Radicals in synthesis
1/92
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
93 Terms
What are radicals?
Atoms or molecules that possess a single electron in a reactive orbital, making them very reactive.
Why are most radicals not observed by spectroscopy?
They are unstable.
How are radicals detected?
By EPR which is very sensitive and can detect radicals at low concentrations. Radicals with lifetimes less than 1 second can be detected, providing information on the nature and structure of the u.p.e.
What is the general reaction pathway for a radical?
What determines the BDE?
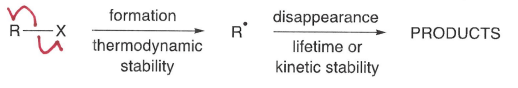
The thermodynamic stability of the radical formed (R*)

What does BDE show about a radical?
As C-H BDE increases, the stability of the radical decreases. This is because it becomes harder to form the radical (via C-H bond breakage).
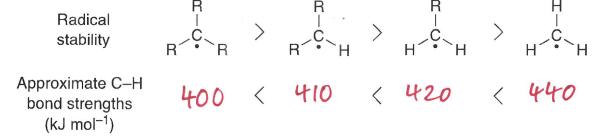
What structural effects influence the BDE?
In double bonds, the pi component doesnt contribute as much strength as a sigma, therefore the BDE is lower than expected.
A radical next to an aromatic is also more stable.
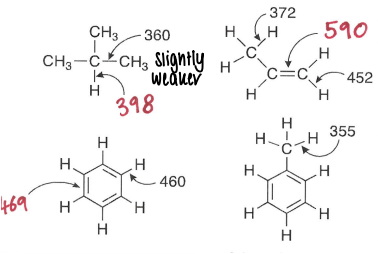
What is the trend in carbon radical stability?
A tertiary radical is more stable.
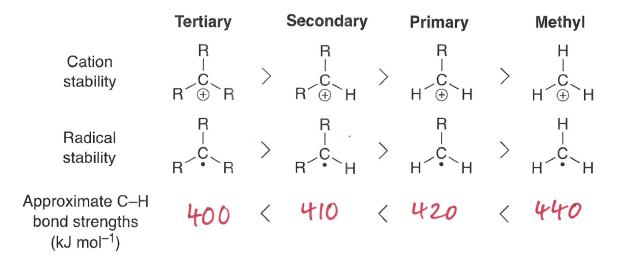
How do inductive effects influence radical stability?
The more +I groups (electron donating), the more stable. This is because the radical prefers to have 8 outer electrons.
How does hyperconjugation influence radical stability?
The p orbital of the radical can interact with a pair of bonding electrons in a neighboring sigma bond, donating these electrons into the partially filled p orbital, therefore stabilising it.
This also causes the planarity of the carbon-centred radicals and their trigonal planar geometry, ensuring maximum orbital overlap.
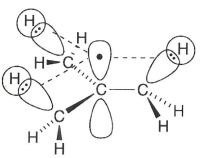
What is needed for radicals to form resonance forms?
The radical p orbital must be in the same plane as the p orbitals of a C=C pi bond.
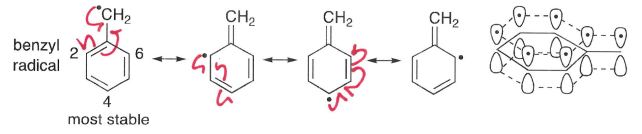
How does resonance influence radical stability?
A -M effects remove electron density from on the radical and causes a decrease in BDE, therefore stabilising the radical.

How do +M effects stabilise radicals?
+M effects donate electron density onto the radical, providing some stavilisation. BDE decreases as electron donation increases.
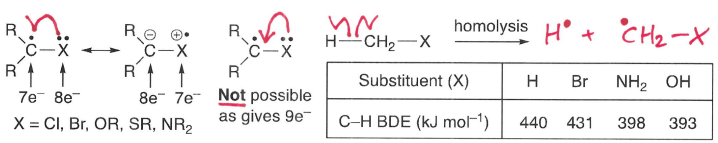
How do -I effects destabilise a radical?
A -I effect causes an increase in BDE.
How does p character affect radical stabilisation?
An increase in p character increases radical stabilisation as homolysis of the sigma C-H bond in alkyls yields a radical with the partially filled orbital furthest from the nucleus. Increasing s character is destabilising.
Holding the radical orthogonal to the pi system prevents resonance stabilisation.
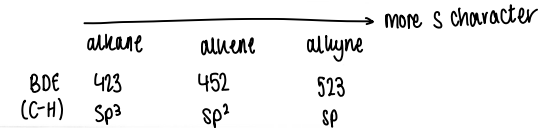
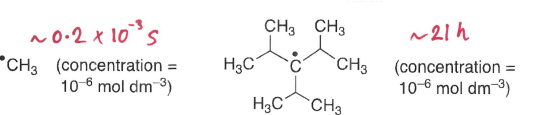
How does sterics influence radical lifetime?
The larger the substituents attached to the radical centre, the more stable it is and therefore the longer its lifetime.
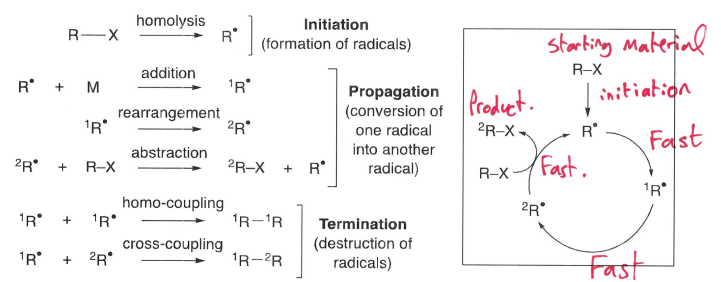
What is a chain reaction?
The series of reactions that occur following the initiation of the starting material, forming the radical. This radical then engages in multiple propagation steps, forming a cycle. Termination causes the destruction of radicals, and therefore end of the cycle.
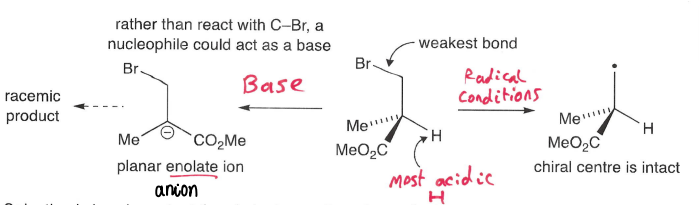
Why are radicals used in synthesis?
Neutral reaction conditions can be used (Avoids unwanted reactions of acid or base sensitive reactants).
Solvation is less important as radicals are not strongly influenced by nature/polarity of solvent.
Synthesis of polar/sterically hindered compounds is possible.
Ability to perform chemistry regular methods cannot.
What is thermolysis?
Compounds with relatively weak bonds (>160 kJmol-1) undergo homolysis using temperatures below 150 degrees celsius.
How do peroxides undergo thermolysis?
Heat at 50-150 degrees, breaking the weak RO-OR bond.
Also undergo photolysis and absorb radiation >250 nm.
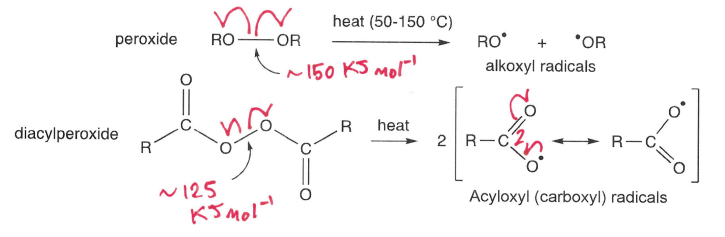
How do azo compounds undergo thermolysis?
Heat at around 80 degrees, breaking the weak N-R bonds.
Also undergo photolysis and absorb radiation >250 nm.
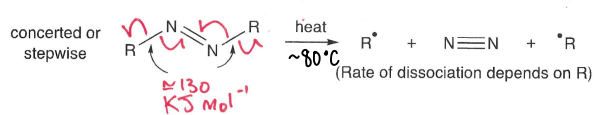
What is photolysis?
Generates radicals at r.t. via the compound absorbing light of a specific wavelength and undergoing dissociation of the excited state.
Energy of the photon must be sufficient to rupture a bond, covalent bonds are cleaved by irradiation with visible (400-800nm) or UV radiation (200-400nm).
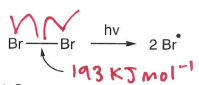
How do halogens undergo photolysis?
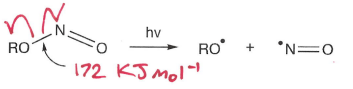
How do nitriles undergo photolysis?
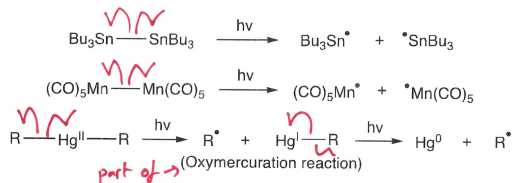
How do organometallics undergo photolysis?
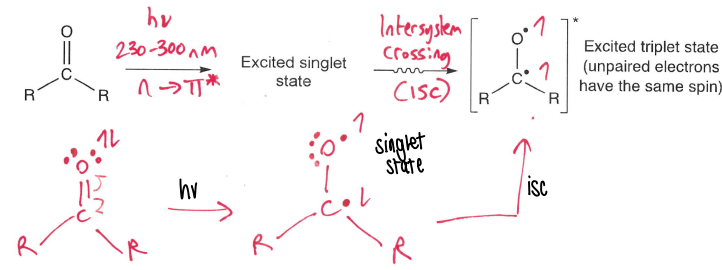
How do carbonyls undergo photolysis?
They absorb at 230-300nm, promoting an electron from the n to pi* orbital and forming a singlet excited state. Isc then occurs forming the triplet state.
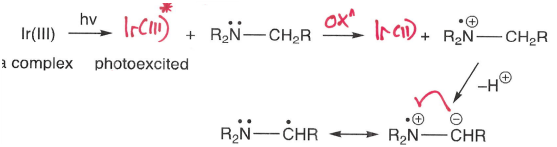
How are radicals formed from oxidation?
A metal oxidant (or anode) is used, forming a radical cation which fragments into a R* radical and X+ cation.
The radical cation fragments to form the most stable radical or cation.
A high standard electrode potential is needed for anode oxidation.
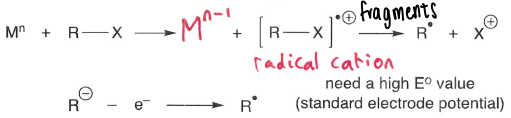
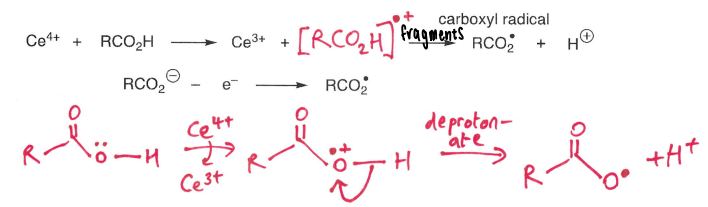
What is the Kolbe reaction?
The oxidation of carboxylic acids using Ce4+.
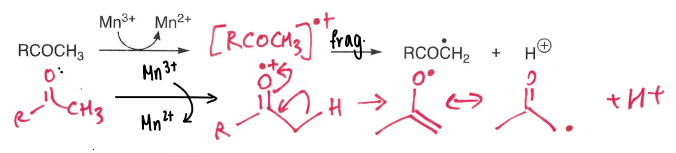
How are carbonyls oxidised into radicals?
Using an Mn3+ oxidant.
How are amines oxidised into radicals?
Using an Ir(III)* photoexcited complex with 2 unpaired electrons.
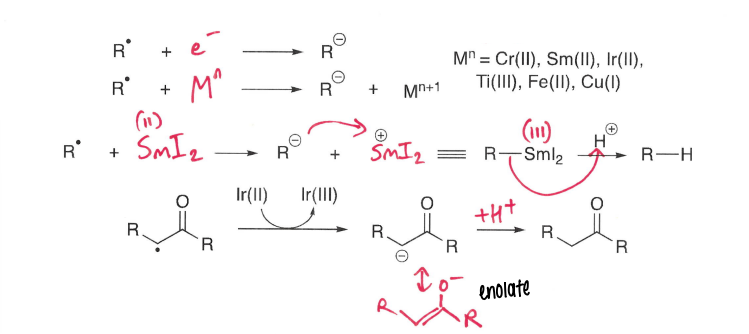
How are radicals formed via reduction?
A metal reduces the R-X compound into a radical anion, which fragments.
A low standard electrode potential is required.
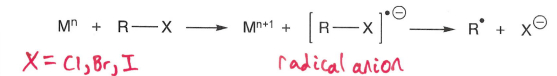
How are peroxides reduced to form radicals?
A Fe2+ (usually FeSO4) metal is used.

How are halides reduced to form radicals?
A Sm2+ (usually SmI2) metal is used as it wants to be in the +3 O.S., making it a good reducing agent.
An Ir(III) photoexcited complex can also be used.
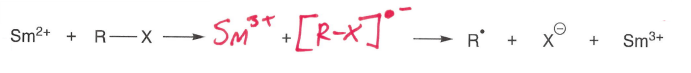
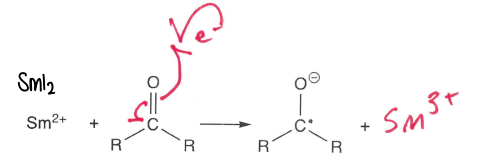
How are carbonyls reduced to form radicals?
Using a SmI2 reducing agent.
The most stable resonance form has the negative charge on the oxygen.
How are diazonium salts reduced to form radicals?
A Cu(I) reductant (usually CuCl) is used, forming the radical. This can further react with more CuCl2 to form an R-Cl molecule.
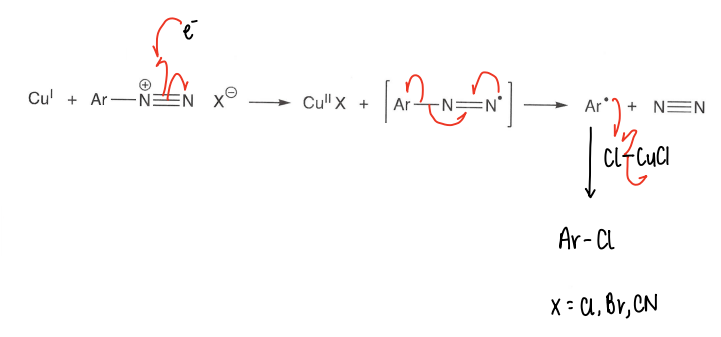
What are intramolecular abstraction reactions?
A form of propagation in which a radical reacts with an X-R molecule, forming a new radical.
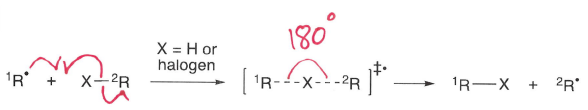
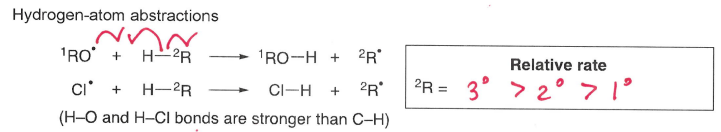
What influences the rate of hydrogen atom abstractions (involving C-H bonds)?
As H-O and H-Cl bonds are stronger than C-H, these bonds are formed and C-H is broken.
The rate increases if the H-R molecule is tertiary.
What influences the rate of hydrogen atom abstractions (involving Si-H or Sn-H bonds)?
C-H bonds are stronger than H-Sn or H-Si, therefore these are preferrable formed.
The rate increases if the R is primary.
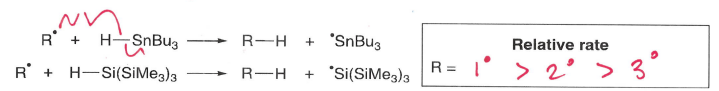
What influences the rate of halogen-atom abstractions?
A radical can react with an X-R molecule to form a more stable R* radical and a molecule of R-X containing a weaker bond.
Rate increases when R is tertiary and X is a larger halogen.
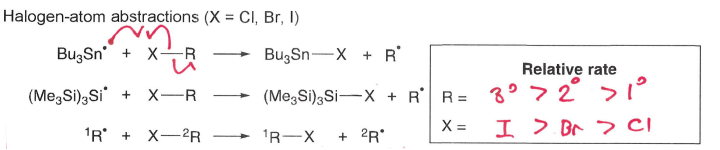
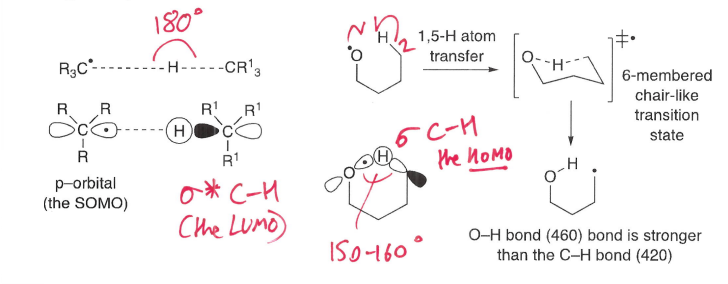
What are intramolecular atom transfers?
1,5 are most common, in which a radical at position 1 reacts with a group at position 5, forming a stronger bond.
A ring intermediate is formed.
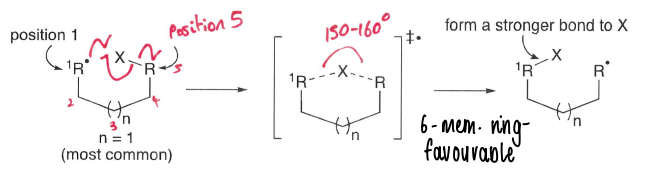
What type of intramolecular atom transfers are most favoured?
1,5 transfers are most common as they form a 6 membered ring, this is favourable.
1,4 transfers have a considerable deviation from linearity in the transition state.
1,6 and 1,7 are less common due to entropic factors, as the length of the chain increases it becomes less likely that the two reactive centres will come together.
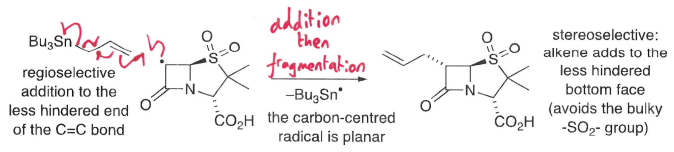
What are radical addition reactions?
Radicals add regioselectively to unsymmetrical C=C and C≡C bonds.
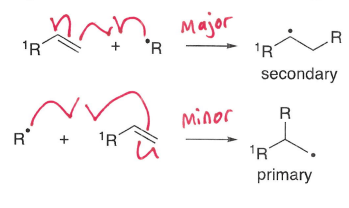
How do intermolecular addition reactions occur with alkenes/alkynes?
For alkenes and alkynes, regioselective additions occur with terminal alkenes at the least hindered position, forming the most stable radical.
A more stable sigma bond is formed at the expense of a weaker pi bond.
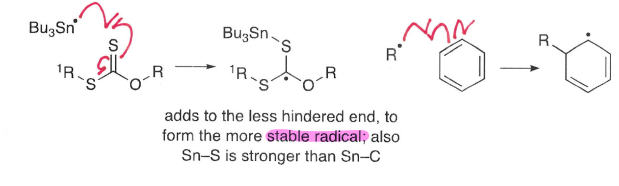
How do radicals add to other unsaturated bonds via intermolecular addition?
Radicals can add to unsaturated bonds such as C=N, C≡N, C=O, C=S and benzene rings.
The radical adds to least hindered end.
What are intramolecular addition reactions?
A radical reacts with a double bond within itself, forming a ring.
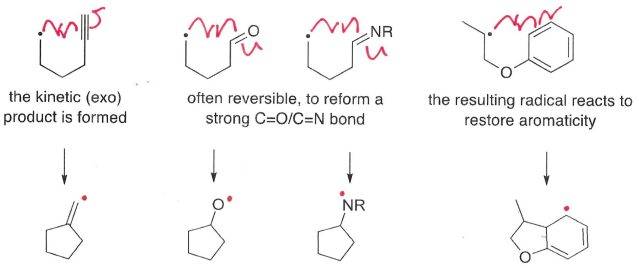
What are the different products that can be formed by intramolecular additions?
Two different products can be formed by reaction of the radical with either side of the double bond.
The exo product is favoured, formed by attack at the more substituted carbon. This is explained by orbital interactions.
The endo product forms by attack at the least hindered end.
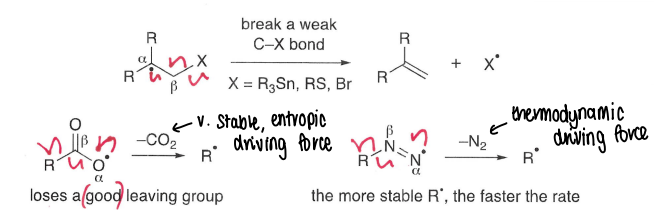
What are fragmentation reactions?
The reverse of radical addition, this is entropically favoured.
A weak bond is broken, forming a radical and often forming a stable gas molecule. This is the entropic and thermodynamic driving force.
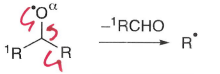
Why do oxygen radicals fragment easily?
Radicals on oxygen are unstable, they fragment to give a more stable radical.
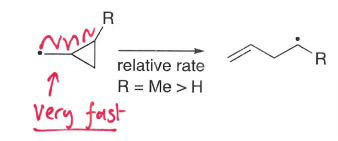
How does fragmentation cause ring opening?
Fragmentation of rings is very fast due to the release of strain, forming a more stable radical molecule.
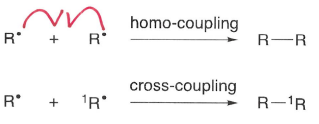
What are termination reactions?
Reactions which involve two radicals reacting to form a non-radical compound.
What are recombination reactions?
The reverse of bond homolysis, two radicals react to form a non radical compound.
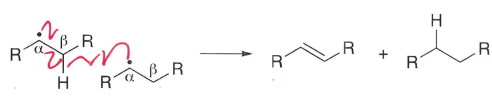
What are disproportionation reactions?
Transfer of a hydrogen atom between two radicals to form two non-radical compounds.
How does oxidation lead to termination?
An electron is removed from the radical, forming a cation. This cation then has a H+ removed, forming a neutral molecule.
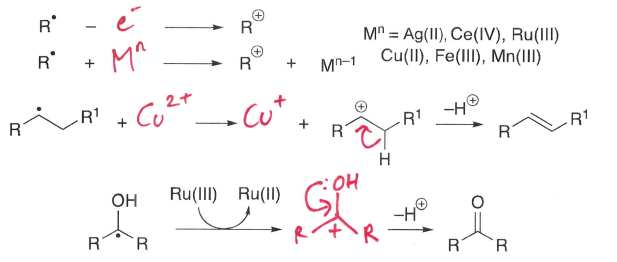
How does reduction lead to termination?
An electron is transferred to the radical, forming a anion. This then reacts with a H+, forming a neutral molecule.
How do you minimise termination?
Aim for a low concentration of radicals and fast propagation steps.
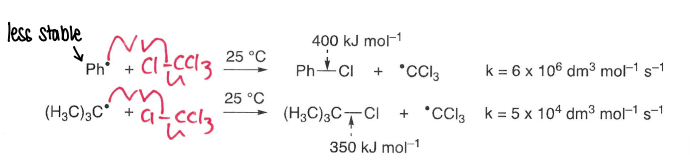
What kind of bonds tend to be weaker?
2 heteroatoms bonded tend to be weaker, e.g. N-S
How is enthalpy used to predict whether a reaction will take place?
Exothermic reactions result in the formation of strong bonds, therefore these reactions can proceed rapidly and often spontaneously.
Endothermic reacts lead to products with weaker bonds, therefore are generally very slow.
For radicals to react, we want to break weak bonds and form strong bonds.
How does entropy affect reaction favourability?
Reactions that increase entropy by increasing the number of molecular species are favourable, therefore homolysis of a bond to give two radicals is favoured.
How do steric effects affect a reaction?
Sterically hindered radicals require very strained transition states with high enthalpies of activation to react, this is disfavoured.
Sterics is also used to explain regioselective addition of radicals to alkenes and the stereoselective formation of adducts.
What orbitals react in a radical reaction?
The radical SOMO can interact with the HOMO or LUMO of a non-radical reactant, these orbitals must overlap.
Why is a linear transition state desired for an abstraction reaction?
This geometry allows maximum orbital overlap.
For a 1,5-HAT, a near linear geometry is achieved via the chair transition state.
What does the transition state look like for a radical addition to a C=C bond?
The transition state has an angle of attack close to 190 degrees.
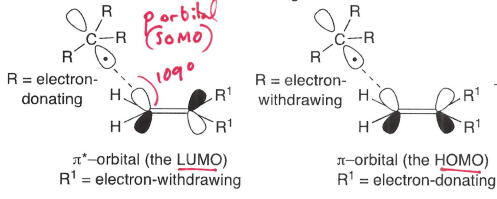
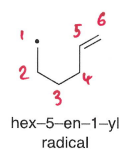
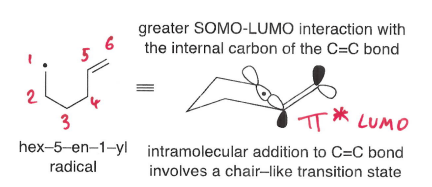
How does stereoelectronic factors explain the reactivity of the radical with the more hindered side of the alkene?
The chair-like transition state formed means there is greater SOMO-LUMO interaction with the internal carbon of the C=C bond, which is able to attack at an angle close to 109 degrees.
In the chair-like TS, substituents are typically equatorial.
What is required for a reaction to occur?
The interacting orbitals must overlap efficiently and have similar energies.
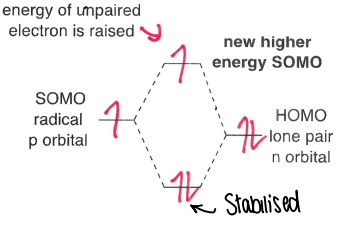
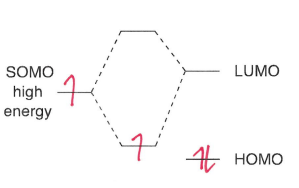
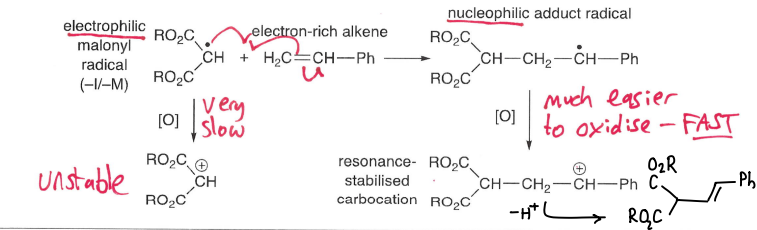
What are nucleophilic radicals?
Radicals adjacent to an electron donating group, therefore having a higher energy SOMO. This means they are more willing to give up an electron than accept one.
Although the energy of the u.p.e is raised, the two electrons in the lower MO are stabilised.
These groups include +I and +M groups.
What are electrophilic radicals?
Radicals adjacent to an electron withdrawing group, therefore the SOMO is lower energy. This means they are more willing to accept an electron.
These groups include -I and -M groups.
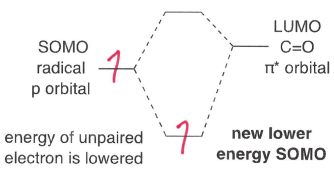
Why dies the SOMO-HOMO interaction dominate in electrophilic radicals?
The SOMO is lower energy, so it is closer in energy to the HOMO. The radical therefore reacts with the high energy HOMO at the most electron rich sites.
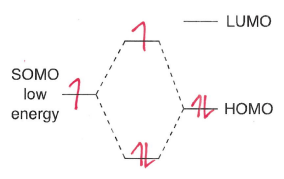
Why does the SOMO-LUMO interaction dominate in nucleophilic radicals?
The SOMO is higher energy, so is closer in energy to the LUMO. The radical therefore reacts with the low energy LUMO of the most electron poor site.
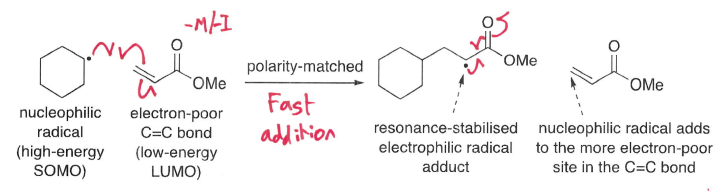
How can regioselectivity of radical addition be explained by polarity?
Nucleophilic radicals prefer to add to the most electron-poor site of a C=C bond, whereas electrophilic radicals prefer to add to the most electron-rich site.
What determines the rate of oxidation/reduction of radicals?
The redox potential of the redox agent and radicals.
Nucleophilic radicals are more easily oxidised, whereas electrophilic radicals are more easily reduced.
What is Baldwin’s rules?
It states that favoured cyclisations are those in which the length and nature of the linking chains enable the orbitals of the terminal atoms to achieve the proper geometries for orbital overlap and reaction.
What are exo reactions?
The bond is broken outside of the ring formed.
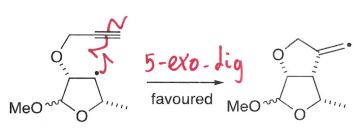
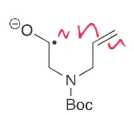
What are endo reactions?
The bond broken is inside the ring formed.
What are the terms used for the geometry at the reaction centre?
tet: sp3
trig: sp2
dig: sp
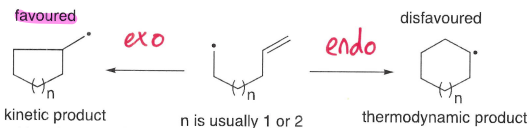
Which product is typically formed by radical reactions?
The kinetic is typically formed (the product of the faster reaction).
The thermodynamic can sometimes form when secondary reactions are possible to form a non-radical.
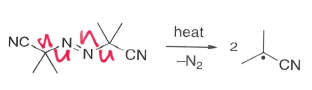
What is AIBN?
An initiator of radical reaction which fragments under heat to form nitrogen gas and a radical.
How can stereochemical outcome be explained by a transition state?
A chair like transition state can form, which places the R group in either the equatorial or axial position, leading to a product with specific stereochemistry.
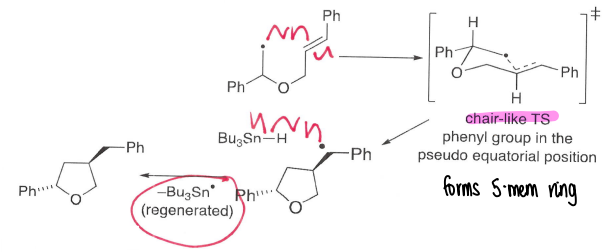
What is SnBu3H used for?
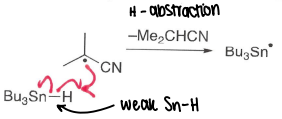
It is a good hydrogen atom source for radical chemistry due to the weak Sn-H bond.
It can react with the AIBN radical in a H-abstraction to form a tin radical, this is good at doing halogen atom abstractions, forming strong Sn-X bonds.
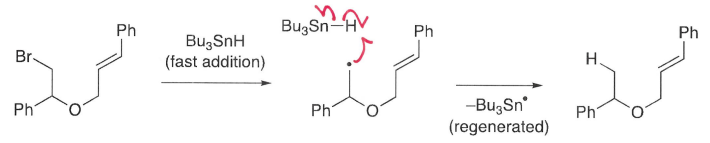
Why is Bu3SnH and AIBN added slowly?
This minimises simple reduction occurring, in which the C-X is replaced by a C-H. This reaction is often not wanted.
This ensures low concentration of Bu3SnH allowing time for cyclisation.
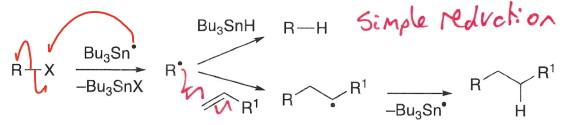
What pathways are possible in Bu3SnH mediated reactions?
The radical can either undergo addition or simple reduction.
How do you ensure fast rate of radical addition?
Match polarity of the radical and alkene.
How are steric effects used to direct stereoselectivity?
A chiral auxiliary can be used to direct radical addition to one face of the planar C=C bond.
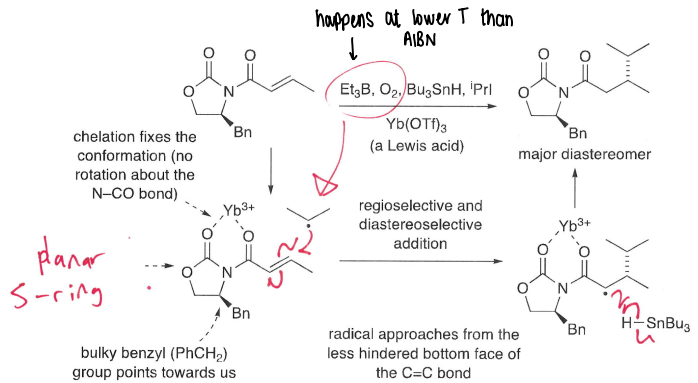
Why is Et3B used in place of AIBN?
Can react at lower T, also less prone to explosion.
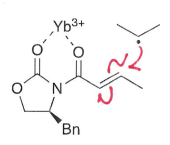
How does chelation of a metal with C=O increase rate of reaction?
The chelation can withdraw electron density from the double bond, reducing energy of the LUMO and so faster addition of the nucleophilic radical.
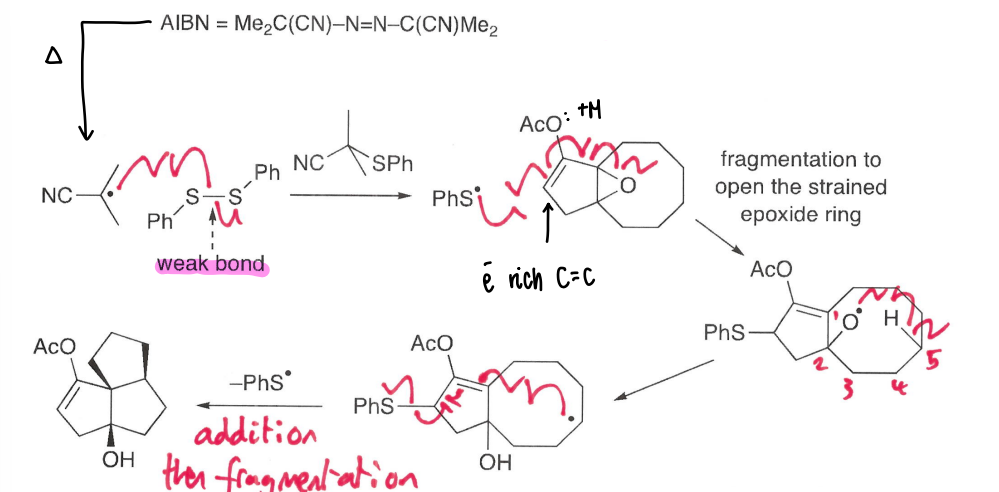
How does epoxide ring opening occur?
AIBN acts as an initiator, breaking down into the radical and N2.
This radical forms a PhS* radical which attacks the double bond, causing the epoxide ring opening.
This forms an oxygen radical, which undergoes a 1,5-HAT to form the stronger O-H bond.
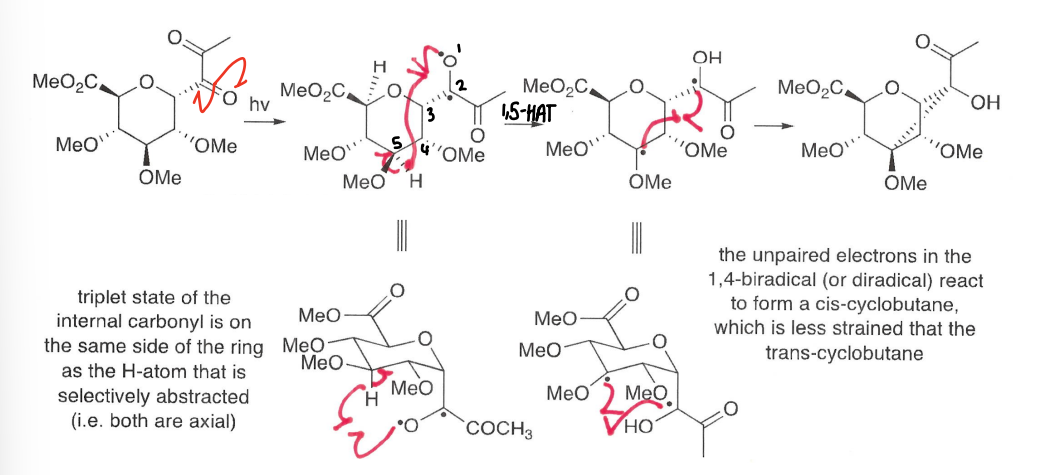
How do carbonyls undergo a 1,5-HAT following photolysis?
The carbonyl is excited to the triplet state radical, this then reacts with the hydrogen in the 5 position forming an OH bond.
Further reaction of the two radicals forms the end product.
What is the general mechanism for AIBN/organotin reactions with halogenoalkanes?
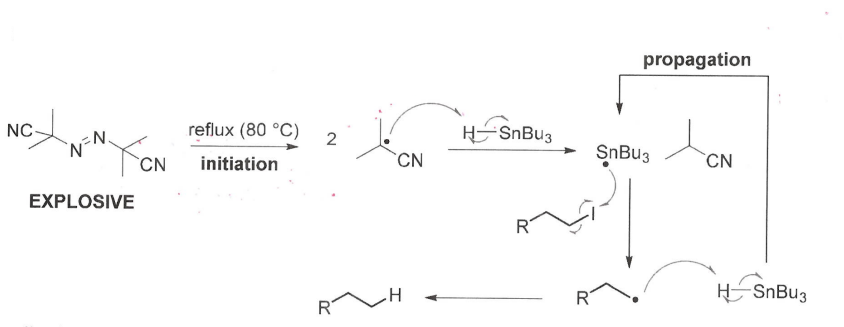
What are the drawbacks of AIBN/organotin radical chemistry?
AIBN is explosive
SnBu3H is very toxic
Benzene is required as the solvent, which is also very toxic.
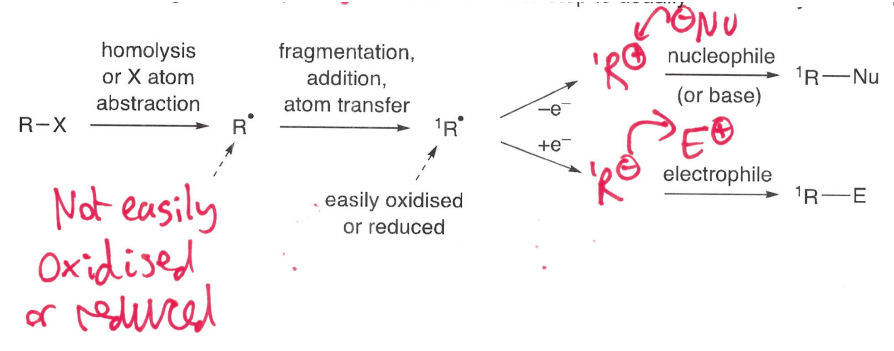
What are radical polar crossover reactions?
Reactions that involve sequential radical and ionic reactions.
E.g. a radical fragmentation/addition/atom transfer is usually followed by an oxidation or reduction.
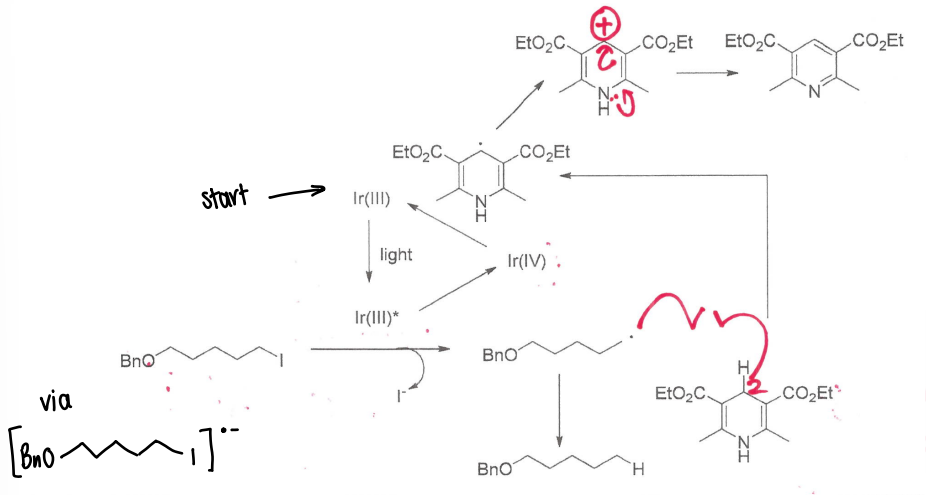
How can radicals be used in photoredox catalysis?
Exciting a photocatalyst causes an electron to be promoted to a higher energy orbital. This forms a radical, which therefore allows the photocatalyst to act as an oxidant or reductant.
How are photoredox catalysts used in place of AIBN/organotin reagents?
The Ir catalyst is excited, reacting with the reagent to form a radical.
How is SmI2 used as a reductant?
Acts as a powerful reducing agent, reducing carbonyls and forming the radical. The SmI2 remains coordinated to the oxygen. This can then further react, the chelation of the Sm ensures a cis-relationship between the OH and ester.
The O-Sm bond is hydrolysed to form the product.