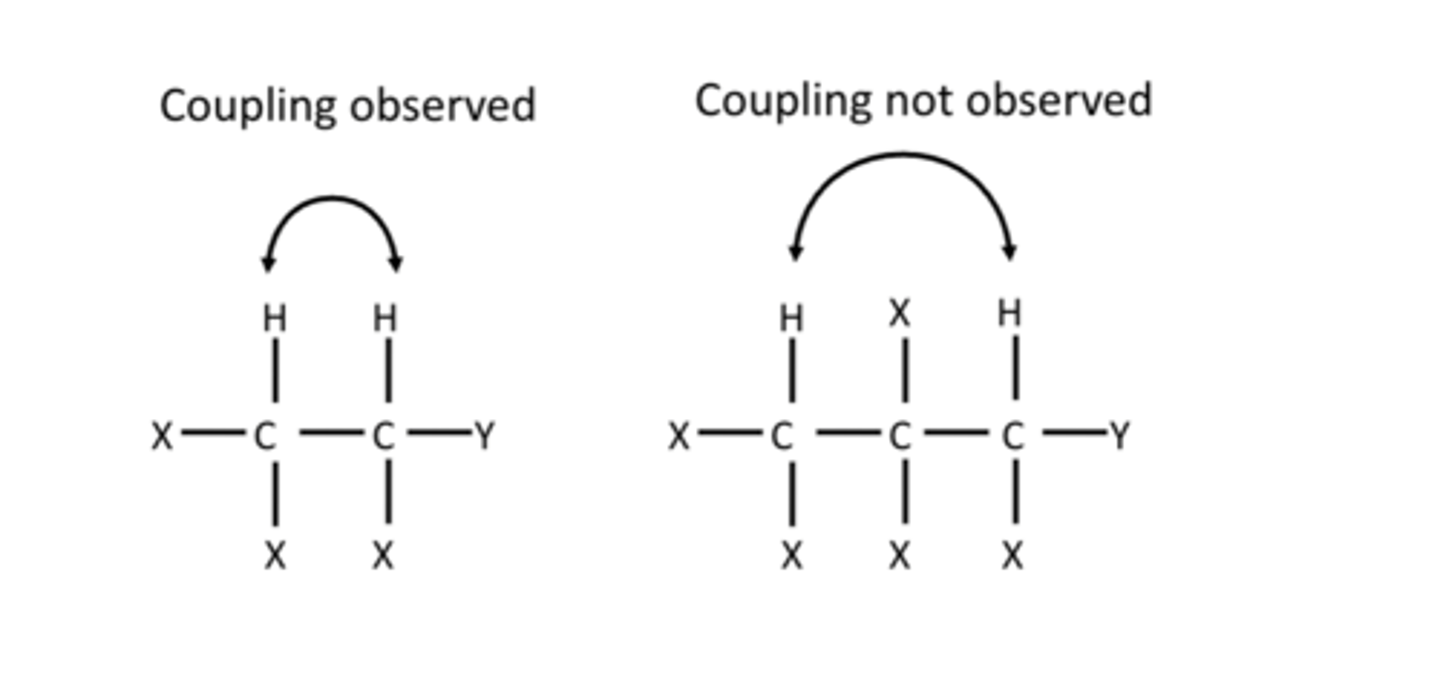
what does signal splitting pattern reveal
Signal splitting pattern reveals the proton's environment
coupling
Generally, the signal for equivalent Hatoms on one carbon atom will be splitinto (n+1) peaks where n is the numberof chemically equivalent H atoms on anadjacent C atom.
Note there is no splitting betweenchemically equivalent H atoms
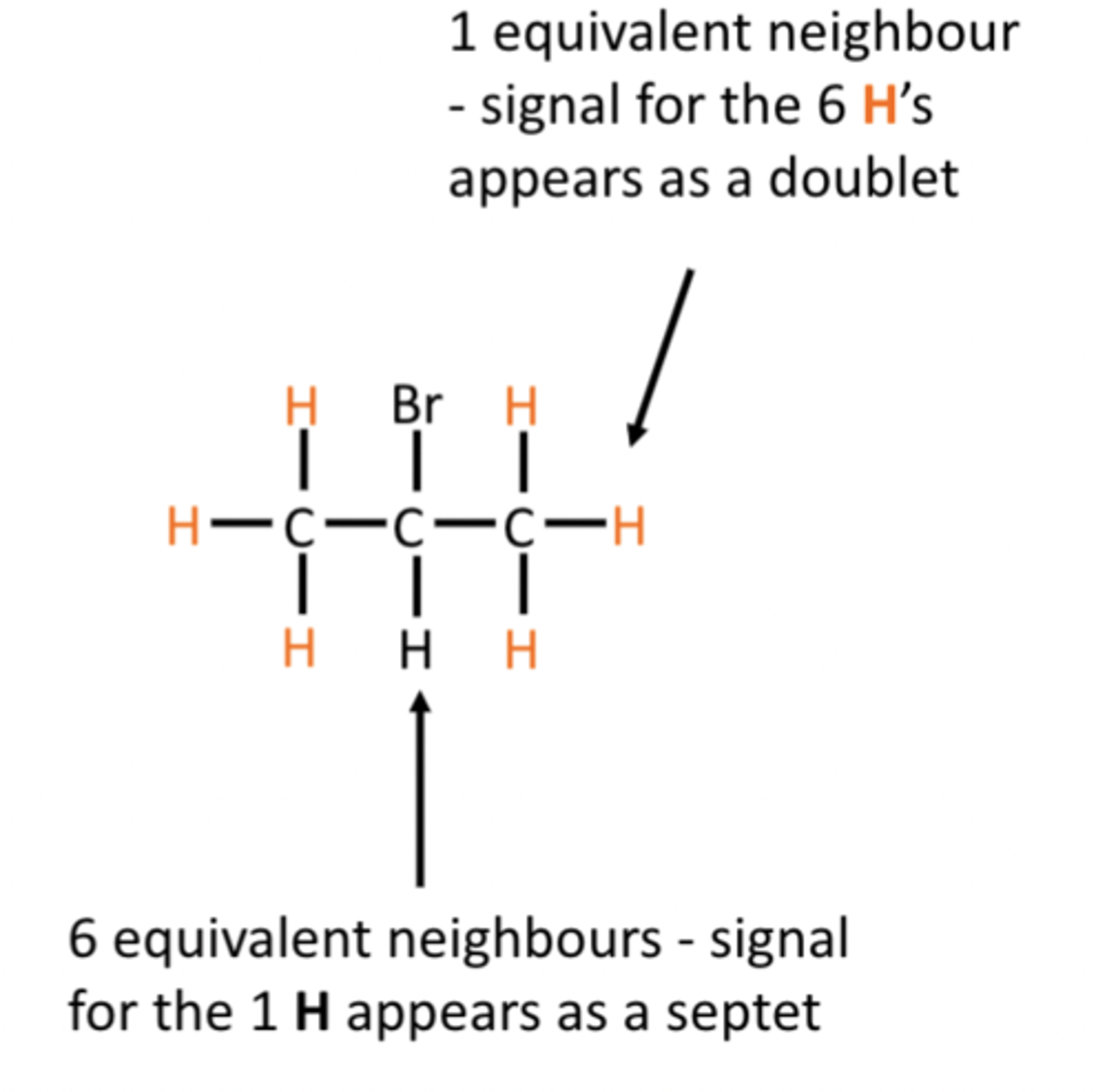
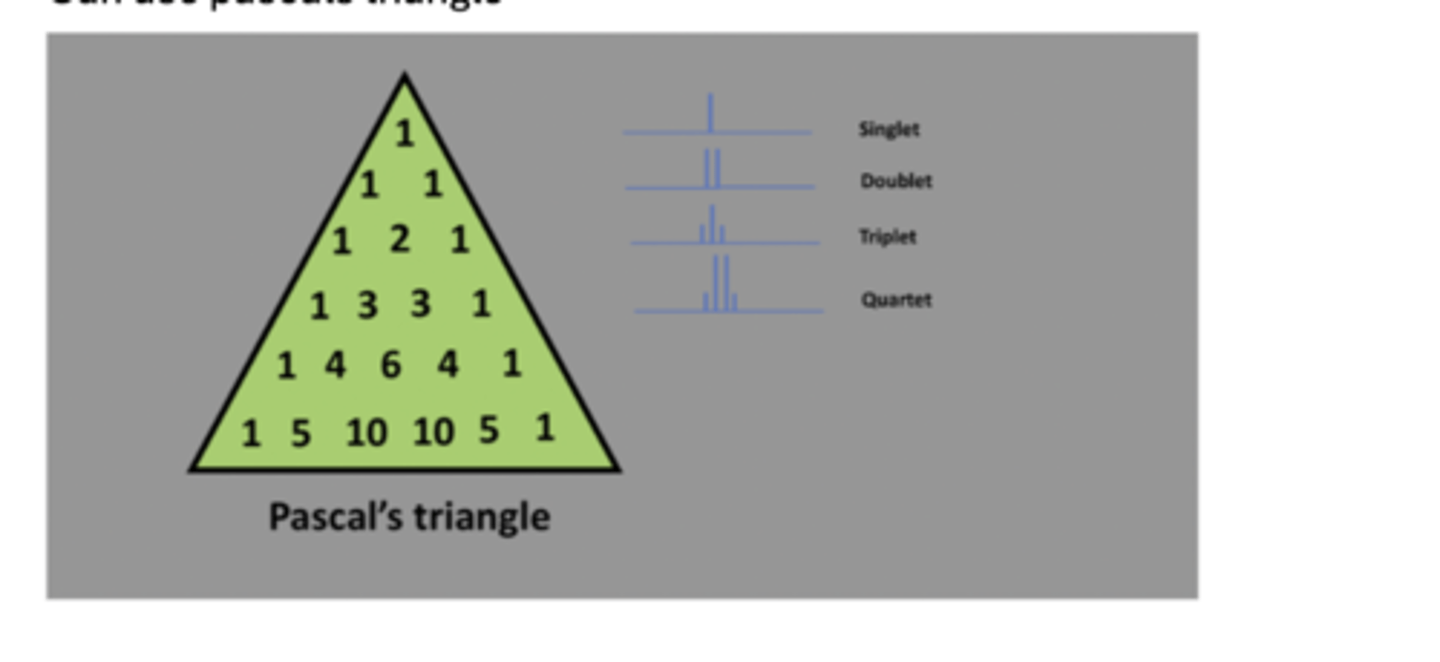
using pascals triangle
Can use pascals triangle to calculate singlet, doublet, etc.
coupling constant
The coupling constant is the space between the peaks in the multiplet
The size of the coupling constant will vary depending on the relationshipof the two coupling atoms
The size of the coupling constant will vary depending on the relationshipof the two coupling atoms
coupling to more than one non-equivalent atom
Results in signals being split multipletimes with different coupling constants