Chemical Logic of Metabolism: Deconstructing Glycolysis
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
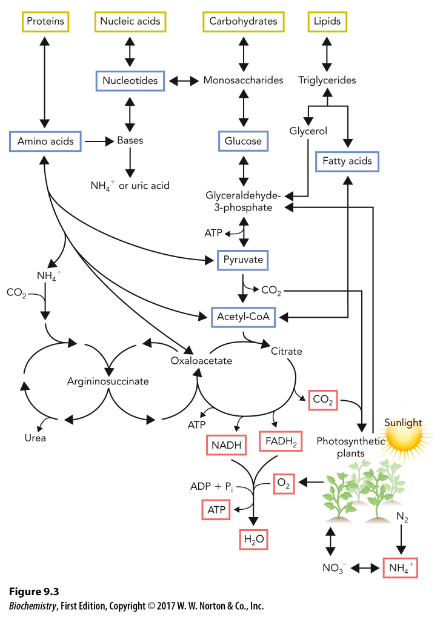
Metabolism
the collection of biochemical reactions which convert chemical energy into work to maintain cellular operation.
metabolite
small molecule intermediate or product of metabolism.
Catabolism
the sequences of enzyme-catalyzed reactions by which large molecules are broken down.
Anabolism
the reverse process to build complex molecules from simple structures.
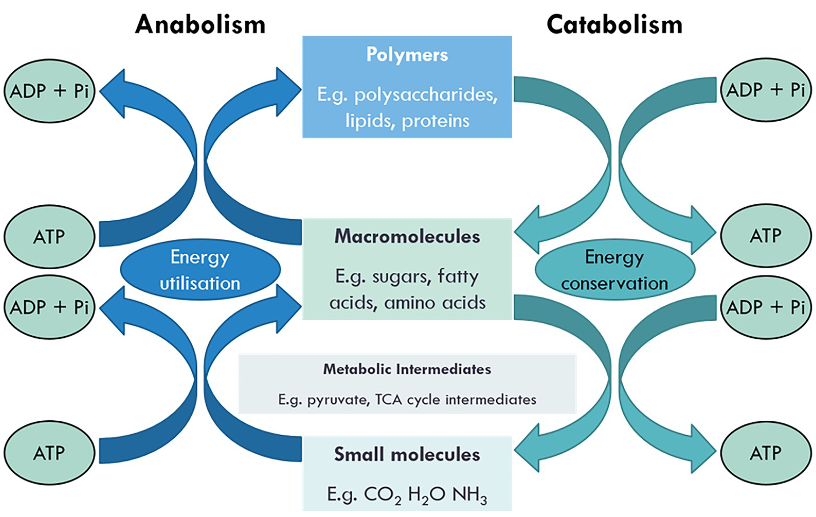
Coupling of anabolic and catabolic pathways
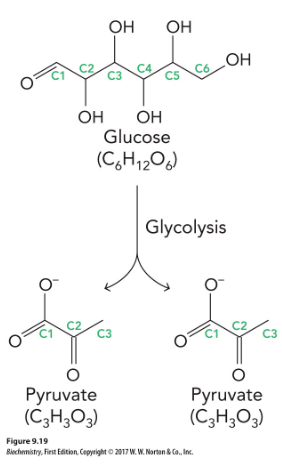
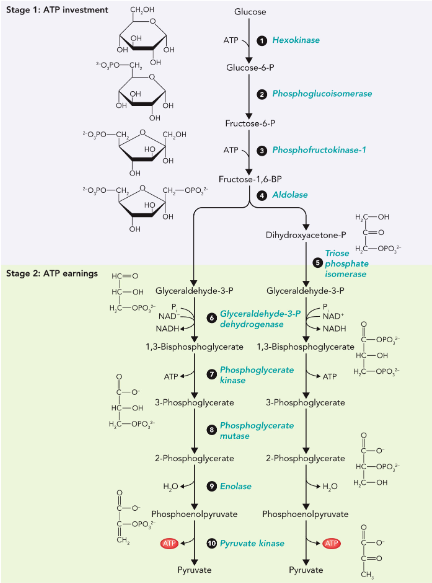
Glycolysis
a series of reactions that extract energy from glucose by splitting it into two three-carbon molecules called pyruvates
produce two molecules of ATP
Consists of 10 enzymatic reactions
occurs in nearly all organisms, both aerobic and anaerobic
does not use oxygen (anaerobic) and predates photosynthesis
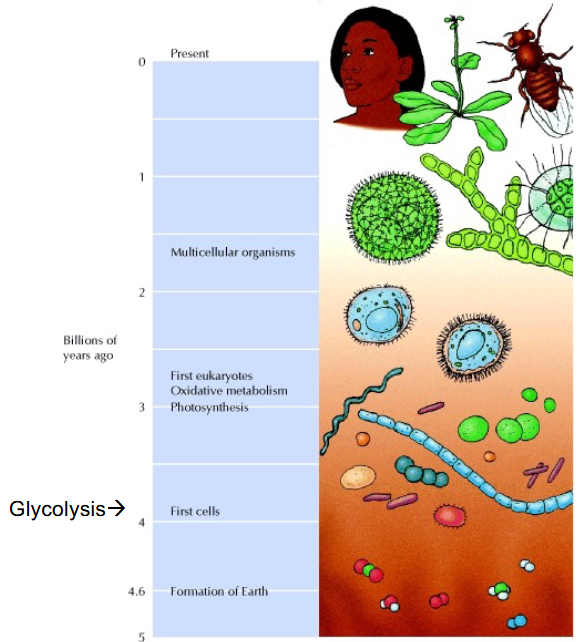
History of glycolysis
~3.9 billion years old, just after the first cells
Glycolytic metabolites
precursors for a large number of independent pathways, including mitochondrial ATP synthesis.
What does glycolysis accomplish for the cell?
alone can sustain all activity in anaerobic cells (ATP, amino acids, nucleotides, lipids, sugars)
In aerobic organisms, glycolysis feeds into TCA cycle and oxidative metabolism.
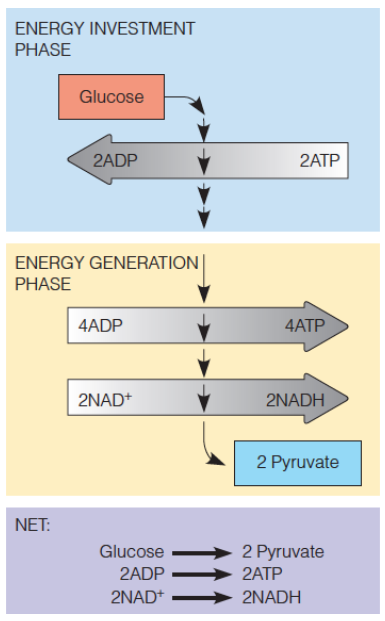
What is the overall net reaction of glycolysis?
Glucose + 2 NAD+ + 2 ADP + 2 Pi => 2 Pyruvate + 2 NADH + 2 H+ + 2 ATP + 2 H2O
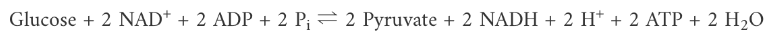
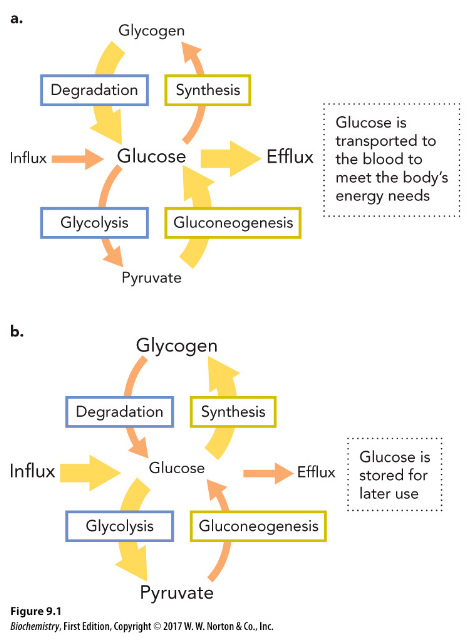
fate of glucose
controlled by the concentration of glucose and the body’s energy requirement
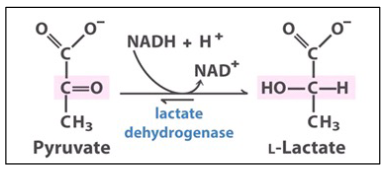
Lactic fermentation
Under anaerobic conditions (such as muscle cells during exercise), pyruvate is converted to lactate. Some microbes also dothis under anaerobic conditions (Lactococcus lactis produce yogurt/ cheese).
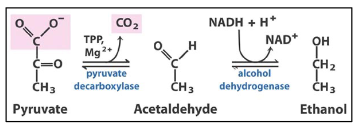
Alcoholic fermentation
Under anaerobic conditions, in some microbes, such as Brewer’s yeast (Saccharomyces cerevisiae), pyruvate is converted to ethanol and CO2.
central energy pathway
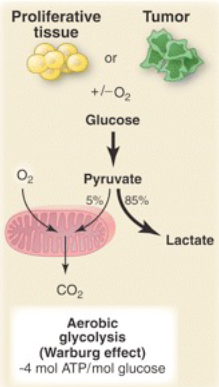
Under aerobic conditions, the majority of pyruvate is metabolized by the citric acid cycle and electron transport chain.
Warburg Effect
Metabolism of cancer cells is adapted to facilitate the uptake and incorporation of nutrients into the biomass (e.g., nucleotides, amino acids, and lipids) needed to produce a new cell → strongly favours glycolysis over oxidative phosphorylation → lactate production
energetics of glucose metabolism
investment and generation phases
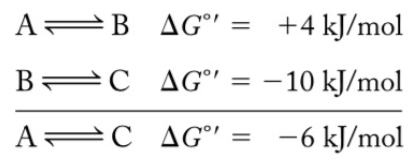
Coupled Reactions
Favorable metabolic reactions are used as a driving force to make unfavorable reactions proceed through a common intermediate.
ATP hydrolysis/synthesis
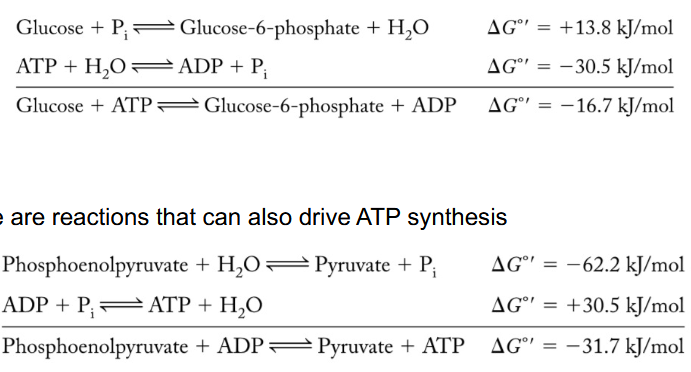
Why did nature come up with such an elaborate scheme to burn up glucose?
glucose was present.
Why phosphorylate glucose?
Hexokinase
drive glucose gradient into cell; make sure G6P doesn’t leave cell
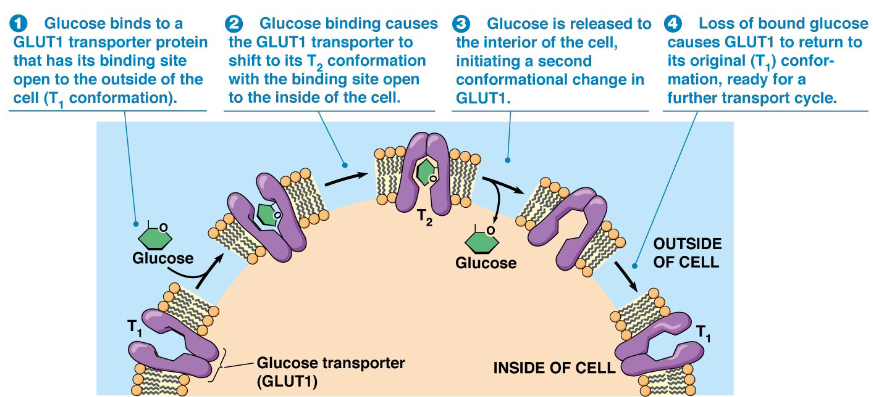
GLUT transporter
transporters glucose into cells, conformational change when bound to glucose
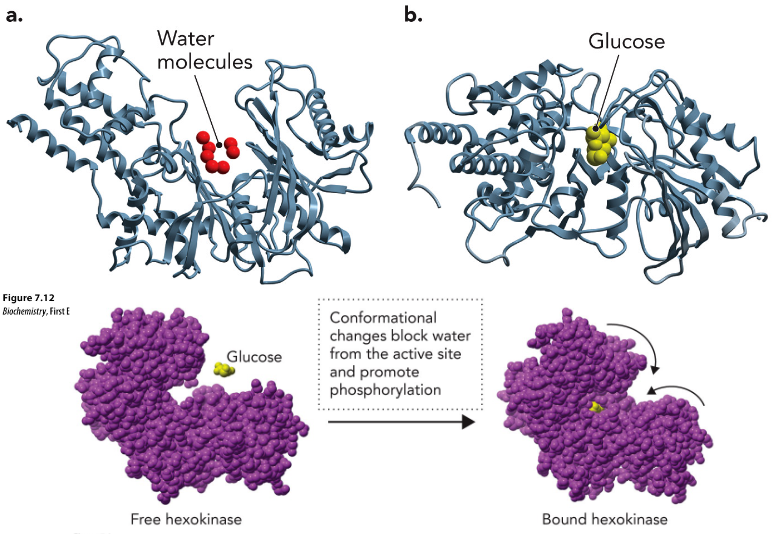
Hexokinase
Conformational changes block water from the active site and promote phosphorylation
uses 1 ATP
Hexokinase regulation
(-) G6P - allosteric regulation
Phosphofructoisomerase
general base in enzyme, activates aldolase cleavage
makes F6P
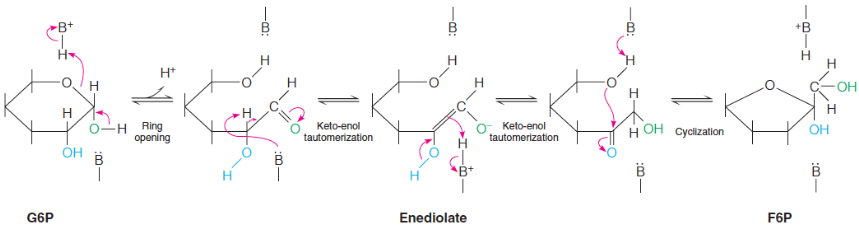
phosphofructokinase-1 (PFK-1)
both cleavage products should be phosphorylated
produces F16BP
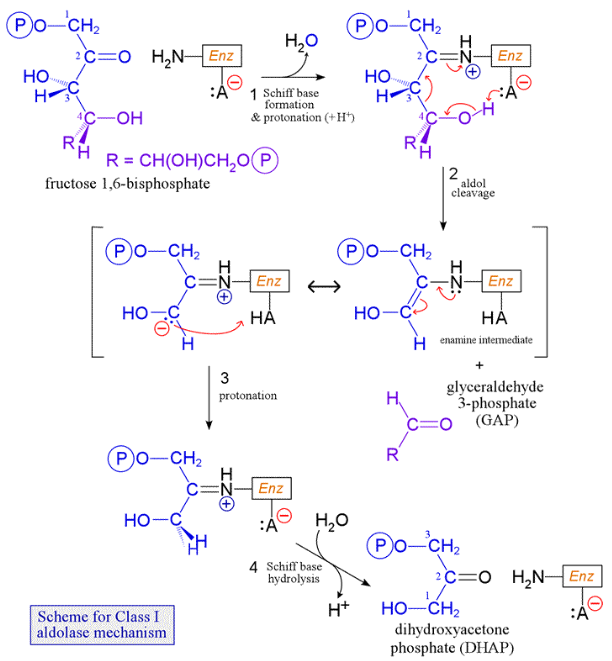
aldolase
adjacent carbonyl activates cleavage
iminium - covalent catalysis to enzyme → reduces entropy + favourable cyclic electron arrangement → control of reaction site
produces DHAP and GAP
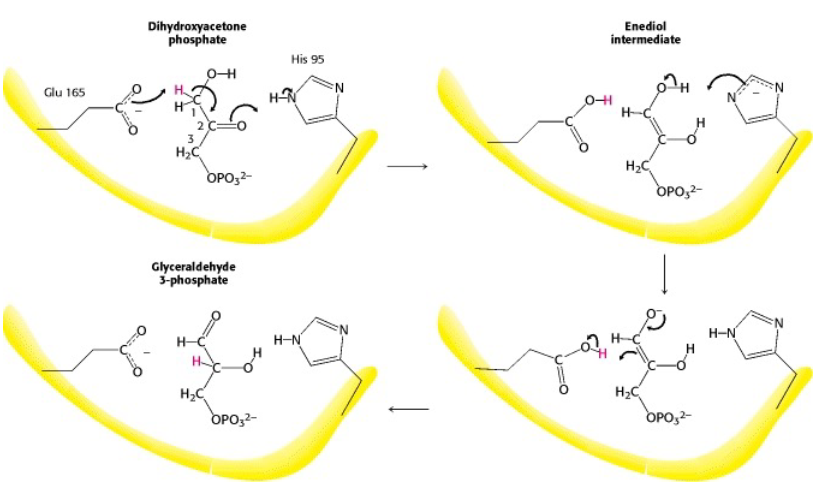
triose phosphate isomerase
enediol intermediate
10^8 kcat/Km- near perfect
Glyceraldehyde-3-phosphate Dehydrogenase (GAPDH)
use of NAD bypasses toxic metabolites - aldehyde
Phosphoryl group transfer requires oxidation of
the aldehyde
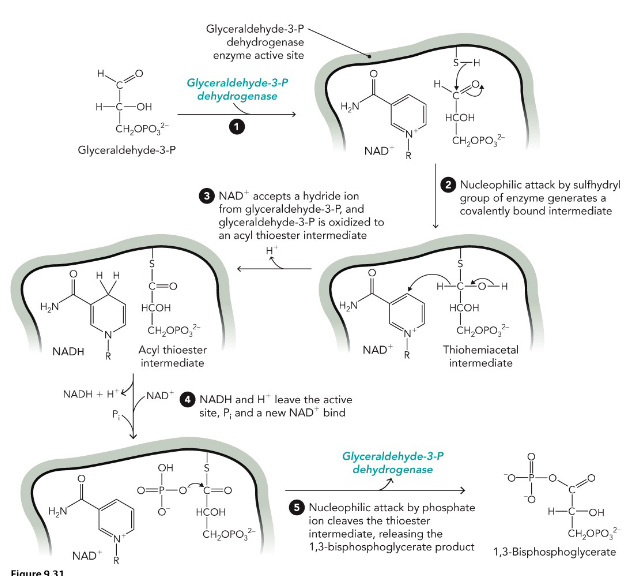
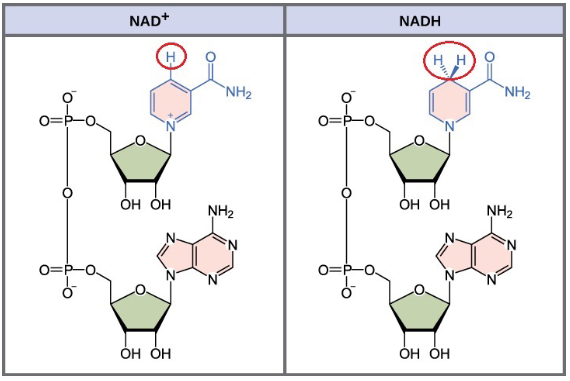
Nicotinamide adenine dinucleotide (NAD+)
cofactor; oxidizing agent; becomes reduced to NADH