Genetic Therapeutics and the Multi-Hit hypothesis of neurological disease
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
What is the function of the SMN1 gene? What does mutation in SMN1/2 cause in terms of protein function? Why is SMN2 beneficial?
Involved in spliceosome assembly and mRNA transport (possibly neuronal development)
telomeric gene (susceptible to rearrangement/deletion (damage)
SMN2 is a duplicate of SMMN1, with C>T at 840 resulting in deleted splice site in exon 7 > non-functional protein
SMN2 produces a small amount of functional protein, which may compensate for SMN1 loss in SMA
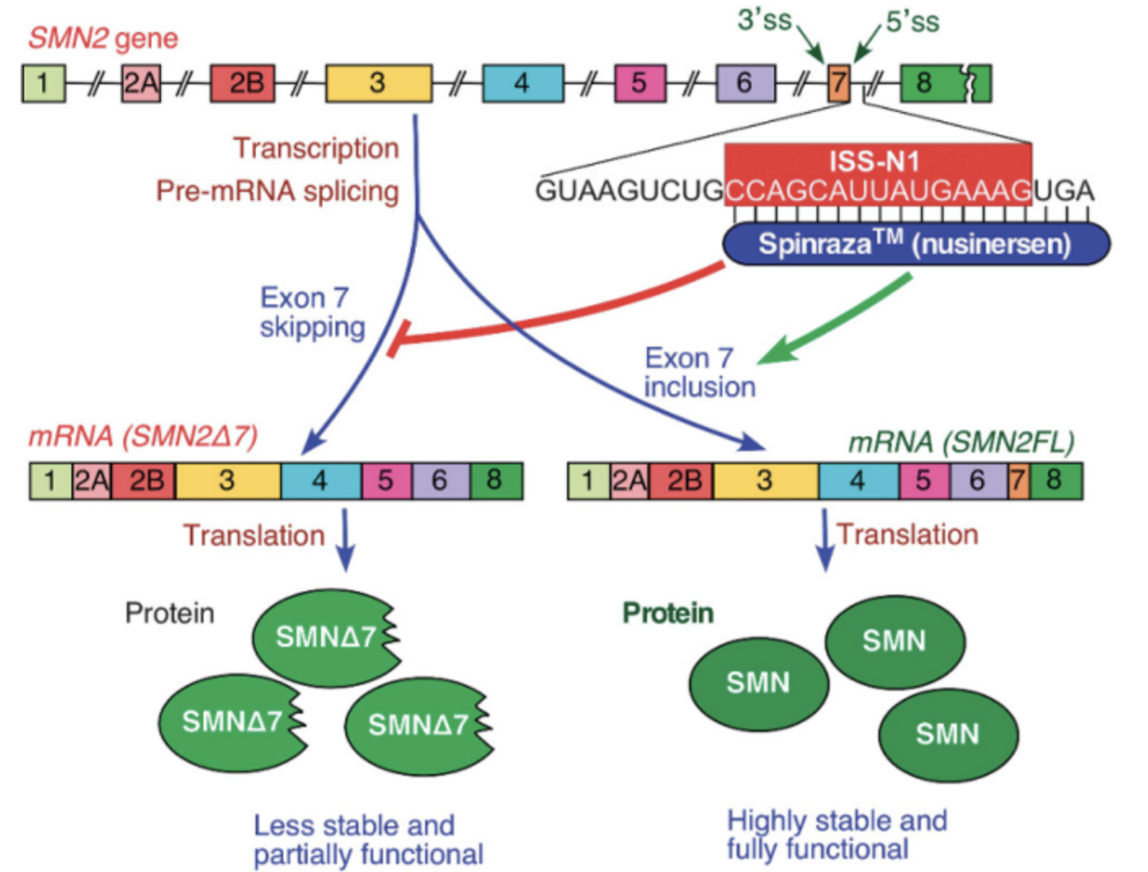
How have ASOs shown promising results in transforming mutated SMN2?
Intronic splicing silencer N1 (ISS-N1) regulates exon 7 in SMN2 - when ISS-N1 is deleted, exon 7 inclusion is promoted
ASO blocking ISS-N1 (Nusinersen) showed ability to convert truncated SMN2 into full-length, funcitonal SMN2
administered intrathecally (due to BBB)
Clinical trial: extended event free and overall survival, and improved achievement of motor milestones.
FIRST APPROVED SMA TREATMENT
initially 4 monthly intrathecal injection followed by 1 every 4 months - discounted on NHS
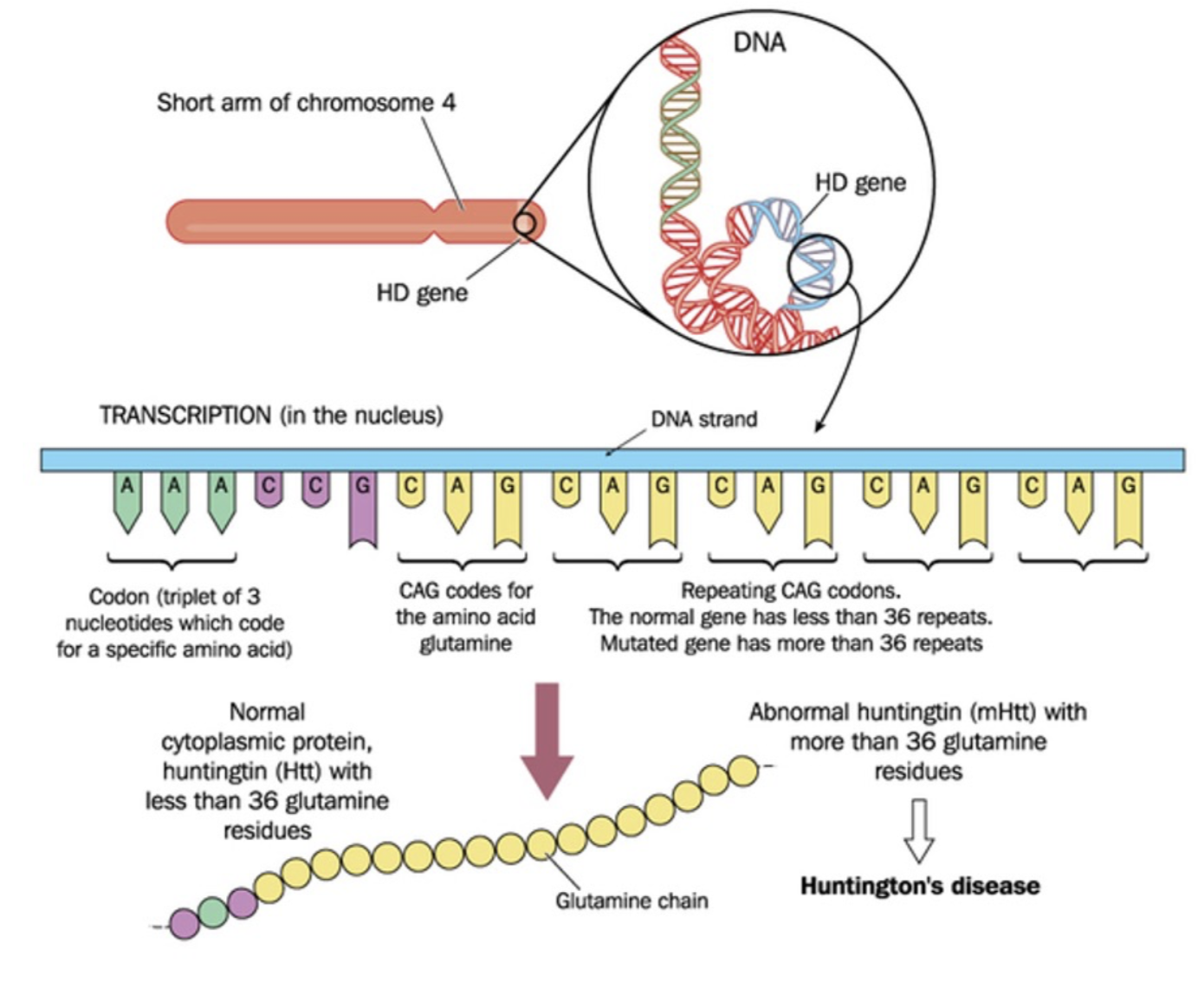
What makes Huntington’s disease a good candidate for ASOs?
HD caused by high penetrance of CAG trinucleotide repeat expansion of Huntingtin (HTT) in exon1 - extends multiple glutamines in protein chain > neurotoxic
HTT thought to be involved in transcription cell signalling and intracellular transport proteins
recent research shows faulty DNA replication increases repeats, leading to later-life onset
How does the formation of HTTexon1 promote HD pathology?
HHT pre-mRNA with high CAG-repeats can undergo cryptic polyadenylation, activating sites in intron 1 - forms pathogenic HTTexon1
HTT + HTTexon1 have the capacity to aggregate and form inclusion bodies - begins in dorsal striatum in basal ganglia, impairing cognitive and motor function > eventually spreads through cortex
Describe the effects and efficacy of AMT-130 miRNA. What is a key limitation of this treatment?
AMT-130 is an ARV containing an artificial miRNA targeting mutant HTT
reduced mutant protein level in mouse models up to 7 months post-injection
Gene is expressed constitutively as it is not integrated into DNA (or under chromatin control) - as neurons are quiescent, AAV remains in cell indefinitely
REQUIRES INVASIVE SURGERY - not available in many locations
administered in one MRI-guided stereotactic surgery via cannulas direct into the caudate and putamen in the striatum via 3 infusions per hemisphere over 8-10 hours - then transported into other brain regions through axon tracts
early results show improvement in function over 3 years in Phase I/II - used historical HD data as control - opposed to sham control -
Define the diagnosis problem in ALS. What is the leading prospective biomarker for ALS diagnosis?
Generic initial symptoms of most neurodegenerative disease, as well as a lack of target for genetic testing and targeting - diagnosis generally comes from ruling out all other neurodegenerative disease
~1 year after first symptom, often too late for treatment
Highlights importance of biomarkers - non identified, leading candidate is neurofilament, however this is still generic to several neurological disease
How has SOD1 ASO done in clinical trials?
ASO against human SOD1 (Tofersen) in SOD1G93A mice decreased levels of target RNA and protein - accompanied by extension of survival by ~37%
clinical trial (VALOR to open label) suggests clinical benefit
Give examples of gene prevalence in different ethnic populations? Why is it important to understand these genetic differences?
SOD1 particularly prevalent in ALS in Asian populations
C9oprf72 more prevalent in Western countries
APOE4 was found to have no effect on memory impairment in African American populations, suggesting ApOE4 may have a protective effect in this cohort
Highlights importance of diverse studies for optimal personalised medicine.
Define mono/polygenic, penetrance and expressitivity and oligo/polygenic.
Monogenic disease: gene mutation causes disease
Polygenic disease: multiple genes play a role in development of disease, often coordinated with other influential factors such as environment
Penetrance: whether someone with a gene mutation will develop disease
Expressitivity: degree to which the mutation will cause symtoms
Oligogenic: diseases are those where very few genes determine if a disease is present
Polygenic: diseases are those where many genes are required to cause disease - disease state is often more heterogenous
Describe the oligogenic link of DYNCH1H1 with ALS genes.
DYNCH1H1 mutations + Loa mutations + SOD1 mutations > extended survival in mice
DYNCH1H1 modifies disease - slowing neuronal transport delays buildup of neurotoxic SOD1, prolonging life expectancy
TDP-43 show a different oligogenic link
TDP-43M337V mouse model normally show limited aggregation of TDP-43, when DYNCH1H1 Loa is introduced, aggregation increases - this is excasserbated in WT mice
Suggests DYNCH1H1 drives pathology but mutant properties of TOP-4343M337V attenuates some pathology
Give an example of evidence for ALS being both oligogenic and polygenic.
C9orf72 and SOD1 are strong oligogenic risk genes, whilst mutations such as ATXN2 may require other genes such as SETX and SQSTM1 for disease, and is therefore polygenic.
Define the multi-hit hypothesis of neurodegenerative disease.
Initially a concept in cancer, carried into neurodegenerative disease
genetic risk for developing disease is fixed, however risk caused by environmental/lifestyle variables can change with time.
accumulation of risk factors is sufficient for the onset of neurodegenerative disease
More risk factors required = later average onset
How many steps are taken to ALS, how is this affected with the presence of mutations? How many steps are required for other neurodegenerative disease?
6 steps to ALS - however reduced with mutation
C9orf72: 3, SOD1: 2, TDP-43: 4
Other neurodegenerative disease:
AD - 13
FTD - 7
PD - 7
CJD - 8
Some diseases, such as MS show no multi-step process - driven by autoimmunity
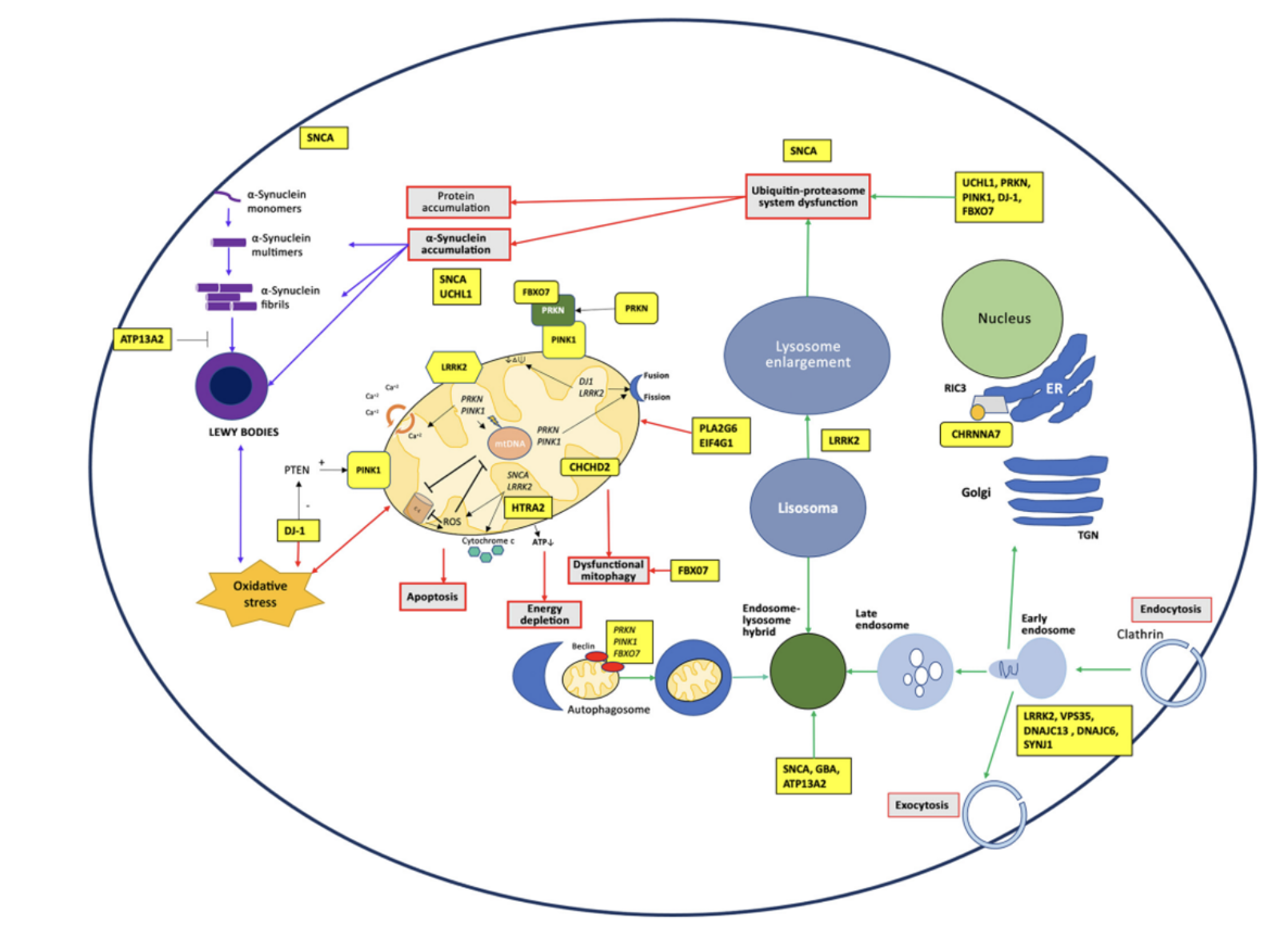
Describe PD pathology
Movement coordination disorder caused by loss of dopaminergic neurons in substantia nigra pars compacta, accompanied by increase in activated microglia - pathology shows aggregated alpha-synuclein in neurons
Describe how cellular pathways are affected in PD
LRRK2 is the most common dominant variant, linked to mitochondria and lysosomes - two pathways most affected in PD
SCNA (codes for a-synuclein) is involved in vesicle trafficking
How is acetylation affected in PD?
Acetylation restricted to midbrain, where dopaminergic neurons are primarily located, showing regional and system specificity - correlated with decreased HDACs
GWAS showed 21 hyperacetylated genes in PD including:
genes causing heritable PD - SCNA, MAPT, APP
involved in disease pathology - SCNA, PRKN, PARK7
involve in complex Parkinsonism - FBOX7, POLG
Some of these genes also indirectly regulate acetylation - also increased in MPTP mouse models
What are some exogenous factors associated with PD risk?
Pesticides - inhibit HDAC activity
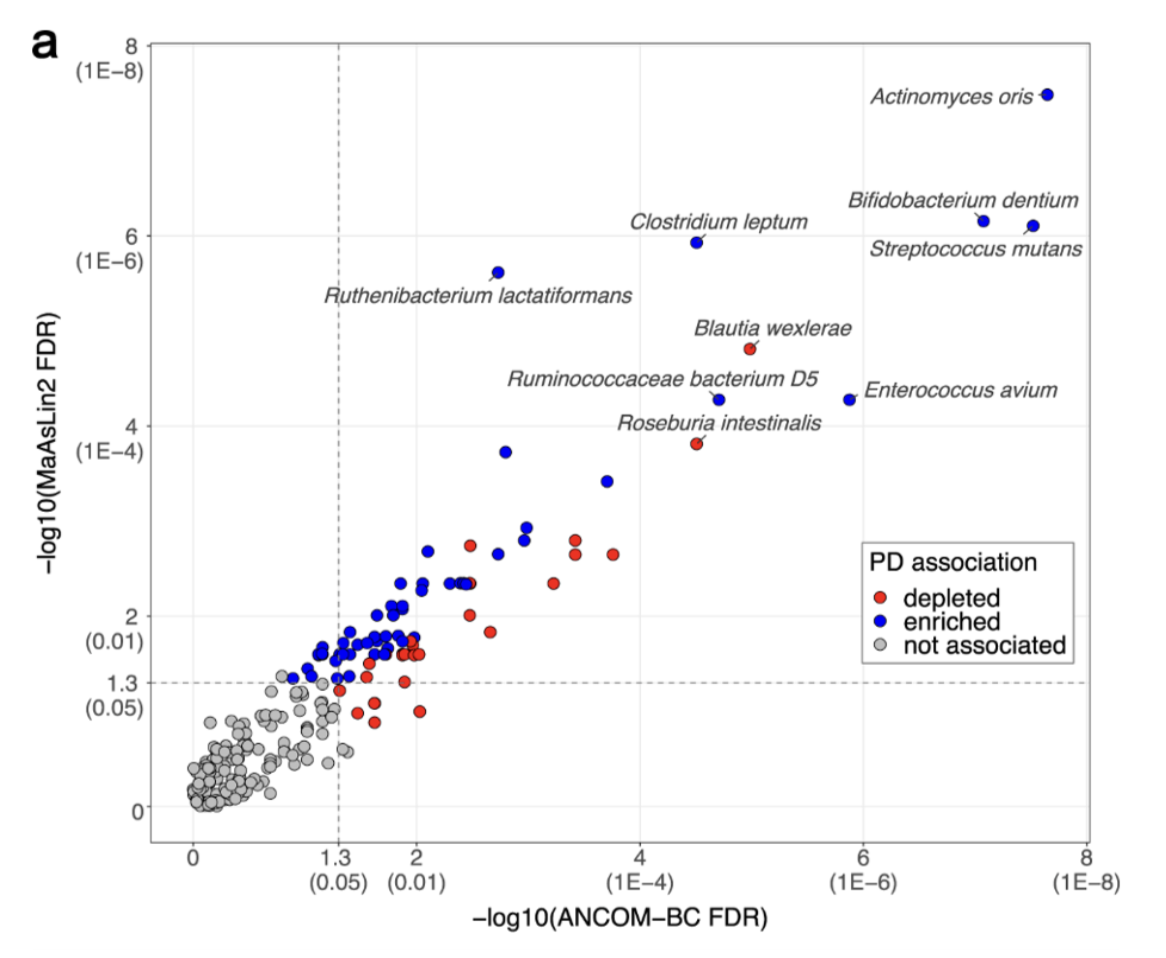
Gut bacteria - promote a-synuclein aggregation, high levels of gut bacteria associated with PD pathology found in PD patients (figure)
Immune system - immune cells express high LRRK2, PD onset is linked with IBD and influenza