26 Non-Visual Pathways
1/57
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
58 Terms
What are non-LGN targets of retinal ganglion cell (RGC) axons, and why are they important?
RGC axons project to several subcortical non-LGN targets that mediate visual functions beyond conscious perception, including attention, eye movements, and affective responses. Key targets include the pulvinar, superior colliculus, and lateral habenula.
What is the role of the pulvinar as a non-LGN target of RGC axons?
The pulvinar is involved in visual attention and selection. It helps filter out distracting stimuli and prioritize stimuli of interest, and may contribute to visually guided reaching and hand coordination.
How does the superior colliculus contribute to visual behavior?
The superior colliculus mediates saccade generation and coordinates eye, head, and neck movements, allowing rapid orientation toward salient visual stimuli (reflexive visual-motor responses).
What visual functions are associated with the lateral habenula?
The lateral habenula links visual input to emotion, pain processing, and learning. It is thought to mediate reflexive avoidance behaviors to visual threats and is implicated in mood regulation (dysfunction associated with depression).
What is the function of the Ventrolateral Pre-optic nucleus?
Sleep initiation and maintenance
What non‑LGN retinal targets are involved in sleep and circadian regulation?
RGC axons project to the ventrolateral pre‑optic nucleus (VLPO) and suprachiasmatic nucleus (SCN).
VLPO: regulates sleep initiation and maintenance
SCN: master clock that synchronizes circadian rhythms to light–dark cycles
How does retinal input to the suprachiasmatic nucleus (SCN) influence behavior?
Retinal signals to the SCN entrain the circadian rhythm, aligning sleep–wake cycles, hormone release, and behavior with environmental light cues (via intrinsically photosensitive RGCs).
Which non‑LGN retinal target mediates the pupillary light reflex, and how?
The pretectum (olivary pretectal nucleus) receives retinal input and mediates the pupillary light reflex by projecting bilaterally to the Edinger–Westphal nuclei, causing direct and consensual pupillary constriction.
What is the neural pathway mediating the pupillary light reflex (from retina to muscle)?
Light → retina → optic tract → pretectal nucleus → bilateral projections to Edinger‑Westphal (E‑W) nuclei → preganglionic parasympathetic fibers via CN III → ciliary ganglion → postganglionic fibers to iris constrictor (sphincter pupillae) muscle.
Why does shining light in one eye cause pupillary constriction in both eyes?
Pretectal neurons project bilaterally to both Edinger‑Westphal nuclei, leading to activation of parasympathetic output to both ciliary ganglia. This produces direct and consensual pupillary constriction.
What is the role of the Edinger‑Westphal nucleus and ciliary ganglion in pupil constriction?
Edinger‑Westphal nucleus: parasympathetic preganglionic neurons (CN III)
Ciliary ganglion: parasympathetic synapse site
Postganglionic fibers innervate iris constrictor muscle, causing miosis
What are the two major functions of the eye beyond image formation?
The eye supports:
Pattern vision – detailed, form‑based visual perception (LGN → visual cortex)
Irradiance detection – sensing overall environmental brightness to regulate pupil size and circadian rhythms (non‑LGN pathways)
What is irradiance detection, and how does it differ from pattern vision?
Irradiance detection measures ambient light intensity, not image detail.
Does not require conscious visual perception
Uses non‑LGN retinal pathways
Provides signals for automatic physiological regulation, unlike pattern vision which encodes shape, contrast, and spatial detail
What physiological processes depend on retinal irradiance signals?
Irradiance information from the retina is used to:
Control pupil size (pupillary light reflex via pretectum)
Regulate circadian rhythms (via SCN)
This allows adaptation to environmental light independent of visual cortex function.
How do ultradian and infradian rhythms differ from circadian rhythms?
Ultradian rhythms: period < 24 hours (e.g., sleep stages, hormone pulsatility)
Infradian rhythms: period > 24 hours (e.g., menstrual cycle, seasonal rhythms)
What causes the symptoms of jet lag?
Jet lag results from misalignment between the internal circadian clock and the external light-dark cycle, typically after rapid travel across time zones, leading to temporary disruption of circadian‑regulated physiology and behavior.
What are common symptoms of disrupted circadian rhythms (e.g., jet lag)?
Symptoms include daytime fatigue, difficulty falling asleep at night, irritability, headaches, digestive disturbances, and reduced ability to concentrate, reflecting widespread circadian control of neural and metabolic processes.
How does the body resynchronize after crossing time zones?
The internal circadian clock gradually realigns to local time, typically at a rate of ~1 day per time zone crossed, primarily through light‑dependent entrainment.
How do circadian rhythms differ between nocturnal and diurnal species?
Both nocturnal and diurnal species show ~24‑hour circadian rhythms, but the phase is shifted:
Nocturnal species: awake at night, sleep during the day
Diurnal species: awake during the day, sleep at night
Physiological rhythms (e.g., body temperature) are aligned with the species’ active phase.
What does body temperature reveal about circadian control?
Body temperature oscillates rhythmically over ~24 hours, peaking during the organism’s active phase and dropping during rest, demonstrating circadian regulation of core physiology independent of behavior.
What was the first experimental evidence for an internal clock in mammals?
Early studies of mice housed under constant conditions showed persistent activity–rest cycles, providing foundational evidence that mammals possess an endogenous circadian clock.
How widespread are circadian rhythms across living organisms?
Circadian rhythms are nearly universal, observed in animals (birds, reptiles, insects, worms) as well as plants, and even some algae and bacteria, indicating a deeply conserved biological mechanism.
What is the core functional advantage of having an internal circadian clock?
An internal clock allows organisms to predict rather than react to environmental changes, coordinating sleep, activity, metabolism, and physiology with the external day-night cycle for maximal efficiency and survival.
Why are human circadian rhythms often “masked” in daily life?
Modern humans are exposed to artificial timing cues (lights, clocks, social schedules) and reduced natural light (“Day Outside”), which can override or obscure the expression of the internal circadian clock, even though it is still present.
What is the approximate free‑running circadian period in humans?
In the absence of external time cues, humans exhibit a free‑running circadian rhythm of ~24.5 hours, indicating the internal clock is slightly longer than the 24‑hour day.
Where is the primary circadian clock located in the mammalian brain?
Lesion studies localized the circadian clock to the hypothalamus, specifically the suprachiasmatic nucleus (SCN), demonstrating that this region is necessary for normal circadian rhythms.
What experimental evidence identified the SCN as the circadian pacemaker?
Lesion experiments in rats (Richter, 1967) showed loss of circadian rhythmicity, and later studies demonstrated that retinal axons project directly to the SCN (Moore & Lenn, 1972), linking light input to clock regulation.
How does retinal input reach the circadian clock?
A small subset (~1–2%) of retinal ganglion cell (RGC) axons projects via the retinohypothalamic tract to the SCN, providing light information used to entrain circadian rhythms, rather than image formation.
Why is the SCN’s connection to the retina functionally important?
Direct retinal input allows the SCN to synchronize (“entrain”) the internal circadian clock to the external light-dark cycle, explaining how environmental light resets daily rhythms despite their endogenous origin.
What happens to circadian rhythms after an SCN lesion?
SCN lesions abolish circadian rhythmicity, producing arrhythmic behavior and physiology even under constant environmental conditions (e.g., constant darkness), demonstrating that the SCN is required for circadian organization.
Why did rod/cone‑loss mice challenge the classical view of irradiance detection?
Mice with complete rod and cone degeneration still entrain normally to day-night light cycles, indicating that rods and cones are not required for circadian photic entrainment.
What happens to circadian entrainment after bilateral enucleation?
With bilateral enucleation (eye removal), animals do not entrain to the light–dark cycle and instead free‑run, demonstrating that ocular input is required for light‑based circadian entrainment.
What surprising result was found in mice with complete rod and cone loss?
Despite lacking rods and cones, these mice still entrain normally to day-night light cycles, indicating that classical photoreceptors are not required for circadian irradiance detection.
What key conclusion follows from circadian entrainment in “blind” mice?
Circadian photic entrainment depends on a non‑classical retinal pathway, distinct from pattern vision, mediated by a specialized class of retinal ganglion cells rather than rods or cones.
What are intrinsically photosensitive retinal ganglion cells (ipRGCs)?
ipRGCs are a small subpopulation of retinal ganglion cells that are directly photosensitive, meaning they can respond to light independently of rods and cones. They project to non‑visual targets, especially the SCN, for circadian regulation.
What key experiments demonstrated that some RGCs are directly photosensitive?
Berson et al. (2002) recorded directly from RGCs projecting to the SCN and showed they exhibited light‑evoked firing even when rods and cones were nonfunctional, proving intrinsic photoreception.
What photopigment enables intrinsic photosensitivity in ipRGCs?
ipRGCs express melanopsin, a photopigment identified by Hattar et al. (2002).
Melanopsin was previously known from frog skin melanophores, indicating a non‑classical phototransduction system distinct from rods and cones.
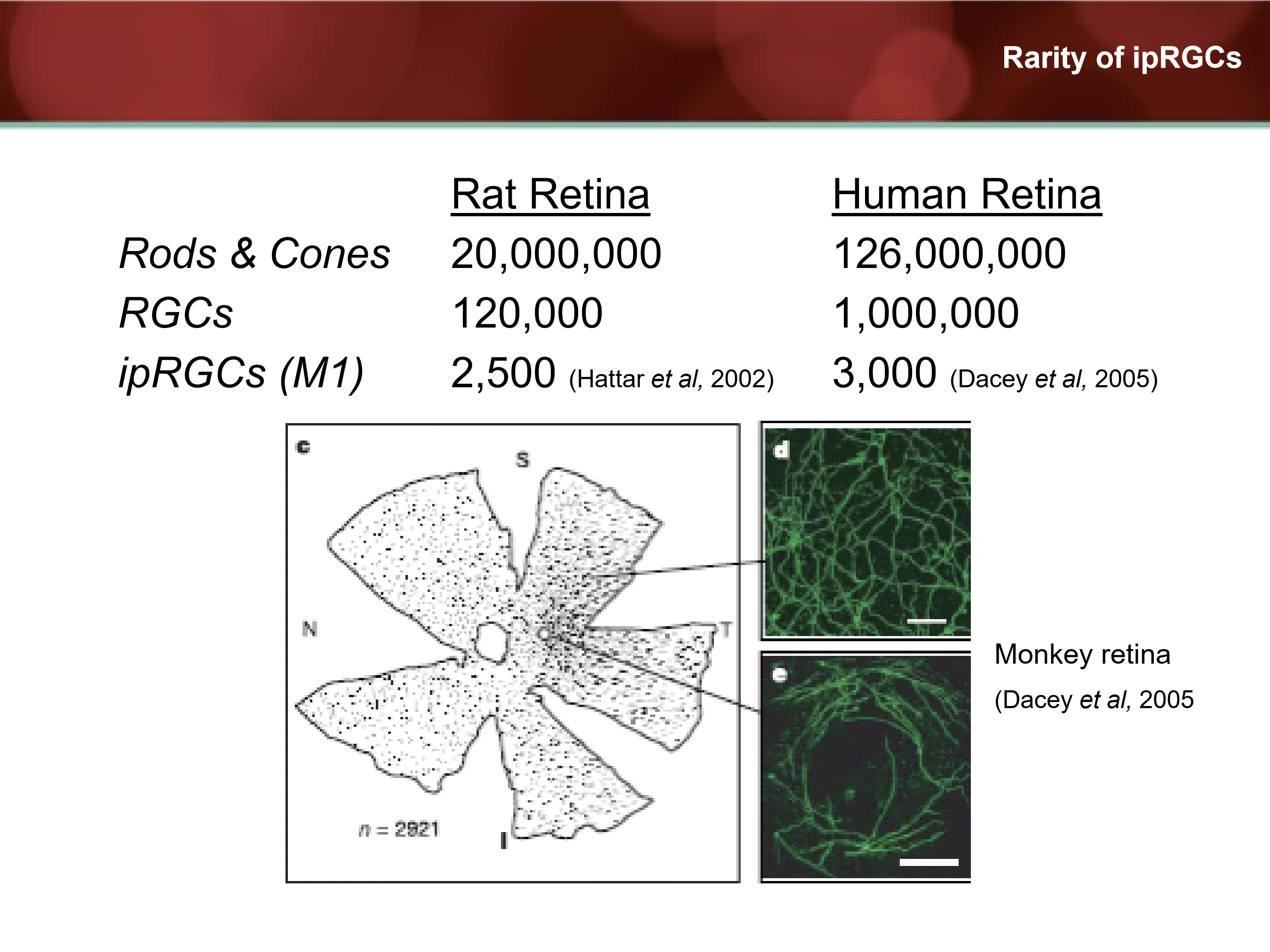
How rare are ipRGCs compared to other retinal neurons?
Very. With only 3,000 in human retina and 2,500 in rat retina.
What wavelength of light most strongly activates melanopsin‑containing ipRGCs?
What wavelength of light most strongly activates melanopsin‑containing ipRGCs?
How does melanopsin sensitivity differ from rod and cone photoreceptors?
Melanopsin requires higher irradiance (brighter light) to activate than rods or cones, meaning ipRGCs are tuned to ambient light levels rather than low‑light image formation.
What functional role does high irradiance threshold serve for ipRGCs?
The high threshold ensures ipRGCs respond primarily to daylight‑level illumination, allowing reliable detection of day-night transitions rather than transient or dim visual stimuli.
What happens to circadian entrainment when ipRGCs are completely ablated?
Animals fail to entrain to light-dark cycles, becoming circadian‑blind, even though rods and cones remain intact, demonstrating that ipRGCs are required for photic circadian entrainment.
Are mice lacking ipRGCs visually blind? What visual functions remain?
No. Mice without ipRGCs retain largely normal visual acuity and visually guided behavior, showing that image‑forming vision (rods/cones → cortex) is intact while circadian photoreception is lost.
What core brain pathway links light exposure to circadian physiology?
Light → ipRGCs (melanopsin‑mediated) → suprachiasmatic nucleus (SCN) → downstream regulation of circadian rhythms, hormone secretion, sleep–wake cycles, and behavior.
Beyond the SCN, which brain regions do ipRGCs project to, and why is this significant?
ipRGCs project to several non‑SCN targets, including the ventrolateral pre‑optic nucleus (VLPO). This demonstrates that ipRGCs influence sleep regulation directly, not solely via circadian timing signals from the SCN.
How does ipRGC input to the VLPO differ from ipRGC input to the SCN?
ipRGC → SCN: regulates circadian phase and timing
ipRGC → VLPO: influences acute sleep-wake regulation
How do ipRGCs regulate melatonin secretion?
ipRGCs provide light input to the SCN, which then regulates the pineal gland via a multisynaptic autonomic pathway. Light activation of ipRGCs suppresses melatonin release, while darkness permits melatonin secretion.
What key human evidence shows that circadian photoreception can be preserved without visual perception?
Zaidi et al. (Curr Biol, 2007) studied patients with cone‑rod dystrophy / retinitis pigmentosa who lacked conscious light perception but still showed light‑dependent physiological responses, demonstrating preserved non‑image‑forming (circadian) photoreception.
What does OCT and ERG evidence indicate in patients with rod–cone dystrophy?
OCT: severe photoreceptor layer degeneration
ERG: absent or markedly reduced rod/cone responses
Yet circadian light responses persist, supporting the conclusion that ipRGCs survive and function despite loss of classical photoreceptors.
What is the effect of light exposure on melatonin secretion at night?
Light exposure suppresses melatonin secretion at night. This is a well‑established physiological response and reflects light input to circadian and sleep‑regulating pathways rather than image‑forming vision.
How effective are common blue‑blocking anti‑reflective (AR) coatings at filtering blue light?
Most commercially available AR blue‑blocking coatings filter only a relatively small percentage of short‑wavelength light, limiting their impact on ipRGC activation and circadian signaling.
What does current evidence show regarding blue‑blocking lenses and visual or sleep outcomes?
A systematic review and meta‑analysis (2017) found:
Few well‑controlled studies (only ~3 at the time)
No convincing evidence that blue‑blocking lenses improve contrast sensitivity, eyestrain symptoms, sleep quality, or circadian outcomes
What role do ipRGCs play in the pupillary light reflex?
ipRGCs project to the olivary pretectal nucleus (OPN) in the midbrain. This pathway contributes to the pupillary light reflex in vivo, particularly sustained pupil constriction in bright light.
What is meant by redundancy in the pupillary light reflex (PLR) circuit?
The PLR circuit is redundant, meaning either rods/cones OR ipRGCs (melanopsin) can independently mediate pupillary constriction in response to light, ensuring robust reflex function.
Under what condition is the pupillary light reflex abolished?
The PLR is abolished only when both systems are disabled (melanopsin (ipRGCs) AND rods/cones are knocked out) demonstrating that these pathways provide parallel and redundant inputs to the reflex circuit.
How do ipRGCs differ from rods and cones in their temporal response to light?
ipRGCs respond sluggishly: they show slow onset, prolonged firing during illumination, and persistent activity after light offset. In contrast, rods and cones have high temporal resolution with rapid on/off responses.
What functional consequence follows from the sluggish temporal dynamics of ipRGCs?
ipRGCs have poor temporal resolution but are well‑suited for encoding ambient light intensity and duration, making them ideal for circadian entrainment and sustained reflexes rather than fast image formation.
Why does blue light cause slower pupil re‑dilation than red light in humans?
Blue light preferentially activates melanopsin‑containing ipRGCs, whose persistent firing produces a prolonged post‑illumination pupil response (PIPR). Red light relies more on rods/cones, which recover faster.