Comprehensive Organic Chemistry and Reaction Mechanisms
1/347
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
348 Terms
What are the types of intermolecular forces?
Dispersion forces, dipole-dipole attractions, and hydrogen bonding.
What are dispersion forces?
The force of attraction between an instantaneous dipole and an induced dipole.
How does polarizability affect intermolecular forces?
Greater polarizability leads to stronger intermolecular forces.
What is the relationship between molecular size and polarizability?
Larger molecules are more polarizable due to loosely bound outer electrons.
What is dipole-dipole attraction?
Attraction between the positive and negative dipoles of neighboring polar molecules.
What defines a permanent dipole?
A permanent dipole occurs when one end of a molecule has a partial positive charge and the other has a partial negative charge.
How do dipole-dipole forces compare to London dispersion forces?
Dipole-dipole forces are stronger than London dispersion forces.
What is hydrogen bonding?
Attraction between a highly electronegative atom in one polar bond and a slightly positive hydrogen atom in another polar bond.
What properties of water are influenced by hydrogen bonds?
High heat capacity, high heat of evaporation, high cohesion surface tension, and excellent solvent properties.
What are adhesive forces?
Forces between unlike substances.
What are cohesive forces?
Forces between like substances.
Define viscosity.
The state of being thick, sticky, and semifluid in consistency due to internal friction.
What is surface tension?
The tension of the surface film of a liquid caused by the attraction of particles in the surface layer.
What is capillary rise?
The ability of a liquid to flow in narrow spaces without external forces.
How do intermolecular forces affect viscosity?
Stronger intermolecular forces lead to higher viscosity.
What are phase transitions?
Transitions between solid, liquid, and gaseous states of matter.
What is the significance of phase transition temperatures?
They are the temperatures required to induce a phase transition in a substance.
How do intermolecular forces relate to phase transition temperatures?
Stronger intermolecular forces result in higher phase transition temperatures.
What is a triple point in a phase diagram?
The point at which the three states of matter: gas, liquid, and solid coexist.
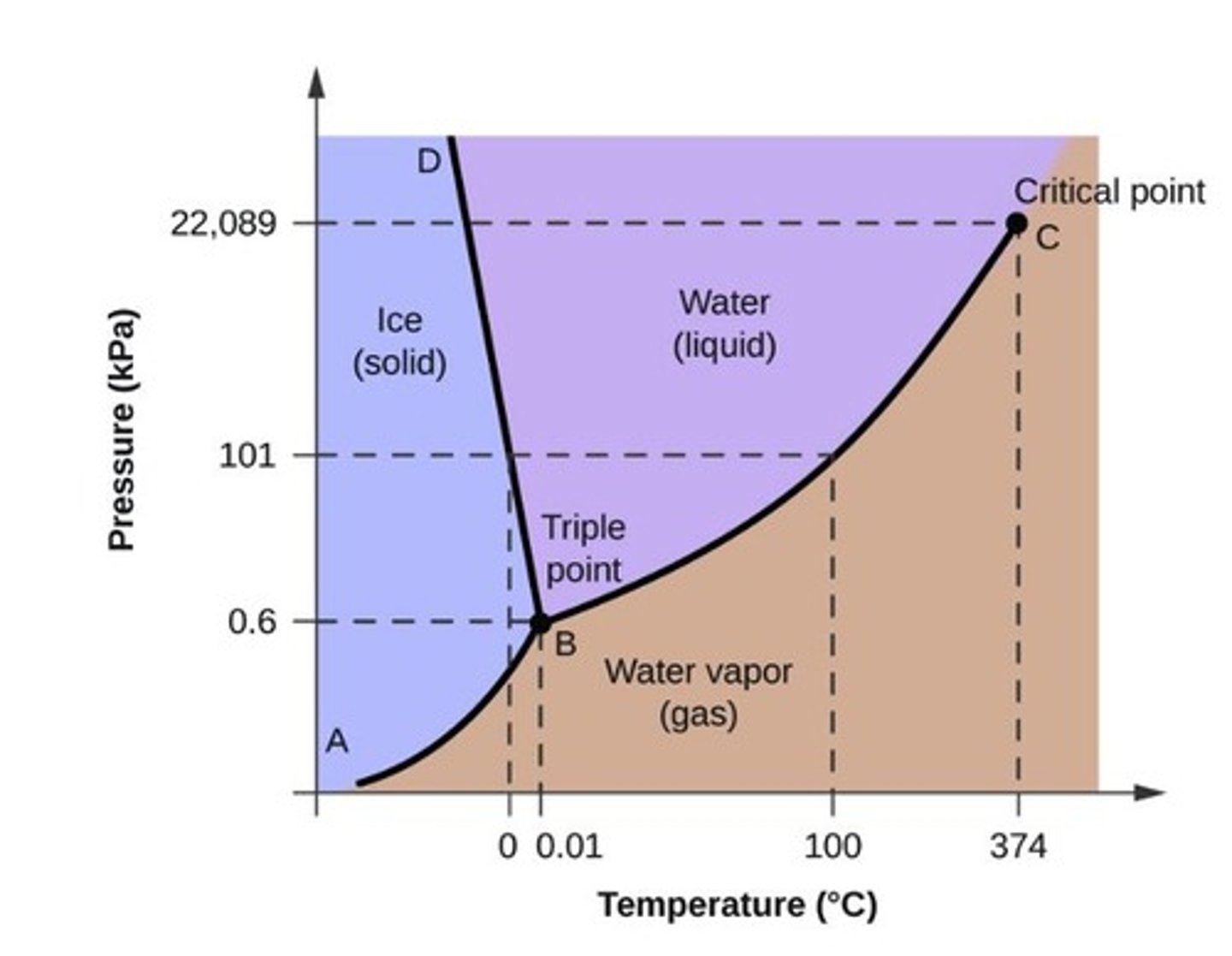
What is the critical point in a phase diagram?
The point at which the substance is indistinguishable between liquid and gaseous states.
What does the fusion curve represent in a phase diagram?
The transition between liquid and solid states.
What does the vaporization curve represent in a phase diagram?
The transition between gaseous and liquid states.
What does the sublimation curve represent in a phase diagram?
The transition between gaseous and solid states.
How can phase diagrams be used?
To identify the physical state of a substance at given temperatures and pressures.
What happens to water at a pressure of 50 kPa and a temperature of -10 °C?
Water exists only as a solid (ice) under these conditions.
What happens to water at a pressure of 50 kPa and a temperature of 50 °C?
Water exists only as a liquid under these conditions.
What is a supercritical fluid?
A substance at a temperature and pressure above its critical point, where distinct liquid and gas phases do not exist.
What unique behaviors do supercritical fluids exhibit?
They can effuse through solids like a gas and dissolve materials like a liquid.
What are the properties of ionic solids?
High melting point, brittle, hard, and held together by electrostatic forces.
What type of bonding occurs in molecular solids?
Hydrogen bonding, dipole-dipole interactions, and London Dispersion forces.
What are the characteristics of metallic solids?
Variable hardness and melting point, conducting, with valence electrons delocalized over many atoms.

What defines covalent network solids?
They are formed by networks of atoms held together by covalent bonds, resulting in high melting points and hardness.
How do crystal defects occur in solids?
Through imperfections in the geometrical shape of atoms, often caused by rapid cooling or high energy radiation.
What is lattice energy?
The energy required to separate 1 mole of a crystalline ionic solid into its component ions in the gas phase.
What role does X-ray diffraction play in crystallography?
It is used for phase identification of crystalline materials and provides information on unit cell dimensions.
What is solvation?
The process of breaking apart a solute in a solvent to form a homogeneous solution.
What factors affect the rate of solubility?
Greater solubility leads to faster solvation; concentration increases with a larger ratio of solute to solvent.
What is the heat of solution?
The sum of the enthalpy changes in separating solute and solvent molecules and forming solute-solvent interactions.
What are strong electrolytes?
Substances that completely dissociate into ions in solution, such as NaCl.
What distinguishes weak electrolytes from nonelectrolytes?
Weak electrolytes partially dissociate into ions, while nonelectrolytes do not produce ions but dissolve due to polarity.
How does temperature affect the solubility of solids and gases?
Higher temperature generally increases solubility for solids and liquids, while lower temperature increases solubility for gases.
What is the effect of pressure on gas solubility?
Higher pressure increases the solubility of gases in liquids.
What happens to the solubility of gases at lower temperatures?
Lower temperatures can increase the solubility of gases, preventing them from evaporating.
What is the relationship between electrolyte strength and solute-solvent attractive forces?
Stronger electrolytes have stronger solute-solvent attractive forces, allowing complete dissociation into ions.
What are the properties of molecular solids?
Soft, low melting, and easily vaporized due to weak intermolecular interactions.
What is the significance of ionic radii in crystal structures?
Ionic radii can be computed using unit cell dimensions to understand the arrangement of ions in a crystal lattice.
What defines the arrangement of atoms in crystalline structures?
Atoms and ions are bound by electrostatic attraction, with the arrangement depending on the size of the ions.
What is the role of temperature in the dissolution process?
Higher temperatures generally increase the solubility of solids and liquids.
How do intermolecular forces influence the heat of solution?
The heat of solution is affected by the intermolecular forces involved in separating solute and solvent molecules.
What is the difference between physical and chemical changes in dissolution?
Ionic compounds completely dissociate into ions, while covalent compounds interact with water through dipole-dipole interactions.
What does Henry's Law state?
The solubility of a gas in a liquid is proportional to the pressure of the gas over the solution.
What is the formula for Henry's Law?
c = kP, where c is concentration in molarity, k is a constant depending on temperature, and P is the pressure of the gas.
What is the unit for the constant k in Henry's Law?
mol/L*atm
Define immiscible liquids.
Two liquids that do not have any possible intermolecular forces with one another and will not mix or dissolve.
Give an example of immiscible liquids.
Nonpolar liquids are immiscible in water.
Define miscible liquids.
Two liquids which share intermolecular forces and are able to dissolve or mix with one another.
What types of substances are typically miscible in water?
Polar liquids or substances that form hydrogen bonds.
What is mole fraction?
The number of moles of a single solute divided by the total moles of all solutes/solvents.
How is mole fraction calculated?
Xi = moles of i / moles of total solution.
What is the significance of mole fractions in a solution?
All mole fractions of individual solutes should total to 1.
Define molality.
Calculated by taking the moles of solute and dividing by the kilograms of solvent.
What is the unit for molality?
Designated by a lower case m, with units of moles of solute/kg of solvent.
Why is molality useful?
It reports true concentration independent of temperature.
What are colligative properties?
Physical properties of solutions dictated by the number of solute particles present, not by chemical structures.
How does solute concentration affect vapor pressure?
The addition of a nonvolatile nonelectrolyte solute lowers vapor pressure.
What is the effect of adding a nonvolatile solute like sugar to a solution?
It lowers the vapor pressure of the solution.
What is the unit for molarity?
moles/L
What is the difference between molality and molarity?
Molality is based on the mass of solvent, while molarity is based on the volume of solution.
What happens to the volume of solutions when temperature changes?
Temperature changes affect the volume of solutions, but masses are not affected.
What is the relationship between solute concentration and intermolecular forces?
Miscible liquids share intermolecular forces, while immiscible liquids do not.
What is the formula for calculating molality?
m = moles of solute / kg of solvent.
What happens to the vapor pressure of a solvent when solute is added?
The vapor pressure decreases due to fewer solvent molecules being able to evaporate.
What is Raoult's Law?
Psolvent = Xsolvent * Po solvent, where Po solvent is the vapor pressure of the pure solvent.
What is the formula for calculating the change in vapor pressure?
ΔP = Xsolute * Po solvent.
What is osmosis?
The movement of solvent from an area of lower solute concentration to an area of higher solute concentration through a semipermeable membrane.
What occurs to red blood cells in a hypertonic solution?
Water flows out of the cells, causing crenation (shriveling).
What occurs to red blood cells in a hypotonic solution?
Water flows into the cells, causing hemolysis (bursting).
How does solute concentration affect boiling points?
An increase in solute concentration elevates the boiling point of the solution.
What is the formula for boiling point elevation?
ΔT = Kb * msolute.
How does solute concentration affect freezing points?
An increase in solute concentration decreases the freezing point of the solution.
What is the formula for freezing point depression?
ΔT = Kf * msolute.
What is osmotic pressure?
The pressure required to stop the flow of solvent through a semipermeable membrane.
What is the formula for calculating osmotic pressure?
Π = MRT, where M is molarity, R is the gas constant, and T is temperature in Kelvin.
What does the Van't Hoff Factor (i) represent?
It represents the number of particles in solution per mole of solute.
How does the presence of electrolytes affect freezing point depression?
ΔT = i Kf msolute, accounting for dissociation.
What is a colloid?
A dispersion of particles of one substance throughout another, intermediate between homogeneous and heterogeneous mixtures.
What is the Tyndall effect?
The scattering of light by particles in a colloid, which helps differentiate colloids from solutions.
What is the definition of reaction kinetics?
The study of the rates of chemical reactions and the steps involved in the reaction process.

What is the formula for calculating reaction rate?
Rate = -Δ[A]/Δt for reactants and Rate = Δ[B]/Δt for products.
What are initial rates in reaction kinetics?
Rates measured at the beginning of the reaction, dependent only on initial concentrations.
What is activation energy?
The minimum amount of energy needed for a reaction to occur.
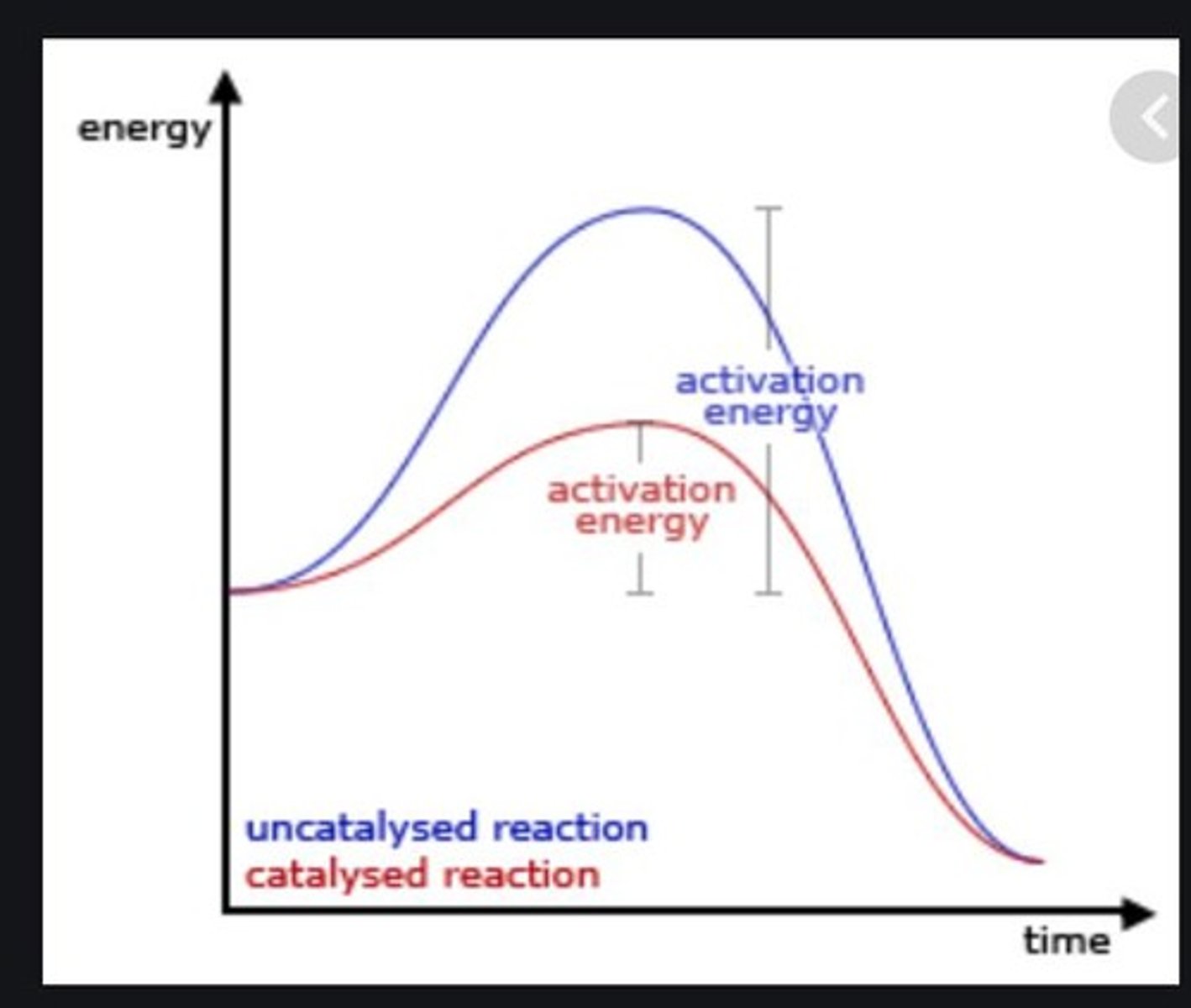
What factors affect the rate of a chemical reaction?
Concentration of reactants, temperature, physical state of reactants, presence of a catalyst, and light.
What is a rate law?
An equation that shows how the rate of a reaction depends on the concentrations of reactants.
What is the order of a reaction?
The exponent of a reactant concentration in the rate law, indicating how the rate depends on that reactant.
What is the difference between differential and integrated rate laws?
Differential rate laws show how the rate depends on concentrations, while integrated rate laws show how concentrations depend on time.
What is the significance of the rate constant (k)?
It is specific to a particular reaction at a given temperature and influences the rate of the reaction.
What is the formula for calculating the rate constant?
Units for k depend on the order of the reaction, which can be determined from experimental data.
What is the relationship between concentration and reaction rate?
Higher concentrations typically lead to increased reaction rates due to more frequent collisions.
What happens to reaction rates when temperature increases?
Reaction rates generally increase because particles collide more frequently and with greater energy.
How does surface area affect reaction rates?
Increased surface area allows for more collisions between reactant particles, speeding up the reaction.