Unit 7 Test Reagents
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
benzene + X2/FeX2 —>
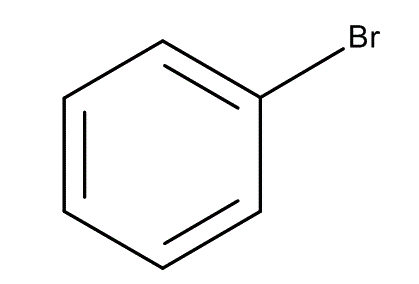
benzene + HNO3 / H2SO4 —>
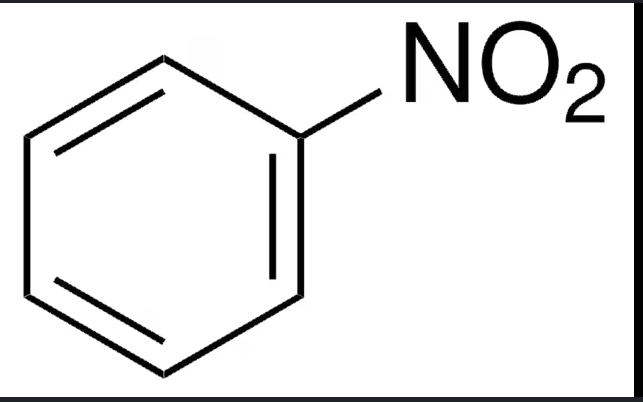
benzene + SO3/H2SO4 —>
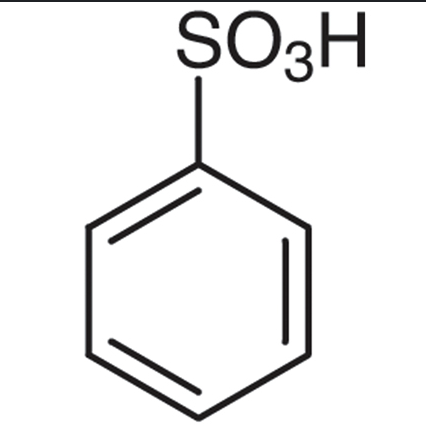
benzene + ROH / H2SO4
benzene with R group attached
(use an alcohol over an acid to oxidize)
benzene + RCl / AlCl3 —>
benzene with R group attached
**friedel-crafts alkylation
benzene + Cl-COR / AlCl3
bezene with COR attached
**friedel-crafts acylation
benzene with side chain + Br2 / hv OR NBS / hv
brominates the side chain at carbon attached to benzene
benzene with side chain + KMNO4 (STRONG OXIDIZER)
oxidizes WHOLE side chain to COOH
benzene with COR attached + Zn(Hg) / HCl or NH2NH2 / -OH
reduces to just CH2R
benzene with NO2 attached + Fe/HCl or Sn/HCl
benzene with NH2 attached
carbonyl + NaBH4 / CH3OH
alcohol (C=O) becomes C-OH
Carbonyl + 1. LiAlH4 / 2. H2O
alcohol (C=O) becomes C-OH
carbonyl + CrO3 / H3O+
NO REACTION
THIS IS JONES REAGENT
carbonyl + Ag2O / ethanol
NO REACTION
THIS IS TOLLENS REAGENT
carbonyl + 1. R—Li / 2. H2O or 1. R—MgBr / 2. H2O
C=O turns to C—OH and add an R group to the center carbon
carbonyl + NaCN / H—Cl
C=O becomes C—OH
add CN to center carbon (carbon now has 4 things)
carbonyl + RNH2 / mild acid such as CH3COOH
C=O becomes C=N—R
*(makes an imine)
carbonyl + R2NH / mild acid such as CH3COOH
C=O becomes NR2
make one of the C—C bonds next to C=O become a C=C bond
*(makes an enamine)
carbonyl + H2O / H+ or OH-
carbonyl becomes C(OH)2
carbonyl + CH3OH, H+ / (2 equiv.)
C=O becomes C(O-)2
R—X + PBr3
RBr
RBr + Mg / diethyl ether
R—MgBr
R—X + 2Li
RLi + LiX
Benzene + I2 / CuCl2
benzene with I added
benzene/nucleophile + alkene / h2SO4
Add the alkene to the benzene / nucleophile
MAKE SURE U HAVE RIGHT AMOUNT OF CARBONS