Carbohydrates_part1_-_Copy-combined
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What are Carbohydrates
The three main nutrients required for human survival, primarily used as an energy source and structural component.
Monosaccharides
Simple sugars that contain 1 sugar unit, e.g. glucose, galactose, fructose.
Disaccharides
Sugars that contain 2 monosaccharide units, e.g. sucrose, lactose, maltose.
Oligosaccharides
Carbohydrates that contain 2 to 10 sugar units, e.g. fruco-oligosaccharides.
Polysaccharides
Complex carbohydrates that contain more than 10 sugar units, e.g. starch, glycogen, chitin, and cellulose.
Aldose
Monosaccharide that contains an aldehyde functional group, typically found at the terminal end of the molecule.
Ketose
Monosaccharide that contains a ketone functional group found between carbon atoms in the molecule.
Glycosidic linkage
A bond formed between monosaccharides when the -OH group from one sugar combines with the -OH from another sugar. This bond results in the formation of a disaccharide or polysaccharide, releasing a molecule of water in the process.
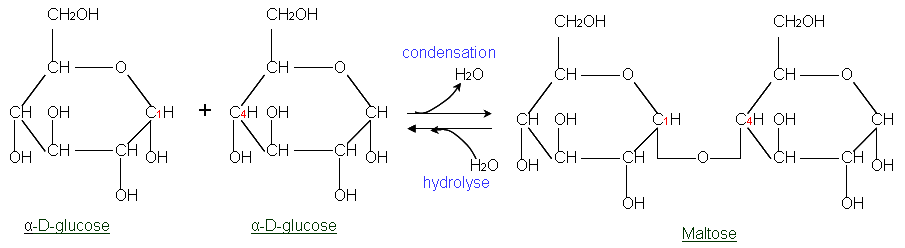
Anomerization
The formation of anomers, which are stereoisomers of a sugar that differ in configuration around the anomeric carbon. This process involves the conversion between the alpha and beta forms of a cyclic sugar.
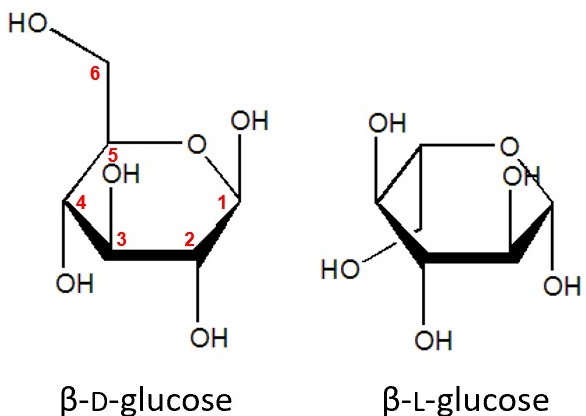
Glucose
A six-carbon sugar (C6H12O6) that serves as a key energy source in the body.
Fructose
A six-carbon sugar (C6H12O6) commonly known as fruit sugar, present in high quantities in sweet fruits.
Galactose
A component of the disaccharide lactose that is found in milk.
Amylose
A type of starch consisting of long unbranched chains of D-glucose residues joined by α-(1,4) glycosidic linkages.
Amylopectin
A branched form of starch containing both linear glucose chains and α-(1,6) glycosidic linkages.
Hydrolysis
The chemical breakdown of a compound due to reaction with water, particularly important for digestion of carbohydrates.
Cyclic form (Haworth structure)
A more stable representation of sugars with four or more carbons in a ring structure.
D-sugar
A sugar that has the -OH group on the right in the Fischer projection.
L-sugar
A sugar that has the -OH group on the left in the Fischer projection.
α-(alpha) anomer
A cyclic sugar form where the OH group on the anomeric carbon points down.
β-(beta) anomer
A cyclic sugar form where the OH group on the anomeric carbon points up.
Reducing sugar
A sugar that can donate electrons to another chemical, reducing it.
Non-reducing sugar
A sugar that cannot act as a reducing agent due to the absence of a free aldehyde or ketone group.