GCSE Chemistry: Bonding, Structure, and Properties of Matter
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
What is a compound?
A substance in which two or more elements are chemically combined.
What are the three types of strong chemical bonds?
Ionic, covalent, and metallic.
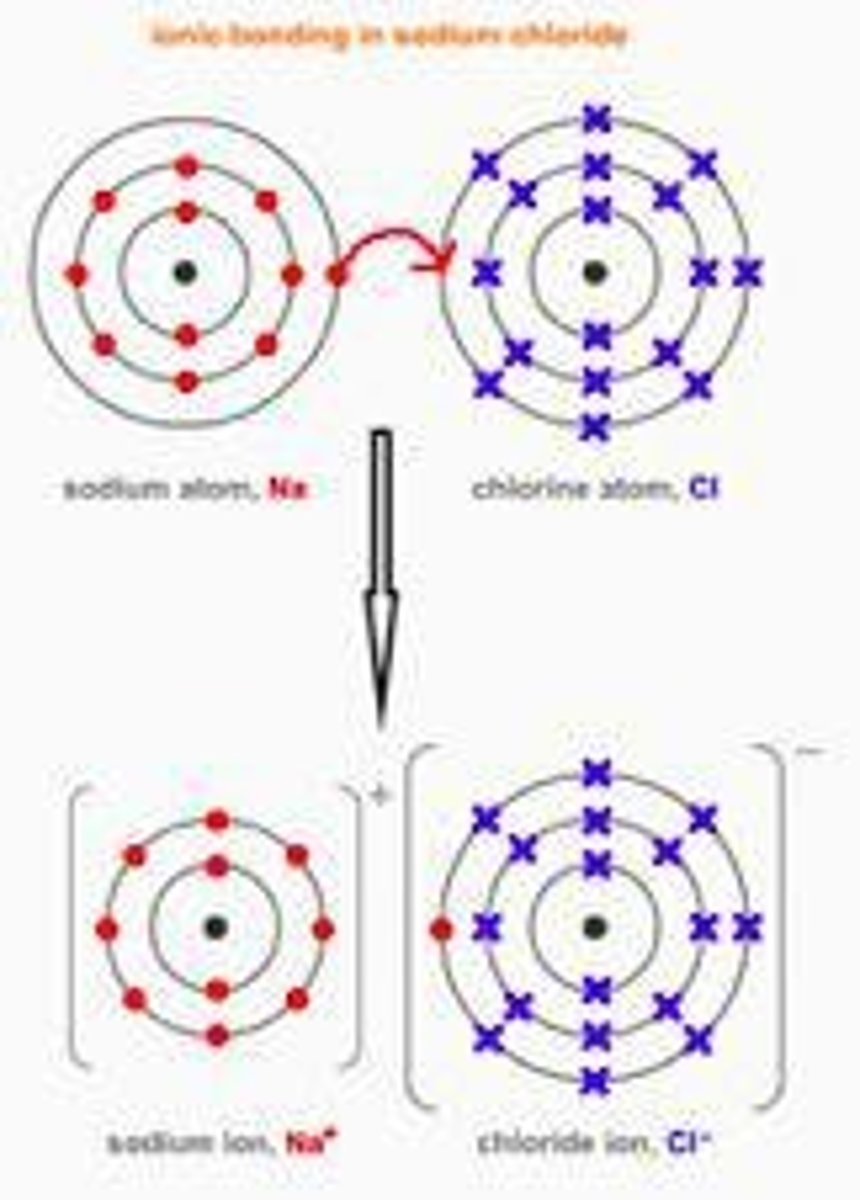
What characterizes ionic bonding?
Particles are oppositely charged ions formed from metals combined with non-metals.
What characterizes covalent bonding?
Particles are atoms that share pairs of electrons, occurring in non-metallic elements and compounds.
What characterizes metallic bonding?
Particles are atoms that share delocalised electrons, occurring in metallic elements and alloys.
What happens to metal atoms during ionic bonding?
Metal atoms lose electrons to become positively charged ions.
What happens to non-metal atoms during ionic bonding?
Non-metal atoms gain electrons to become negatively charged ions.
What is an ion?
An atom that has lost or gained electron(s).
Which groups of elements typically form ions in ionic compounds?
Metals in Groups 1 and 2, and non-metals in Groups 6 and 7.
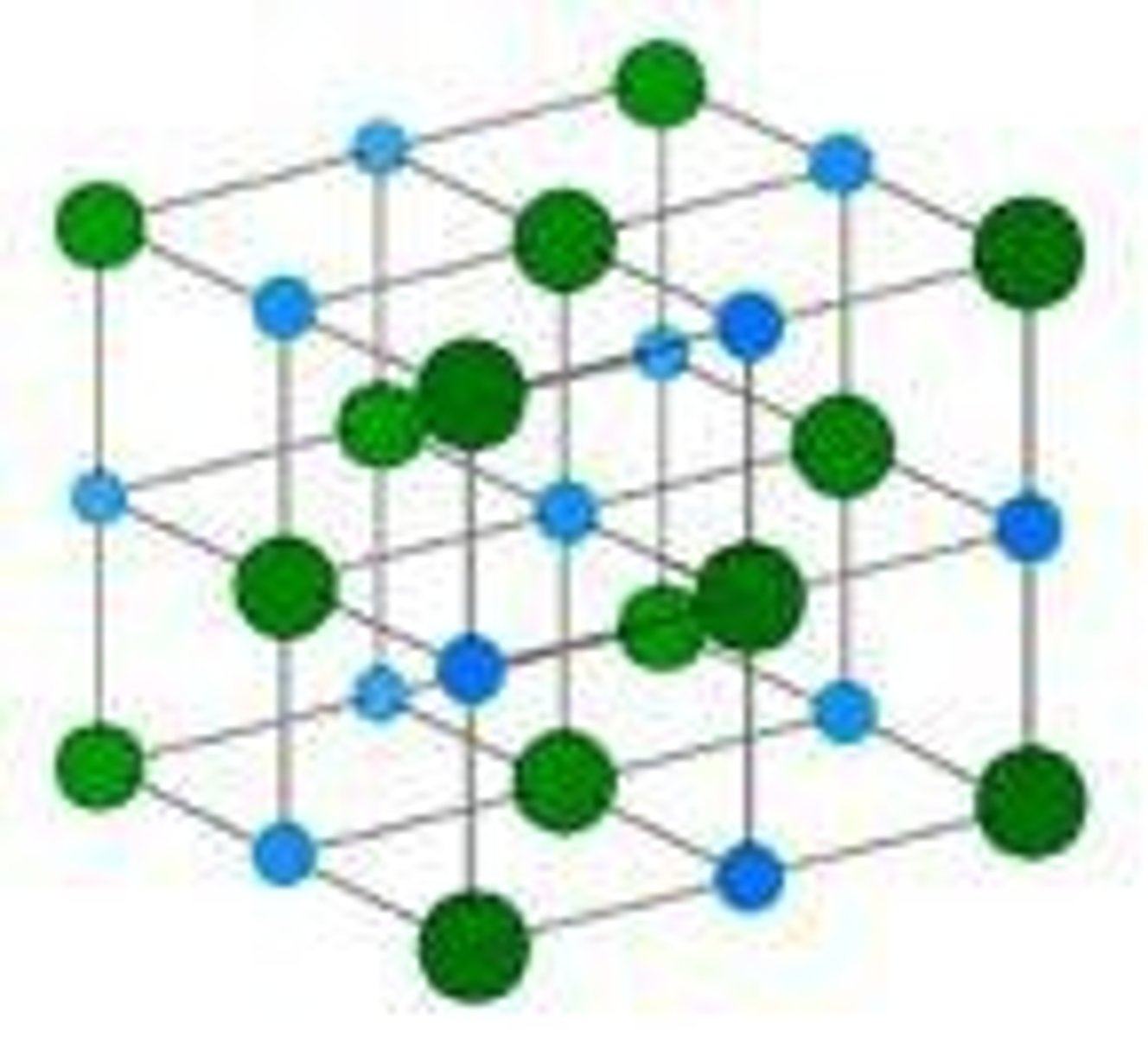
What is the structure of ionic compounds?
A giant structure of ions held together by strong electrostatic forces of attraction.
What is a characteristic of covalent bonds in small molecules?
They are strong within the molecules but weak between them.
What are giant covalent structures?
Structures consisting of many atoms covalently bonded in a lattice, such as diamond and silicon dioxide.
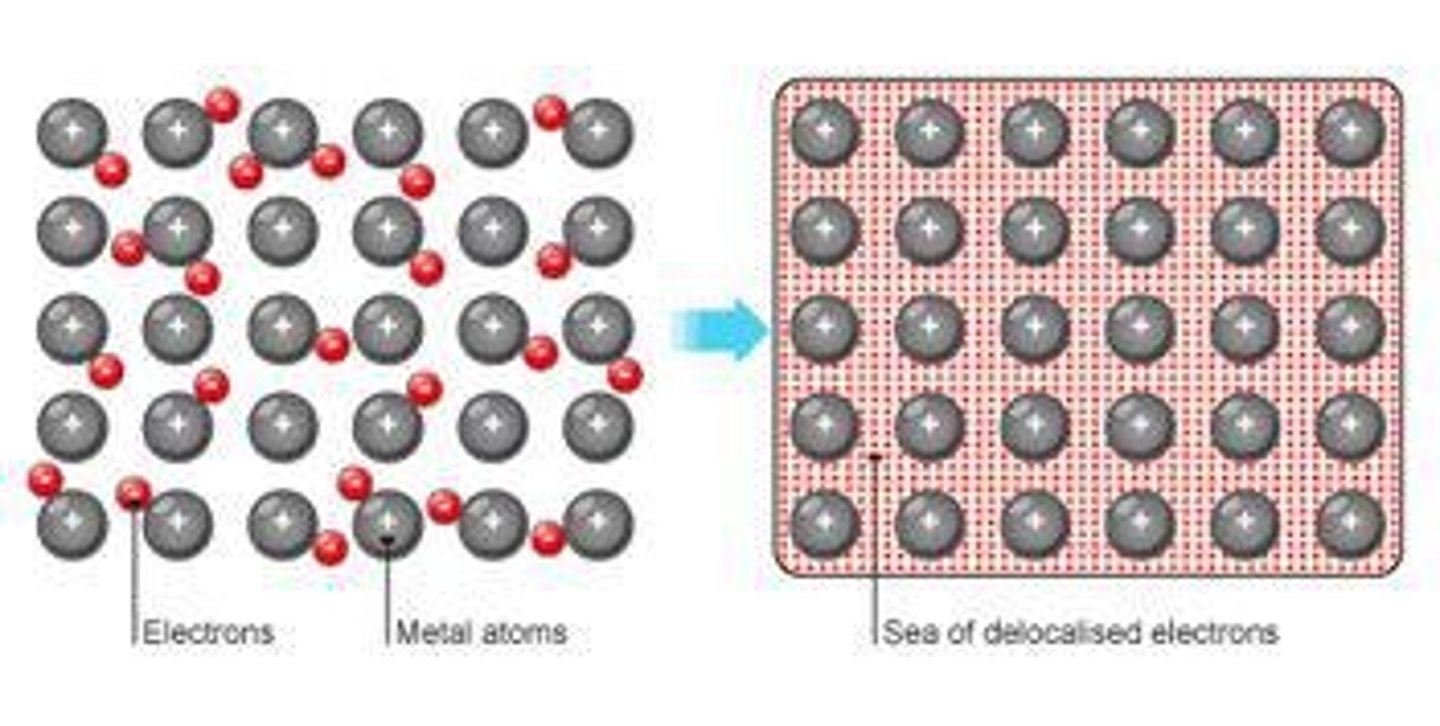
What is the arrangement of particles in metallic bonding?
Positive ions and delocalised electrons arranged in a regular pattern.
What allows metals to conduct electricity?
Delocalised electrons that are free to move through the structure.
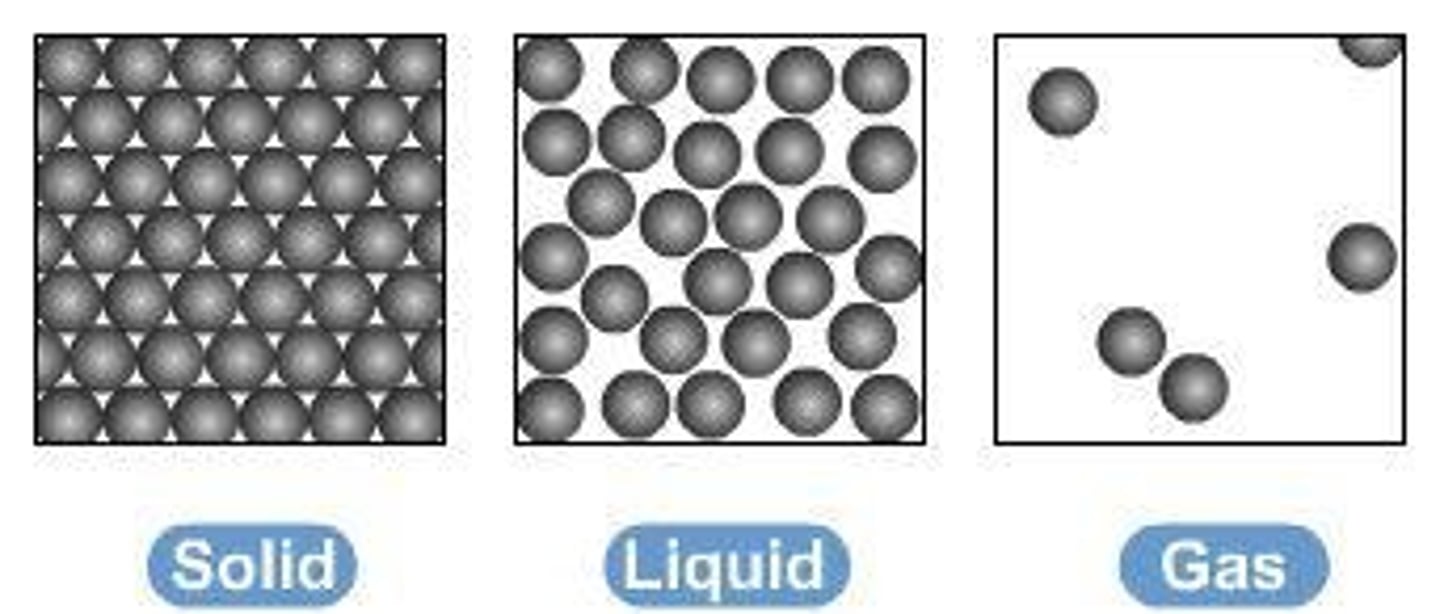
What are the three states of matter?
Solid, liquid, and gas.
What occurs at the melting point?
Melting and freezing take place.
What occurs at the boiling point?
Boiling and condensing take place.
How does particle theory explain state changes?
The amount of energy needed to change state depends on the strength of the forces between particles.
What are state symbols in chemical equations?
They indicate the states of matter: solid (s), liquid (l), gas (g), and aqueous (aq).
What are the properties of ionic compounds?
They have high melting and boiling points and conduct electricity when melted or dissolved.
What are the properties of small molecules?
They usually have low boiling and melting points and do not conduct electricity.
What happens to intermolecular forces as the size of molecules increases?
Intermolecular forces increase, leading to higher melting and boiling points.
What are polymers?
Substances with very large molecules linked by strong covalent bonds.
What type of forces exist between polymer molecules?
Relatively strong intermolecular forces.
What state are polymers typically in at room temperature?
Solids.
Give examples of giant covalent structures.
Diamond, graphite, and silicon dioxide (silica).
What is a key property of metals?
They have giant structures of atoms with strong metallic bonding.
Why do metals have high melting and boiling points?
Due to strong metallic bonds.
What allows metals to be bent and shaped?
The layers of atoms can slide over each other.
What are alloys?
Materials made from two or more different types of metals.
How do alloys compare to pure metals in terms of hardness?
Alloys are harder than pure metals due to distorted layers.
What makes metals good conductors of electricity?
Delocalised electrons carry electrical charge through the metal.
How do metals conduct thermal energy?
Energy is transferred by delocalised electrons.
What is diamond's structure?
Each carbon is joined to 4 other carbons covalently.
What are the properties of diamond?
Very hard, very high melting point, and does not conduct electricity.
Describe the structure of graphite.
Each carbon is bonded to 3 others, forming layers of hexagonal rings.
Why is graphite slippery?
Layers can slide over each other due to weak intermolecular forces.
What is a unique property of graphite compared to diamond?
Graphite can conduct electricity due to delocalised electrons.
What is graphene?
A single layer of graphite with unique properties useful in electronics.
What are fullerenes?
Molecules of carbon atoms with hollow shapes based on hexagonal rings.
What is Buckminsterfullerene (C60)?
The first fullerene discovered, with a spherical shape.
What are carbon nanotubes?
Cylindrical fullerenes with high length to diameter ratios.
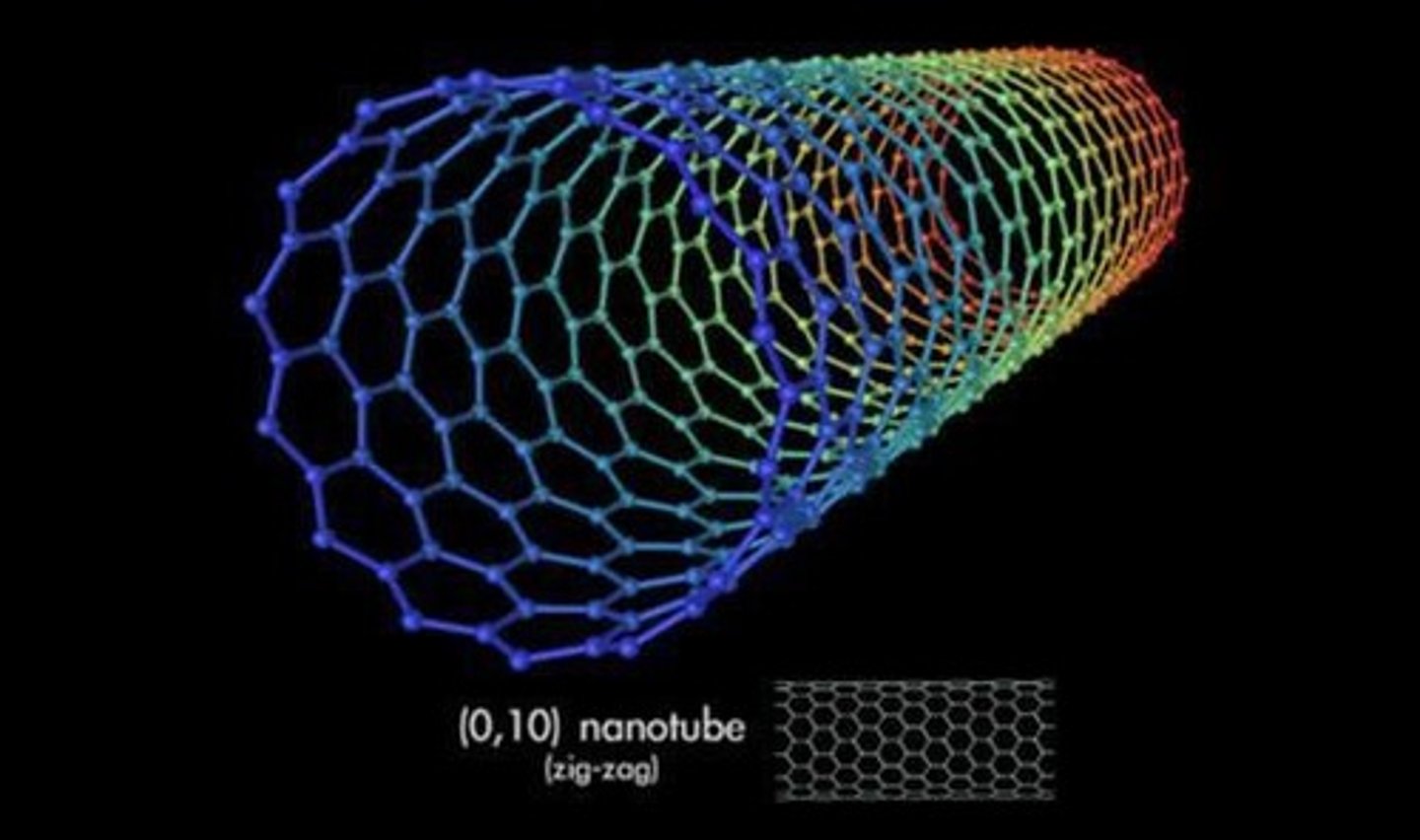
What are some uses of carbon nanotubes?
Lubricants, drug delivery, catalysts, and reinforcing materials.
What are nanoparticles?
Particles that are 1-100 nanometers across, containing a few hundred atoms.
How do nanoparticles differ from fine particles?
Nanoparticles are smaller than fine particles (PM2.5).
What happens to the surface area to volume ratio as the size of a cube decreases?
It increases by a factor of 10.
What are some advantages of nanoparticles?
High surface area to volume ratio makes them good catalysts and useful in sensors.
What are potential disadvantages of nanoparticles?
Concerns about toxicity and the ability to enter the bloodstream and brain.