4.3 ALCOHOLS AND PHENOLS
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Halogenoalkane to alcohol
Reaction: Halogenoalkane + OH- → Alcohol + Halide ion-
Condition: Aqueous NaOH Heat under reflux
Nucleophilic substitution
OH- nucleophile (lone pair donor)
Aldehyde to Primary alcohol
Reagent: NaBH4
Conditions: Aqueous ETHANOL Room temperature
Hydride attack +carbon → potonation
Ketone to secondary alcohol
Reagent: NaBH4
Conditions: Aqueous ETHANOL Room temperature
Carboxylic acid to primary alcohol
Reagent LiALH4
Conditions: Dry ether (no water ) Dilute acid
Alcohol → Halogenoalkane
Alcohol + HX → Halogenoalkane + H2O
HCl needs ZnCl2 catalyst
HBr made in situ KBr + conc H2SO4
Tertiary → immediate cloudy (form carbocation more easily)
Secondary → Slow cloudy
Primary → No change
Alcohol + ethanoyl chloride
Alcohol + COCL → Ester + HCl
Steamy white fumes HCl gas
Fast, no catalyst
Alcohol + Carboxylic acid → Ester
Alcohol + Carboxylic acid ⇌ Ester + H2O
Conc H2SO4 catalyst
Heat under reflux
Fruity smell
Phenol and NaOH
Phenol + NaOH → Sodium phenoxide + H2O
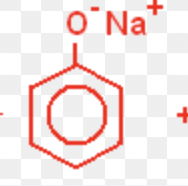
Phenol and bromine water
Phenol + Br2 → 2,4,6 tribromophenol
Orange brown bromine → Colourless
White precipitate
Room temp no catalyst
No catalyst faster than benzene
Phenols acidity
Weak acid phenol > alcohol
React with NaOH not Na2CO3
Phenoxide ion stabilised by delocalisation
Negative charge spread over benzene ring
Phenol + ethanoyl chloride
Phenol + COCL → Phenyl COCH3 + HCL
Steamy white fumes HCl
No catalyst
FeCl3 test phenols
Purple violet colour