Acids, Bases and Salt Preparations (2d)
1/24
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Solubility: Sodium, Potassium and Ammonium Salts
All soluble
Solubility: Nitrates
All soluble
Solubility: Chlorides
most, except Silver and Lead(II)
Solubility: Sulfates
most, except Barium, Calcium and Lead(II)
Solubility: Carbonates
most insoluble, except Sodium, Potassium and Ammonium
Solubility: Hydroxides
most insoluble, except Sodium, Potassium and Ammonium
(Calcium Hydroxide is slightly soluble)
Proton Transfer: Acids
proton donors
ionize in solutions → produce protons, H+ ions
H+ ions make aqueous solution acidic
ex: HCl
HCl (aq) → H+ (aq) + Cl– (aq)
Proton Transfer: Bases
proton acceptors
ionize in solutions → produce OH- ions that accept protons
OH- ions make aqueous solution alkaline
ex: NaOH
NaOH (s) → Na+ (aq) + OH– (aq)
Metal + Acid Reactions
only metals above hydrogen react with dilute acids
more reactive metal = more vigorous reaction
K and Na: dangerous, explosive reaction
reaction forms salt and hydrogen gas
Metal + Acid general equation
metal + acid → salt + hydrogen gas
Acid + Base Reactions
produces salt and water
identity of salt depends on acid used and base’s positive ions
2HCl + CuO ⟶ CuCl2 + H2O
H2SO4 + 2NaOH ⟶ Na2SO4 + 2H2O
HNO3 + KOH ⟶ KNO3 + H2O
Acids + Metal Carbonates
forms metal salt, carbon dioxide and water
distinguishable because of effervescence caused by carbon dioxide gas
2HCl + Na2CO3 ⟶ 2NaCl + H2O + CO2
H2SO4 + CaCO3⟶ CaSO4 + H2O + CO2
Bases vs Alkali
water soluble base = alkali
alkaline pH: >7
turn red litmus paper blue
bases: usually oxides, hydroxides or carbonates of metals
OH- ions makes aqueous solutions alkaline
ammonia is an exceptional base because it produces OH- ions
Soluble Salt Preparation
reaction of acid and insoluble base
insoluble reactant added in excess to ensure all the acid has reacted
if not, unreacted acid becomes dangerously concentrated during evaporation and crystallisation
excess reactant is filtered to ensure only salt and water remain
since all acid has reacted, excess solid base removed → solution left can only be salt and water
water evaporated until small crystals appear
usually when half the water is left
evaporating slowly over many days = larger crystals
if carbonate used, CO2 released into atmosphere
ex: preparing copper(II) sulfate from copper(II) oxide and dilute sulfuric acid
CuO (s) + H2SO4 (aq) ⟶ CuSO4 (aq) + H2O (l)

Soluble Salt Preparation with a Metal
acid can also be reacted with a metal to produce a salt, where:
metal is above hydrogen in the reactivity series
no too reactive that a dangerous reaction takes place
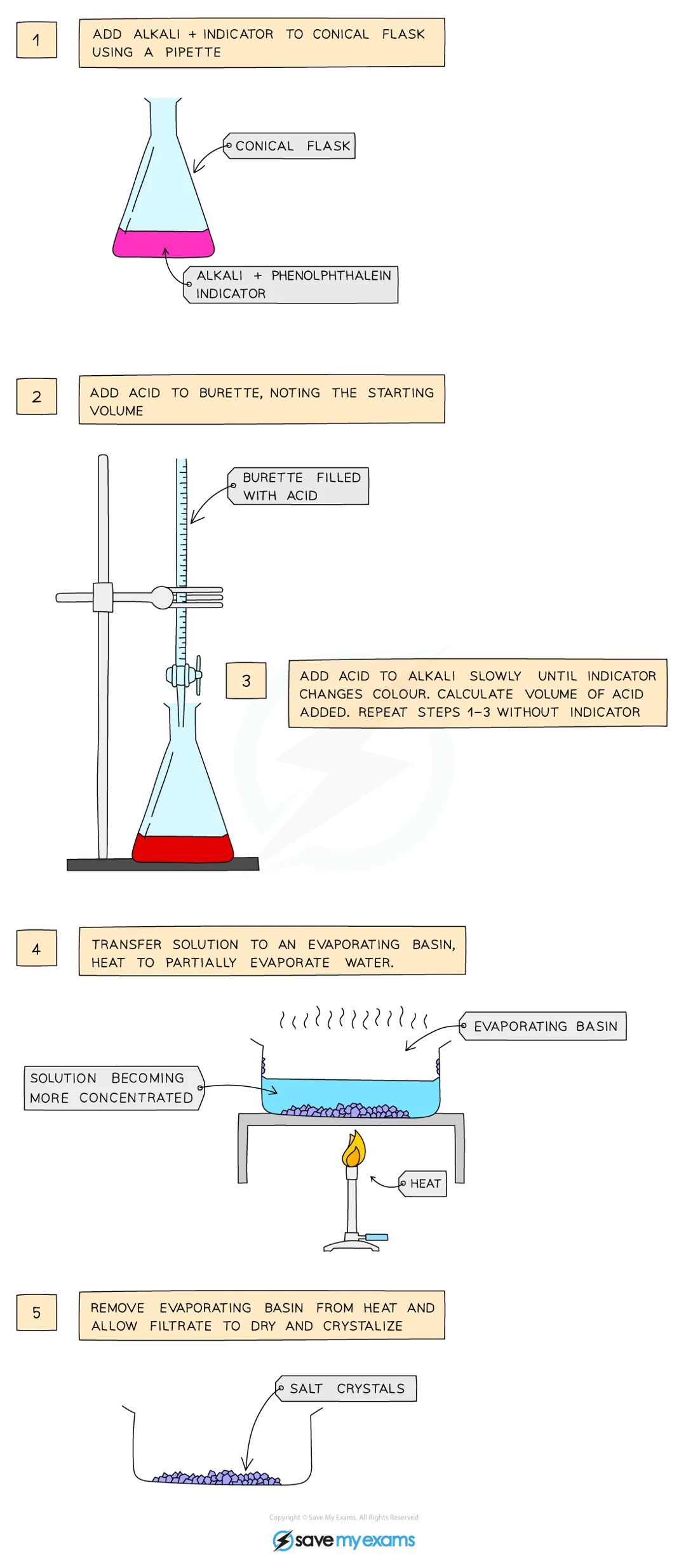
Soluble Salt Preparation using Titration
use pipette to measure alkali into conical flask
add drops of indicator (phenolphthalein or methyl orange)
add acid into burette, note starting volume
add acid very slowly from burette until indicator changes to correct colour
note and record final volume, calculate volume of acid added (starting - final acid volume)
add this volume of acid into same volume of alkali without indicator
heat to partially evaporate, leaving saturated solution
leave to crystallise decant excess solution, allow crystals to dry
Insoluble Salt Preparation
precipitation reaction
solid salt is precipitate → solid salt formed must be insoluble
pattern:
soluble salt 1 + soluble salt 2 ⟶ insoluble salt + soluble salt 3
AB + CD ⟶ AD + CB
measure out fixed volume of one solution and add second salt solution until in slight excess → ensures max. amount of precipitate obtained
precipitate recovered by filtration, washed with distilled water to remove residue (recovering solid) contaminating reactants → left to dry
good way to prepare silver and lead(II) salts which are often insoluble → starting material = usually nitrates of silver or lead(II) since all nitrates are soluble
Practical (Preparing Copper(II) Sulfate): Aim
to prepare a pure, dry sample of hydrated copper(II) sulfate crystals
Practical (Preparing Copper(II) Sulfate): Materials
1.0 mol / dm3 dilute sulfuric acid
copper(II) oxide
spatula & glass rod
measuring cylinder & 100 cm3 beaker
Bunsen burner
tripod, gauze & heatproof mat
filter funnel & paper, conical flask
evaporating basin and dish.
Practical (Preparing Copper(II) Sulfate): Diagram
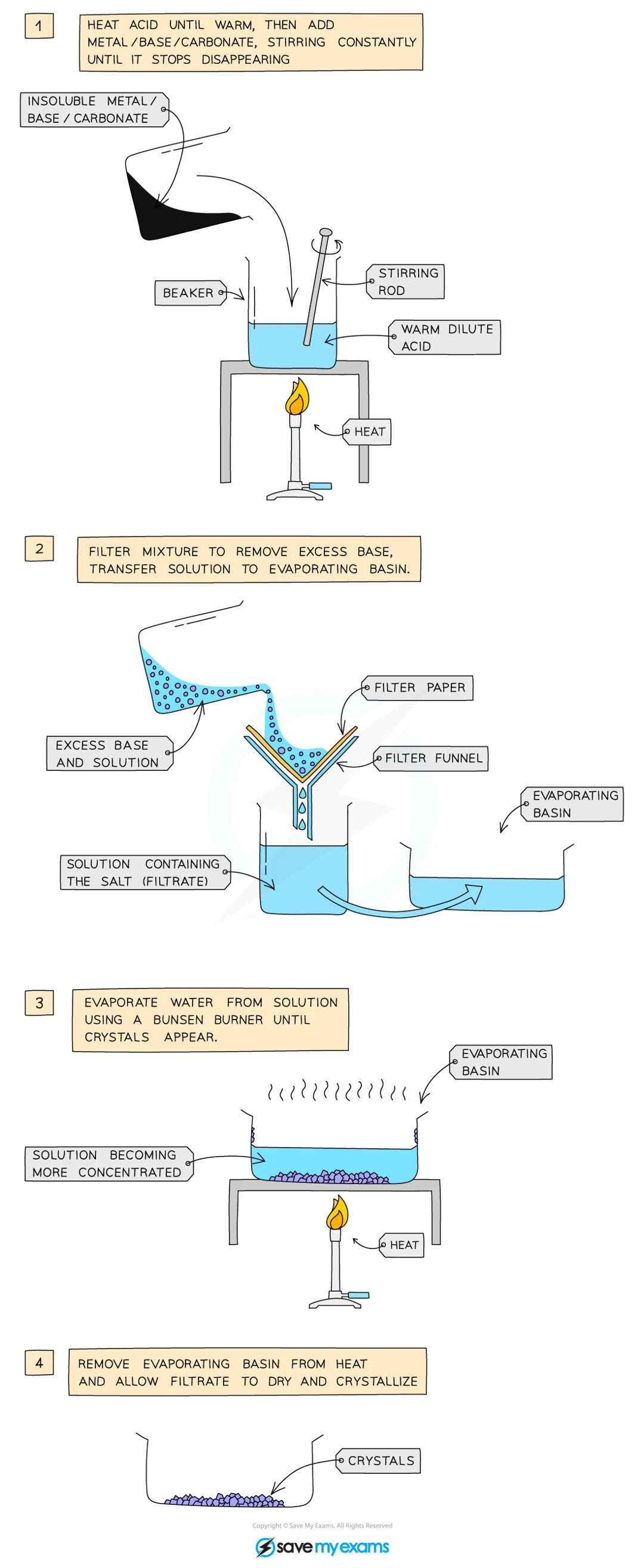
Practical (Preparing Copper(II) Sulfate): Method
add 50 cc dilute acid into beaker
warm gently with a Bunsen burner
add copper(II) oxide slowly into hot dilute acid
stir until base is in excess (stops dissolving, suspension forms in acid)
filter mixture into evaporating basin → remove excess bases
gently het solution in water bath or with electric heater → evaporate water and saturate solution
check solution saturation by dipping cold glass rod and seeing if crystals form on the end
leave filtrate in a warm place to dry and crystallise
decant excess solution and allow to dry
Practical (Preparing Copper(II) Sulfate): Excess Base Added
added in excess to use up all the acid
acid would become dangerously concentrated during evaporation and crystallisation
Practical (Preparing Copper(II) Sulfate): Results
hydrated copper(II) sulfate crystals should be bright blue, regularly shaped
Practical (Preparing Lead(II) Sulfate): Aim
prepare a dry sample of lead(II) sulfate
solid salt obtained = precipitate → solid salt formed must be insoluble, reactants must be soluble
Practical (Preparing Lead(II) Sulfate): Method
add 25 cc of 0.5 mol dm3 lead(II) nitrate solution to a small beaker
add 25 cc of 0.5 mol dm3 potassium nitrate to the beaker and mix with stirring rod
filter to remove precipitate from mixture
wash filtrate with distilled water to remove traces of other solutions
leave in oven to dry
Soluble salt 1 = lead(II) nitrate
Soluble salt 2 = potassium sulfate
lead(II) nitrate + potassium sulfate → lead(II) sulfate + potassium nitrate
Pb(NO3)2 (aq) + K2SO4 (aq) → PbSO4 (s) + 2KNO3 (aq)