hydrocarbons
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What is crude oil?
Straight and branched chain alkanes
Less amount of cycloalkanes and arenes
What is the uses of the petroleum gases fraction?
fuels
Feed stocks in some petrochemical processes
What is the uses of gasoline fraction in a fractional distillation?
petrol
Petrochemicals
What is the uses of naptha fraction in a fractional distillation?
Feedstock for petrochemical manufactures
What is the uses of kerosene fraction in a fractional distillation?
production of aviation fuel
Some Chemical processes
What is the uses of gas oil fraction in a fractional distillation?
diesel fuels
Heating
Lubricating oils
What is the uses of residue in a fractional distillation?
bitumen
Waxes
Less volatile lubricating oils
What is cracking?
Process that splits long chains of alkenes into a shorter chain alkanes and alkenes and hydrogen
What are the uses of cracking?
increases amount of gasoline
Increases branching in chains - factor for petrol
Produces alkenes - important feedstock for chemicals
How does combustion in alkanes occur?
chemicals react with oxygen to give out heat
Makes CO2 and H2O
Is there is an incomplete combustion then what happens?
lack of oxygen
Creates CO and 2H2O
How are alkanes chlorinated?
presence of UV light
UV light needed to break bonds inn chlorine molecule
How is the chlorine bonds broken?
chlorination - homolytic fission making chlorine free radical
How is free radicals reaction with alkanes?
reactive
In a free radical substitution reaction
draw a free radical mechanism.
during initiation - need homolytic fission + UV light for it to work
Can contain ethane - when 2 free radicals collide

Explain the bonding in ethene.
overlap of s orbital creates a sigma bond
Sideway overlap creates pi bond - joined together on top
Pi bond in a region of high electron density
Draw an electrophilic addition with bromine and hydrogen
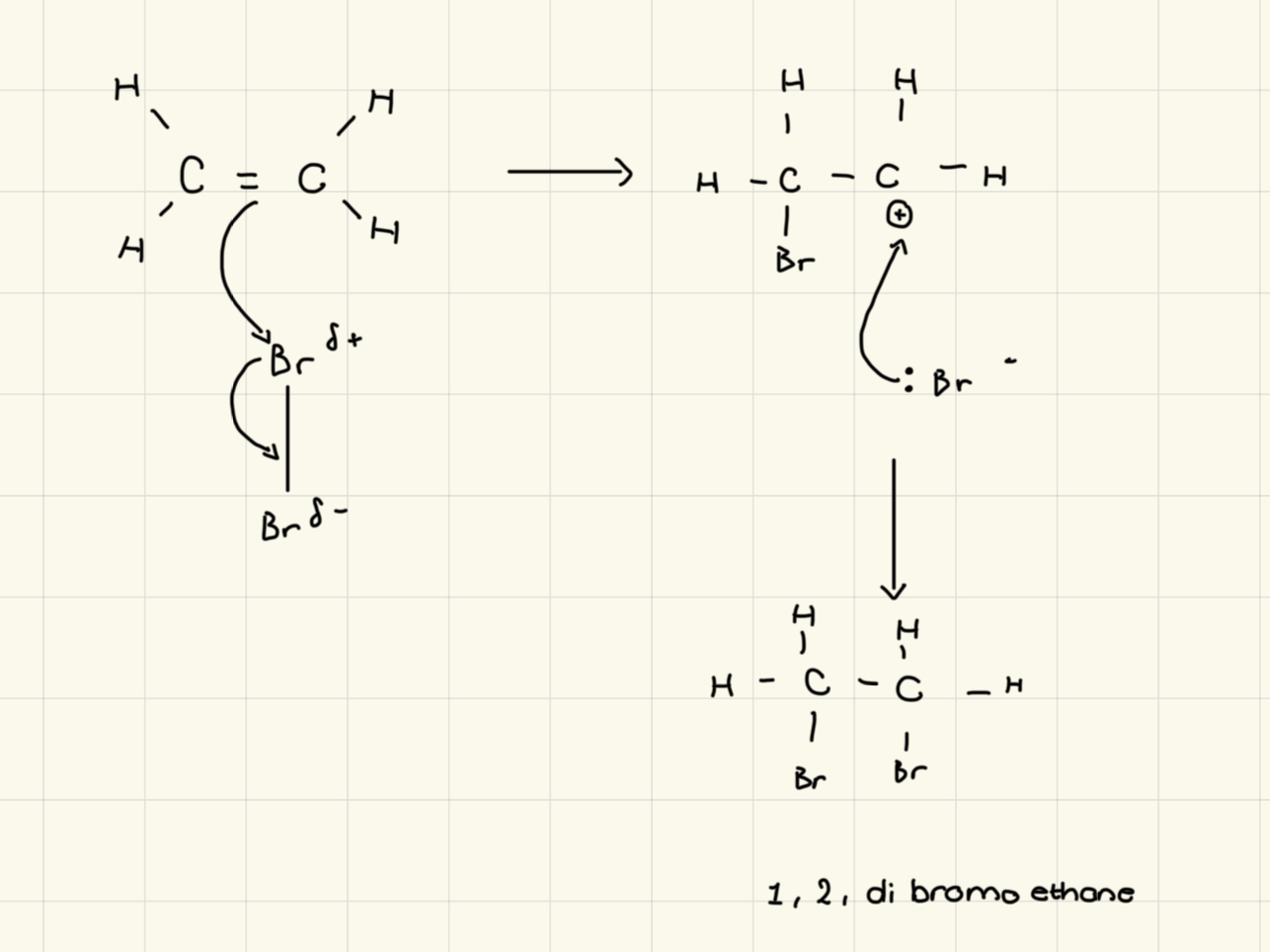
positive test for alkenes?
Bronine water
Brown —> colourless
Another way to test for alkenes?
potassium manganates (vii)
under cold
Acid - dilute sulphuric acid - purple —> colourless
Alkaline - sodium carbonate - purple —> dark green
What is carbocations stability and why?
increases as the number of r groups increase ( h groups decrease)
R groups contains greater electron density than h atoms - density attracted towards - reduces positive charge of carbon atoms
What is hydrogenation?
hydrogen added to carbon double bond using nickel catylyst
(Used in hydrogenated fats)
What is polymerisation?
Addition reactions to create a long chain polymer