MII2 killing pathogens
1/82
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
83 Terms
what is the role of CD4+ T cells
Help activate B cells and macrophages by releasing cytokines.
what is the role of CD8+ T cells
killing infected target cells, macrophage activation
what are the 4 subsets of Th cell
Th1, Th2, Th17, Tfh
what is the purpose of the different Th subsets
specialised for dealing with different infection types
what is the first signal required for Th cell subset differentiation
T cell receptor recognises an Ag presented by MHC
what is the second signal required for Th cell subset differentiation
Ag presenting cell upregulates B7 to signal to the T cell that the Ag is from a microbe
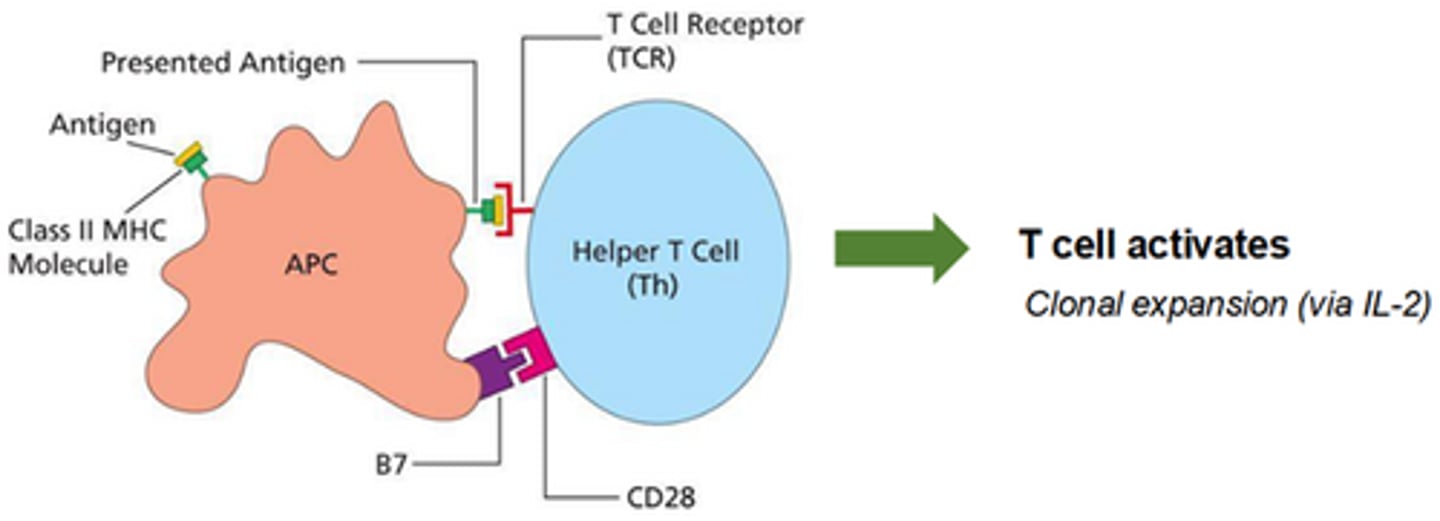
what is the third signal required for Th cell subset differentiation
usually given by cytokines-- tells the T cell the infection type (eg bacteria, virus, parasite etc), and this which subset to differentiate into
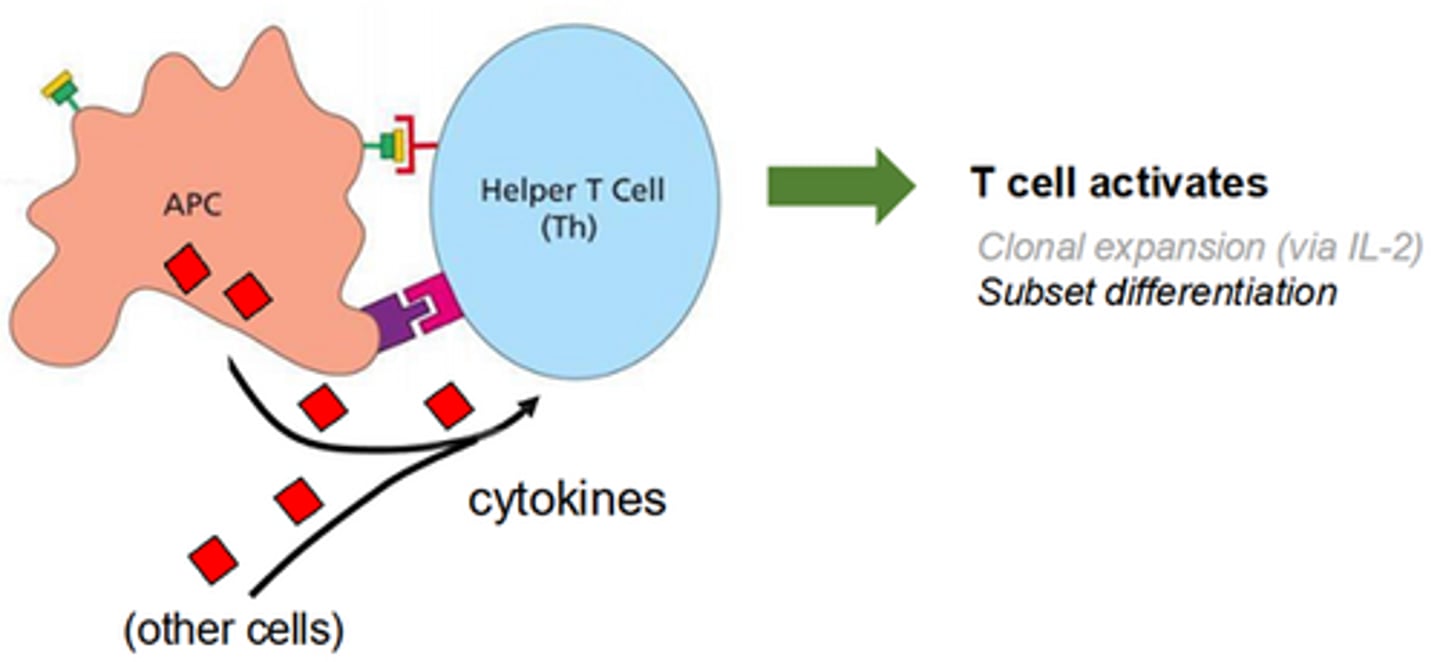
which subset T cells migrate to infection sites once differentiated
Th1,2,17
where does Tfh go once differentiated
remains in the lymph node for interaction with B cells
what are activated (antibody producing) B cells called
plasma cells
what are B cell effector functions defined by
the class/isotype of antibody it produces
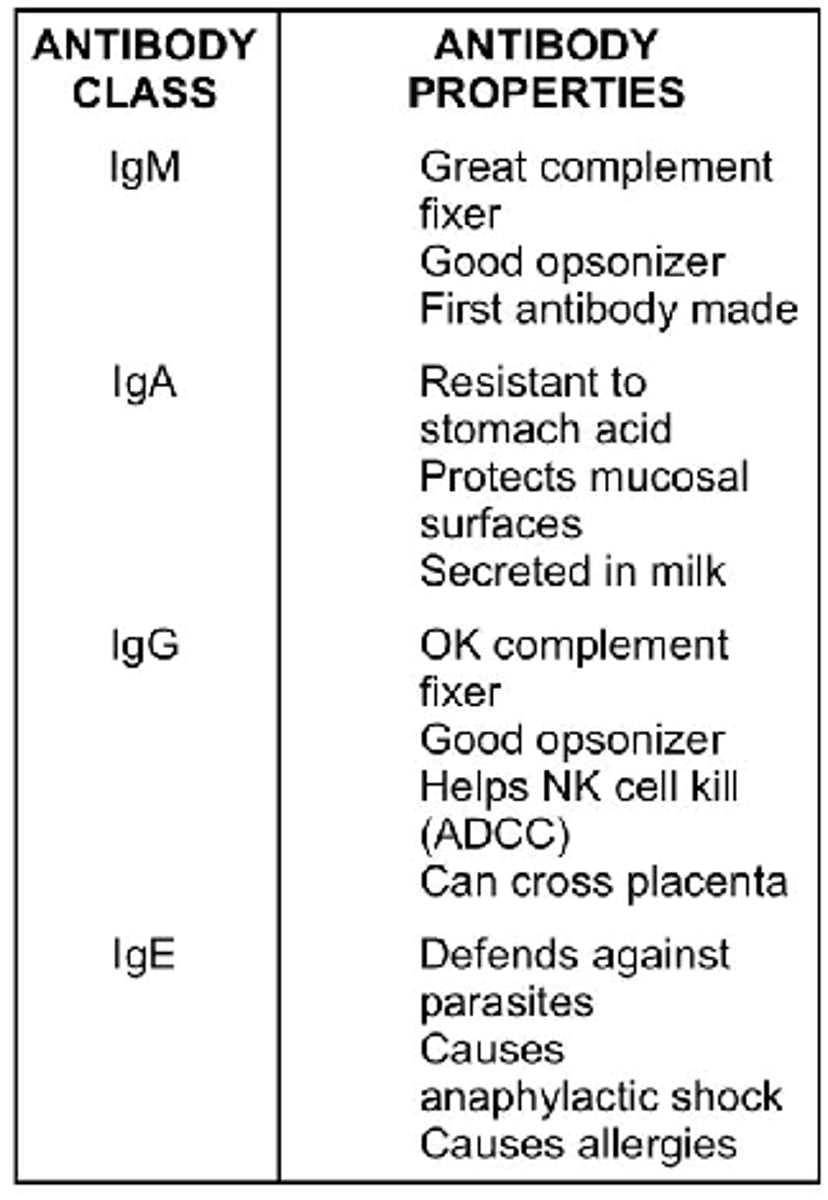
what are the types of antibodies (reminder)
what are the three things antigens can help block to prevent pathogen infection
1. penetrating epithelial barrier
2. binding of microbe for infecting of cells
3. binding of toxins to cellular receptors
what does the antibody in the SARS Cov-2 vaccine do
prevents entry to cells by acting on the spike protein
what does the antibody in the tetanus vaccine do
blocks the function of the tetanus toxin
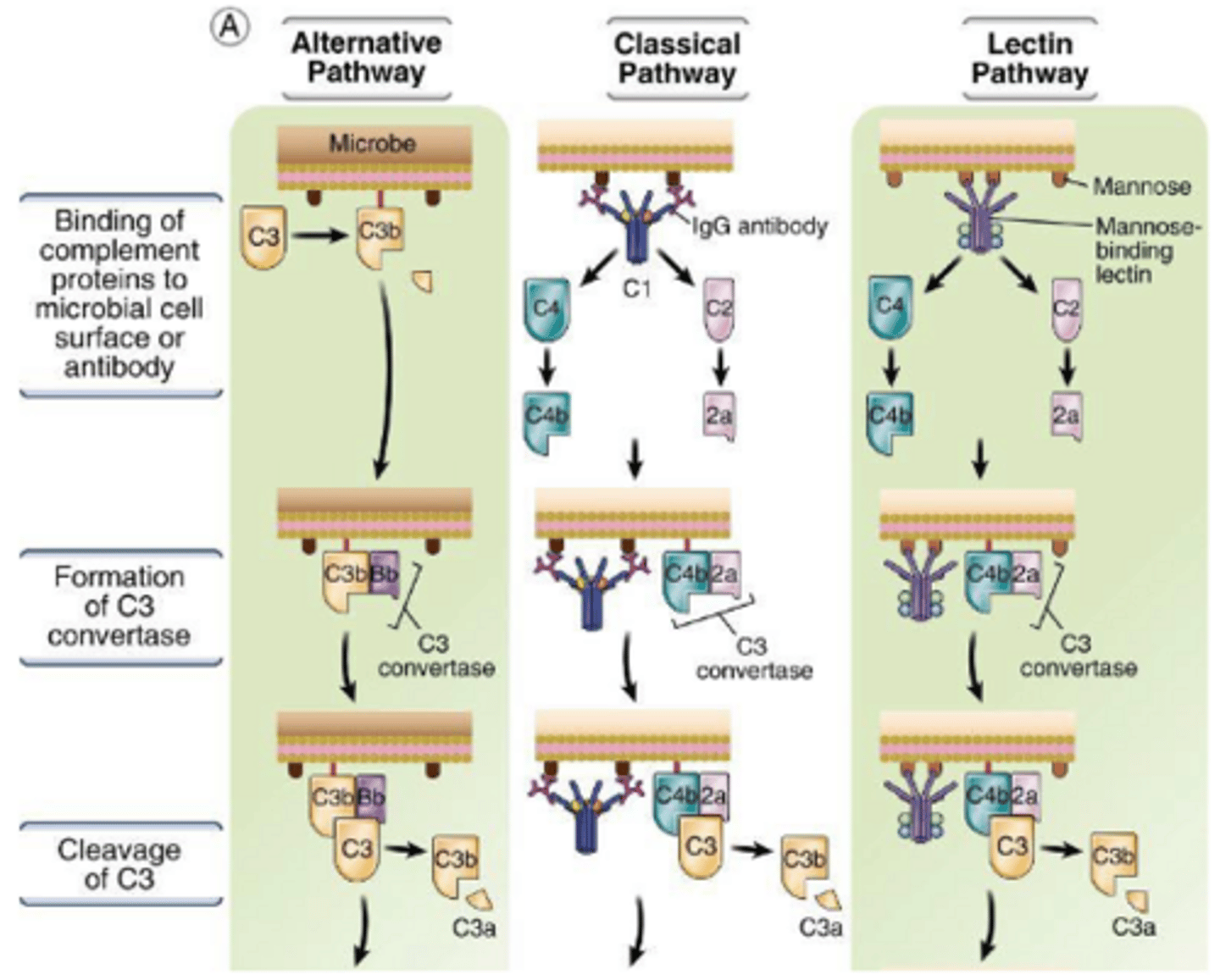
What does the classical complement pathway use for pathogen recognition and what is the advantage of this
Uses antibody (IgM/IgG) to identify the pathogen.
The Ab has very high affinity for its Ag, increasing the specificity and efficacy of complement activation.
what are the three main effector functions of the complement
1. opsonisation to enhance phagocytosis
2. stimulating inflammation by recruiting and activating immune cells
3. lysing microbes and cells
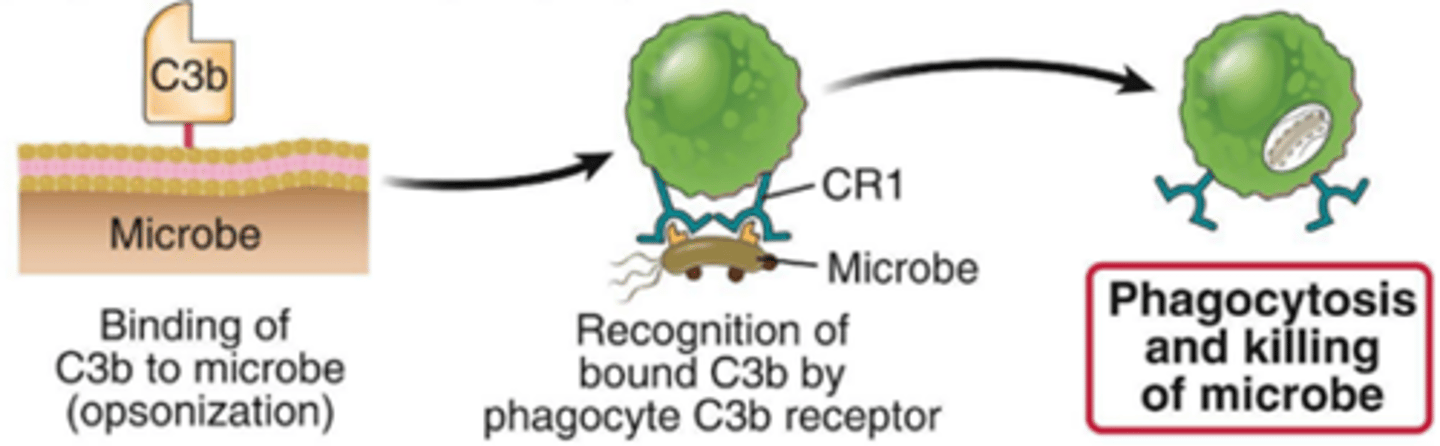
describe opsonisation
pathogens are coated with opsonins (such as C3b),
recognition of C3b by phagocyte,
phagocytosis of pathogen
describe how the complement mediates inflammation (what complement molecules are involved (3) and how do they behave)
C3a, C4a, C5a release during complement activation. They act locally similar to inflammatory cytokines. Eg:
- Recruit cells to infection site
- Activate cells
describe how the complement mediates cytolysis
The membrane attack complex (MAC) forms in the membrane of bacteria. Water enters, ions exit, causing the microbe to burst.
This process can also kill host cells.
reminder: where do macrophages reside
in tissues and blood
reminder: where do neutrophils reside
the blood
what are the 4 steps of phagocytosis
1) Phagocyte detects microbe via PRR, complement, or Ab, and extends pseudopodia to engulf microbe.
2) Membrane invaginates forming an inside out vesicle called a phagosome
3) The phagosome fuses with a lysosome to form a phagolysosome. The lysosome contains the toxic molecules that degrade microbes.
4) The chemicals in the phagolysosome activate and digest the microbe.
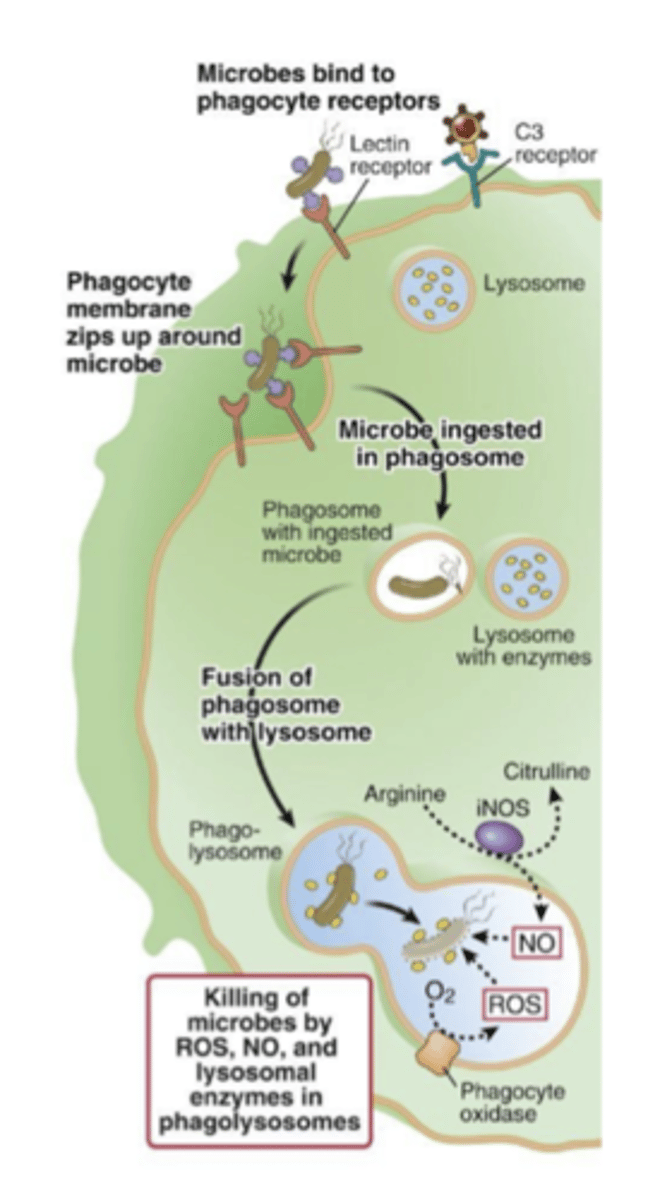
what is the action of vacuolar ATPases in the phagosome
Pump H+ into phagosome to acidify the environment
what is the action of phagocyte oxidase and cofactor NADPH oxidase in the phagosome
They initiate the respiratory burst, converting O2 into reactive oxygen species
- Eg hydrogen peroxide
what does myeloperoxidase make in the phagosome and which cells express this
neutrophils express myeloperoxidase which makes HOCl (bleach)
name a proteolytic enzyme produced by neutrophils in the phagosome that degrades bacteria
elastase
how can phagosomes starve the pathogen
withhold nutrients
what are defensins
microbicidal proteins
what enzyme mediates production of peroxynitrite radicals in the phagosome
nitric oxide synthase
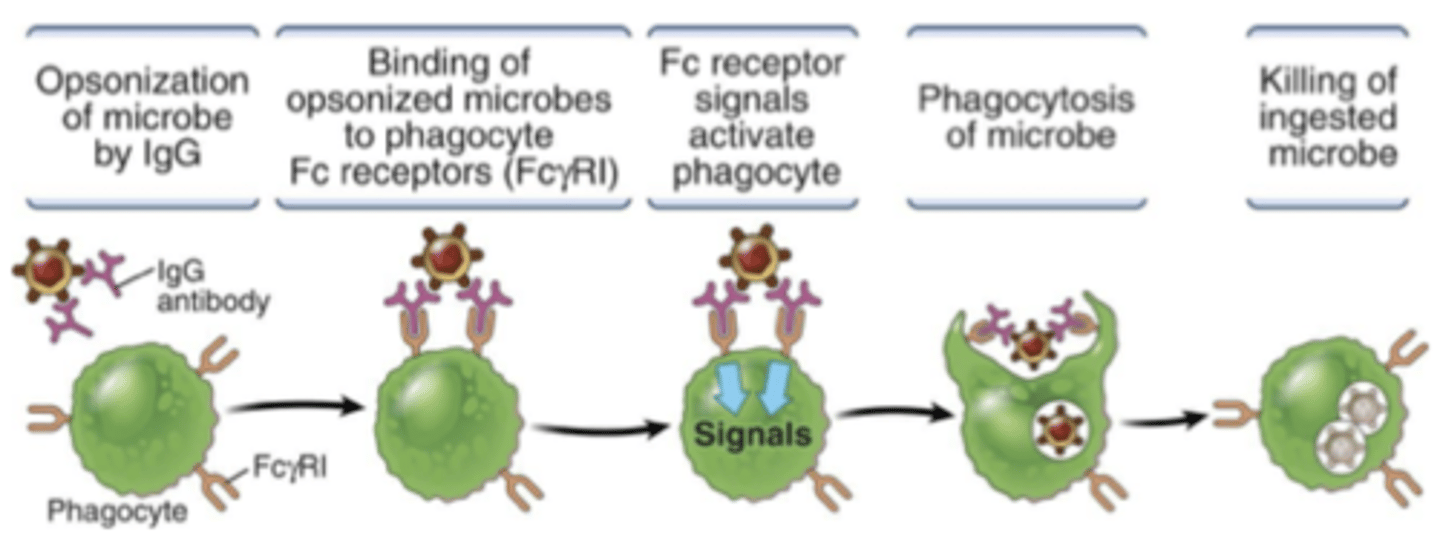
how do antibodies make phagocytosis more efficient
Ab bind to microbe (opsonisation) with high specificity and affinity.
Phagocyte binds to Ab via Fc Receptor (FcR)
This is a more efficient detection system than PRRs
what immune process takes advantage of neutrophil death (name and describe this process)
Neutrophil extracellular traps (NETs)
Neutrophil dies via a process called NETosis
Nucleus swells and burst extruding DNA like a net.
The DNA has anti-microbial molecules attached (e.g. defensins, proteases)
Traps and kills bacteria, fungi, and viruses (pus)
What cytokines do T cells use to enhance macrophage and neutrophil killing
IFN-y and IL-17
Phagocytes can bind to the Fc region of an Ab bound to a microbe. Is this detection system more or less efficient than via PRRs?
more efficient
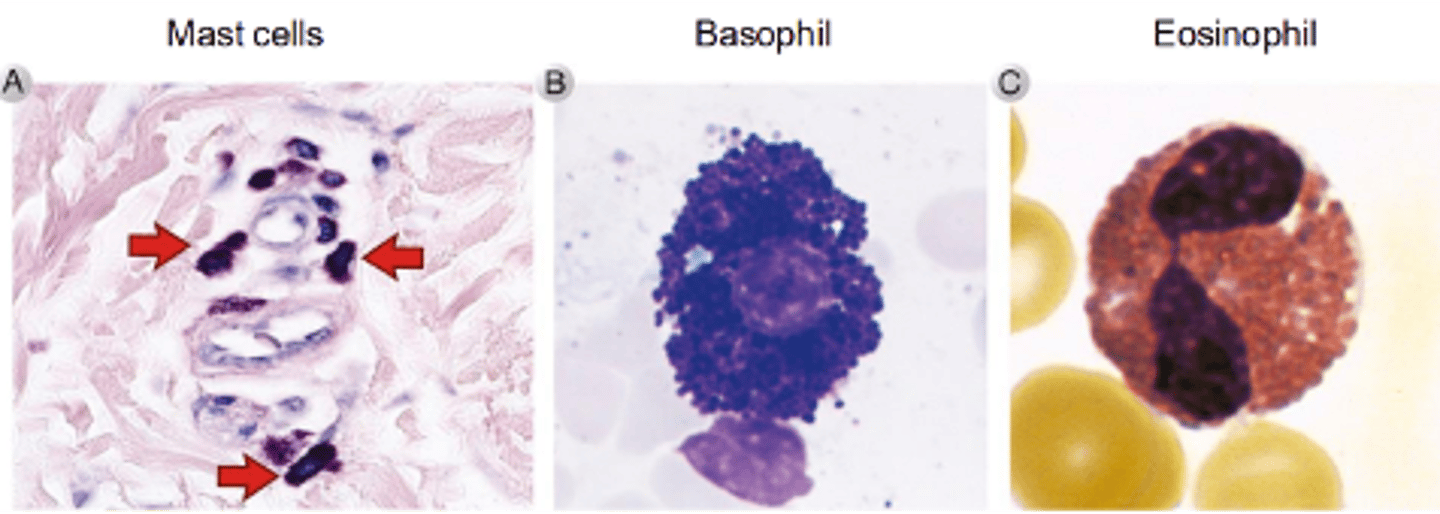
what are granulocytes
White blood cells originating from bone marrow:
neutrophils, eosinophils, basophils
How do granulocytes uniquely help in immune responses
they pre-store effector molecules in granules in the cytoplasm, so that they are ready to release when activated.
describe antibody-dependent cell-mediated cytotoxicity (ADCC)
1. Antibodies bind to the target cell or microbe
2. Fc portion of the Ab binds to the Fc receptor on an innate cell
3. The cell is activated if multiple Fc receptor-Ab interactions occur
4. The cell releases its granules at the target
- This is destructive and causes collateral damage.
which granulocyte can use ADCC on extracellular microbes
eosinophils
what is the weep and sweep response
Tissue dumps fluids then fluids are swept out of body (washes pathogens out of body)
what is the benefit of T and B cells differentiating
if they become memory cells they retain their effector functions (eg Th1/IgM etc) for a faster response
what are 2 benefits for a pathogen infecting immune cells
1. immune cells migrate readily around the host
2. easier to manipulate immune pathways from inside the immune cell
where can intracellular PRRs be found
endosomes (including phagosomes) and the cytosol
what PRRs are found in endosomes
Toll-like receptors TLRs
what PRRs are found in the cytosol
NOD-like and RIG-like receptors
how does Legionella avoid phagocytosis
prevents lysosome fusion with the phagosome
it replicates in the phagosome and eventually the cell will burst
How does Listeria avoid phagocytosis
disrupts the phagosome membrane with Listeriolysin O (LLO) to break out
Lives and replicates in the cytosol and moves between cells with host actin
what cytokine fully activates macrophages
IFN-γ
high levels of which type of T cell is best for macrophage activation
Th1
which cells can produce IFN-gamma
NK cells, T cells Th1 and CTLs (cytotoxic)
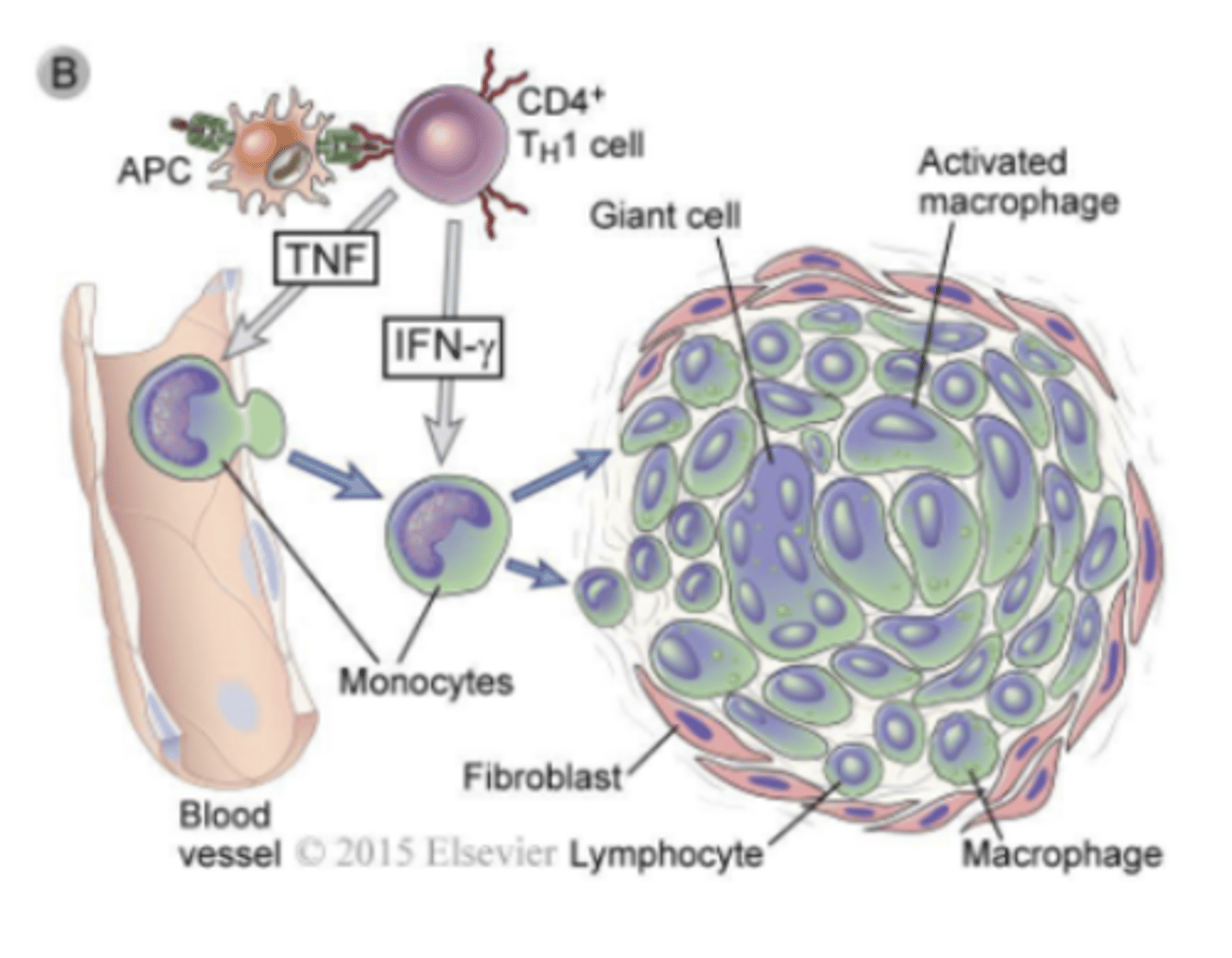
what is the result of TB evading phagocytosis so well
chronic infection with continual production of IFN-γ and macrophage activation
reminder: what is the immune response to TB bacteria
granuloma formation
which interferons are stimulated by viral infection (2)
Type 1 interferons: IFN-alpha and IFN-beta
what are the 3 functions of type 1 interferons
1. inhibition of viral gene expression
2. induces apoptosis
3. promote T cell and NK cell activation
how do type 1 IFNs inhibit viral gene expression (3)
- block viral transcription and translation
- viral RNA degradation
- autophagy
what is autophagy
The cell recycles its own organic material
how do type 1 IFNs induce apoptosis (2)
- misfolded viral proteins triggers protein response causing apoptosis
- alters cell response to TNF-alpha from pro inflammatory to apoptosis
How do type 1 IFNs promote T cell and NK cell activation (4)
• Sequester lymphocytes in LN
• Increase cytotoxicity of CTL and NK cells
• Promote Th1 differentiation
• Upregulate MHC class 1
what types of infection do NK cells target (3)
bacteria, viruses, protozoa (and tumour)
how do NK cells compare to cytotoxic T cells
they are the innate counterpart -- they are faster but less precise
why might NK cells be essential over cytotoxic T cells
some pathogens can evade cytotoxic T cells by inhibiting the MHC class 1 presentation
what is necessary for full NK cell activation
IFN-gamma
what do NK inhibitory receptors recognise
ligands on healthy cells such as self MHC class I
what do NK activating receptors recognise
ligands on infected or injured cells -- stress signals
give an example of something that an activating receptor would recognise
MHC class I downregulated and/or stress molecule expression
what is the difference between necrosis and apoptosis
necrosis: uncontrolled cell death, cell rupture releases contents, highly inflammatory
apoptosis: programmed, apoptotic cells cleared by phagocytosis, non inflammatory
how do NK cells use Abs to recognise infected cells
if a microbe leaves an antigen on the surface of a cell upon entry, Abs can bind to that antigen, then the NK cell can bind the Fc region and kill the cell
how do Th1 cells help cytotoxic T cells (2)
produce IFN-gamma and cytokines that stimulate CTL differentiation
what method of cytotoxic T cell identifying infected cells makes them more specific than NK cells
the ability to recognise Ag presented by MHC class 1
how do cytotoxic T cells interact with infected/tumour cells
via forming a synapse between the cells to allow precise control over which cell Is killed
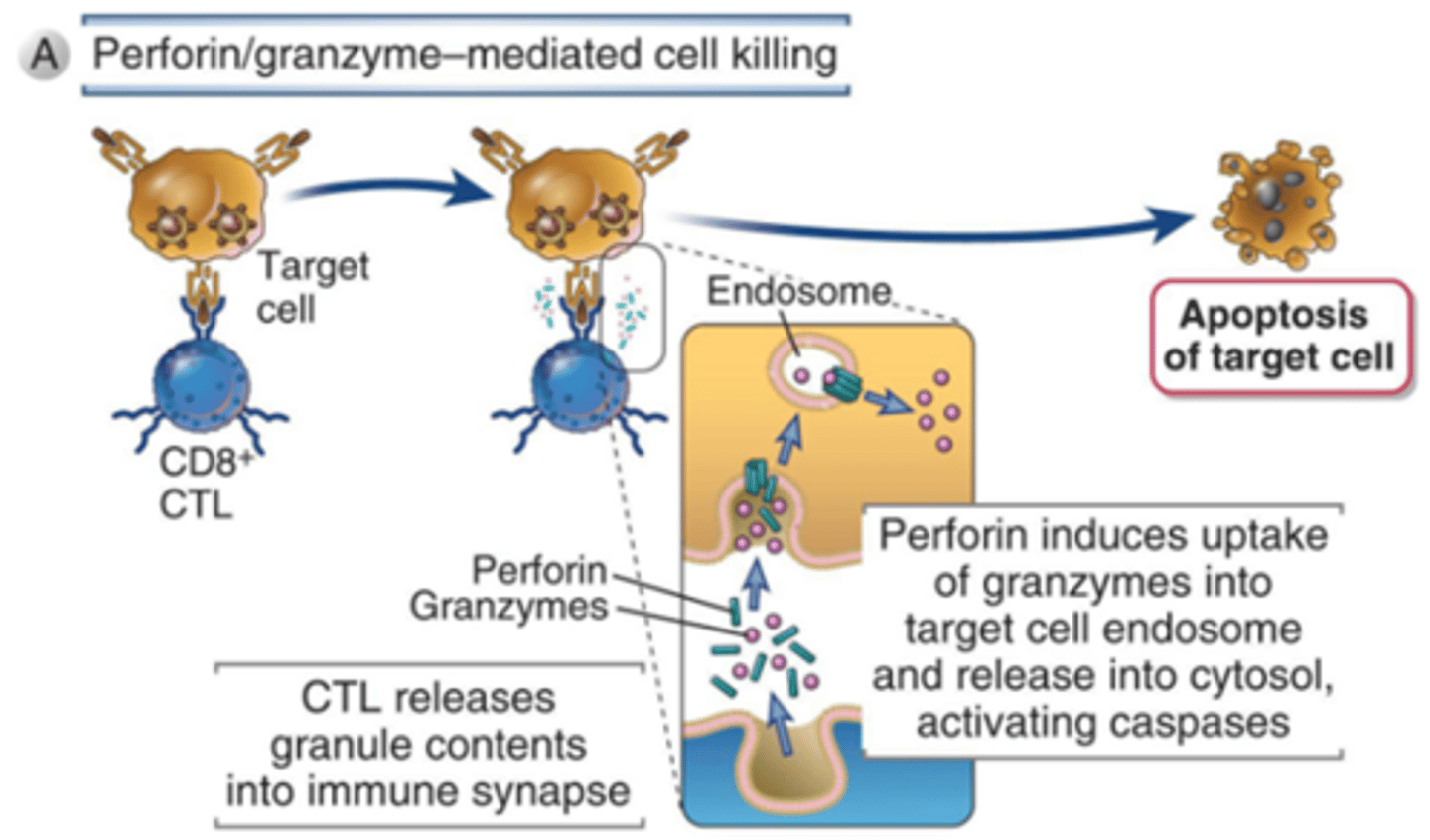
do NK and cytoT cells have the same killing mechanisms
yes
what protein do NK and cytoT cells use to breach an infected cell membrane
perforin
what is released into the infected cell via the perforin pores
granzyme
what does granzyme do
induce apoptosis
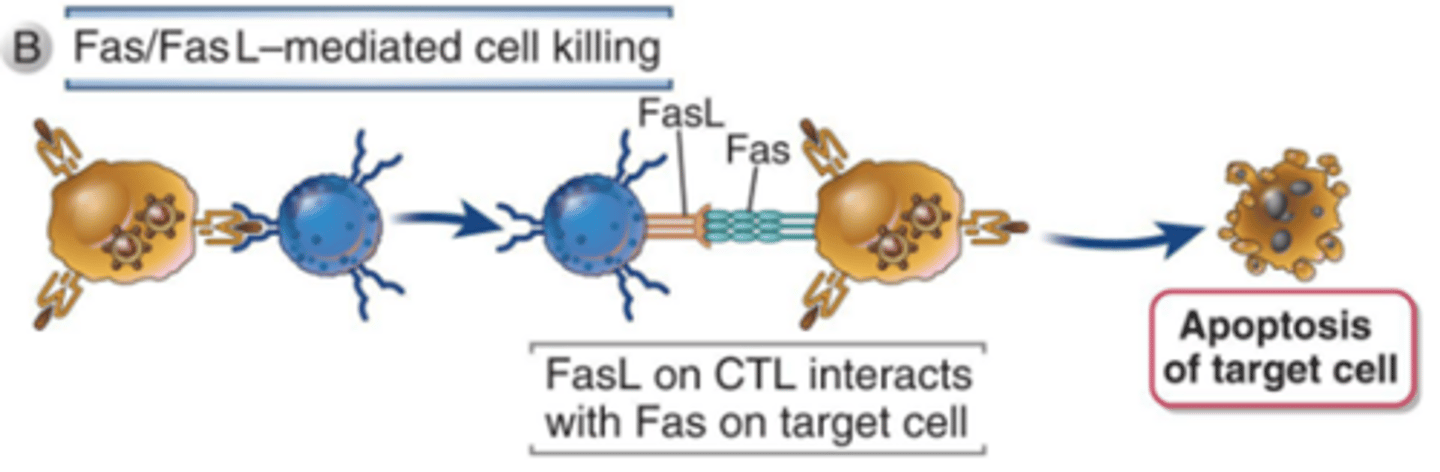
what is the second mechanism for NK/CTL cell killing (name)
Fas/FasL mediated
describe this mechanism
Target cells express Fas.
CTL/NK cells express FasL (ligand) which activates Fas.
Fas activation induces apoptosis.
which cells recognise MHC class II
CD4+ Th cells
what is cross-presentation
a process that allows specialised dendritic cells to take up extracellular antigens and present them on MHC class I instead of class II
what is the purpose of cross-presentation
be able to activate CD8+ (cytotoxic) T cells
When can Abs detect intracellular pathogens
during movement between cells
which complement molecules are involved in the membrane attack complex (4)
C6-C9
which complement molecules are involved in activating the classical pathway
C1
which complement molecules are involved in activating the alternative pathway
C3
which complement molecules are anaphylatoxins.
and what are anaphylatoxins
C3a, C4a, C5a
they are proinflammatory peptide fragments