Organic Chemistry Final Review (Mechanisms, Acids & Bases, Functional Groups)
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
SN2 reactions
Primary or Secondary Substrates ONLY
Utilizes Strong Nucleophiles
Backside attack only!
E2 Reactions
Primary, Secondary, but mostly Tertiary Substrates preferred
Large strong bases
Utilizes stealing a H off of a nearby C bond to displace the Pi electrons
SN1
Multistep reaction
Loss of a leaving group
Deprotonation
Tertiary
Racemic mixture
EI
Multistep Reaction
Typically observed in Polar Solvents (ETOH)
Utilizes Base
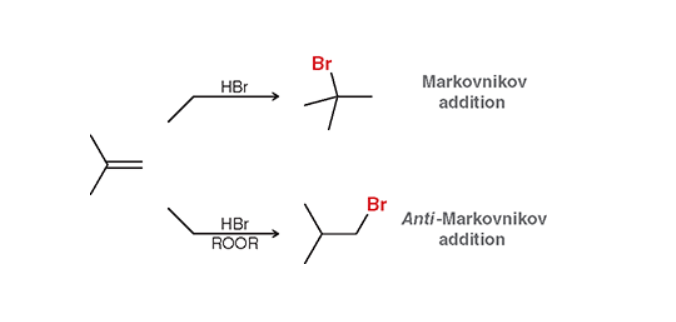
Hydrohalogenation
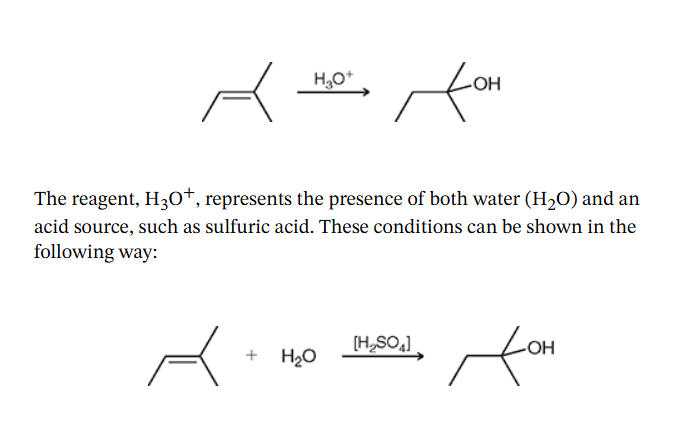
Acid Catalyzed Hydrations
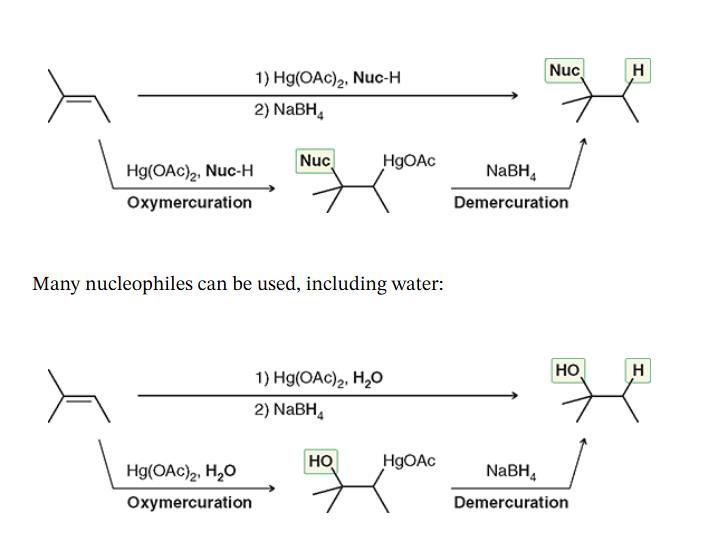
Oxymercuration - Demurcuration
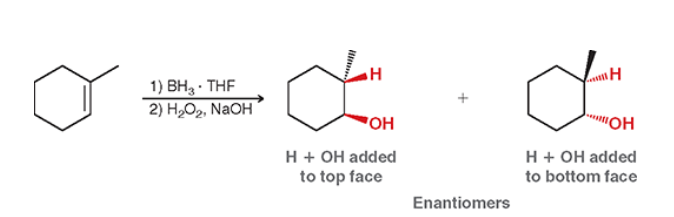
Hydroboration-Oxidation
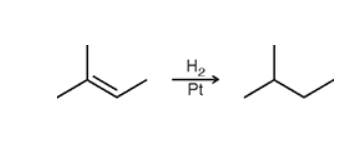
Catalytic hydrogenation
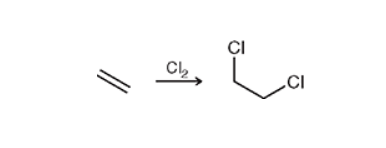
Halogenation
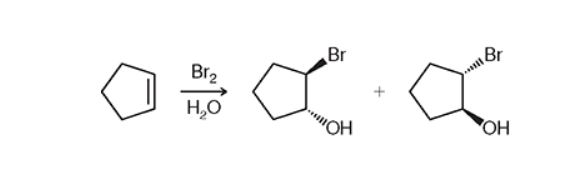
Halohydrin
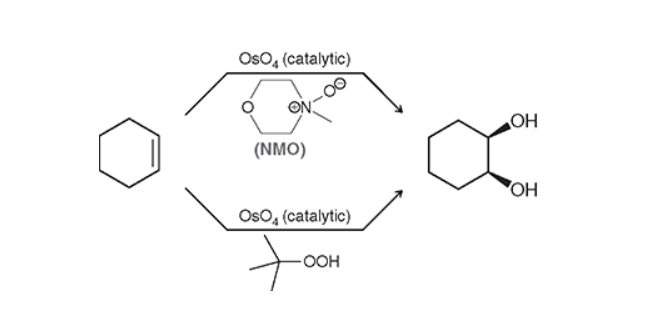
Syn Dihydroxylation (OSO4)
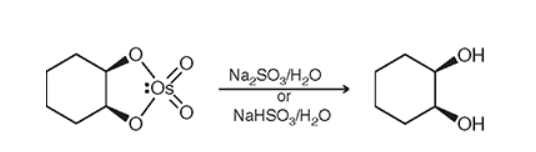
Syn Dihydroxylation (Na2SO3/H20 or NaHSO3/H2O)
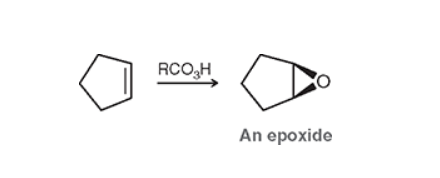
Epoxidation
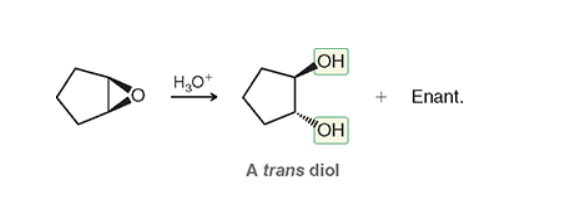
Ant Dihydroxylation
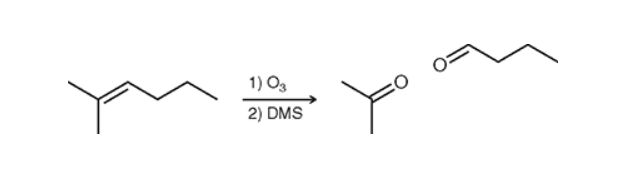
Ozonolysis
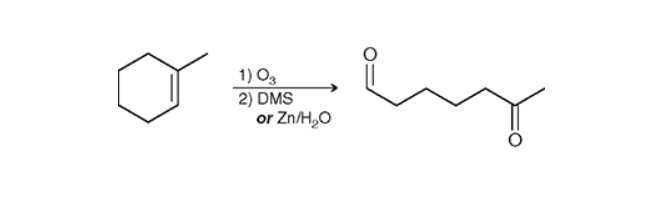
Breaking a Ring with Ozonolysis
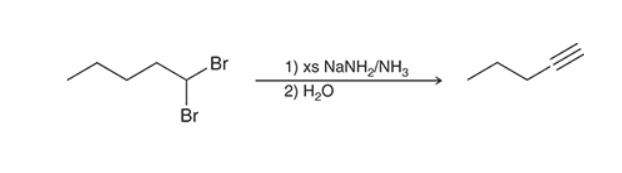
Preparation of Alkyne
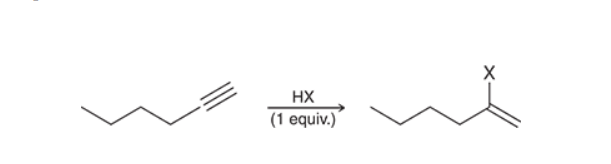
Hydrohalogenation of an Alkyne
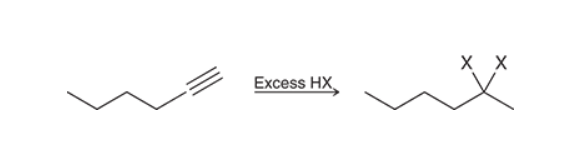
Hydrohalogenation of Alkyne with excess reagent
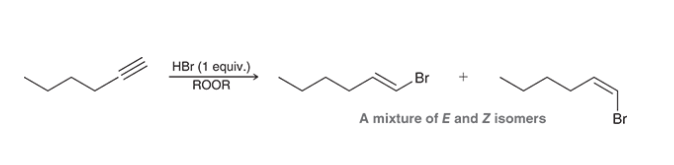
Anti Markonikov Hydrohalogenation of Alkyne
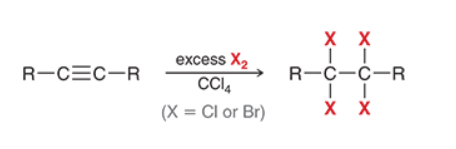
Halogenation of Alkyne (xs)
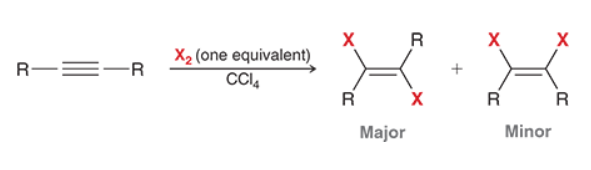
Halogenation of Alkyne
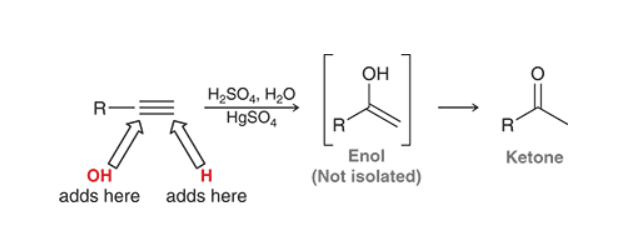
Acid Catalyzed Hydration of Alkyne
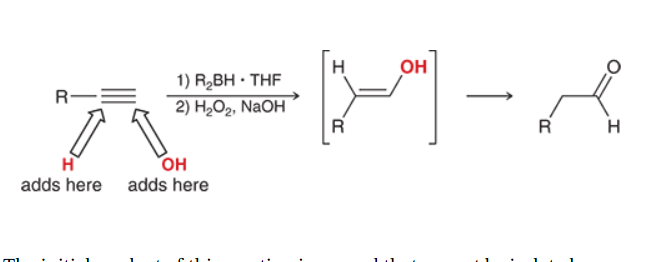
Hydroboration of Alkynes
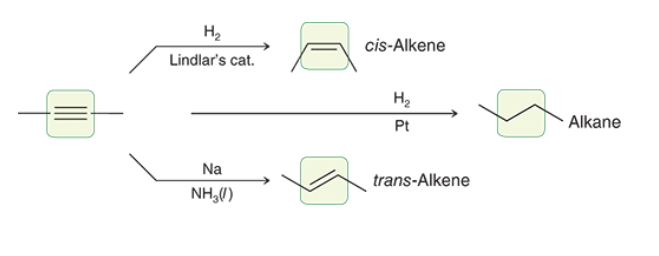
Catalytic Hydrogentation, Lindlars Treatment, and Dissolving Metal Reduction
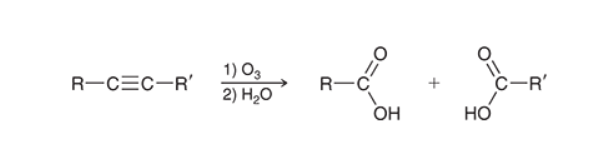
Ozonolysis of Alkynes (Internal)
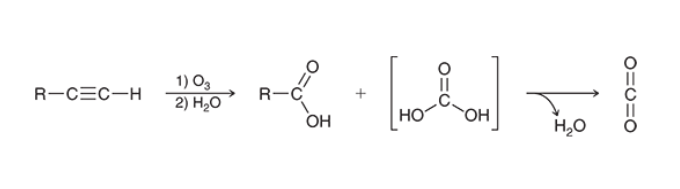
Ozonolysis of Alkyne (Terminal)
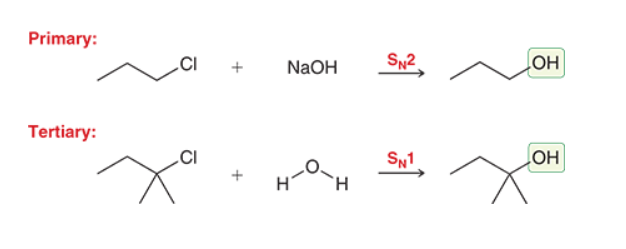
Preparation of Alcohols using Primary or Tertiary atoms
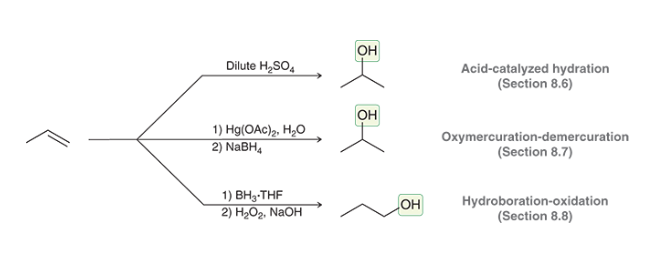
Addition Reactions of Alkenes making Alcohols
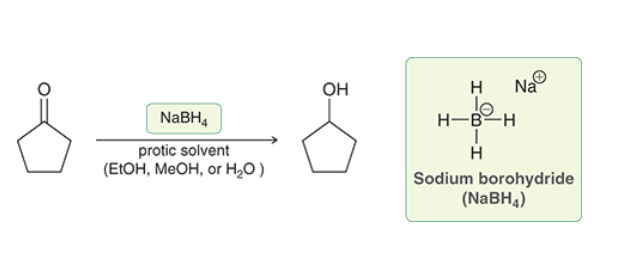
Reduction of Alcohols
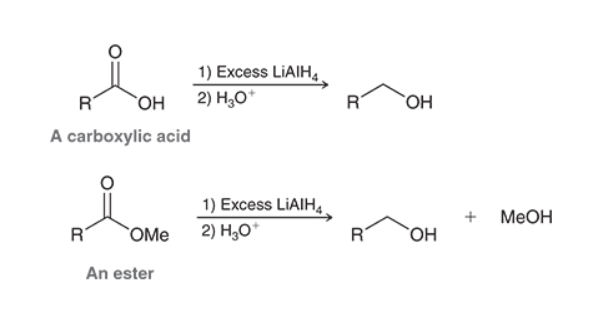
Reduction with LiAlH
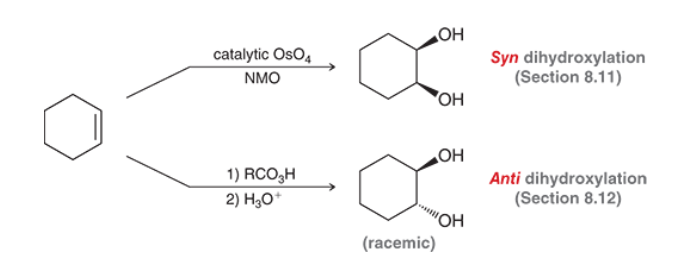
Prep of Diols (Syn vs Anti)
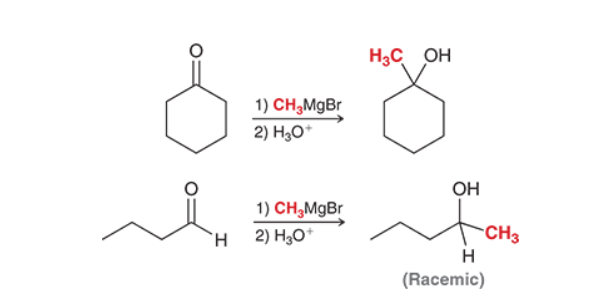
Prep of Alcohols via Grignards
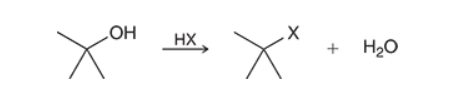
Substitutions of Tertiary Alcohols
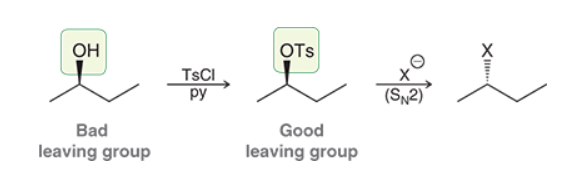
Substitutions of Primary or Secondary Alcohols
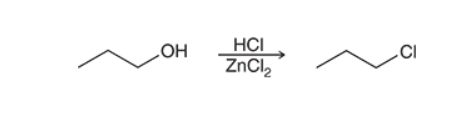
Substitutions of Primary Alcohol (Lucas Reagent)
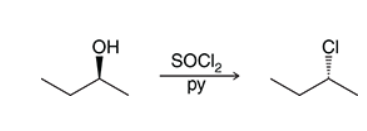
Substitution of Primary and Secondary Alcohols (Thionyl Choride)
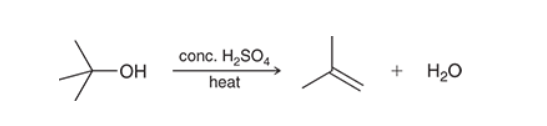
E1 reaction of Alcohols
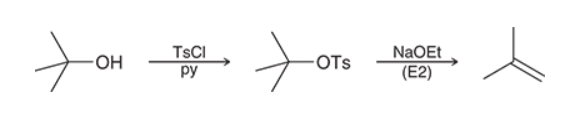
E2 Reactions of Alcohols
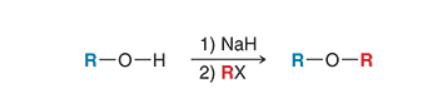
Williamson Ether Synthesis
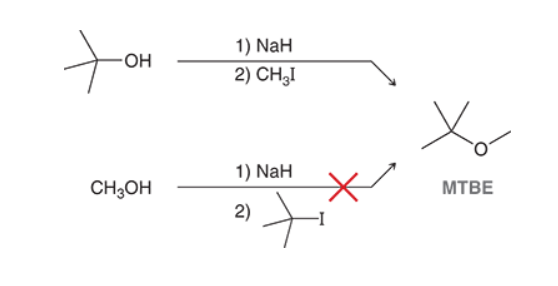
Creation of MTBE
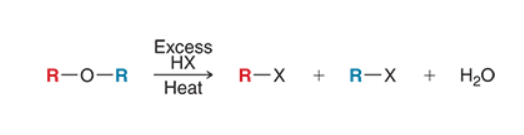
Acidic Cleavage
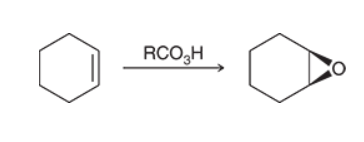
Preparation of Epoxide
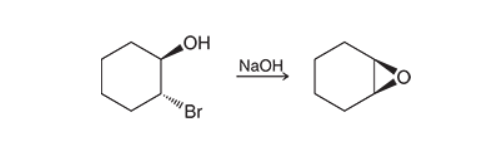
Preparation of Epoxides with Halohrydrins
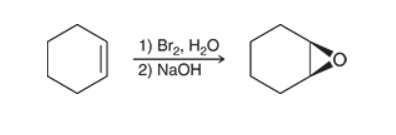
Preparation of Epoxide with alkenes
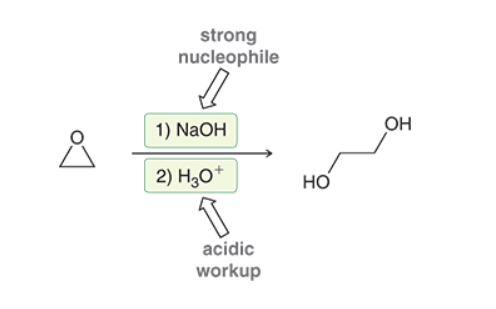
Ring Opening of Epoxides