E109 Midterm 1
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
Intracellular fluid
Fluid inside cells
Extracellular Fluid
Fluid not inside cells (includes plasma and interstitial fluid)
Plasma
Liquid fraction of blood (blood - cells)
Interstitial fluid
extracellular fluid not inside cardiovascular system
Most common cation/anion in extracellular fluid
Na+ (Sodium), Chloride (Cl-)
Most common cation in intracellular fluid
K+ (potassium)
Homeostasis
stability of internal environment, maintained by exchange between internal and external environments.
Mass Balance
Total = Existing + Intake + Production - Excretion - Broken down
Electroneutrality
Positive charges = Negative Charges
A cell has an internal solute concentration of [Na+] = 14mM, water enter the cell and the cell volume doubles. What is the final internal [Na+] after the water enters the cell?
7mM ( CV= CV)
How to find unknown volume of a solution to which you have added a known amount of something
concentration = mass added/ volume (C = m/v)
Concentration = how much solute in liquid
Volume = how much liquid
Molecules that easily cross membrane
Nonpolar, small molecules (O2, CO2, steriods)
Molecules that cross membrane but not as readily
Small polar molecules (H2O, urea)
Molecules that do not cross membrane
Ions (Charged) and Large polar molecules (Glucose)
What affects membrane permeability?
Polar/Non polar
Large/Small
Changed/ Uncharged
Diffusion
Driven by random movement (no energy needed)
High concentration →low concentration
Which direction will a single molecule move?
Can equally move left or right (random movement)
Which direction will many molecules move?
High concentration →low concentration (net movement)
When there are the same amount of molecules in each side:
Both sides exchange the same amount of molecules (no net diffusion).
When one side has more molecules than the other side:
The side with more molecules will have its molecules moving to the other side. But the side with less molecules will not have as many molecules to move to the other side. So more molecules will be entering the side with less molecules (high → low concentration).
Fick’s Law (UNDERSTAND CONCEPTUALLY)
J = - DA (C avg /X avg)
J = Rate of diffusion across a membrane (mol/s)
D = diffusion coefficient (m²/s^-1)
A = surface area (m²)
C/X = concentration gradient (mol/m³)
C avg = C1 - C2
Greater the area = greater the diffusion rate
Greater the concentration gradient = greater the diffusion rate
If C1=C2, C avg= 0, so J =0
Simple Diffusion
Can pass right through membrane lipids without protein
Channel facilitated diffusion
Molecules go straight through channel
Carrier facilitated diffusion
Molecule enters through opening side (leaving side closed), once molecule inside protein, leaving side opens and opening side closes. Conformation change occurs.
Cell keeps facilitated diffusion going without using ATP when transporting glucose by:
Converting glucose/ using it up so there’s still a lower concentration of glucose inside the cell
Properties of protein mediated transport across membranes
specificity - protein is specific about what it is transporting
affinity - strength of binding (binding too strong = slow down transport, no binging = no transport)
saturation - percent of all binding sites occupied
competition - if two different solutes can bind to same protein
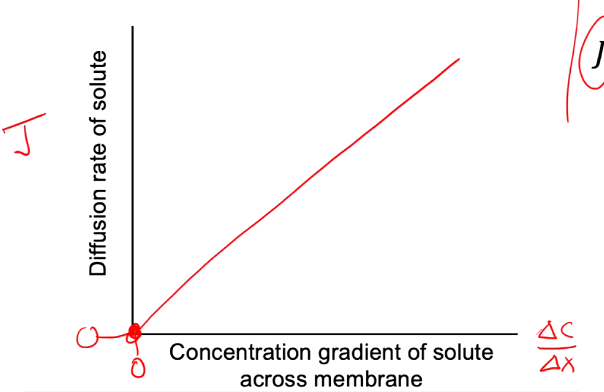
Simple Diffusion
J and (C/X) are linear
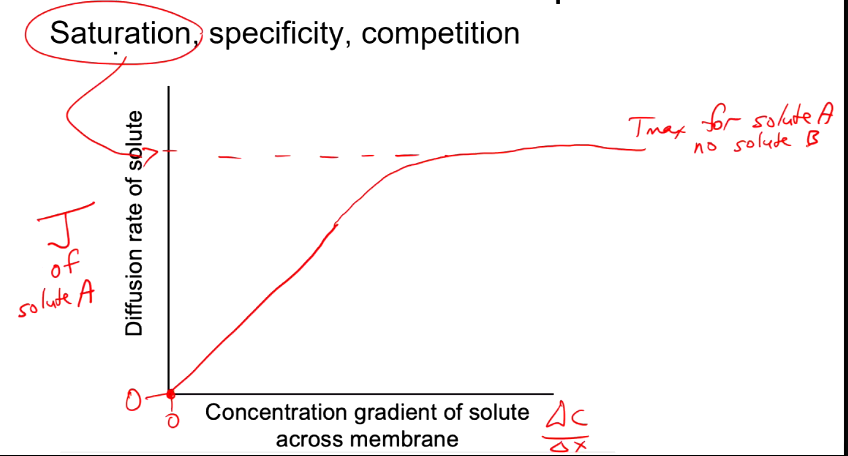
Carrier facilitated diffusion (Saturation)
Reaches max rate of transport (saturation is 100%) when all protein carriers are full
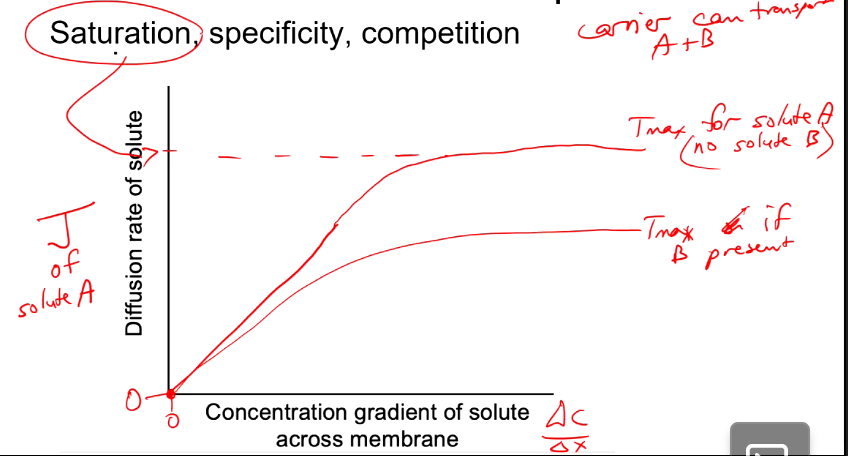
Carrier facilitated diffusion (Specificity)
Reaches max rate of transport earlier because all protein carriers are full
Passive Transport
Molecules move themselves down gradient
Simple diffusion
Facilitated diffusion
Active Transport
Movement in opposite direction, requires energy
Primary active transport (uses ATP at the same time)
Secondary active transport (uses energy stored by action of primary transport)
Water transport across membranes
no carriers for water
no active transport for water
channels for water (aquaporins)
water will move down ITS concentration gradient via diffusion
Molarity
number of moles/liter
Osmolarity
number of