Bio 0150 Exam 4: Metabolism
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
Potential Energy
Chemical energy stored in covalent bonds is released during hydrolysis of polymers
Potential Energy
Electrical gradients across cell membranes help drive the movement of ions through channels
Kinetic Energy
Heat can be released by chemical reactions, and this can alter the internal temperature of an organism
Kinetic Energy
Light energy is captured by pigments in the eye and plant pigments in photosynthesis
Kinetic Energy
Mechanical energy is used in muscle movements within cells
Metabolism
The sum of all chemical reactions in a Biological System
Anabolism
Simple atoms + ATP —> Molecule
Catabolism
Molecule —> ATP + atoms
First Law of Thermodynamics
Energy is Neither Created nor Destroyed
Second Law of Thermodynamics
When energy is converted from one form to another, some energy becomes unavailable for work and lost to disorder
Free energy
Available Energy
Entropy
The measure of disorder in a system
ΔG
Change in free energy (usable energy)
ΔH
Change in enthalpy (Total energy; heat)
TΔS
Change in unable energy = (T);
Change in Entropy = ΔS
Absolute temperature in Kelvin
(temp in celcius + 273k)
What is the change in free energy (ΔG) for a reaction with an enthalpy change (ΔH) of 725 kJ/mol and an entropy (ΔS) change of 5kJ/mol(K) at a physiological temp of 37°C
Equation: ΔG = ΔH - TΔS
-825 kJ/mol
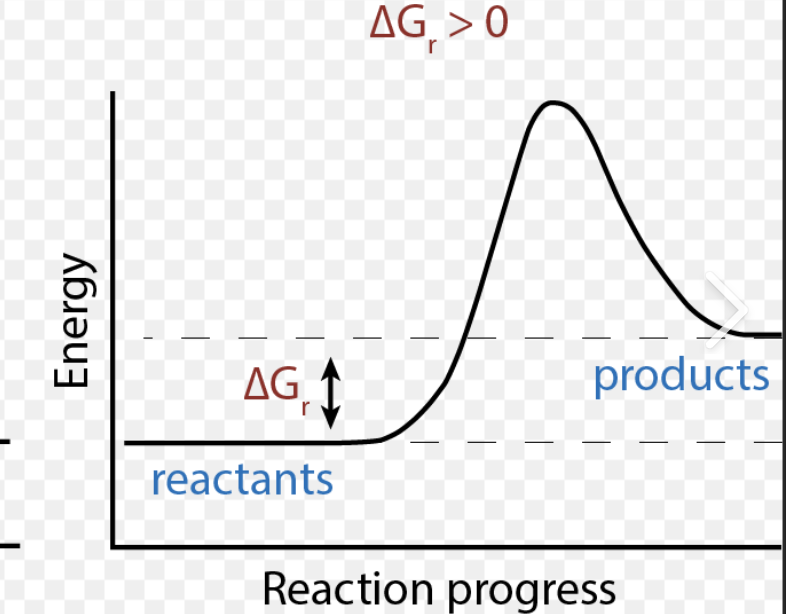
Positive ΔG
Products have more free energy
Free energy is required
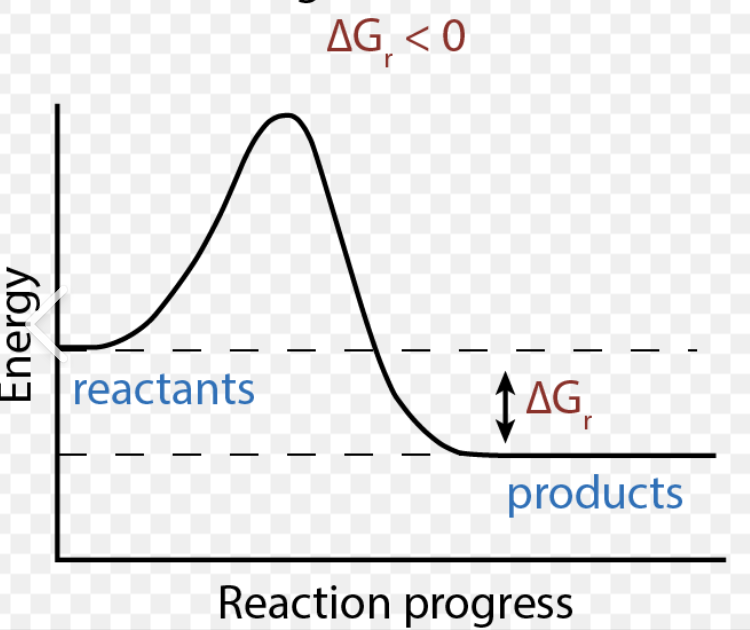
Negative ΔG
Reactants have more free energy
Free energy has to be released
-ΔG
Spontaneous (happens w/o energy input)
Exergonic reaction
+ΔG
Non-spontaneous (Needs energy to occur)
Endergonic Reaction
Chemical reaction that releases energy
Exergonic
Chemical reaction that consumes energy
Endergonic
Endergonic and Exergonic Reactions can be
Coupled
Main Energy Currency of the Cell
ATP
ATP Hydrolysis
ATP + H2O —> ADP + Pi + Free energyΔG for ATP Hydrolysis
= 7.3 kcal/mol (-30kJ/mol)
What Couples Endergonic and Exergonic Reactions?
ATP
Exergonic Processes
Cellular respiration
Catabolism (think large to small)
Endergonic Processes
Active transport
Cell movements
Anabolism (think small to large)
What is coupled to endergonic (non-spontaneous) reactions?
ATP Hydrolysis
The conversion of glycerol to glycerol 3-phosphate is an endergonic reaction with a ΔG of 2.2 kcal/mol. How can this be coupled to ATP in the cell so that the reaction is spontaneous
Equation: Glycerol + Pi → Glycerol 3-phosphate + 2.2 kcal/mol
Remember: ΔG for ATP Hydrolysis = 7.3 kcal/mol (-30kJ/mol)
-5.1 kcal/mol
The formation of ATP in the cell is an endergonic reaction with a ΔG of +30kJ/mol. How could this be tied to the following reaction to become spontaneous?
Equation: Creatine + Pi → Creatine-phosphate + 43.1kJ/mol
ADP + Pi → ATP + 30 kJ/mol
Remember: We have to adjust one of our reactions to make a -ΔG at the end of coupling
Creatine - Phosphate → Creatine Pi = -43.1 kJ/mol
ADP + Pi + Creatine-phosphate → ATP + Creatine + Pi
we have -43.1 + 30 = -13.1 kJ/mol
Explain the characteristics of ATP that account for the high free energy released during is hydrolysis to form ADP and Pi
ATP has high energy bonds b/w phosphate groups that store significant potential energy. When these bonds are broken during hydrolysis the bonds become stable
The three phosphate groups of ATP are negatively charged and repel, which creates high energy. When hydrolyzed, the repulsion is revealed and makes more stable products (ADP and Pi)
The hydrolysis of ATP is reversible, meaning cells can regenerate ATP from ADP and Pi using energy from other metabolic processes. This coupling between exergonic and endergonic reactions is important for cellular function
Catalysts
Speed up chemical reactions without being permanently altered
-Often end in “ase”
-Act as a scaffold for a reaction
Active Site
Where substrates of an Enzyme bind
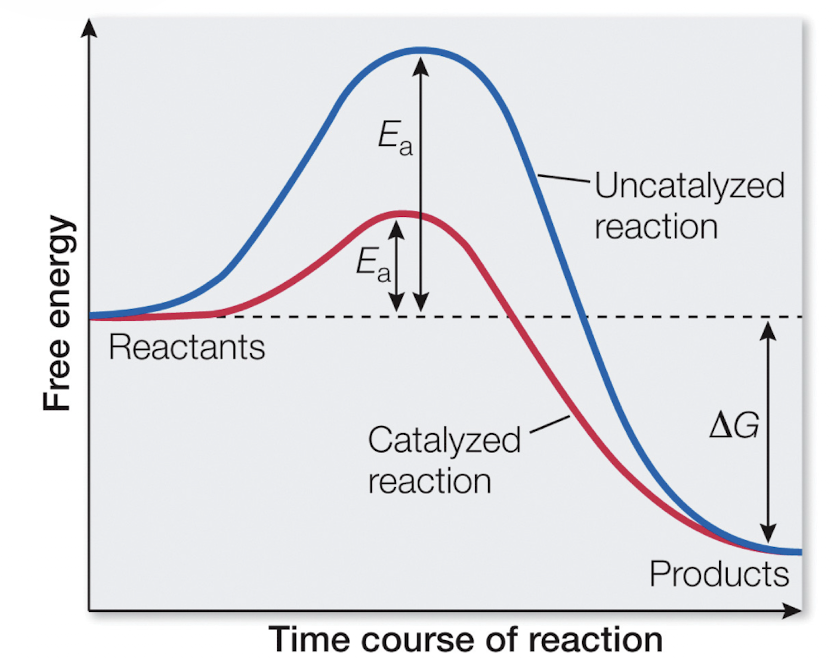
Activation Energy (Ea)
The amount of energy needed to start a reaction
Occurs for all reactions
Puts reactants into a transition state
Does not impact ΔG
What lowers the activation energy required?
Enzymes
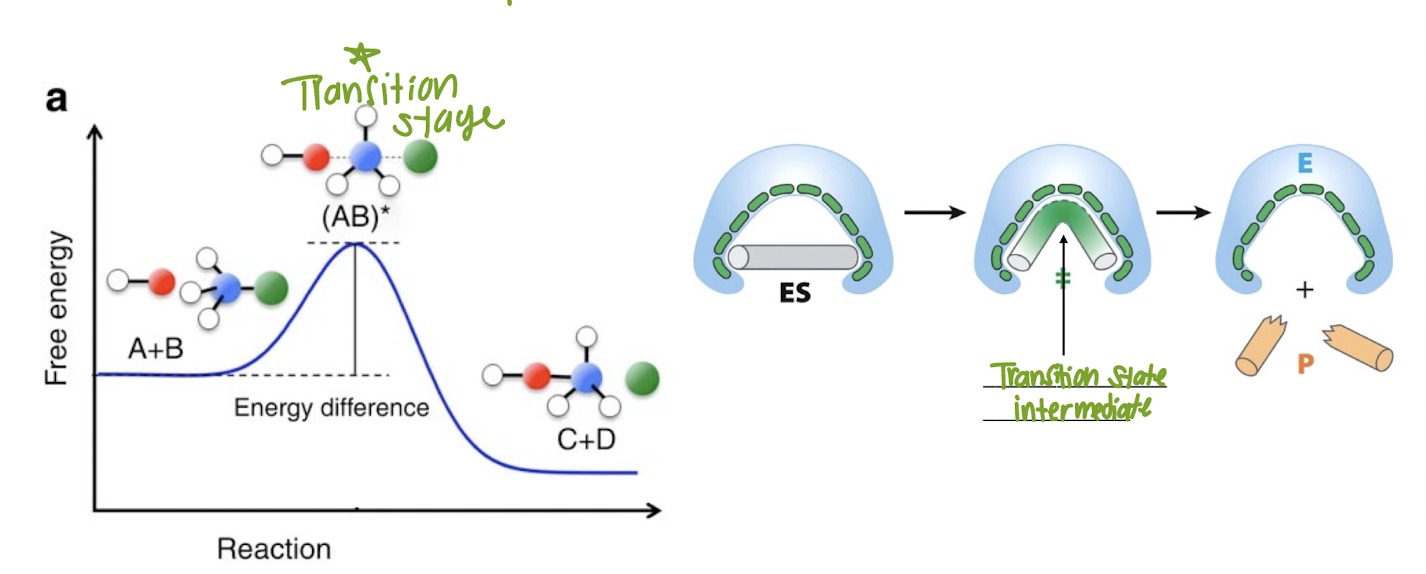
What happens when the Ea has been achieved?
The reaction will proceed
Enzyme-Substrate Complexes
Can use the same enzyme over and over again
Enzymes can lower Ea through
Correct Orientation of Substrates
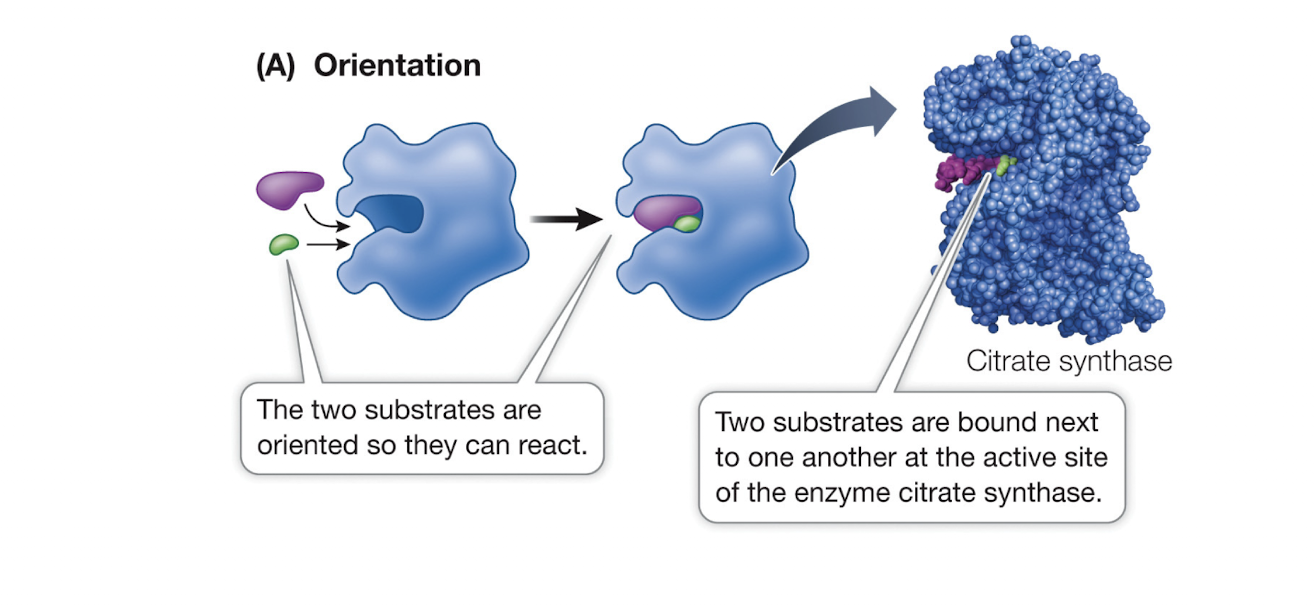
Enzymes can lower Ea by
Placing Physical Strain on bonds
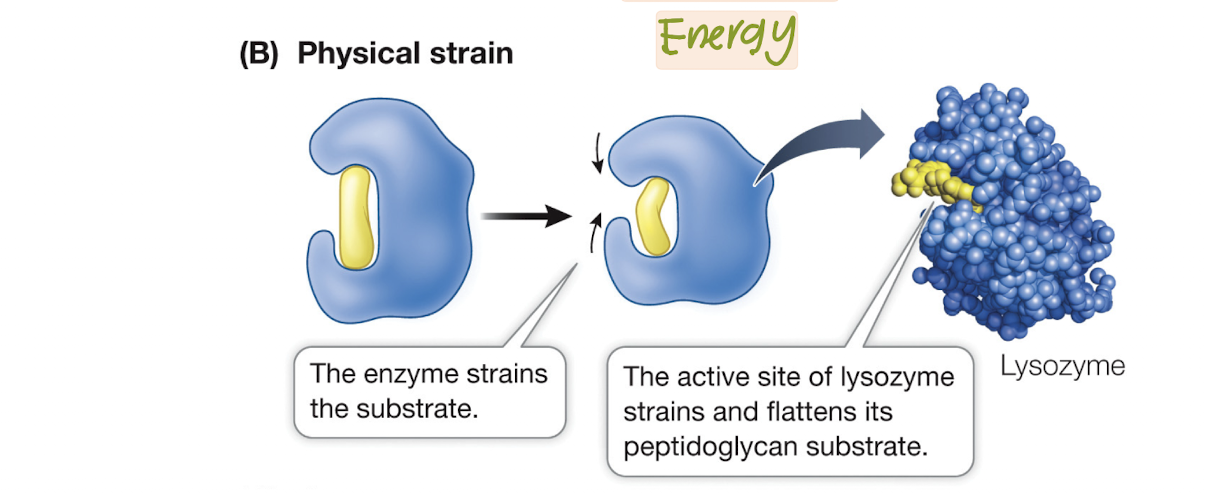
Enzymes can lower Ea by
Adding Chemical Groups
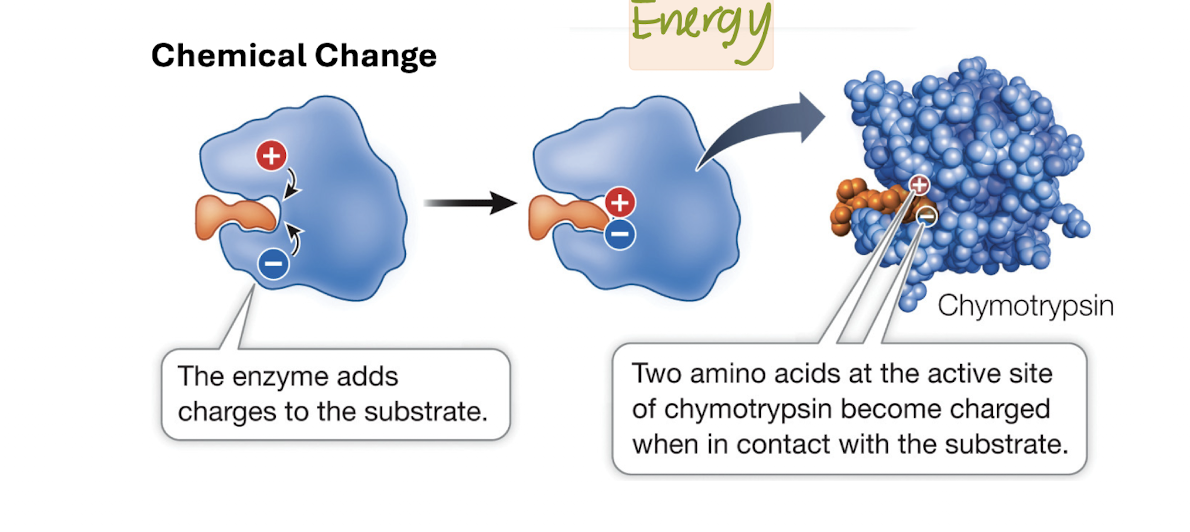
Acid-Base Chemical Catalysis
Acidic or basic R groups transfer H+ to form a substrate
Covalent Chemical Catalysis
R groups form a temporary covalent bond with a substrate
Metal Ion Chemical Catalysis
Metal ions bound to enzyme and gain/lose electrons from the substrate
The active site is specific to the substrates due to
Shape Chemistry
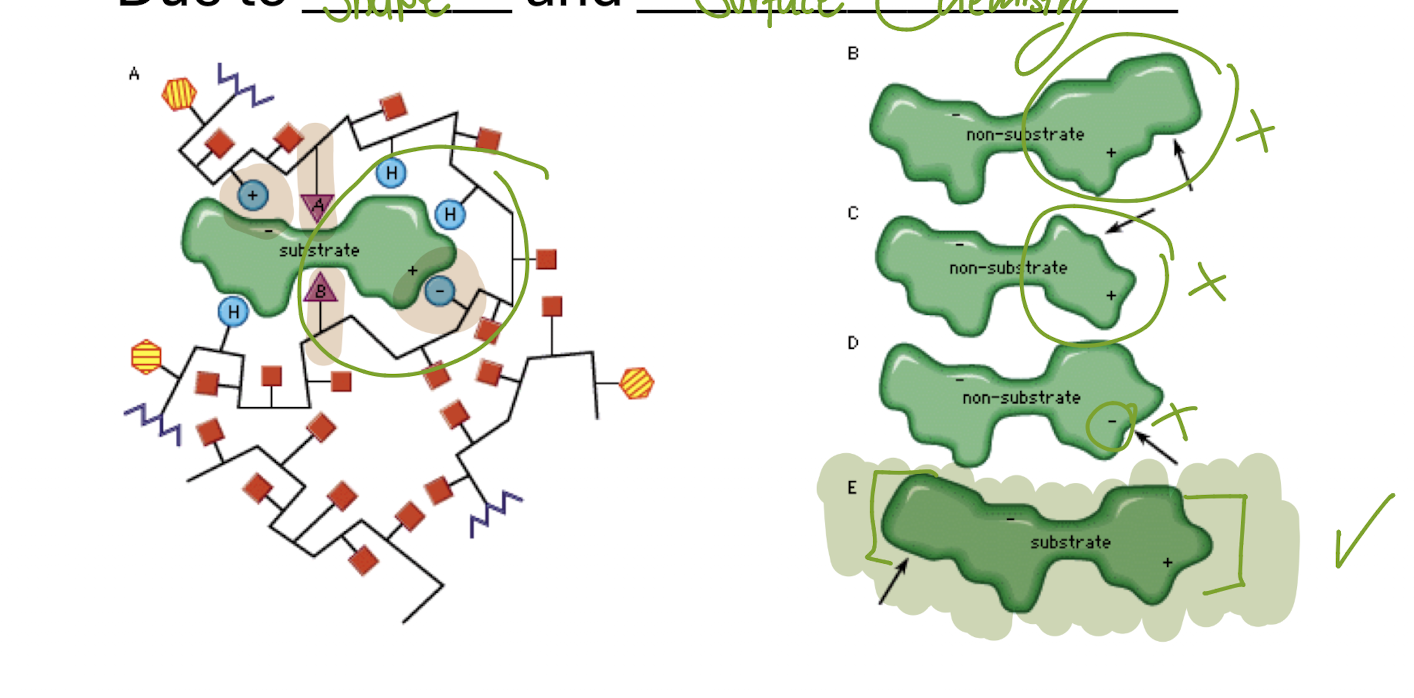
Induced-Fit Model
Active site conforms to it’s substrate’s shape
*Accurate model as opposed tot eh Lock-and-Key
When some enzymes require other molecules to function they are either:
Prosthetic Groups, Inorganic Cofactors, or Coenzymes
Prosthetic groups
Carbon-Based (organic) molecules permanently bound to the enzyme
Ex). Heme, FAD, Retinal
Heme (Prosthetic Group)
Binds ions, O2, and electrons
FAD (Prosthetic Groups)
Carries electrons/protons
Retinal
Converts light energy
Molecule Y is permanently bound to the enzyme in the active site. It will agin and lose electrons over the course of a chemical reaction
Prosthetic Group
Inorganic Cofactors
Not carbon-based (inorganic), permanently bound to the enzyme.
Iron (Inorganic Cofactors)
Oxidation / reduction
Copper (Inorganic Cofactors)
Oxidation / reduction
Zinc (Inorganic Cofactors)
Stabilizes DNA binding structure
Metal Z is permanently bound to the enzyme in the active site. During a chemical reaction, it will pass electrons between substrate 1 and substrate 2.
Inorganic Cofactor
Coenzymes
Bind to active site only during the reaction, some are organic, non-protein molecules.
Molecule X binds to the active site during a chemical reaction. The enzyme will transfer a phosphate group from molecule X to the substrate
Coenzyme