Post Seg Final
1/163
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
164 Terms
4 phases of FA
Pre-arterial: choroidal flush (20-30 sec after injection)
Arterial: arteries fill, veins still dark
Arteriovenous: capillaries and smaller arteries/veins
Venous: laminar flow (rapid flow of plasma along BV walls)
why is macula dark in FA?
Foveal avascular zone: 400-500 microns large, no inner retinal BVs
More columnar RPE cells, increased conc of lipofuscin, xanthophyll, melanin, etc
What causes hypofluorescence in FA?
Vascular filling defect → no fluorescein (eg CRVO/CRAO)
View is blocked (even though fluorescein is present, eg subhyaloid heme blocks view of normal BVs)
What causes hyperfluorescence in FA?
Window defect: RPE is missing so choroid is visible
Leakage: fuzzy borders, gets larger in size
Staining: gradual increase in fluorescein persisting in late stage, size stays the same
Pooling: cavity fills brightly with dye over time, size stays the same
Characteristics of leakage in FA
Edges become increasingly blurry/fuzzy in late phase
Any fluorescein that remains 10-15 minutes after BVs are empty
Either caused by: break in RPE into subretinal space/inner retina OR out of inner retinal BVs or retinal NV into the vitreous
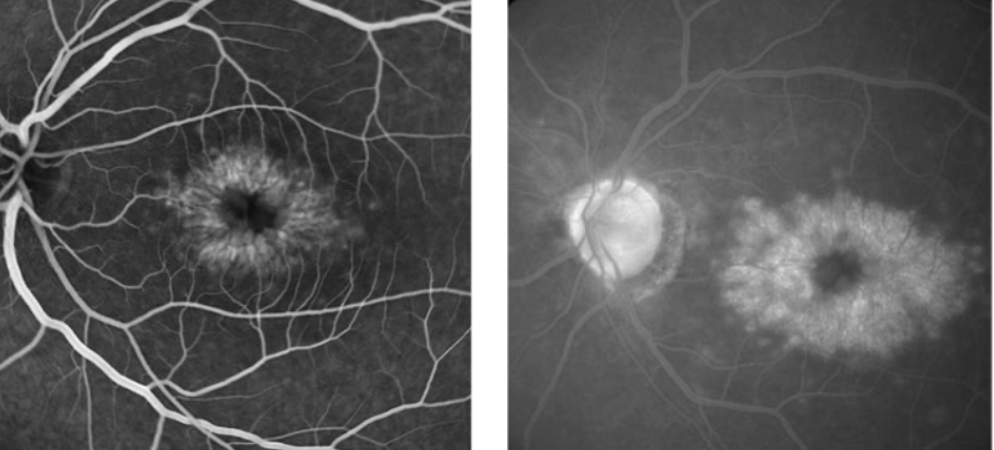
Petalloid pattern around macula = CME
Inner BRB breaks down → leakage into inner retina/vitreous
Caused by retinal vascular diseases: DR, CRVO/BRVO
Subretinal leakage = CNVM
Outer BRB breaks down → leakage into subretinal space/neurosensory retina
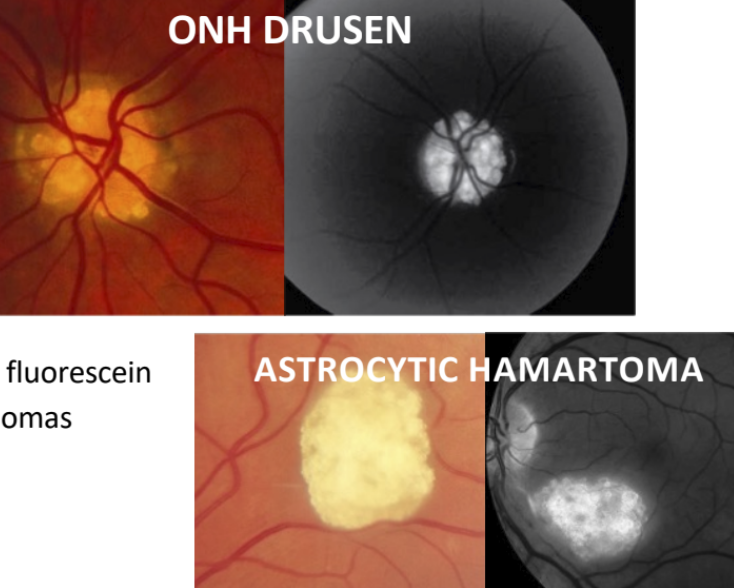
What is autofluorescence in FA?
Fluorescence in the absence of fluorescein dye
Commonly caused by ONH drusen, astrocytic hamartomas
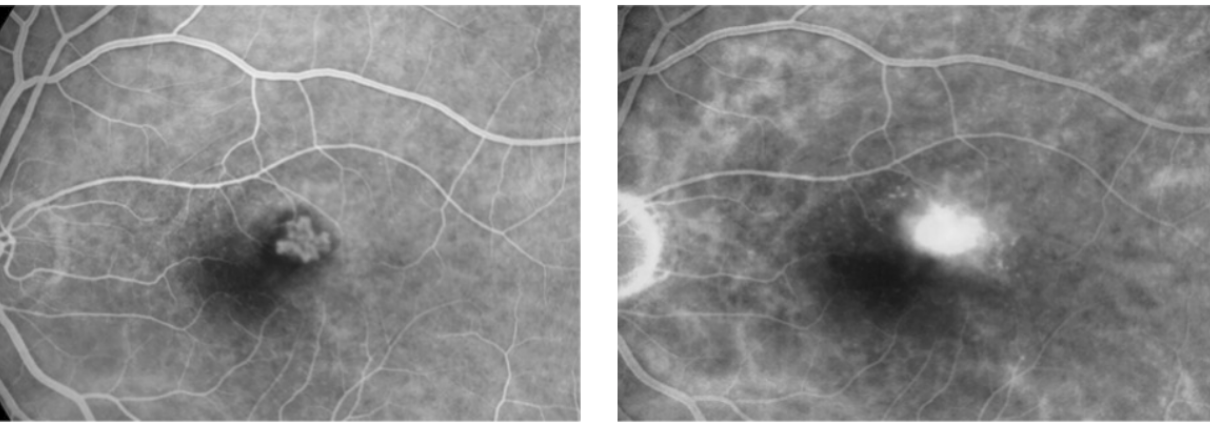
Advantages of ICG angiography compared to FA
Indocyanine green absorbs/emits in IR range → not blocked by normal eye pigments
Best for imaging choroidal circulation, esp thru overlying heme/pigment/fluid
Eg: PED, occult CNVM, central serous
How FAF works?
Fundus autofluorescence: using natural fluorescence of lipofuscin to check RPE/cell health
Large amts of lipofuscin indicates RPE/cell death
Might see hypoautofluorescence areas (RPE atrophy) surrounded by hyperautofluorescence (lipofuscin accumulation)
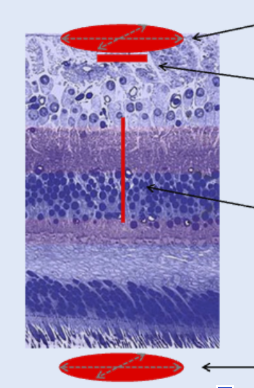
List inner retinal layers, outer retinal layers, predominant layers of foveola, and significance of OPL
Inner retina: ILM, NFL, GC, IPL, INL
Outer retina: OPL, ONL, ELM, PRs, RPE
Foveola (RPE OI): RPE, PRs, ELM, ONL, ILM
OPL: hydrophobic barrier to fluid movement, separates inner and outer retina, mostly perfused by choroid (outer retina)
Which retinal/OCT layers have BV plexi? Which are avascular?
Retinal layers w/ BV plexi: RNFL, GC, INL
OCT layers: RNFL, GC/IPL, INL
IPL contains vertically oriented BVs that connect GC and INL (not plexi)
Avascular (all others): OPL, ONL, ELM, PRs, RPE
Regarding problems with retinal vasculature, what does leakage lead to, and what does ischemia lead to?
Leakage (eg DR/BRVO) → extracellular edema: cysts, schisis, cavities, separation of layers
Ischemia (eg BRAO) → intracellular edema: swelling of cells within inner retina (layer edges look less distinct)
3 most common reasons for decreased vision in recent onset RVO? Tx options?
edema (anti-VEGF, laser), ischemia (no Tx, monitor for NVG), preretinal hemorrhage (wait for hemorrhage to clear → vitrectomy)
Difference between RD, PED, and foveal schisis (which retinal layers separating?)
RD: PRs and RPE
Either tractional (non-rhegmatogenous) or non-tractional (rhegmatogenous, fluid entering)
PED: RPE and Bruch’s membrane
drusen, CNVM, serous exudates, blood
Foveal schisis: ONL and OPL
What happens when blood flow is significantly reduced?
Occlusion
If blood flow is restricted enough → causes increased pressure in veins and leakage
Causes decreased oxygen supply → ischemia
What is the critical window for retinal ischemia to be reversible?
Within 4.5 hours
past critical window → cell death/necrosis occurs which is irreversible!!
Are retinal arterial occlusions a medical emergency? What are the two categories of RAOs?
ALL NEW RAOs within 30 days are considered medical emergency
Goal is to prevent ischemic stroke and death (not to save dead cells, it’s too late for that..)
RAOs are either arteritic (5%, GCA or hypercoagulability), or nonarteritic (95%, embolic)
Classic sx and risk factors of GCA
Sx: jaw claudication, fever, scalp tenderness, malaise, weight loss
Risk factors: white woman >50 yo
Dx tools for GCA, and why is GCA considered a medical emergency? Tx?
Gold standard dx: temporal artery biopsy — also ESR, CRP, platelet count
Medical emergency bc other eye is affected in 50% of patients
If pt doesn’t have a visible embolus → must consider GCA or an arteritic RAO
Tx: high dose IV and oral steroids → purpose is to reduce risk of bilateral vision loss (too late to save vision in affected eye)
Why are non-arteritic RAOs a medical emergency?
Embolic based, usually lodged where lumen size decreases suddenly
Emergency bc increased risk for concurrent ischemic stroke → need to refer to primary stroke center for neuro/embolic workup
Difference b/w embolus, plaque, thrombus
Embolus: any particle/mass traveling thru blood (Hollenhorst plaque, platelet, calcific)
Plaque: buildup on BV wall → narrow lumen
Thrombus: sudden blood clot forms inside vein/artery
CRAO
Retinal whitening caused by intracellular edema/ischemia in the inner retina
Outer retinal layers and choroid look normal
Blot hemes: subretinal/subhyaloid
Flame heme: RNFL
Dot heme: OPL (capillaries leaking)
Subsensory: sheeting, in PRs/RPE (DNVM, will not obscure retinal BVs)
What are RVOs caused by?
NOT caused by emboli → NOT medical emergency
Caused by thrombus (compression of artery against vein due to shared common adventitial sheath)
Thrombus caused by Virchow’s triad:
1. Endo damage
2. Blood stasis
3. Hypercoagulable state
Difference b/w ischemic and non-ischemic CRVOs
Ischemic CRVO (30%):
1. 10 DA of capillary nonperfusion
2. VA worse than 20/200
3. (+) APD
4. Hemes: numerous, large and dark color
more likely to develop sequelae (NVG)
Non-ischemic CRVO (70%):
1. VA better than 20/200
2. (-) APD
3. Hemes: smaller/fewer
How does pressure in BVs change with RVO vs RAO?
RVO: pressure within A/V crossing increases from obstruction → causes leakage
RAO: pressure within vein decreases from lack of blood flow, while pressure in arteries increases next to occlusion (no hemes bc arteries can handle pressure increase)
Ocular sequelae/long-term management of RAOs
BRAO: RNV and anterior NV is rare
CRAO: RNV is rare, anterior NV occurs 15%
Ocular sequelae/long-term management of RVOs
BRVO: RNV 8%, anterior NV rare, CME 5%
CRVO: RNV 9%, anterior NV 35% in ischemic, 10% in non-ischemic, CME 76%
Gold standard in treatment of retinal/anterior NV
PRP (panretinal photocoagulation) → killing retina stops VEGF release
Anti-VEGF is adjunctive therapy with only temporary effect
Risk factors of AMD
Age: >50 vs >80 triples risk of development
FHx: 50% AMD risk if (+) FHx
Smoking: 2-3x increased risk
Diet: high fat, low omega-3s, vitamins, carotenoids
HTN
Lack of exercise
Where does drusen accumulate and what sizes are there?
B/w RPE and Bruch’s membrane, represent sites of focal inflammation (RPE atrophy)
Sizes based on size of CRV (diam 125 um)
Large drusen >125 um
Intermediate drusen b/w 63-125 um
Small drusen < 63 um
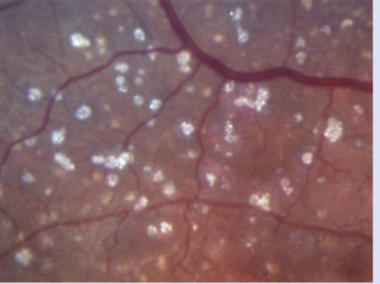
Hard/nodular drusen characteristics
<63 um (small)
Sharp margins, uniform color density
Found in central and peripheral retina
Normal with age
Soft drusen characteristics
>125 um (large)
Soft/fuzzy margins, white center w/ yellow edges
Only in posterior pole, assoc with pigmentary changes
OCT or FA can see if any fluid buildup
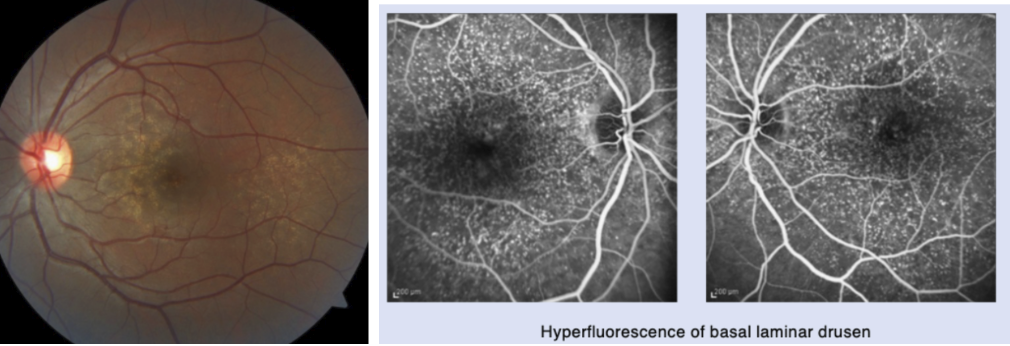
Cuticular/basal laminar drusen
Found in large numbers → cause starry sky in FA, window defect w/ staining
Can appear like soft drusen when they coalesce, but NOT assoc w/ pigmentary changes
Start in Bruch’s → accumulate b/w Bruch’s and RPE
Strongly associated w/ advanced AMD
Characteristics of calcified drusen
Considered end-stage drusen, since all drusen likely contain calcium
Strongly associated w/ advanced AMD
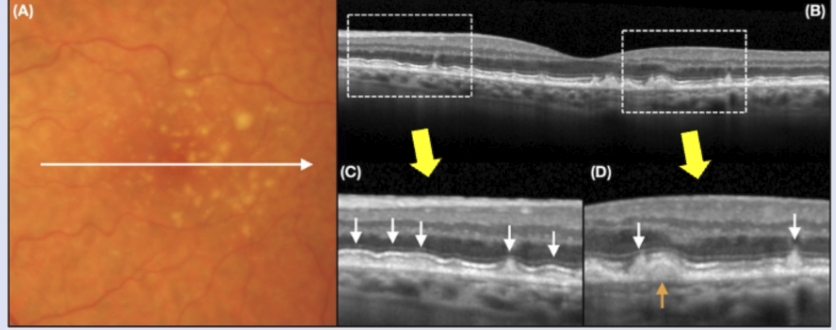
Characteristics of reticular pseudodrusen
Located in subsensory space, b/w PRs and RPE
Can coalesce into ribbons → can cause PIL to go away
White arrows here is pseudodrusen
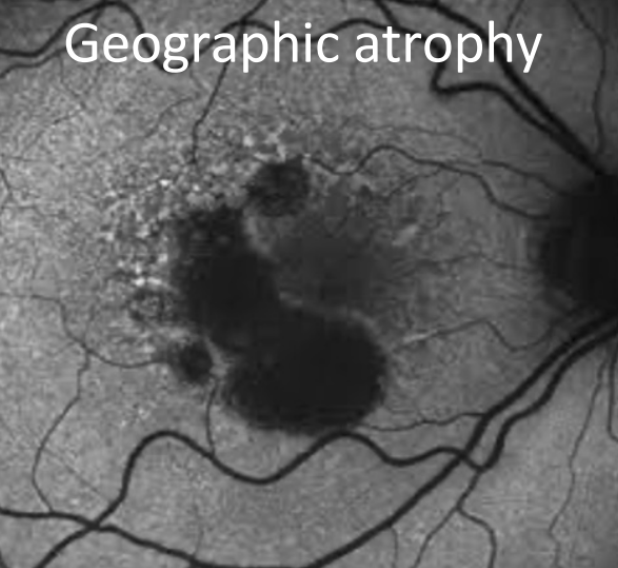
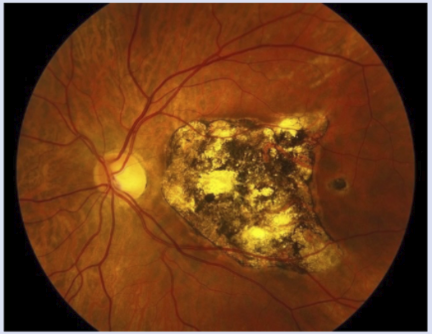
Definition of geographic atrophy and its cause
Large area of RPE atrophy → leads to progressive loss of inner retina
Caused by chronic inflammation: overactive complement system → RPE, PRs, choriocapillaris loss first
Definition of advanced dry AMD
Geographic atrophy of the foveola
Dry AMD makes up 90% of AMD cases
Definition of wet AMD
Any presence of CNVM = wet AMD
AMD is the most common cause of CNVM
wet AMD makes up 10% of AMD cases, but accounts for 90% of vision loss
Type 1 vs Type 2 CNVM
Type 1 CNVM = occult CNVM (most common)
B/w RPE and Bruch’s membrane → double layer sign on OCT
Type 2 CNVM = classic CNVM
Above RPE (subsensory)
Type 3 CNVM characteristics
Retinal NV that connects retinal and choroidal circulation
Starts in inner retina and moves posterior towards choroid
What is a disciform scar?
Endstage wet AMD, when retinal tissue is replaced w/ scarring and fibrosis
CNVM can still bleed/leak
4 stages of AMD
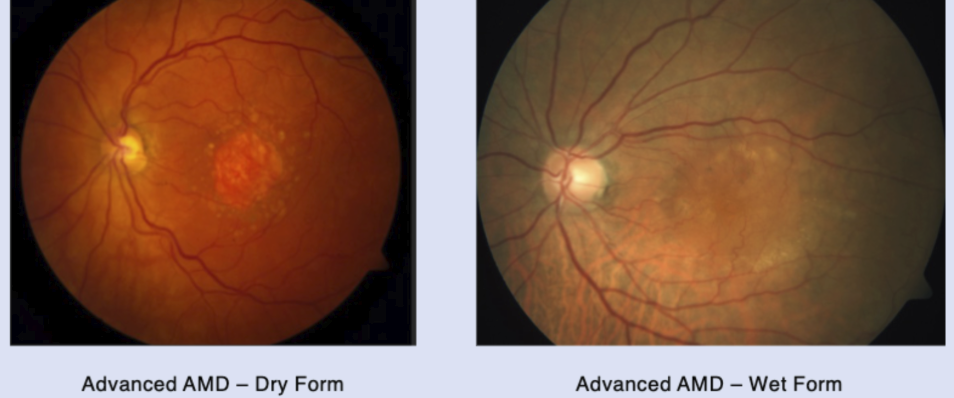
Category 1: no AMD
few small or no drusen
Category 2: early AMD
many small drusen or few medium-sized drusen, in one or both eyes
Category 3: intermediate AMD
many medium sized drusen or one large drusen, in one or both eyes
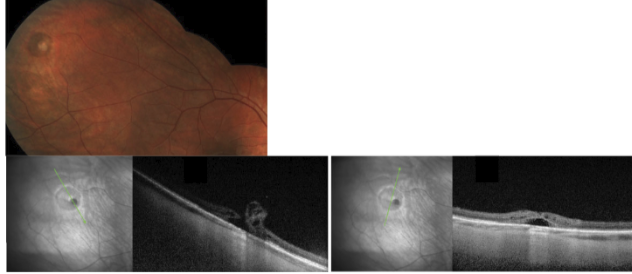
Category 4: advanced AMD
breakdown of any retinal cells or supportive tissue (dry) OR abnormal BVs under retina (wet) - see photo
Risk assessment to quantify risk of advanced AMD in the next five years, what qualifies as 0.5, 1 and 2 points?
In one eye:
Advanced AMD OR wet AMD: 2 points
Pigmentary changes: 1 point (includes GA)
Large drusen: 1 point
Intermediate drusen w/ no large: 0.5 point
2 points: 12% risk of adv AMD
3 points: 25%
4 points: 50%
Difference b/w AREDS and AREDS2
AREDS2 does NOT have beta-carotene (increases lung cancer risk in smokers)
AREDS2 has lutein + zeaxanthin added (tendency to be beneficial after 10 years)
AREDS2 has decreased risk of developing adv AMD by 25% in intermediate or advanced AMD
AREDS2 has NO BENEFIT For EARLY AMD
Clinical findings of CNVM
Hemes and fluid: subretinal or sub-RPE
Hard exudates
Gray membrane
Subretinal fibrosis
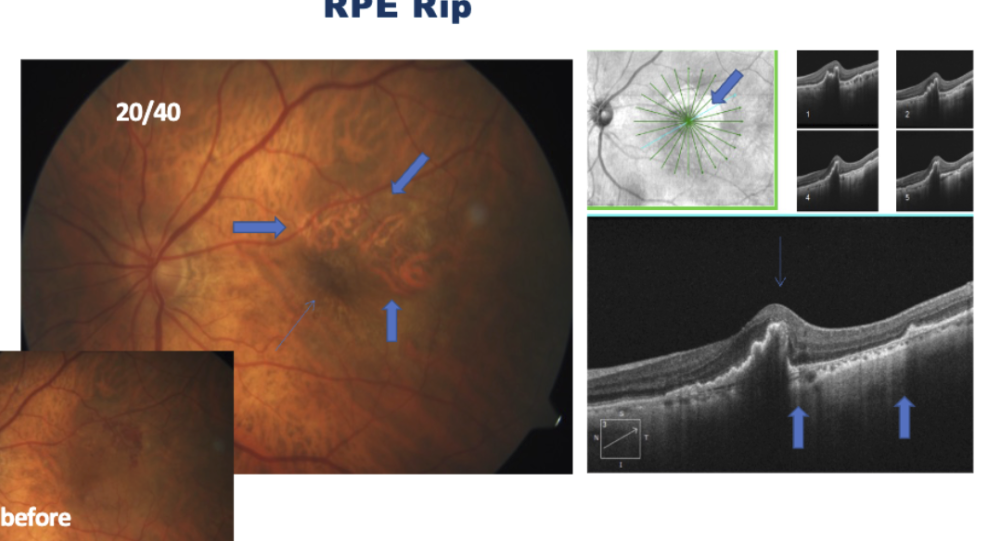
What is an RPE rip?
Consequence of anti-VEGF injections
Common with PED
If choroidal vessels are clear, and there is shadowing in adjacent area → indicates RPE is bunched up after getting displaced
How does a PVD and ERM affect anti-VEGF injection treatments?
PVD can have positive effect on injections (may need fewer)
ERM can have negative effect (may need more injections)
Angioid streaks and systemic associations
Abnormal Bruch’s membrane → can cause crack-like breaks
Assoc with pseudoxanthoma elasticum, Paget’s dz, Ehlers-Danlos
POHS: presumed ocular histoplasmosis syndrome
Caused by histoplasmosis fungus found in Ohio and Mississippi river valleys
Triad: atrophic chorioretinal punched out scars, PPA, no vitritis
Asymptomatic unless CNVM forms
3 symptoms of late/end-stage CNVM
Scotoma
Hallucinations (Charles Bonnet syndrome)
Floaters due to breakthru vitreous heme
2 criteria for a “subclinical” RD
1 DD of retinal detachment from the edge of the break
No more than 2DD posterior to the equator
subclinical means BIO is required to see it, cannot be detected by VF
What two types of vitreoretinal tufts are associated with RD?
Zonular: retinal projection of lens zonules
Cystic: fibroglial projection into vitreous (assoc with cystic degeneration)
Why do retinal breaks look more red when indented?
Light path from BIO hits more pigment, and is silhouetted against the RPE
Light has to take a longer pathway thru the choroid
What causes lattice, and which side of lattice is more prone to tearing?
Vitreous liquefaction over lesion and vitreoretinal adhesion at margins of lattice
Posterior side will tear more than anterior, due to direction of vitreous traction
The border farther from the ora (closer to posterior pole) → more potential for traction
Crater vs atrophic hole
Crater: more moth-eaten, not distinct borders, RPE is not fully present
Atrophic hole: all 9 retinal layers completely missing → looking directly at the RPE
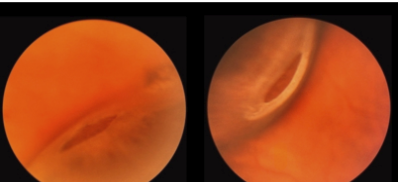
Differentiate b/w recent and chronic RDs
Recent RD: corrugations, edematous retina, bleeding from sheared BVs
Chronic RD: bullous, clear, no bleeding
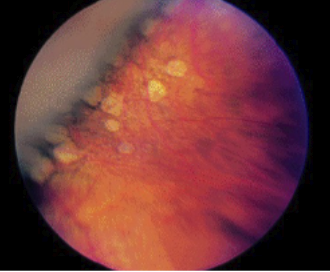
Pavingstone degeneration: multiple round punched out RPE areas
Choroidal BVs visible since RPE is thinner/missing
Common with increasing age
Benign, commonly seen bilateral
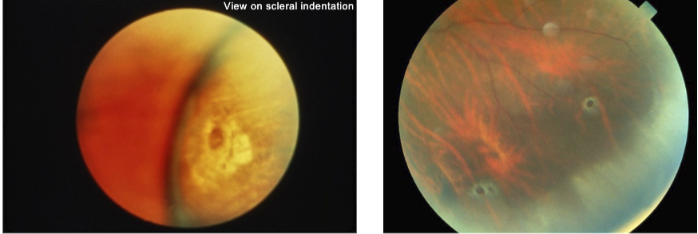
Atrophic retinal hole: break in retina not assoc w/ vitreoretinal traction
Low risk for RD (higher if fluid assoc with hole
Vitreoretinal tuft: location of strong vitreoretinal adhesion
Can result in retinal tears/holes during PVD → rhegmatogenous RD
Meridional fold
Radially oriented fold of the retina at the ora, assoc w/ oral bays
Sometimes can have retinal break at posterior border
White and slightly elevated
When to follow up for an acute asymptomatic PVD without retinal tear/detachment
If no vitreous heme/retinal break → 1 month f/u
If small vitreous heme w/ good view of fundus → 1-2 week f/u
If vitreous heme w/ poor fundus view → B-scan w/ extremely close monitoring
What occurs in a rhegmatogenous retinal detachment
Full thickness hole or tear in the retina → vitreous seeps in → retina detaches from RPE
If lattice with holes → can cause chronic RD with pigment line demarcating
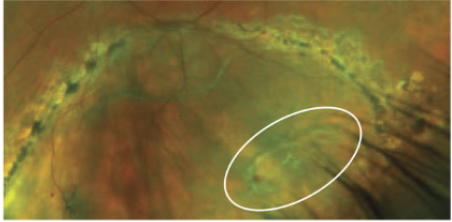
Retinal dialysis: caused by ocular trauma
Avulsion of vitreous base, when retina is pulled off ora
8-15% risk of RD (progresses slowly)
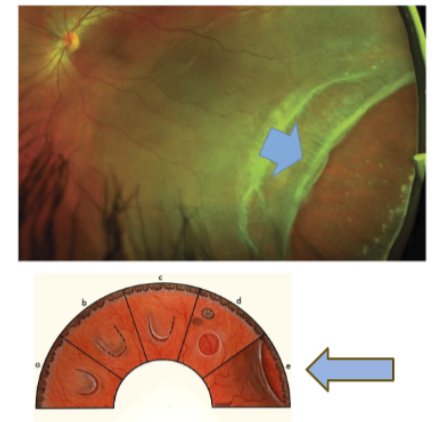
Giant retinal tear: > 3 clock hours
Can have a rolled edge bc the RD is so large
Likely caused by trauma
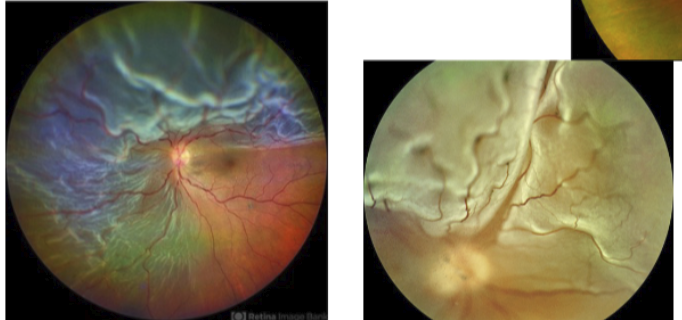
Fresh rhegmatogenous RD
Convex configuation, with an opaque corrugated appearance
Also loss of underlying choroidal pattern
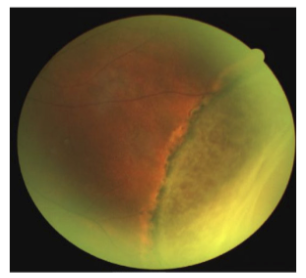
Chronid RC: retinal thinning due to atrophy
Can be confused for retinoschisis
Subretinal demarcation lines caused by RPE proliferation at the junction of flat/detached retina
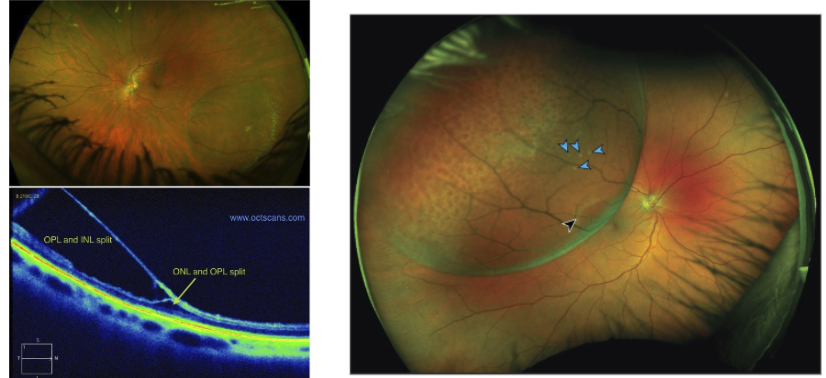
Peripheral retinoschisis: split b/w INL and OPL from viscous fluid accumulation in OPL
Most non-progressive and rarely become RRD
Smooth and dome-shaped with beaten metal appearance, and white flecks on surface
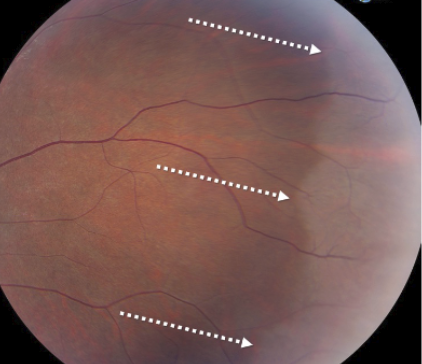
WWOP
Scalloped edges
Found in 30% of normal eyes, younger patients
Unknown cause: could be abnormal reflex or vitreous traction
No correlation with RD/breaks
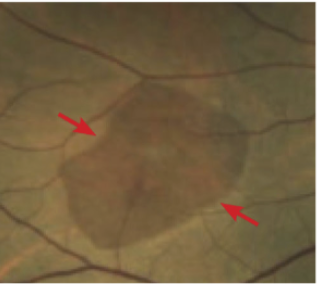
Dark without pressure
Flat, brown fundus lesion with well-defined margins in peripheral retina
Can be posterior to WWOP, can look like retinal tear
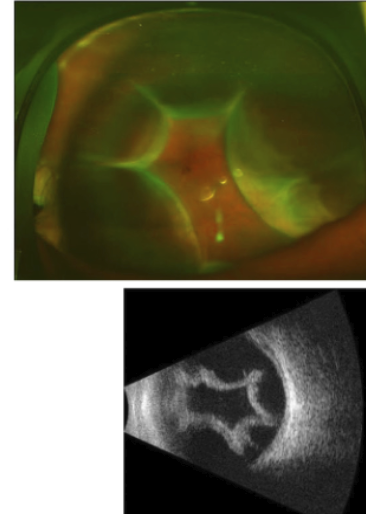
Choroidal detachment: dome-shaped elevation
Usually caused by hypotony → serum or blood accumulates b/w choroid and scleral
Ora seen w/o depression
Pathophysiology of cotton wool spots
Occlusion of precapillary arterial flow
Focal ischemia stops anterograde and retrograde axoplasmic flow → causes RNL swelling locally
Pathophysiology of microaneurysms in DR
From capillary nonperfusion → causes pericyte loss and degen of basement membrane → capillary wall bulges and outpouching forms
Becomes hyalinized over time → leakage can occur around it
Pathophysiology of flame hemes
Nonperfusion of post-arteriolar capillary bed, following NFL contour
If flame hemes only: likely vascular occlusion or HTN
DR is usually a combo of flame and dot hemes/MAs
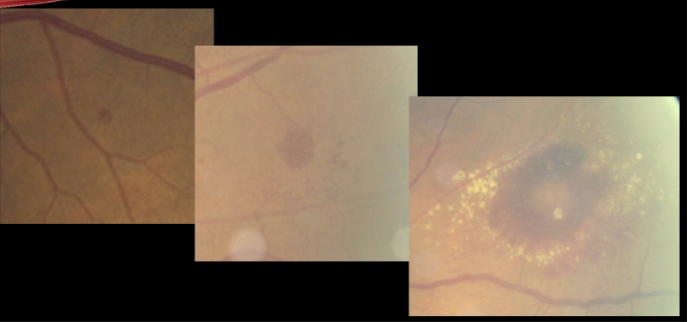
Pathophysiology of intraretinal hemes (dot/blot hemorrhage)
In INL/OPL (deep retinal capillary bed)
indicates deep retinal edema, and a sign of venous stasis
Pathophysiology of hard exudates
Exudates come from compromised deep retinal capillaries
Blood lipids and macrophages will migrate to edge of edema (circling the wagon)
Hard exudates in FAZ indicate severe edema
Pathophysiology of venous beading
Localized venous dilation that indicates severe nonperfusion
Strictures: when venous walls get so thick that blood flow is blocked
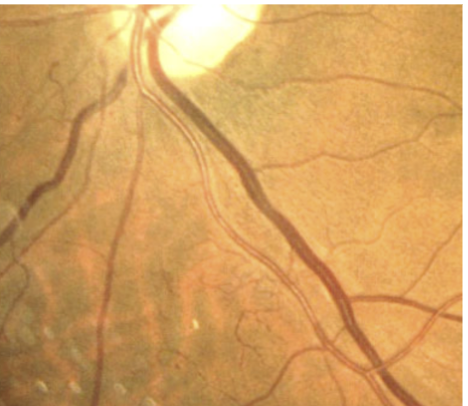
Pathophysiology of IRMA (intraretinal microvascular abnormalities)
Larger area of nonperfusion → existing capillaries dilate and endo cells proliferate
Comes before retinal NV
Pathophysiology of superficial retinal NV
Severe nonperfusion over large area = large hypoxia
VEGF causes new BVs to grow + fibrous proliferation
This causes traction on the retina
When to consider PRP treatment vs vitrectomy
Consider PRP with severe NPDR, low risk PDR
Must tx with PRP if high risk PDR
If active/severe PDR: tx with vitrectomy
Mild, mod, severe, and very severe NPDR requirements
Mild: 1+ MAs or intraretinal hemorrhage
Moderate: b/w mild and severe
Severe (one of following): 4 quadrants of MAs, 2 quadrants of venous beading, or 1 quadrant of IRMA
Very severe: 2+ of above conditions
Requirements for high risk PDR
NVD >1/3 DA
NVD with assoc preretinal/vitreous hemes
NVE >1/2 DA with assoc preretinal/vitreous hemes
Requirements for clinically significant DME
Retinal thickening within 500 um of FAZ
Hard exudates within 500 um of FAZ w/ retinal thickening
Retinal thickening area >1DD within 1DD of FAZ
Features of CRAO
Sudden painless monoc vision loss (20/200 to CF, + APD)
± amaurosis fugax
Cherry red spot, boxcarring (segmenting blood column), delayed arterial filling and AV transit time in FA
Even if retinal circulation is restored, vision loss is irreversible
What changes does CRAO cause to an ERG?
CRAO causes reduction of B wave
A wave: outer retina (PRs)
B wave: inner retina
Features of ophthalmic artery obstruction
VA: usually HM or NLP
No cherry red spot (bc choroidal blood flow also blocked)
causes pigmentary disturbance after few weeks
ERG results: reduced/absent A and B wave
Treatment for CRVO
Goal is to stabilize BRB and decrease vascular permeability
Anti-VEGF and steroids (to decrease inflammation)
Which ocular disease is most commonly associated with CRVO?
Glaucoma most assoc w/ CRVO
Advanced glc increases risk of CRVO 5-7x
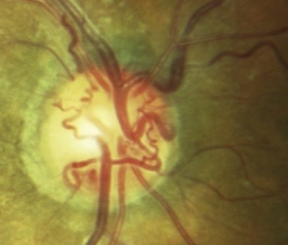
Optociliary shunt vessels
Collateral vessels connecting choroid and retinal vasculature
Most common in RVOs (but also chronic papilledema, GLC, and ON meningioma)
Features of hemi-retinal vein occlusion (HRVO)
Variant of CRVO with 2 quadrants (superior or inferior half of retina)
Caused by anatomic variation of ONH (veins draining each half are merging posterior to lamina cribrosa)
Behaves like CRVO (more than branch)
Features of ocular ischemic syndrome
Severe carotid artery stenosis/occlusion
Causes ischemic ocular pain, mid-peripheral retinal hemorrhages, vision loss
More common elderly men>women
High risk anterior seg NV
Features and side effects of PRP (panretinal photocoagulation)
Tx for ischemic retina: to kill retinal cells and reduce VEGF load and oxygen demand (NOT cauterizing BVs)
Side effects: macular edema, exudative RD, choroidal effusion, VF defects, night vision issues
What is laser creep
When scar created by focal laser expands over time
Can cause delayed vision loss, in first 3 months of surgery or years later
Solar retinopathy (photochemical mechanism of damage)
Causes color change on fundus due to damaged PRs that contain carotenoids
RPE usually no damaged
Mechanical mechanism of damage (eg laser pointer injury)
Both RPE and PRs are damaged (stronger choroidal signal)
Light energy deposits faster than mechanical relaxation
Hydroxychloroquine/plaquenil is used as which treatment, MOA and dosage
Plaquenil treats malaria or autoimmune disorders (RA, lupus)
Helps reduce inflammation and swelling, reduce WBCs, blocks platelet aggregation/adhesion
Dosage 200-400 mg per day
How does plaquenil affect the retina
Affects metabolism of retinal cells
Toxic to RPE → reduces phagocytosis of PRs → PR loss → futher RPE degeneration
Risk factors for plaquenil toxicity
Medication dosage
> 1000 g (cumulative)
> 200-400 mg per day
> 5 mg/kg daily (rare to develop toxicity below 6.5 mg/kg)
Medication duration: > 5-7 years
Monitoring guidelines for plaquenil
10-2 for non-Asian pts, 24-2 for Asian pts
After 5 years: annual screening with HVF, FAF, mfERG, SD-OCT
mfERG extremely sensitive to early HCQ toxicity (when changes are still reversible)
SD-OCT sees structural changes: ONL thickness, PIL, COST → RPE loss
FAF sees early parafoveal hyperautofluorescence (from increased lipofuscin) → becomes hypoautofluorescence due to RPE cell death