Chemistry 3rd Term
1/21
Earn XP
Description and Tags
everything chemistry 3rd term
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What is the kinetic theory of matter?
The fundamental idea that all matter is composed of tiny particles that are constantly in motion. Because these particles are moving, they possess kinetic energy.
What is the relationship between temperature and energy?
Temperature acts as a direct measure of the energy within a substance.
An increase in the temperature of a gas causes a corresponding increase in the average kinetic energy of its particles.
Higher temperatures result in particles moving at greater speeds.
What are the Postulates of the Kinetic Theory of Gases?
These postulates describe the behavior of an Ideal Gas
1. The gas molecules move randomly in straight lines, colliding with one another and with walls of the container.
2. The collisions of the gas molecules are perfectly elastic.
3. The actual volume occupied by the gas molecules is negligible relative to the volume of the container.
4. The cohesive forces between the gas molecules are negligible.
5. The temperature of the gas is a measure of the average kinetic energy of the gas particles.
What does boyles law state?
The volume of a fixed mass of gas is inversely proportional to its pressure, provided that the temperature remains constant.
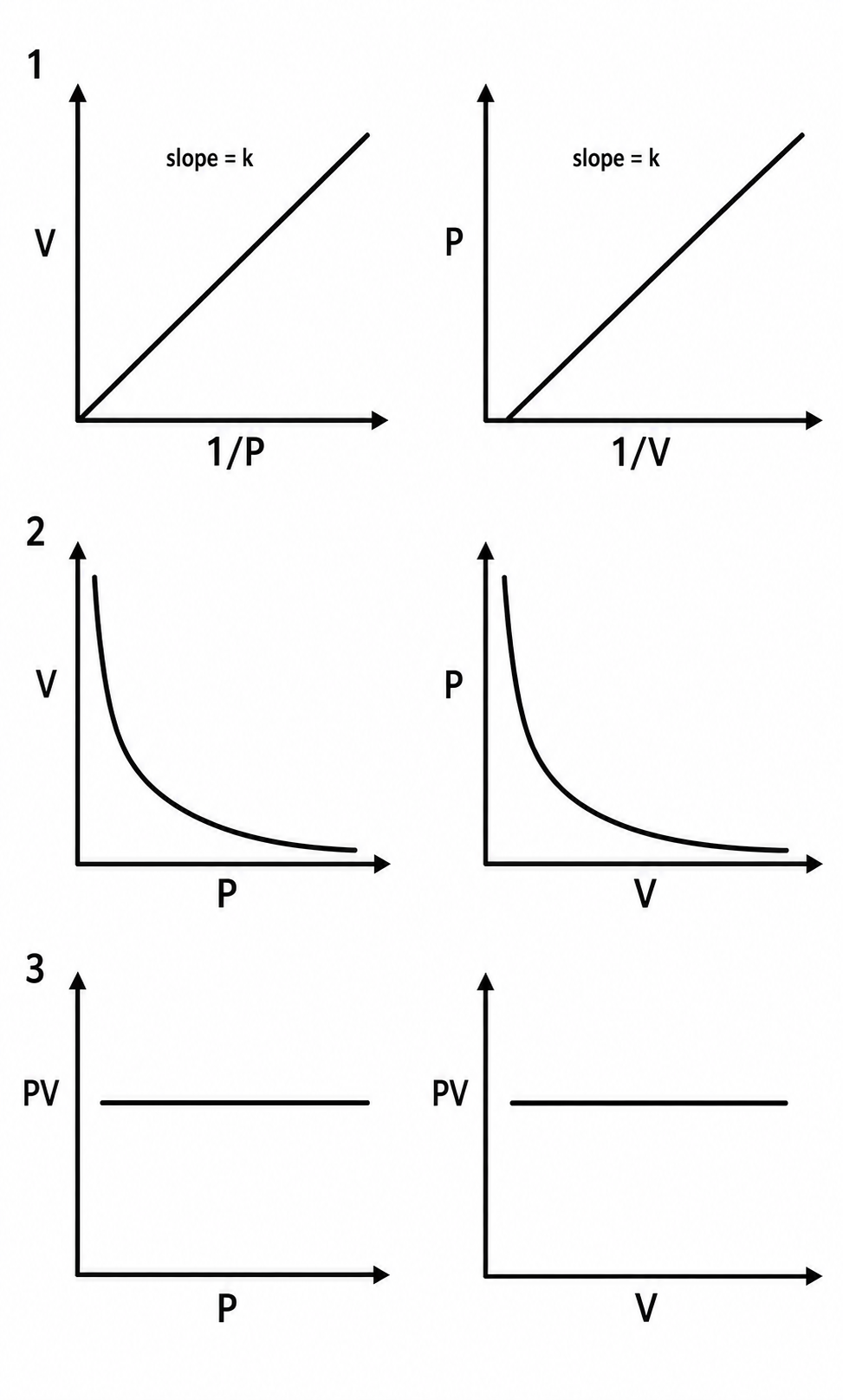
What is the mathematical expression of boyles law?
V ά 1/P
V=K/P
Or PV=K
P1V1=P2V2
What does Charles law state?
The volume of a fixed mass of gas is directly proportional to its absolute temperature, provided that the pressure remains comstant.
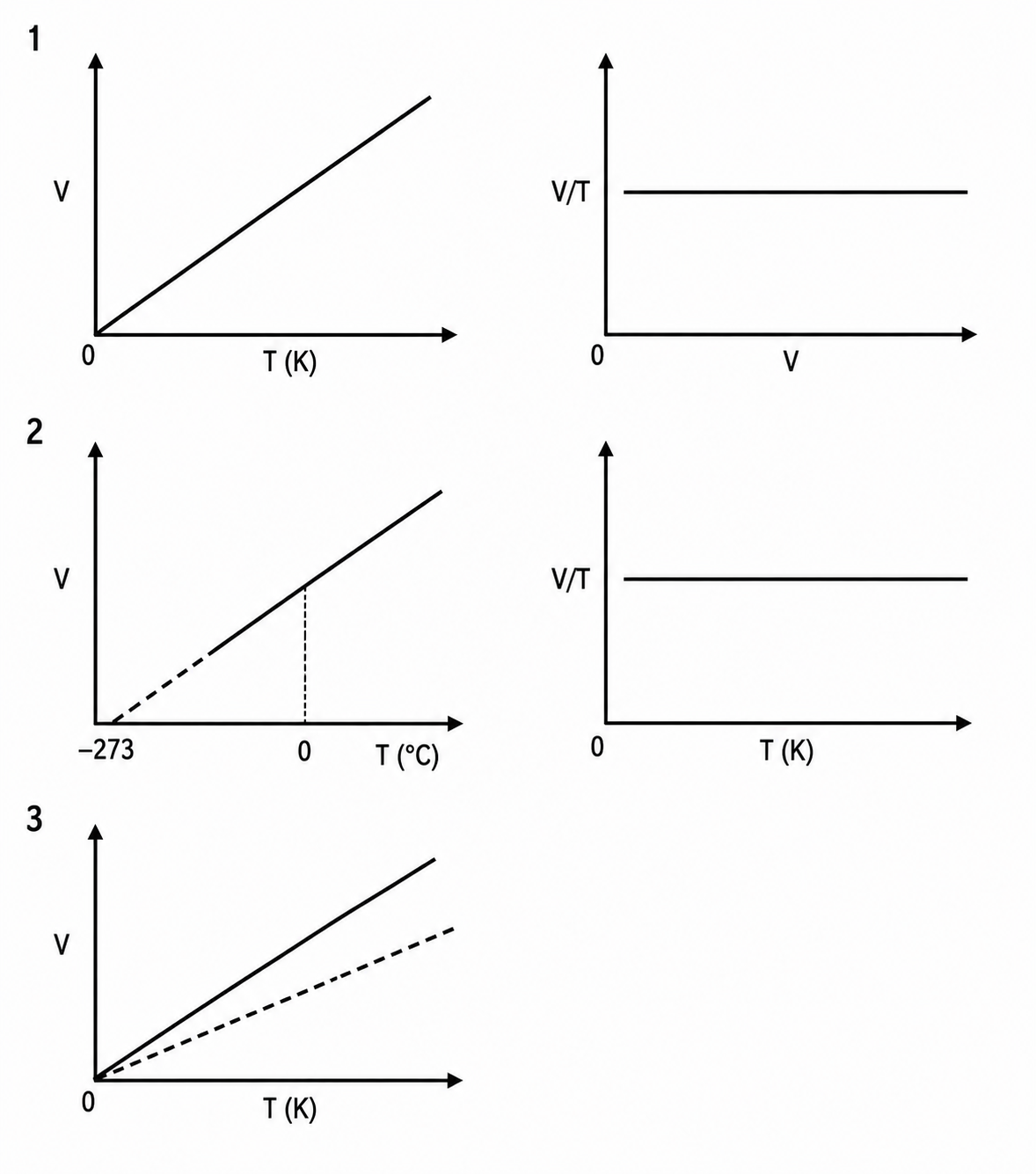
What is the mathematical expression of Charles law?
V ά T
V=KT
Or V/T=K
V1/T1=V2/T2
What is the graphical representation of Charles law?
What does the general gas law state?
For a fixed mass of gas the product of its pressure and volume divided by its absolute temperature is constant.
What is the mathematical formula for the general gas law?
P₁V₁ / T₁ = P₂ V₂ / T₂
What is standard temperature?
273K or 0 C
What is standard pressure?
760 mmHg, 1 atm or 1.01×10⁵Nm⁻²
What does the ideal gas equation state?
For an ideal gas PV/T is constant
PV/T=R (R is constant)
PV=RT
For n moles of gas,
PV= nRT
Where P in atm, V in dm3 , T in K at s.t.p. for 1 mole of gas,
R=PV/nT
= (1atm) (22.40dm3)/(1mol) (273K)
= 0.082atmdm3K-1mol-1
What are the deviations of real gases from ideal gases?
1. Real gases occupy space unlike ideal gases.
2. Real gases have forces of attraction unlike ideal gases.
3. Real gases liquefy when the temperature drops.
What are the definitions of absolute or zero temperature?
The temperature at which the volume of a gas would be theoretically reduced to zero.
The temperature at which all motion of gas molecules is assumed to have ceased
The lowest temperature theoretically possible. Absolute or zero temperature is -273C.
What does Daltons law of partial pressure state?
The total pressure exerted by a mixture of non-reacting gases is equal to the sum of the partial pressures of the individual gases that make up the mixture.
What is partial pressure?
Partial pressure is the pressure that a single gas in a mixture would exert if it were the only gas in the container.
What are the implications of Daltons law?
For a mixture of gases A, B and C, with partial pressures PA, PB and PC respectively; the total pressure
Ptotal of the mixture is expressed as:
Ptotal = PA + PB + PC
When a gas is collected over water, the total pressure is the sum of the partial pressure of the dry gas and the partial pressure of the water vapour at that temperature. i.e. Pgas = PTotal - PVapour
What does grahams law of diffusion state?
The rate of effusion or diffusion of a gas is inversely proportional to the square root of its molar mass.
Express grahams law of diffusion mathematically
Mathematical expression
R α 1/ √D
For two gases: comparing their rates of diffusion, we have:
R1/ √D1= R2/ √D2
R1/ R2 = √D2/√D1 =M2/M1
This expression above is used when the volumes are different.
Rate of diffusion = volume/ time
t1/ t2 = √M1/ √M2
It is used when the volume is the same.
Note: Vapour density is related to relative molecular massV.D = relative molecular mass of gas/2
What does avogadros law state?
Equal volumes of all gases at the same temperature and pressure contain the same number of molecules.
What does Gay Lussacs law state?
When gases react, they do so in volumes which bear a simple whole-number ratio to one another and to their gaseous products, as long as temperature and pressure are kept constant.