Nomenclature
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
define empirical formula
the smallest whole number ration of atoms of each element in a compound
define molecular formula
the actual number of atoms of each element in a compound
define general formula
the condensed formula
define structural formula
the arrangement of atons in the molecule of a compound.
no bonds are shown
define displayed formula
a type of structural isomer that shows all the bonds between every atom
define skeletal formula
the representation of an organic compound which lines represent the carbon chain
give the molecular formula of butane
C4H8
give the empirical formula of butane
CH4
give the general formula of butane
CH3CH2CH2CH3
Give the skeletal formula of butane

give the structural formula of butane
CH3-CH2-CH2-CH3
define homologous series
molecules with the same functionl groups but different carbon chain lengths
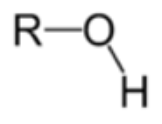
give the functional group
ALCOHOL
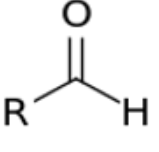
give the functional group
ALDEHYDE
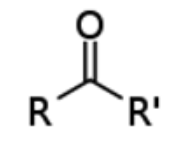
give the functional group
KETONE
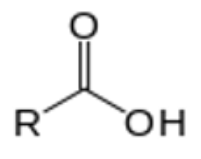
give the functional group
CARBOXYLIC ACID
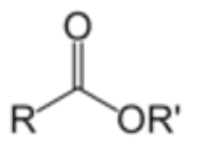
give the functional group
ESTER
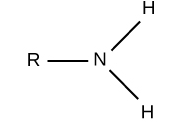
give the functional group
AMINE
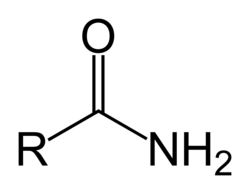
give the functional group
AMIDE
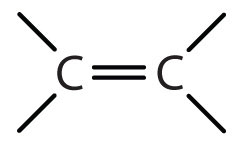
give the functional group
ALKENE
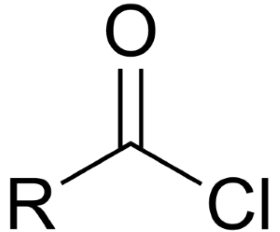
give the functional group
ACYLCHLORIDE
give the formula to work out the percentage by mass of an element in a compound
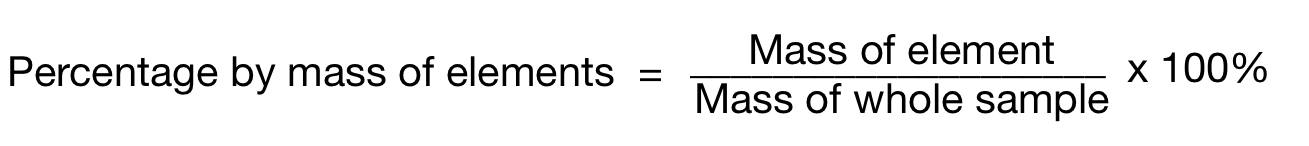
give the percentage mass of Oxygen in Ca(NO3)2
58.5%
give the Mr of Iron(III) carbonate
Fe2(CO3)2