Fluids
1/99
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
100 Terms
Fluid outside cells
Extracellular fluid (ECF)
Cytosol (fluid inside cells)
Intracellular fluid
Most of our body is composed of this
Water
Balance between the amount of water gained and the amount of water lost
Involves regulating the content and distribution of body water in the ECF and the ICF
Fluid balance
Water gain in what 2 ways?
Water loss in what 2 ways?
Gain: Absorption by the digestive system + metabolic reactions
Lost: Excretion in urine and feces + perspiration and breathing
Balance between the amount of electrolytes gained and the amount of electrolytes lost
Electrolyte balance
Ions released through dissociation of inorganic compounds
Can conduct electrical current in solution
Electrolytes
Electrolytes gains (1)?
Electrolyte losses (2)?
Gains: Absorption by the digestive system
Losses: Excretion in urine and feces + perspiration
A balance between the amount of hydrogen ions (H+) gained and the amount of hydrogen ions (H+) lost
Acid-base balance
Water accounts for approximately what % of adult male body weight?
60%
Water accounts for what % of adult female body weight
50%
Intracellular fluid contains about what amount of total body water
2/3
What is the primary intracellular fluid?
Cytosol
What is the primary fluids of extracellular fluid?
What else?
Interstitial fluid of peripheral tissues and plasma of circulating blood
Lymph, cerebrospinal fluid, synovial fluid, serous fluids, aqueous humor of eye, perilymph and endolymph of the inner ear
More solutes in plasma than RBC and losing water is what kind of solution?
Dehydration
Hypertonic solutions
Water flows inside RBC to burst
More solutes inside RBC
Excessive fluids
Hypotonic solution
Same solute/water concentration as inside cells; water moves in + out
Isotonic solution
Solute exchnage between ICF and ECF occurs across selectively permeable plasma membranes by ________and______________(active and passive)
Diffusion + carrier-mediated transport
Solute exchanges between the subdivisions of ECF occur across the endothelial lining of capillaries and from interstitial spaces to plasma via _______________ that drain into the venous system
lymphatic vessels
Principal ions in the ICF (4)
Potassium
Magnesium
Phosphate ions
Negatively charged proteins
Principal ions of ECF (4)
Sodium
Chloride
Calcium
Bicarbonate
All homeostatic mechanisms that monitor and adjust body fluid composition respond to changes in the ______
ECF
Receptors monitor ______ and___________as indicators for fluid and electrolyte balance
Plasma volume and osmotic concentration
Cells cannot move water by active transport, only by ________
"Water follows salt“
osmosis
Body’s water or electrolyte content will ____if gains exceed losses, and will _____if losses exceed gains
rise and fall
Released at the posterior pituitary gland when osmoreceptors of the hypothalamus detect higher osmotic concentration
Stimulates water conservation at the kidneys (late DCT & CD), reducing urinary water loss and concentrating urine (REABSORPTION)
Stimulates hypothalamic thirst center to promote fluid intake
Antidiuretic hormone (ADH)
Released by the adrenal glands in response to:
increasing potassium/decreasing sodium
in response to activating the RAAS
Stimulates sodium reabsorption and potassium loss (secretion) in the kidneys
Also stimulates water retention (water follows sodium)
Aldosterone
Released by cardiac muscle cells in response to abnormal stretching of the heart wall due to increased blood volume and blood pressure
Reduce thirst
Block the release of ADH and aldosterone
Stimulate diuresis (fluid loss by the kidneys) and therefore lower the blood pressure and plasma volume
Natriuretic peptides (ANP and BNP)
Under normal circumstances, is there significant water movement between ECF and ICF?
No
What % of ECF is in interstitial fluid and minor fluid compartments?
What % is in plasma?
80 and 20
Net hydrostatic pressure pushes water ____of plasma
Net colloid osmotic pressure draws water _____plasma
out
into
Movement of too much water from plasma into interstitial fluid
Caused by blockage of lymphatic drainage
Edema (tissue swelling)
Lymphedema
If water is lost (but electrolytes retained) what happens to osmotic concentration?
Osmotic concentration of ECF increases; becomes hypertonic compared to ICF
Water moves from ICF to ECF by osmosis
If water is gained (but electrolytes stay the same), what happens to the osmotic concentration?
The osmotic concentration of the ECF decreases; it becomes hypotonic compared to the ICF
Water moves from the ECF to the ICF (into cells) by osmosis
This happen when water losses are greater than gains
May results in excessive perspiration, inadequate water consumption, repeated vomiting, and diarrhea
Dehydration (water depletion)
Fluid shift causes water to move from where to where with dehydration?
ICF to ECF
What leads to a decrease in urination and increase in thirst?
Treatment?
ADH, aldosterone, and renin secretion
Increase drinking or IV fluids
What are 4 common IV fluids?
Normal saline
Lactated Ringer’s
Dextrose 5% in Water (DSW)
Half Normal Saline
Develops when water gains are greater than losses
May result from ingestion of a large volume of water, injection of hypotonic solution into bloodstream, inability to eliminate excess water in urine (chronic renal failure, heart failure, cirrhosis), excess ADH production
Hyperhydration (water excess)
What happens to ECF volume and osmotic concentration during hyperhydration?
Where does water move to and from?
ECF volume increases and ECF osmotic concentration decreases
Water moves from ECF to ICF
Response mechanisms in cases of severe water gain?
Treatment?
ADH decrease and ANP/BNP increase cause decrease in thirst and increase in water loss with urine
Administer diuretic drugs, IV of concentrated salt solution to promote fluid shift out of cells and treat the hyponatremia
4 IV Fluids for dehydration
Isotonic solutions (equal to blood serum)
Hypotonic solutions (lower concentration than blood)
Hypertonic solutions (higher concentration than blood)
Colloids (larger molecules)
When BV decreases due to sweating, from what source is water taken in by the blood?
Interstitial fluids (ECF)
What happens in tissues when capillary blood pressure is less than osmotic pressure?
Net reabsorption
Solute contributes to the movement of water between cells and the surrounding medium by?
Osmotic pressure
A cation has what charge?
Positive
Review Qs
Body generates acids during normal metabolism
Absorption by the digestive system
Hydrogen gains
Kidneys secrete H+ into urine and generate buffers that enter bloodstream
Lungs affect pH through elimination of CO2
Hydrogen loses
When gains and losses are equal for each electrolyte in the body
Electrolyte balance
Amount of positive or negative ion that supplies 1 mole of electrical charge
1 equivalent = 1000 milliequivalents (m E q)
Equivalent
Main cation in the ECF
Sodium chloride (NaCl) and sodium bicarbonate provide __% of ECF osmotic concentration
Sodium
90%
Are problems with potassium balance more or less common than sodium imbalance?
Less common, but more dangerous
Gains of this involve an uptake across digestive epithelium
Loss of this involve an excretion in urine and perspiration
If gains exceed losses, total content of ECF _________
Na+
increases
What is released when sodium gains exceed loses?
ADH, increasing thirst and water retention
If ECF volume rises, what else increases?
What is released?
Blood volume and blood pressure
Natriuretic peptides released
If ECF volume drops, what else decreases?
What is activated and what increases?
Blood volume and blood pressure decreases
The renin-angiotensin-aldosterone system is activated
Sympathetic stimulation increases
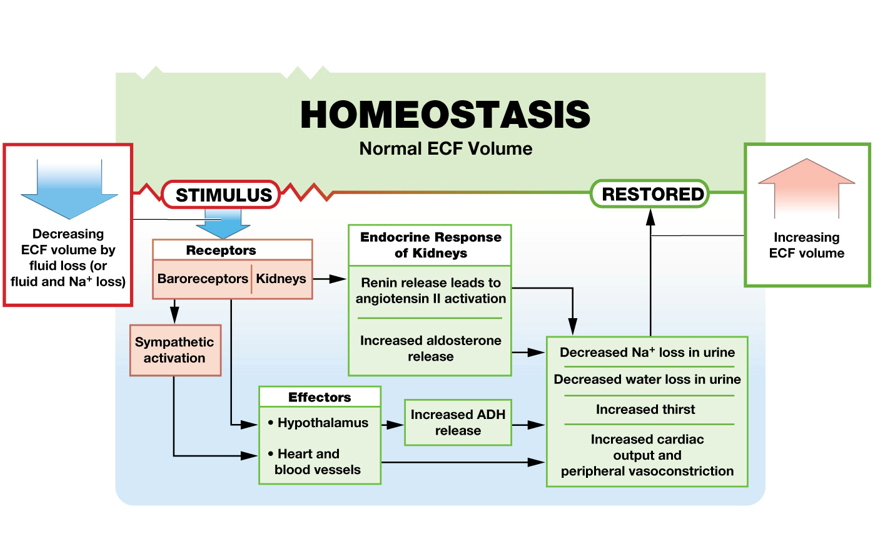
Na+ concentration of ECF < 135mEq/L
Caused by hyperhydration
Na+ concentration of ECF > 145mEg/L
Caused by dehydration
Hyponatremia
Hypernatremia
What percent of potassium in the body is in the ICF?
98%
Potassium gains include uptake across what?
Potassium loss in where?
digestive epithelium
Urine
Higher K+ concentration in ECF leads to higher rate of ______
Na+ exchanged for ___ rather than K+ so rate of K+ secretion declines
Increase in aldosterone leads to more ____ excretion in exchange for Na+
secretion
H+
K+
Low K+ in blood
Caused by certain diuretic drugs and excess aldosterone secretion
Results in muscle weakness and paralysis
Hypokalemia
Elevated level of K+ in blood
Caused by acidosis (low blood p H), kidney failure, and certain diuretic drugs
Results in over-excitable muscles and nerves, and cardiac arrhythmias
Hyperkalemia
This is the most abundant mineral in the body
98% is deposited in the skeleton
Calcium
Calcium is important for what 4 things?
For muscular and neural activities
For blood clotting
As cofactors for enzymatic reactions
As second messengers
Raise calcium concentrations in ECF
Decrease calcium concentrations in ECF
Parathyroid hormone (PTH) and calcitrol
Calcitonin
Ca+ gains result in absorption in ______
Reabsorption in ______
Stimulated by ____ and _____
Ca+ losses in _____, ____, or feces
usually small
digestive tract
kidneys
PTH and calcitriol
bile, urine
Ca+ concentration in ECF > 5.3 mEq/L
Caused by hyperparathyroidism (oversecretion of PTH), malignant cancers (breast, lung, kidney, and bone marrow) or excessive calcium or vitamin D supplementation
Results in fatigue, confusion, cardiac arrhythmias, calcifications in the kidneys and soft tissues
Hypercalcemia
Ca+ concentration in the ECF < 4.3 mEq/L
Caused by hypoparathyroidism (undersecretion of P T H), vitamin D deficiency, and chronic renal failure
Results in muscle spasms, convulsions, weak heart beats cardiac arrhythmias, and osteoporosis
Hypocalcemia
About ____% of magnesium deposited in skeleton
Where are magnesium ion primary found, the ECF or ICF?
Reabsorbed by what organ?
60%
ICF
Kidneys
A measure of the acidity or alkalinity of a solution
Depends on the amount of dissolved acids, bases, and salts
What is normal in the ECF/blood?
pH - 7.35-7.45
Abnormally low blood pH (pH<7.35)
Abnormally high blood pH (pH>7.45)
pH=7 is what?
Acidosis
Alkalosis
Neutral
Acid-base imbalance affect all body system but mostly which?
Is acidosis or alkalosis more common?
Nervous and cardiovascular systems
Acidosis
Acids that do not leave the solution (ex. sulfuric acid and phosphoric acid), eliminated through the kidneys
Acids (ex. pyruvic acid, lactic acid, ketone bodies) that can be metabolized without accumulation
Acids that leave the body by being breathed out by CO2 (*EQUATION*)
Fixed acids
Metabolic acids
Volatile acids
CO2 is converted to ________
carbonic acid
Carbonic acid dissociates into what 2 ions?
Hydrogen ions
Bicarbonate ions
Partial pressure of ___ is most important factor affecting blood pH
CO2
When CO2 levels increase, H+ and HCO3- increase and blood pH _____
*And vice versa
decreases
Hydrogen ions gained at ______
Lost at ______
neutralized by ______ to avoid tissue damage
digestive tract
kidneys and lungs
buffers
These dissociate completely in solution (ex. hydrochloric acid, HCl)
These do not dissociate complete in the solution, release fewer H+ (ex. carbonic acid
Strong acids and strong bases
Weak acids or weak bases
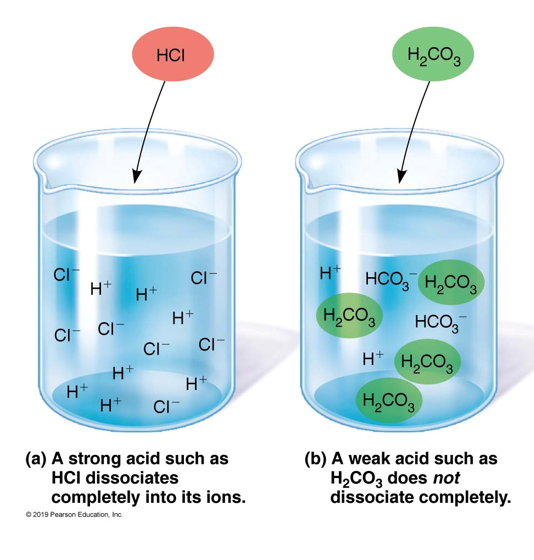
Dissolved compounds that stabilize the pH of a solution by adding or removing H+
Weak acids can donate H+ and weak bases can absorb H+
Buffers
Combination of weak acid and the anion released by its dissociation
Buffer systems
Most important buffer for the ICF and urine
Phosphate buffer system
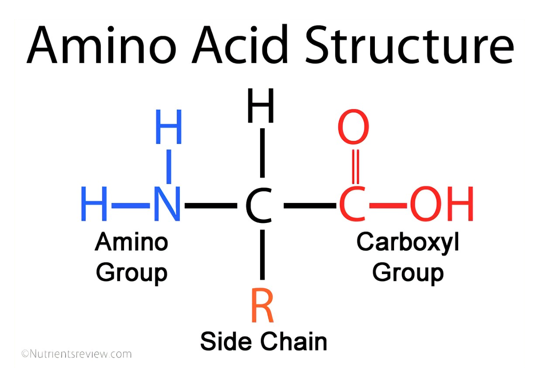
_______ group of amino acid dissociates to release H+
________ groups and __ groups of amino acids can accept H+
Carboxyl
Amino, R
CO2 in RBCs converted to carbonic acid
Hemoglobin buffer system
Carbon dioxide is constantly generated by body cells and converted to carbonic acid
Most important buffer in ECF
Functions only when the respiratory system and the respiratory control centers are working normally
Carbonic acid-bicarbonate buffer system
Buffer system coordinate with these 2 mechanisms?
Respiratory and renal
Changing rate of ____ helps regulate CO2 levels and therefore pH
respiratory levels
Compensation for low pH (acidosis)
Decrease _____ by breathing faster
*Vice versa for alkalosis
PCO2
Buffers in urine are required to eliminate a lot or a little H+?
If pH of tubular fluid reaches 4-4.5, H+ _______ stops
a lot
secretion
Disturbance of acid-base balance from what conditions?
Disorders affecting circulating buffers, respiratory performance, or renal function, such as emphysema or renal failure
Cardiovascular conditions such as heart failure or hypotension
Conditions affecting the CNS, such as neural damage or disease, can disturb reflexes essential to p H regulation
Initial phase when pH moves rapidly out of the normal range
Physiological adjustments occur and return pH to the normal range, but compensatory mechanisms need to be active to keep it there
Acute phase
Compensated phase
Results from imbalance between CO2 generation in peripheral tissues and CO2 excretion at lungs. Cause abnormal CO2 levels in the ECF
Results form the generation of metabolic or fixed acids (increases H+ in ECF) or conditions affecting HCO3 concentration in the ECF
Respiratory acid-base disorders
Metabolic acid-base disorders
Low blood pH due to hypercapnia
Develops when respiratory system cannot elimination enough CO2
Primary cause is hypoventilation
Response:
Respiratory compensation + renal compensation
Respiratory acidosis
Develops if decline in pH is severe and the lungs can not respond
Immediate, life-threatening condition
Develops when normal respiratory function has been compromised
Compensatory mechanisms have not failed completely
Examples: emphysema, congestive heart failure, pneumonia, and pneumothorax
Acute respiratory acidosis
Chronic respiratory acidosis
High blood pH due to hypocapnia (low blood PCO2)
Primary cause is hyperventilation (abnormally high respiratory rate)
Examples: pain, stress, anxiety, high altitude, and patients on respirators
Responses:
Respiratory compensation: decrease respiratory rate (ex. breathing into paper bag)
Renal compensation: secrete bicarbonate ions and reabsorb hydrogen ions
Respiratory alkalosis
Major causes:
High production of fixed or metabolic acids (H+ overload buffer system)
Lactic acidosis after strenuous exercise or hypoxia
Ketoacidosis due to starvation or diabetes mellitus
Impaired H+ excretion at kidneys
Severe bicarbonate ion loss (chronic diarrhea)
Responses:
Respiratory compensation: increase respiratory rate
Renal compensation: secrete hydrogen ions and reabsorb bicarbonate ions
Metabolic acidosis
Caused by elevated HCO3 concentrations and decreased H+ concentration
Examples: alkaline tide after a meal and concentration
Responses:
Respiratory compensation: decrease respiratory rate
Renal compensation: secrete bicarbonate ions and reabsorb hydrogen ions
Metabolic alkalosis
These ions are required for bone mineralization
Found in ICF or ECF?
Reabsorbed where and lost where?
Phosphate
ICF
Kidneys, lost in urine and feces
The most abundant anion in the ECF
Absorbed in digestive tract and reabsorbed with what other ion
Chloride
Na+