Chemistry 11 - Chapter 5
0.0(0)
Studied by 57 peopleCard Sorting
1/67
Last updated 6:04 PM on 4/3/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
1
New cards
Who were Leucippus and Democritus and what did they do?
They were ancient Greek philosophers that reasoned that if a sample were cut into smaller and smaller pieces, the eventual result would be a particle so small that it couldn’t be cut into anything smaller.
2
New cards
What did Democritus call these particles and what did he imagine about them?
He called these particles “atomos” and imagined that particles of all different shapes and sizes might be components of all different types of matter.
3
New cards
Who was Aristotle and what idea did he propose?
A philosopher who proposed that all types of matter were made up of different proportions of four basic elements: earth, air, fire, and water. This was embraced for 2000 years in the practice of alchemy, though this notion was later proved false.
4
New cards
What did alchemists contribute to real science?
While searching for a way to change base metals to gold, alchemists discovered useful chemical processes such as distillation, separation of metals from ores, and the preparation of some acids.
5
New cards
Who was Robert Boyle and what did he do?
The first real scientist and the first person to perform and publish the results of true quantitative experiments. He dispelled Aristotle’s proposition of the four elements and provided an experimental definition of an element as being an “unmingled body”. He also outlined what we know today as the scientific method.
6
New cards
Who was John Dalton and what did he do?
He was an English schoolteacher who presented what we now know as Dalton’s Atomic Theory. He provided a major step forward in the world’s understanding of matter.
7
New cards
What are the four main points in Dalton’s Atomic Theory?
1. All matter is composed of extremely small particles called atoms, which cannot be broken into smaller particles, created, or destroyed. (as Democritus suggested)
2. The atoms of an given element are all identical to each other and different from the atoms of other elements.
3. Atoms of different elements combine in specific ratios to form compounds.
4. In a chemical reaction, atoms are separated, rearranged, and recombined to form new compounds.
8
New cards
Who was William Crookes and what did he do?
Scientist that created the cathode ray tube experiment. While he did not yet have an explanation as to why, he concluded through the location of the shadow in the tube and the cathode’s glow that something must be travelling from the cathode to the other end of the tube even though all the air was gone.
9
New cards
What is the cathode ray tube experiment?
A sealed glass tube connected to a vacuum pump. The tube itself contained a negatively charged electrical terminal (cathode) embedded in one end, a positive terminal (anode) located about halfway, and a broad flat cross placed two-thirds down. When a high voltage is applied across the electrodes as the air pumped out, any air remaining in the tube began to glow (through fluorescence).
10
New cards
Who is Jean Baptiste Perrin and what did he do?
A French scientist who discovered that a magnet placed next to the tube was able to deflect the mysterious beam in the CRT. As only a beam of charged particles can be deflected by a magnetic field, he deduced that the cathode ray must be a stream of charged particles.
11
New cards
Who was Joseph John “J.J” Thomson and what did he do?
An English physicist who modified the original Crookes’ tube and determined that the charges on the moving particles in the beam must be negative. He then calculated the charge-to-mass ratio of these negative particles and found the value to be 1.76 x 10^8 C/g (2000x smaller than the hydrogen ion). Thus, he discovered a particle smaller than the atom, the electron.
12
New cards
What was JJ Thomson’s most important discovery on the nature of the atom?
He concluded through further experimentation that electrons were present in atoms of different elements. He reasoned that if an intact atom has no charge, there must be a positive charge balancing the negative charge on the electron. Therefore, the building blocks of matter are electrical entities, and he developed his model to reflect this.
13
New cards
What was Ernest Rutherford’s background?
He was a farmer who won a scholarship to study with JJ Thomson, working with him during the discovery of negative electric charges.
14
New cards
What did Rutherford and Frederic Soddy discover together?
They discovered three types of radioactive emissions as well as their characteristics: alpha particles, beta particles, and gamma rays.
15
New cards
What are the characteristics of the three types of emissions?
* Alpha particles: helium nuclei; least penetrating; big and heavy
* Beta particles: electrons; mid-level penetration abilities; lighter than alpha particles
* Gamma rays: high energy waves moving at the speed of light; highly penetrating
* Beta particles: electrons; mid-level penetration abilities; lighter than alpha particles
* Gamma rays: high energy waves moving at the speed of light; highly penetrating
16
New cards
What did Rutherford use alpha particles for?
He used them to test the makeup of the atom. He expected there to be little to no deflection of alpha particles from the set-up gold foil, but a few particles deviated heavily or bounced back to where it came from. Nothing in Thomson’s model of the atom should be capable of knocking the alpha particle back, so this led to a major revision of the atomic model.
17
New cards
How did Rutherford explain the results of his experiment?
He suggested that virtually all the mass of an atom is concentrated in a very tiny, dense, positive structure at the centre of the atom called the nucleus. The nucleus is surrounded by a cloud of negatively charged electrons that account for the volume of the atom, but not the mass. He then predicted the existence of a neutral particle to account for the rest of the atom’s mass.
18
New cards
What did James Chadwick do?
He discovered the neutron, as Rutherford predicted.
19
New cards
What is an element’s atomic number (Z) ?
The number of protons and the positive charge of the nucleus of each of its atoms. **The number of protons in an atom determines the identity of an atom.**
20
New cards
What is an element’s mass number (A) ?
The sum of the number of protons and neutrons in the nucleus.
21
New cards
What is an element’s neutron number (N) ?
A (mass number) - Z (atomic number)
22
New cards
What is 1 AMU?
One atomic mass unit is exactly 1/12 the mass of a carbon-12 atom.
23
New cards
What is atomic mass?
The average mass of an element’s atoms.
24
New cards
How does this information apply to chemical changes?
They never involve change to the nucleus but often involve electrons being added or removed from neutral atoms that form ions.
25
New cards
What are isotopes?
They are different forms of the same element with different numbers of neutrons and therefore different mass numbers. The relative masses and abundances of isotopes can be determined by using a mass spectrometer.
26
New cards
How do we know which isotopes are most or least deflective?
We observe the charge-to-mass ratio (e/m). The greater the ratio, the more deflection occurs.
27
New cards
What was the big issue with Rutherford’s model of the atom?
According to classical physics, accelerating charges should radiate energy, and since orbiting electrons are accelerating charges, electrons in atoms should lose energy and the atom should collapse upon itself as the electrons spiral into the nucleus.
28
New cards
Was Rutherford’s model of the atom correct? What was the missing piece?
It was correct, but the classical physics of the day had not yet advanced far enough to explain the behavior of electrons in atoms. Physics needed to catch up instead.
29
New cards
Who was Planck and what did he suggest?
A German scientist who suggested that energy was not strictly a wave phenomenon, but actually could be shown to behave like particles in the form of small, discreet packets called quanta. According to Planck, energy could only be absorbed or emitted in whole numbers of quanta.
30
New cards
What was the equation for the energy associated with one energy packet or quantum?
E = h/v, where e = energy, v = frequency, and h = a very tiny proportionality constant known as Planck’s constant.
31
New cards
Why was Planck’s discovery so revolutionary and how did it gain acceptance in the scientific community?
Before this revelation, wave and particle behaviors were strictly defined as two distinct, separate things. To suggest that either could act like the other was crazy. However, Planck’s theory finally gained acceptance after Einstein used it to explain the photoelectric effect.
32
New cards
Who was Niels Bohr and what did he do?
A Danish scientist who found a way to save Rutherford’s nuclear model of the atom using Planck’s quantum theory. He suggested that if energy was quantized and could only exist in certain values, perhaps this applied to the energies of electrons orbiting the nucleus.
33
New cards
What did Bohr study to explain his theory?
Hydrogen’s bright-line spectrum. From there he formed his postulates to explain the behavior of the bright-line spectrum and the nuclear model of the atom.
34
New cards
What were Bohr’s postulates?
1. The hydrogen atom had only certain allowed energy levels or stationary states. Each corresponds to a circular electron orbit of a fixed size. The larger the orbit, the greater the chem. potential energy associated with it. Each energy level was given a quantum number referring to by increasing energy. The ground state is n = 1, and the larger orbits were considered excited states.
2. As long as the electron remains in an allowed stationary state, the electron and thus, the atom, does not radiate or absorb energy.
3. The electron can only move from one allowed state to another if it absorbs or emits the amount of energy exactly equal to the energy difference between the two states.
35
New cards
What is the quantum phenomenon observed by Bohr?
By quantizing the energies allowed for the atom, it was restricted to existing in only certain energy states and not others.
36
New cards
How did the postulates explain the bright-line spectrum of hydrogen?
An atomic spectrum could not be continuous (i.e., a complete rainbow of colors) because an atom’s energy states could only be certain values and not others. When dropping between energy states, electrons would emit a certain, quantized amount of energy. If the frequency of that emitted energy corresponds to any part of the visible spectrum, then a bright line of that specific color would be seen.
37
New cards
What was the problem with Bohr’s theory?
While it established the concept of fixed atomic energy levels, Bohr’s model only worked for hydrogen, and no other atom. Thus, he knew that the fixed atomic energy levels were there, but he could not explain why.
38
New cards
What fixed Bohr’s problem?
Quantum mechanics
39
New cards
Who was Louis de Broglie and what did he propose?
A French scientist who proposed particle-wave duality. If waves behaved like particles, could vice versa be true? This explained why electrons had fixed energy levels.
40
New cards
How would wave nature explain electron’s fixed energy levels?
Stable or “standing” waves must have a wavelength (λ) such that a whole number (n) of half-wavelengths (λ/2) fit within the length L of the string. Any fractional amount of half-wavelengths will cause the wave to break down by a process known as destructive interference. De Broglie considered what the requirements would be for a stable orbit if electrons did exhibit wave motion. He proposed that the only stable electron orbits were those whose size, and corresponding energy, allowed for standing electron waves to be maintained.
41
New cards
What is De Broglie’s equation and how did he derive it?
De Broglie used Einstein’s E = mc^2, Planck’s E = hv, and c = λv, to derive the equation λ = h/mv. It represents the wavelength of a particle.
42
New cards
What is the significance of De Broglie’s equation?
* It tells us that any moving particle, whether it’s an electron or a baseball, has a wavelength that we can calculate.
* It shows us that a particle’s wavelength is inversely proportional to its mass: as the mass of a particle decreases, its wavelength increases and vice versa.
* Because the value of Planck’s constant is so very small (6.626 × 10–34J.s), virtually any particle big enough for us to see has a wavelength so incredibly small that it can’t even be measured. However, particles with a mass as small as an electron have wavelengths that are very significant.
* It shows us that a particle’s wavelength is inversely proportional to its mass: as the mass of a particle decreases, its wavelength increases and vice versa.
* Because the value of Planck’s constant is so very small (6.626 × 10–34J.s), virtually any particle big enough for us to see has a wavelength so incredibly small that it can’t even be measured. However, particles with a mass as small as an electron have wavelengths that are very significant.
43
New cards
What proved De Broglie’s hypothesis?
Clinton Davisson and Lester Germer working at Bell Labs in the United States confirmed that electrons did indeed behave like waves. A beam of electrons directed at a nickel crystal generated a diffraction pattern similar to that produced by electromagnetic waves such as X-rays.
44
New cards
Who were Heisenberg and Schrodinger and what were their respective approaches to the behavior of an electron?
Heisenberg was a German physicist who treated the electron as a pure particle with quantum behaviour. Schrodinger was an Austrian physicist who viewed the electron mathematically as purely a wave to eliminate strange quantum aspects of electron behaviour.
45
New cards
How did Heisenberg and Schrodinger contribute to quantum mechanics?
Both of their theories were correct, and eventually had their theories united as key contributors in a single theory.
46
New cards
What is the Heisenberg uncertainty principle?
It was impossible to know exactly both where any particle was located and where it was going at the same time. Furthermore, the more certain you became of one measurement, the less certain you were of the other. (equation: (m∆v)(∆x) ≥ h/2π) the uncertainty in where a particle of constant mass is going (∆v) multiplied by the uncertainty in where a particle is located (∆x) is greater than or equal to a very tiny constant (h/2π). This means that the two values are inversely related. As we become more certain about an object’s position ∆x decreases. As ∆x decreases, the uncertainty in where the object is going, ∆v, increases and vice versa.
47
New cards
What is the reasoning behind Heisenberg’s uncertainty principle?
For quantum systems, the very act of measuring something causes a significant change in what we’re measuring due to how small the thing we are measuring is. This applies to any measuring method we know. For example, if we tried to see an electron, the energy of the photons hitting the electron would cause a significant change in its location, making it impossible to locate for certain.
48
New cards
How can we describe electrons in atoms after knowing the uncertainty principle?
Schrödinger represented the electron as a wave function he called ψ. The wave function is obtained by solving the equation using the allowed energy states associated with electrons in atoms. Its square, ψ2, gives the probability of finding those electrons within a region of space around the nucleus. The regions in which there is the highest probability of finding electrons are called atomic orbitals, which are how we describe electrons in atoms.
49
New cards
How does quantum mechanics describe electron behaviour?
It uses the mathematics of waves to determine the probability of finding an electron around the nucleus in a particular orbital.
50
New cards
What are the three important aspects of the quantum mechanical view of the atom?
1. The energies of electrons in atoms are quantized because of their wave nature. This relates to the idea that only certain allowed energy states associated with standing electron waves can exist.
2. The Heisenberg uncertainty principle states that it is impossible to simultaneously state both where an electron is and where it’s going.
3. Atomic orbitals are those regions in 3D space around a nucleus where electrons with a particular energy are most likely to be found.
51
New cards
What are quantum numbers?
They describe atomic orbitals. Each quantum number specifies something different about the orbitals and electrons.
52
New cards
What does each quantum number represent?
1. Principal quantum number (n) = relative size of the orbital
2. Angular momentum quantum number (l) = shape of the orbital
3. Magnetic quantum number (ml) = orientation in space
4. Spin quantum number (ms) = the spin of the electron
53
New cards
What is radial probability?
The probability of locating the electron is higher closer to the nucleus and lower further away from the nucleus. As we move out from the nucleus, we find that hydrogen’s electron in its ground state is likely to spend most of its time a slight distance from the nucleus, rather than at the nucleus itself. Quantum mechanics describes this as the most probable distance from the nucleus for the electron
54
New cards
Where can electrons with greater energy be found?
It will spend most of its time farther from the nucleus.
55
New cards
Why do the lobes of each p orbital not reach the nucleus?
The amplitude of the electron wave at the nucleus is zero. A wave amplitude of zero is called a node, and it tells us that there is a zero probability of locating the electron here.
56
New cards
What do the first three quantum numbers tell us?
The three quantum numbers, taken together, will always specify a particular atomic orbital because they tell us all we need to know about that orbital: its size, shape, and orientation in space.
57
New cards
What is the pattern for the number of orbitals going from s → p → d → f?
One s orbital, three p orbitals, five d orbitals, seven p orbitals.
58
New cards
How many electrons fit in one orbital and what directions must they spin in?
Any atomic orbital can hold a maximum of two electrons. When two electrons are in the same orbital, their spins must be opposite.
59
New cards
What is the Pauli exclusion principle?
No two electrons in the same atom can be described by the same set of four quantum numbers.
60
New cards
Why do orbital sublevels have different energies?
Repulsive forces cause a sublevel with a greater number of orbitals to have a greater energy. In several cases energies get closer together as n increases. In these cases, repulsive forces are such that some sublevels containing smaller orbitals actually have higher energies than some larger orbitals.
61
New cards
What is the Aufbau principle?
When filling orbitals, the lowest energy orbitals available are always filled first.
62
New cards
How can we represent the arrangement of electrons in an atom?
An electron configuration or an orbital diagram.
63
New cards
What is Hund’s rule?
When orbitals of equal energy are being filled, the most stable configuration is the one with the maximum number of unpaired electrons with the same spin.
64
New cards
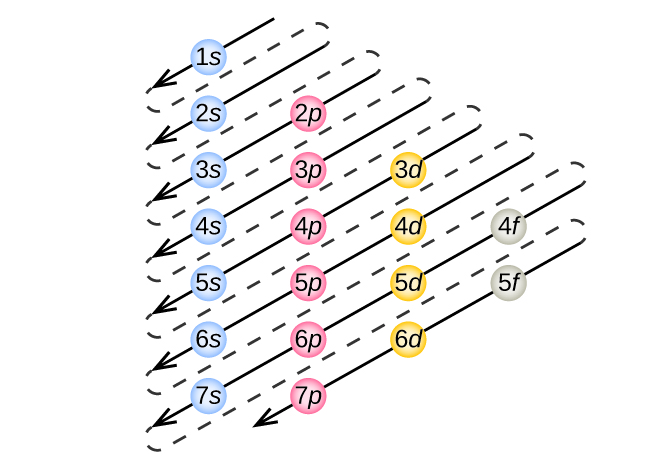
What is this?
The order to fill orbital sublevels.
65
New cards
What does it mean for ions to be isoelectronic?
The two species have the same number and configuration of electrons.
66
New cards
What are the five exceptions in electron configurations?
Cr, Cu, Mo, Ag, and Au.
67
New cards
What are degenerate orbitals?
Orbitals on the same sublevel. They are of equal energies.
68
New cards
Why do electrons like to be paired in orbitals?
The magnetic fields cancel out.