PHR 946 - Block 3: Heart Failure
1/65
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
66 Terms
heart failure (HF)
inability of the heart to meet metabolic demands of the body
HF epidemiology
- risk increases with age, most common 65+
- more common in men (more men have MIs and damage to heart)
- poor prognosis: 50% will not survive 5 years
disorders causing HF
- ischemic heart disease
- hypertension
- valvular heart disease
- other cardiomyopathies (genetic, viral, alcoholic)
> all lead to decreased cardiac output = poor exercise tolerance, fatigue, dizziness, muscle weakness, and edema
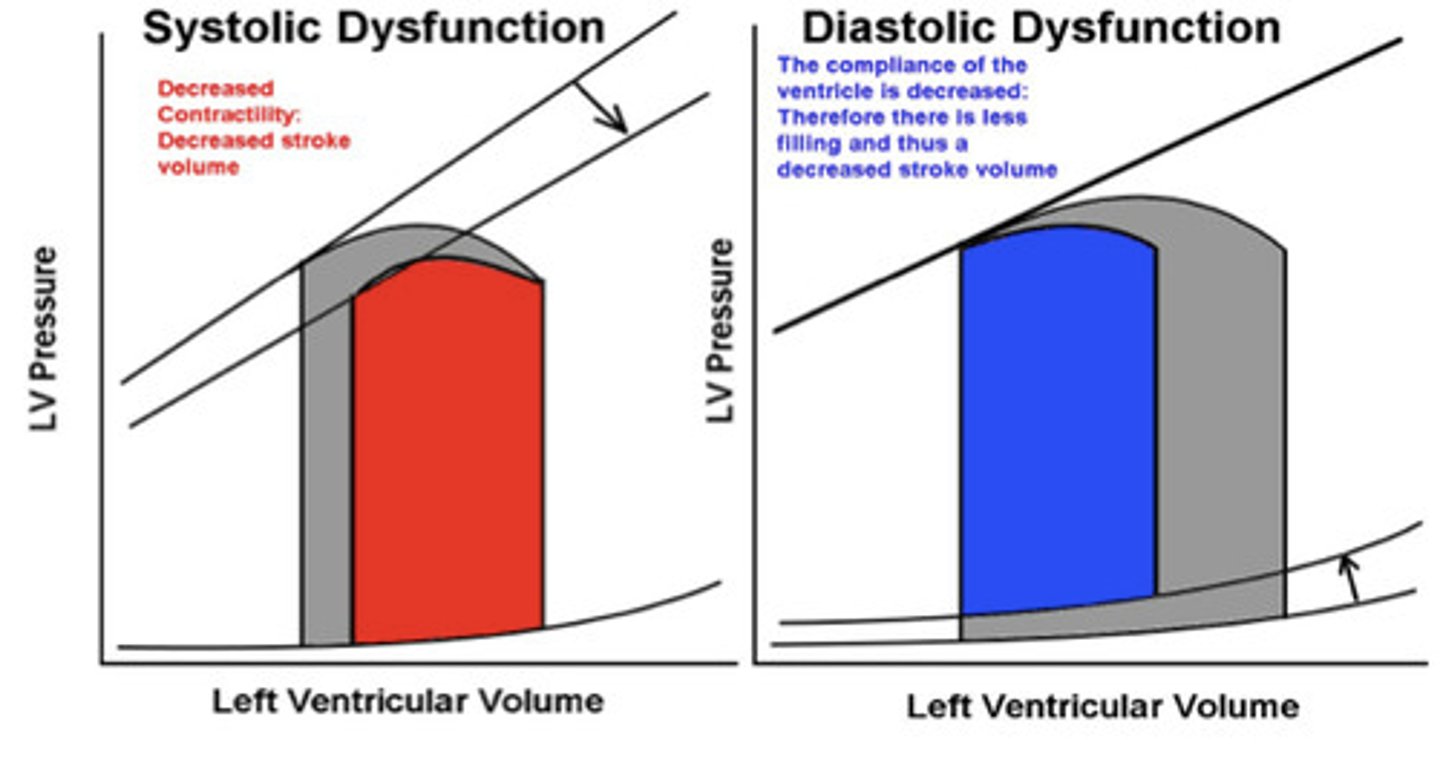
systolic and diastolic dysfunction
systolic: heart can't pump
- used to be more common
diastolic: heart can't fill
- slightly more common now
- pretty much seen equally now
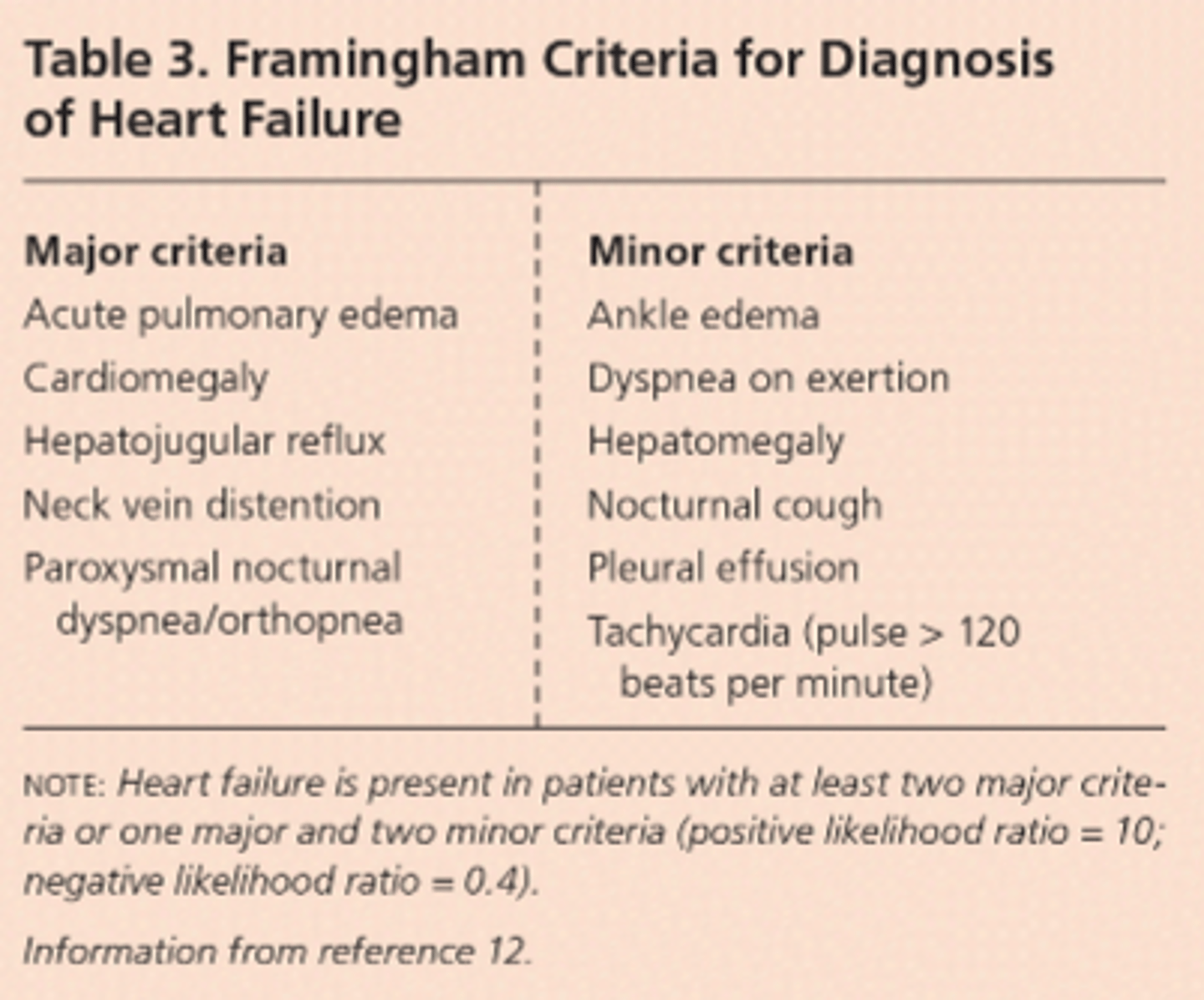
HF criteria
major criteria (more specific for HF)
- nocturnal dyspnea
- neck jein distention
- pulmonary edema
- radiographic cardiomegaly
- hepatojugular reflux (palpate liver, jugular vein distends)
minor criteria (less specific)
- bilateral ankle edema
- nocturnal cough
- dyspnea on ordinary exertion
- hepatomegaly
- tachycardia
normal cardiac function
cardiac contraction/ relaxation cycle
1. systole (contraction): pumps blood out of the ventricles to the body, increasing blood pressure
2. diastole (relaxation): allows the ventricles to fill with blood, lowering blood
> Ca²⁺ taken up by sarcoplasmic reticulum
- contractile proteins of cardiomyocytes must function properly in both
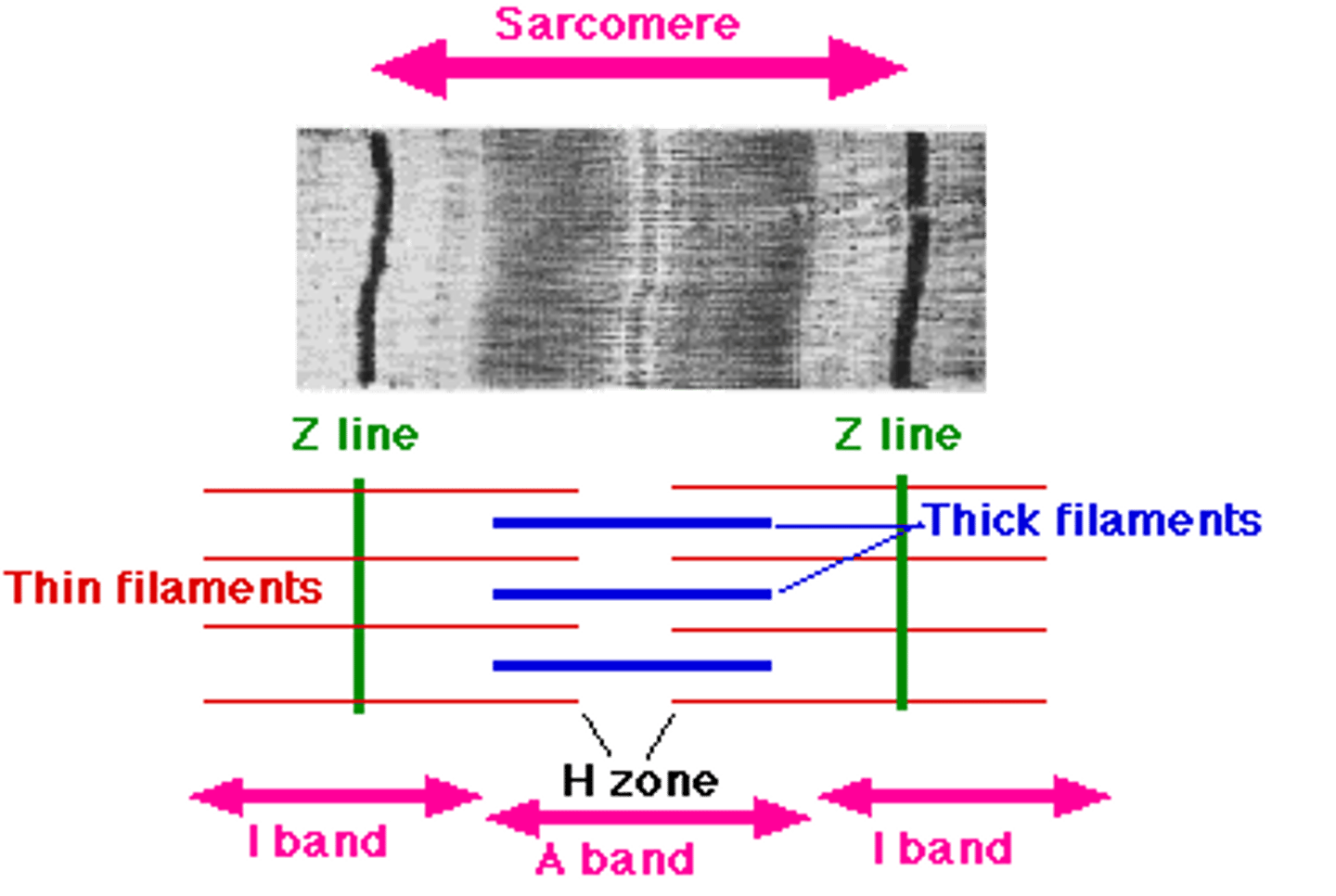
> sarcomere: fundamental contractile unit
major contractile proteins
- thin actin filament
- thick myosin filament
cardiac muscle
sarcomeres
- basic contractile unit of cardiac muscle
- organized repeating units that create striations
- contain actin (thin filaments) and myosin (thick filaments)
band of actin–myosin overlap
- region where thin (actin) and thick (myosin) filaments overlap
- site where cross-bridge cycling occurs
- responsible for force generation during contraction
myosin filament
- thick filament within the sarcomere
- contains myosin heads that bind to actin
- myosin heads use ATP to pull actin filaments, producing contraction
mitochondria
- very abundant in cardiac muscle cells
- provide large amounts of ATP for continuous contraction
- cardiac muscle relies heavily on aerobic metabolism
troponin
- regulatory protein on the actin (thin filament)
- binds Ca²⁺, causing tropomyosin to move
- exposes myosin-binding sites on actin → allows contraction
- levels increase in myocardial infarction (MI)
physiology of cardiac muscle contraction
1. action potential
- electrical signal spreads through cardiac muscle cell membrane
2. Ca²⁺ influx
- action potential opens voltage-gated Ca²⁺ channels
- Ca²⁺ enters the cell
3. calcium-induced calcium release
- incoming Ca²⁺ triggers additional Ca²⁺ release from the sarcoplasmic reticulum
4. troponin activation
- Ca²⁺ binds to troponin
- moves tropomyosin, exposing myosin-binding sites on actin
5. cross-bridge formation
- myosin heads bind actin
- ATP is used to pull actin → sarcomere shortens
6. relaxation (begins diastole)
- Ca²⁺ pumped back into sarcoplasmic reticulum
- troponin returns to resting state
- muscle relaxes
- contraction is strengthened by: concentration of Ca²⁺ and length of the muscle fibers at the end of diastole
preload and afterload
preload
- load present before the contraction
- venous filling pressure that fills the ventricle
- ↑ preload = ↑ ventricle distension during diastole (stronger contraction), ↑ heart rate
afterload
- systolic load on the left ventricle after it has started to contract
- produced in the artery leaving the ventricle
- primary determinant is total peripheral resistance

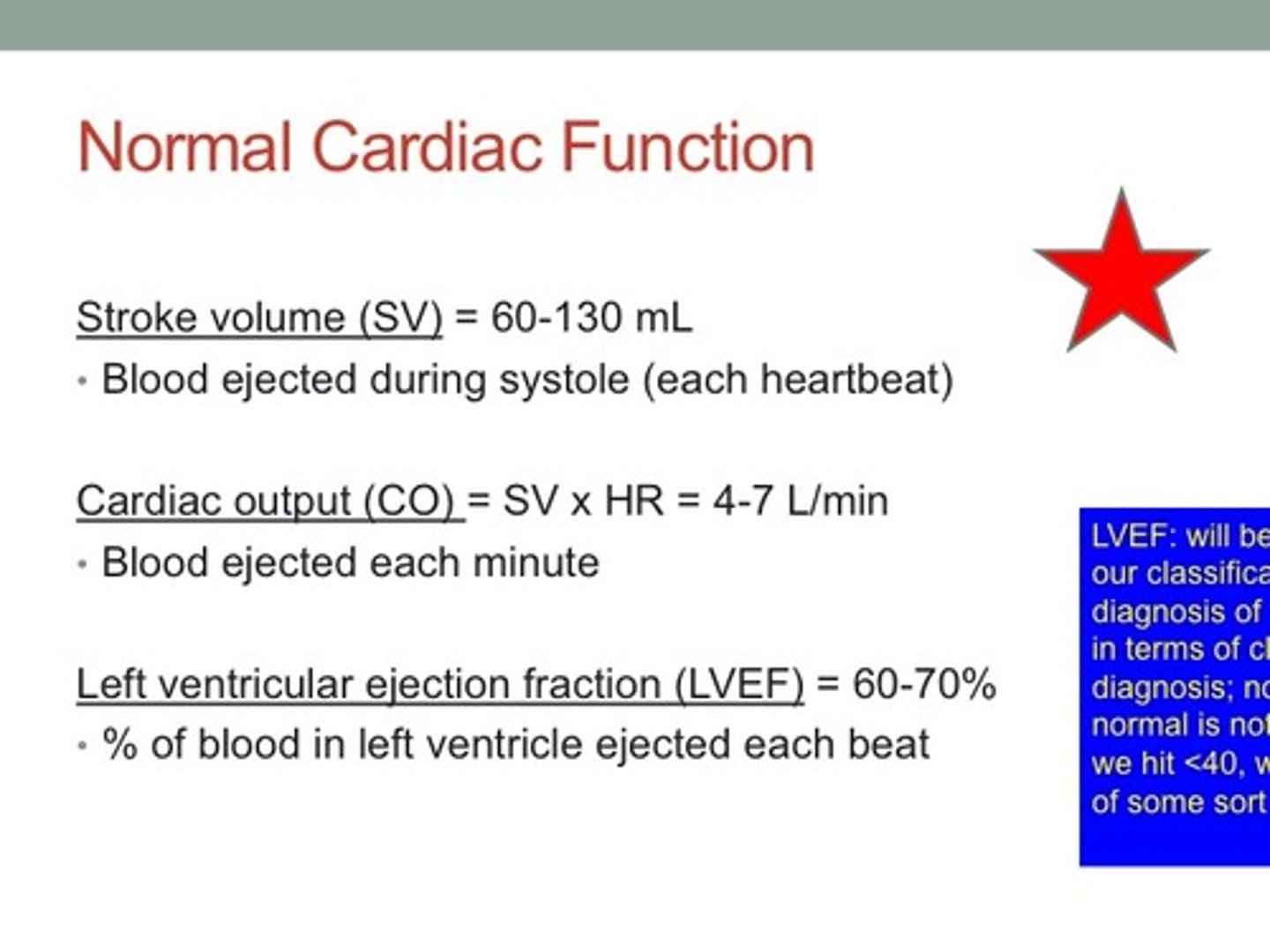
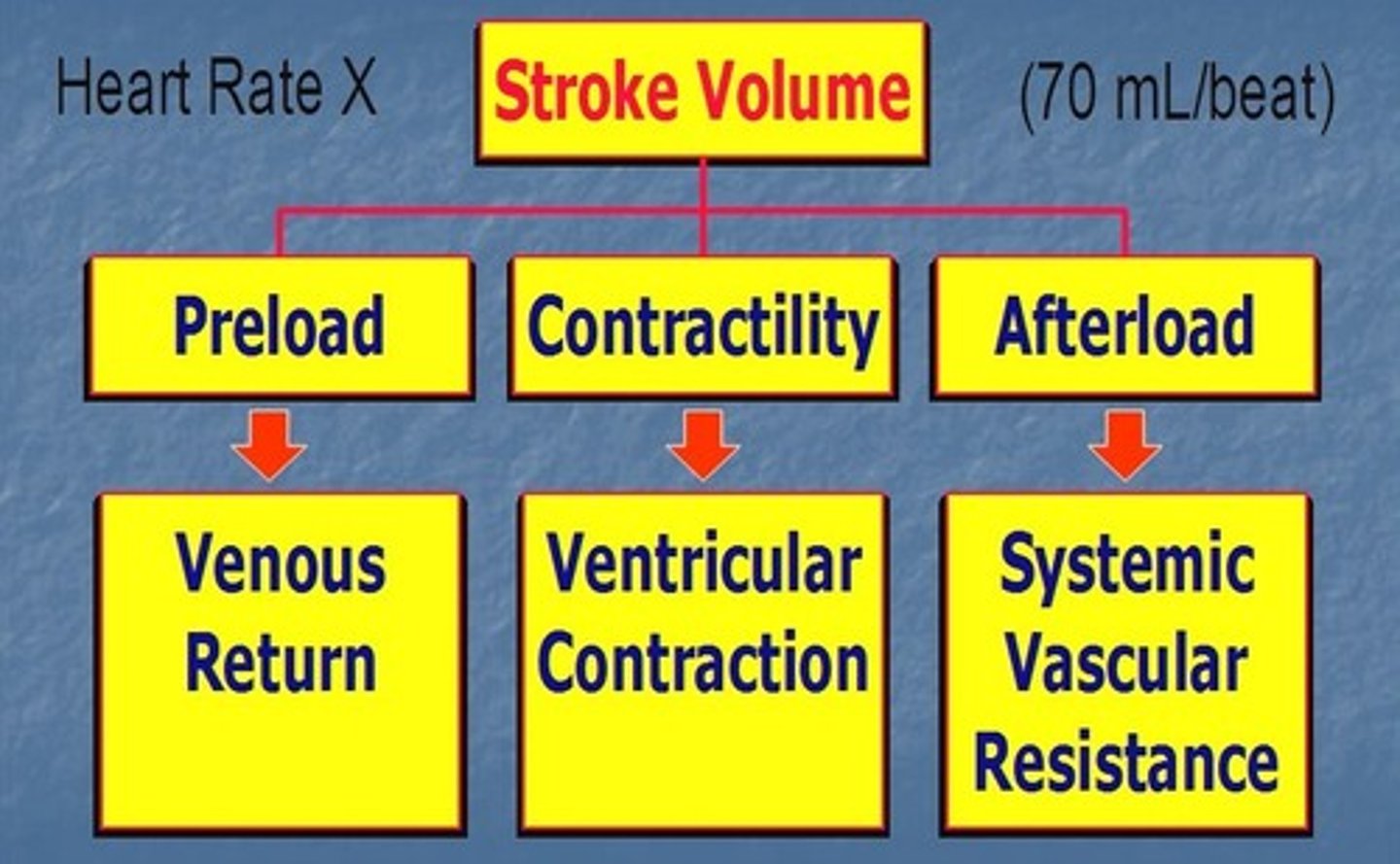
cardiac contraction measurements
can measure volumes directly with imaging
- end systolic volume: blood volume remaining in the left ventricle at the end of systole
- end diastolic volume: volume of blood in the ventricle at the end of diastole
- stroke volume (volume/ beat): volume of blood ejected from the ventricle during systole
> end diastolic volume - end systolic volume
- ejection fraction (EF): percentage of ventricular volume expelled during systole
> normal is 50%+
systolic vs diastolic heart failure
systolic HF
- an impaired inotropic state
- heart failure with reduced ejection fraction (HFrEF)
- inadequate cardiac output and diminished expulsion of blood
- symptoms: cardiomegaly, peripheral edema, jugular venous distention, left ventricular dilation
- more common in men
diastolic HF
- reduced ability of the ventricles to accept blood (failure to relax)
- heart failure with preserved ejection fraction (HFpEF)
- slowed/ incomplete ventricular relaxation
- filling volume = low; stroke volume = low
- more common in women

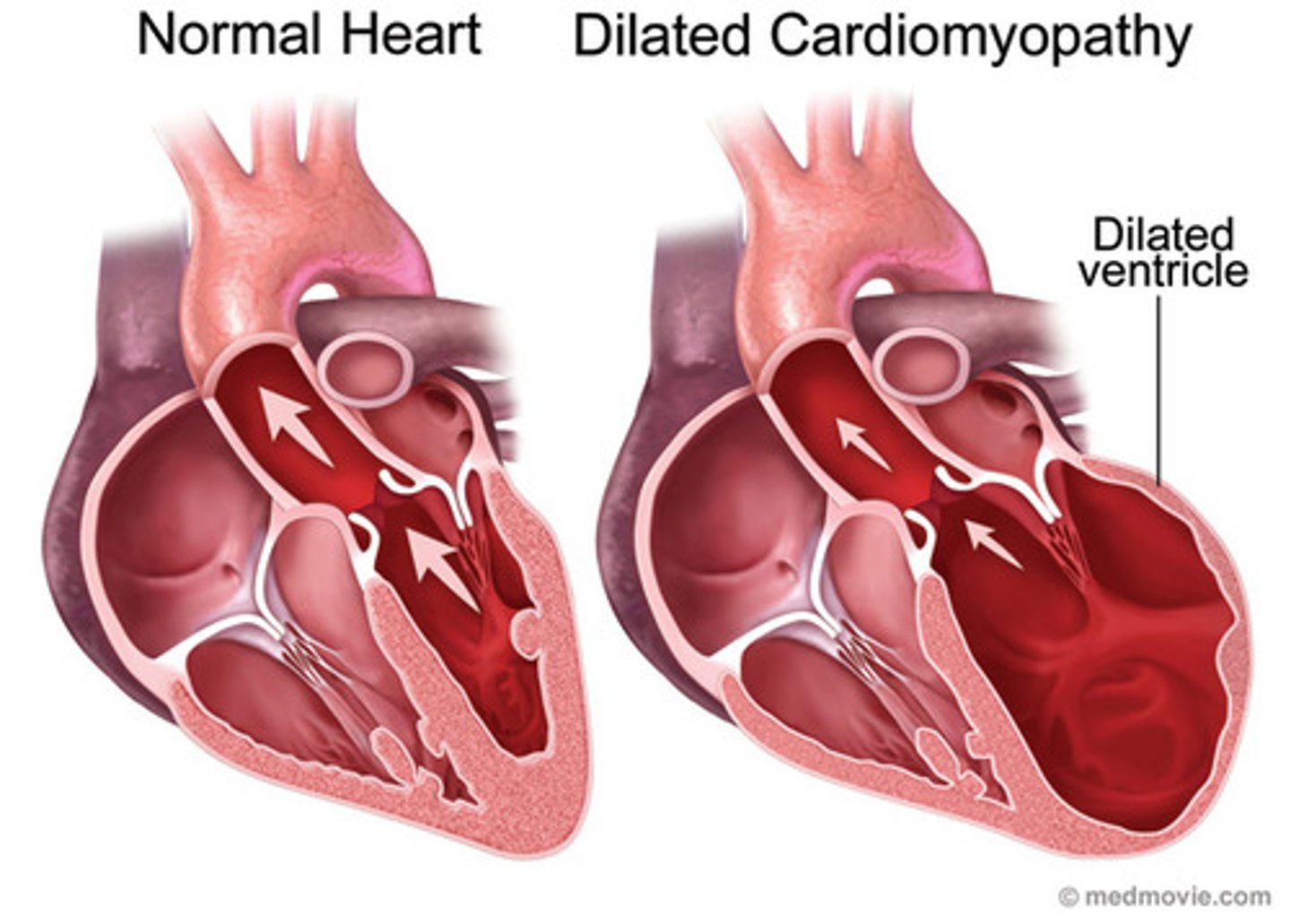
hypertrophy in HF
- dilated left ventricular hypertrophy greater with systolic HF (heart can't pump)
- an adaptive increase in muscle mass and heart wall thickness, initially effective
- over time leads to poor contractility, increased O2 needs, poor coronary artery circulation, and risk for dysrhythmias
regulation of myocardium Ca²⁺ flux
during systole
- Ca²⁺ enters the cell during depolarization through L-type channels
- this triggers the release of large amounts of Ca²⁺ from the sarcoplasmic reticulum
during diastole
- multiple mechanisms to remove cytosolic Ca²⁺
sodium-calcium exchanger
- does not use ATP
- uses the Na+ exchange gradient produced by Na+/K+/ATPase
sarcoplasmic reticulum
- actively uptakes Ca²⁺
- SERCA (sarcoendoplasmic reticulum Ca²⁺ ATPase)
in heart failure
- Ca²⁺ uptake by SERCA is depressed
SERCA
sarcoendoplasmic reticulum Ca²⁺ ATPase
- pump located on the sarcoplasmic reticulum (SR) membrane
function
- uses ATP to pump Ca²⁺ from the cytosol back into the SR
role in cardiac muscle
- lowers cytosolic Ca²⁺ → allows cardiac muscle relaxation
- stores Ca²⁺ in the SR for the next contraction
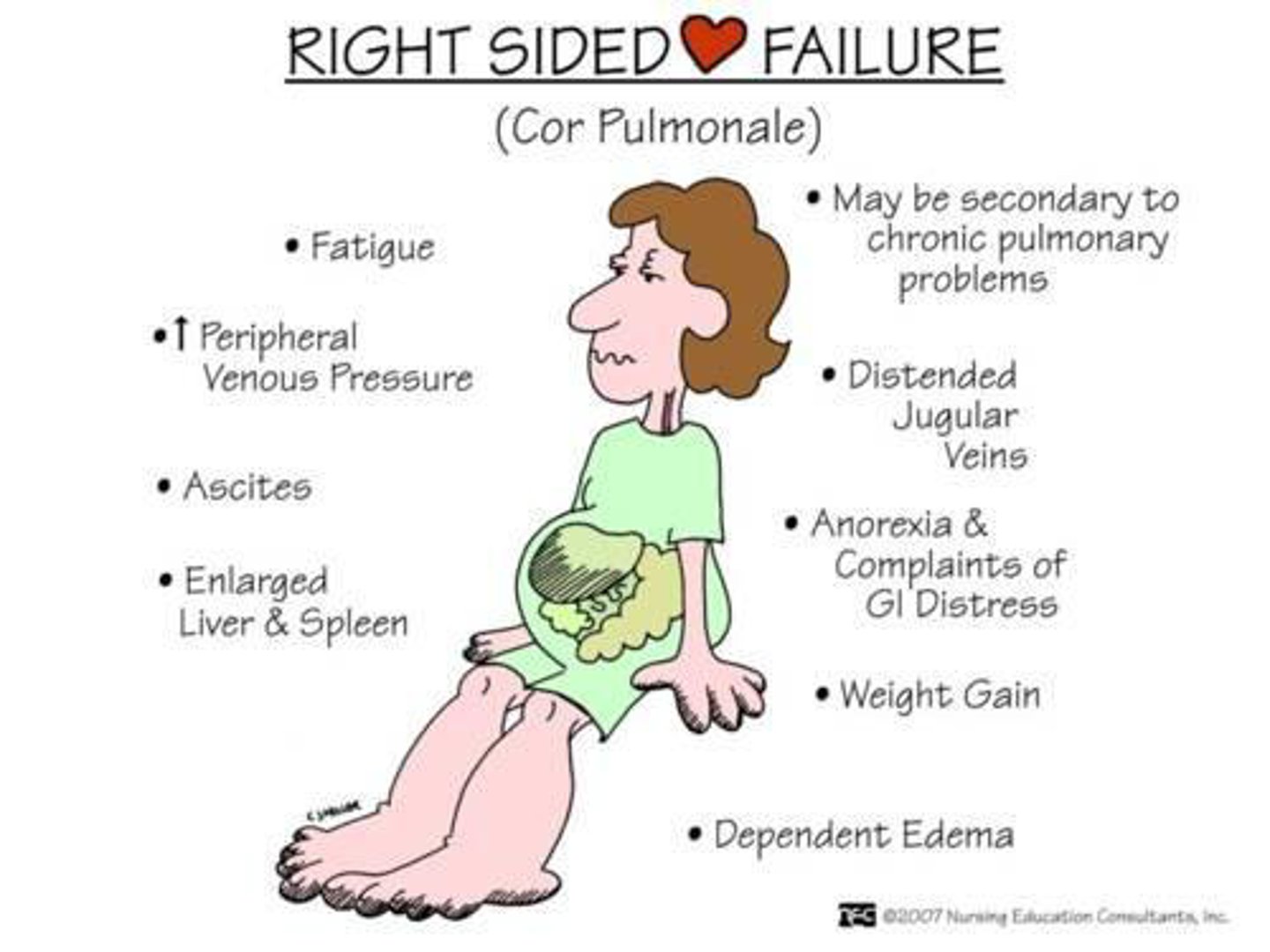
right sided HF
the right ventricle fails
- less common
- right ventricle cannot accept or eject the returning blood from the periphery
- blood backs up in the periphery
- leads to increased pressure in the periphery and loss of fluids to tissues
- RAAS system is activated → peripheral edema
left sided HF
the left ventricle fails
- left ventricle cannot accept or eject blood delivered from the lungs
- increases the blood volume in the lungs
- leads to pulmonary congestion and edema
- symptoms: dyspnea, orthopnea (dyspnea when laying), cough from pulmonary congestion

adaptive mechanisms in HF
heart depends on these to maintain cardiac output
- Frank-Starling mechanism: increased preload, helps to sustain cardiac performance
> occurs rapidly
- activation of neurohumoral systems: release of norepinephrine by adrenergic cardiac nerves; activation of the RAAS to maintain arterial pressure and perfusion of vital organs
> occurs rapidly
- myocardial remodeling: mass of contractile tissue is increases
strengthening of cardiac muscle
cardiac output varies with demand
- intracellular Ca²⁺ regulation influences contraction strength
- sarcomere length at the end of diastole affects force of contraction
Frank-Starling law of the heart
- greater ventricular filling (stretch) = greater force of contraction
- increased venous return stretches cardiac muscle
- stretch creates more optimal actin-myosin overlap and greater Ca²⁺ sensitivity
normal heart
- in a normal, nonfailing heart, ↑ blood volume in = ↑ blood pumped out
4 compensatory mechanisms during HF
1. sodium/water retention
- decreases renal perfusion, increases aldosterone release
- benefits: ↑ blood volume and venous return = Frank-Starling mechanism
- adverse effects: pulmonary and systemic edema
2. vasoconstriction
- ↑ SNS activity, Ang II, and vasopressin
- benefits: helps maintain BP when CO is reduced
- adverse effects: ↑ afterload and myocardial O2 demand
3. tachycardia
- ↑ SNS activity, baroreceptor response decreases BP
- benefits: helps maintain CO
- adverse effects: ↑ myocardial O2 demand, arrhythmias, downregulation of beta-1 receptor
4. ventricular hypertrophy
- ↑ afterload and preload
- benefits: helps maintain CO, reduces myocardial wall stress
- adverse effects: diastolic dysfunction, valve dysfunction, arrhythmias
vasoconstriction compensation
maintenance of arterial pressure with reduced cardiac output = an effective compensatory mechanism
- occurs mostly in areas not vital for immediate survival (skin, skeletal muscle, GI, kidneys)
- increase of activity of vasoconstrictor systems: sympathetic NS, RAAS, endothelin
- redistribution maintains the delivery of oxygen to vital organs (brain and heart)
- adverse effects: anaerobic metabolism, lactic acidosis, oxygen debt ,weakness, fatigue
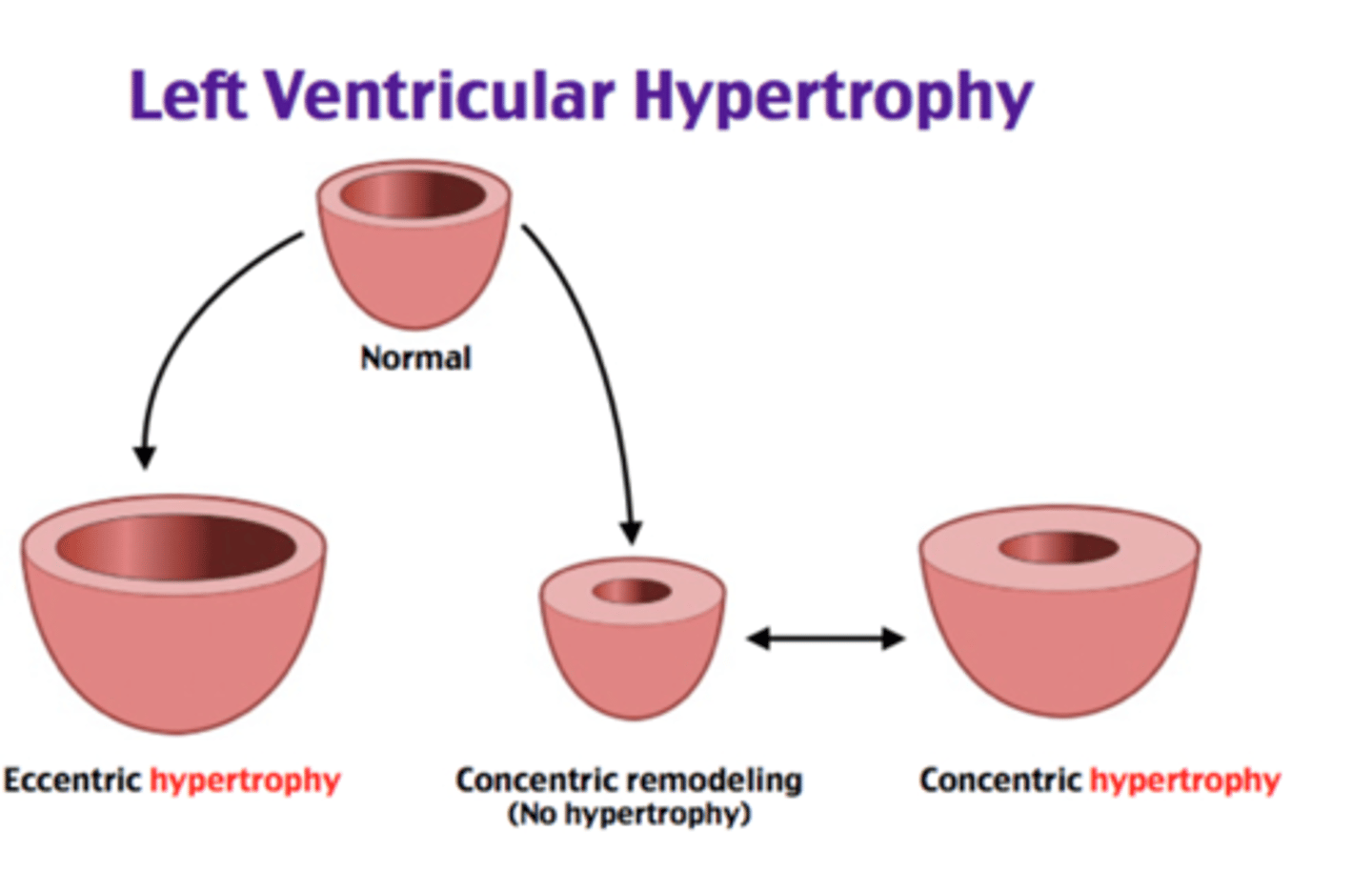
chronic myocardial remodeling
changes in mass, volume, and shape allows heart to compensate for increased lode
- primarily stimulated by pressure overload
- increased wall stress leads to: thickening of individual myocytes, replication of sarcomeres, elongation of myocytes, and ventricular dilation
- with chronic volume-overload, the ventricle becomes more spherical and causes valve defects
changes in Ca²⁺ excitation-contraction coupling
Ca²⁺ is essential for regulating cardiac contraction
- cAMP increases the activity of the L-type Ca²⁺ channel
- cAMP increases Ca²⁺ ATPase of the SERCA
> increased Ca²⁺ reuptake into the SR accelerates diastolic relaxation
- during HF, there is a negative force-frequency relationship
> impaired Ca²⁺ reuptake due to decreased SERCA activity
> disturbances in Ca²⁺ conc = reduced contractile and dilatory function
- expression of the Na+/Ca²⁺ exchanged increases during HF
> potential for arrhythmias is increased
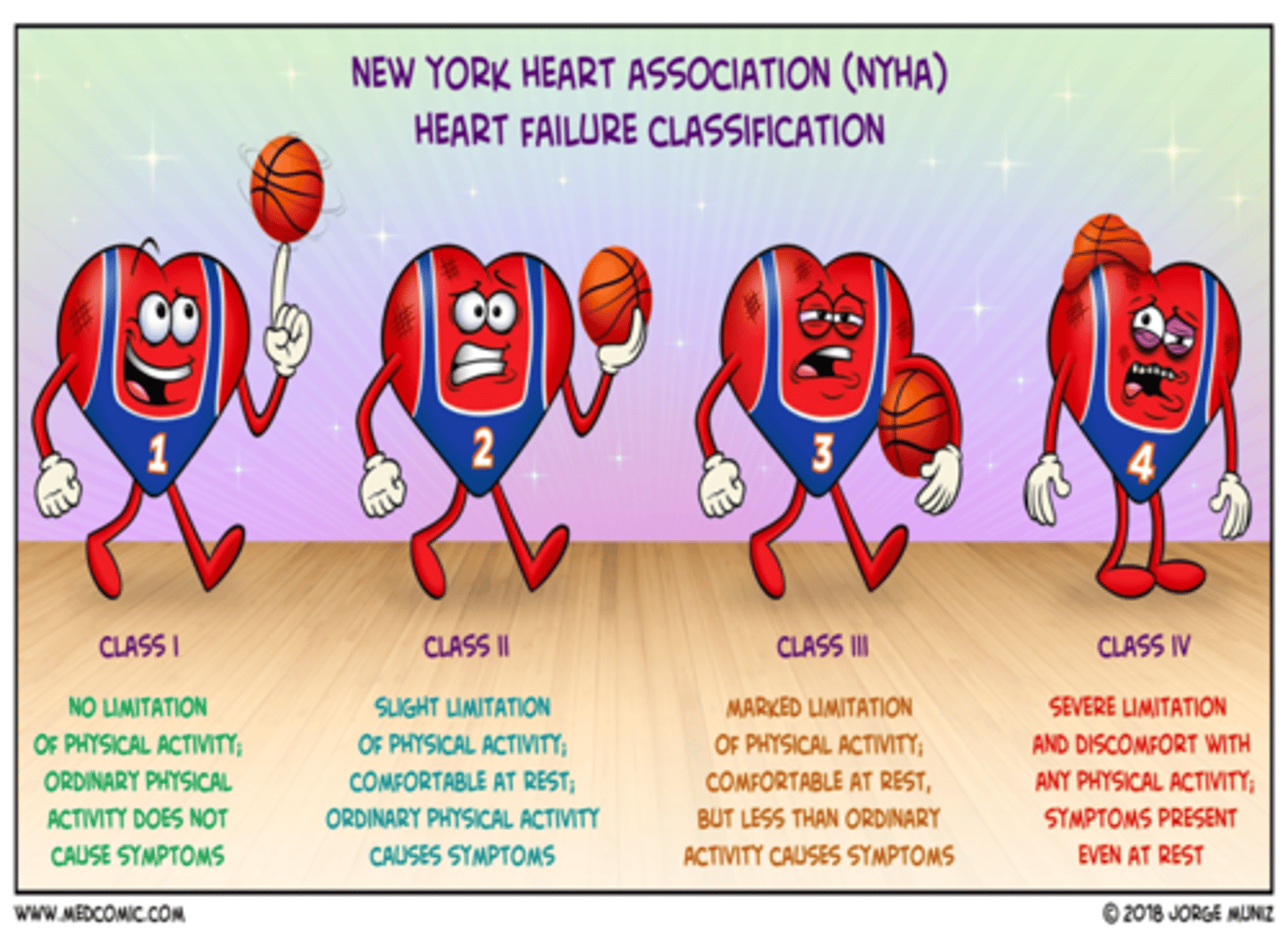
NYHA HF classification
class I: no limitation
- only evident during stress test
class II: slight limitation
- ordinary physical activity results in fatigue, palpitation, dyspnea, or angina
class III: marked limitation
- less than ordinary physical activity leads to symptoms
class IV: severe limitations
- symptoms of HF present even at rest
adrenergic signaling in HF
acute increases in sympathetic activity
- normal regulatory mechanism for increasing cardiac output (exercise)
chronic increases in sympathetic activity
- stimulated by reduction in cardiac output (toxic)
- increases HR, excess vasoconstriction, and excess retention of salt and water
long term sympathetic stimulation
- further depresses ventricular function
- increases heart's demand for energy
NE and beta receptors
- HF is associated with increased levels of norepinephrine (NE), which is directly toxic to the heart
> excess stimulation of beta adrenergic receptors + increased energy consumption by the heart
- alpha 1 receptors: much lower density than beta receptors
- beta 1 receptors: 80% are in the heart
- beta 2 receptors: 20% are in the heart (mostly in the lungs)
> beta receptors ↓ during HF
antiadrenergics for HF
- cause downregulation of beta receptors
- ↓ adenylyl cyclase activity and cAMP levels
3 classes of beta blockers:
1. 1st gen: non-selective without alpha-1 activity
- not approved for HF, makes symptoms worse
- propranolol, nadalol
2. 2nd gen: beta-1 selective
- approved for HF
- metoprolol, bisoprolol
3. 3rd gen: block beta and alpha-1 receptors
- approved for HF
- alpha-1 antagonism: reduces afterload, causes vasodilation
- carvedilol
effects of antiadrenergics for HF
beneficial
- reduces adverse effects from excessive NE
> ↓ cardiac remodeling, myocardial death, HR
- reverses hyper-phosphorylation of the Ca²⁺ release channel in the SR (ryanodine receptor)
- stabilizes cardiac rhythm
adverse effects
- may decrease systolic function (ejection fraction) immediately after starting
> need to start w lose dose and titrate
> recovers over a few months of use
vasodilators in HF
- vasoconstriction is important at first in HF as it redistributes blood flow to important tissues (brain and heart)
- eventually, the constriction increases afterload and places excessive work on the heart + compromises other tissues that are receiving less blood flow
main classes of vasodilators in HF:
1. nitrovasodilators
2. hydralazine
3. ACE inhibitors

nitrovasodilators (organic nitrates)
vein dilation: +++ artery dilation: +
- MOA: biotransformation to NO = smooth muscle cell relaxation (mainly reduces preload)
- NOT monotherapy for HF, used with hydralazine
- isosorbide dinitrate/ hydralazine (BiDil) is the only organic nitrate approved for HF (HFrEF class III-IV)
> not 1st line, adjunct to standard therapy

hydralazine
Apresoline
vein dilation: + artery dilation: +++
- MOA: reduces afterload in HFrED patients by relaxing arterial smooth muscle (minimal effects on preload)
- combined with isosorbide dinitrate
> isosorbide reduces preload, hydralazine prevents nitrate tolerance
> hydralazine has antioxidant properties that help prevent NO degradation = restored balance between NO and reactive oxygen species

vericiguat
Verquvo
soluble guanylate cyclase (sGC) stimulator
- MOA: directly stimulates soluble guanylate cyclase (sGC) to increase sensitivity to endogenous NO → increased cGMP levels → smooth muscle relaxation and vasodilation
- DDI: PDE-5 inhibitors (↑ hypotension)
- [black box]: embryo-fetal toxicity
extracellular fluid volume in HF
- compensatory changes lead to ↑ in salt and water retention
> adrenergic stimulation: beta-adrenergic receptor-mediated release of renin & renin-angiotensin system
- treatment: diuretics
- goal: reduce edema, pulmonary congestion, and ventricular pressure
- may use: loop diuretic, thiazide-like diuretic, or K+ sparing diuretic
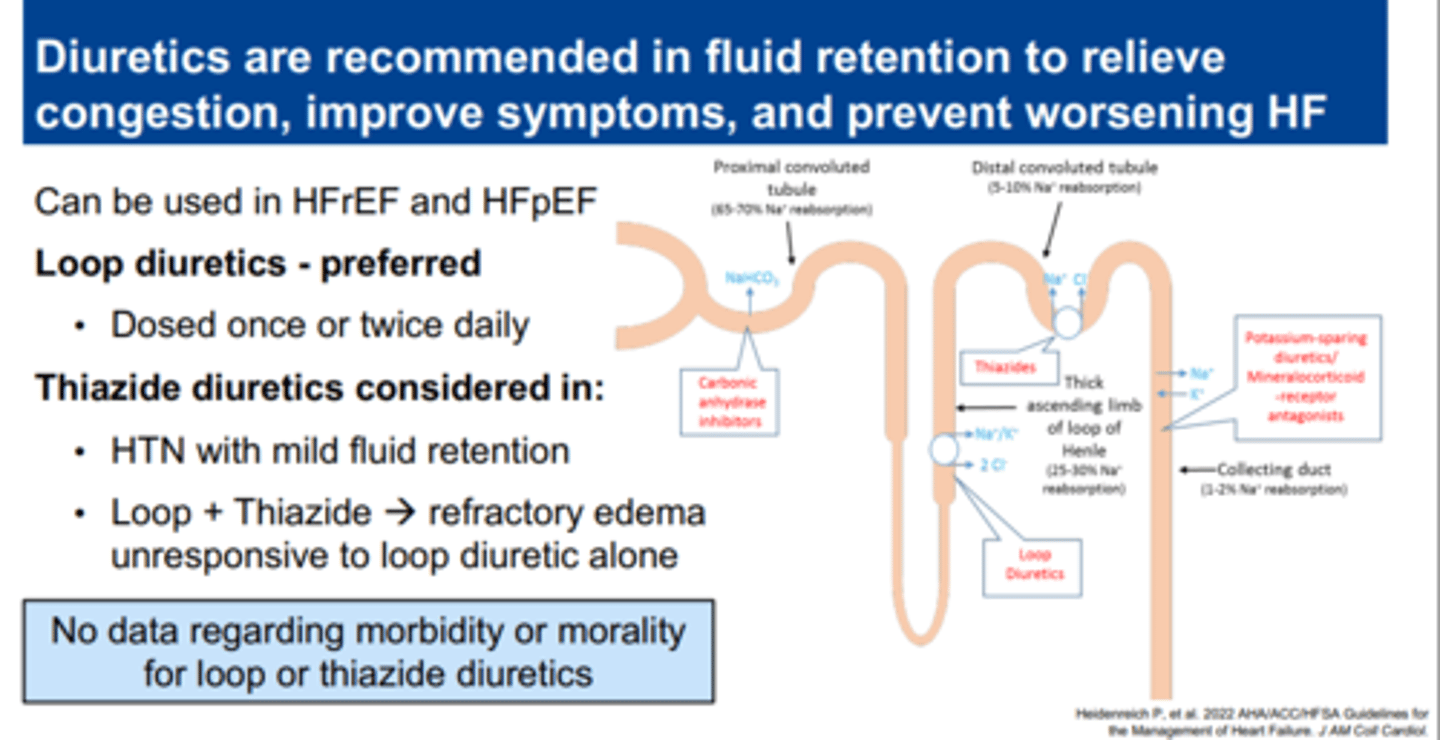
diuretics in HF
loop diuretics
- furosemide (Lasix), bumetanide (Bumex), torsemide (Soaanz)
- inhibit the Na+/K+/2Cl- cotransporter in the thick ascending loop
- most effective class in treating edema for HF patients
- caution: hypokalemia, digoxin toxicity
thiazide-like diuretics
- chlorthiazide (Diuril), hydrochlorothiazide, indapamide, chlorthalidone (Thalitone), metolazone
- inhibit the Na+/Cl- cotransporter in the distal convoluted tubule
- may be used as monotherapy in early stages
- caution: hypokalemia, reduced effectiveness in renal failure
K+ sparing diuretics
- spironolactone (aldactone), eplerenone (Inspra)
- competitively inhibit the binding of aldosterone to the mineralocorticoid receptor
- improves overall survival when added to conventional therapy
- caution: hyperkalemia, blocks androgen & progesterone receptors (spiro)
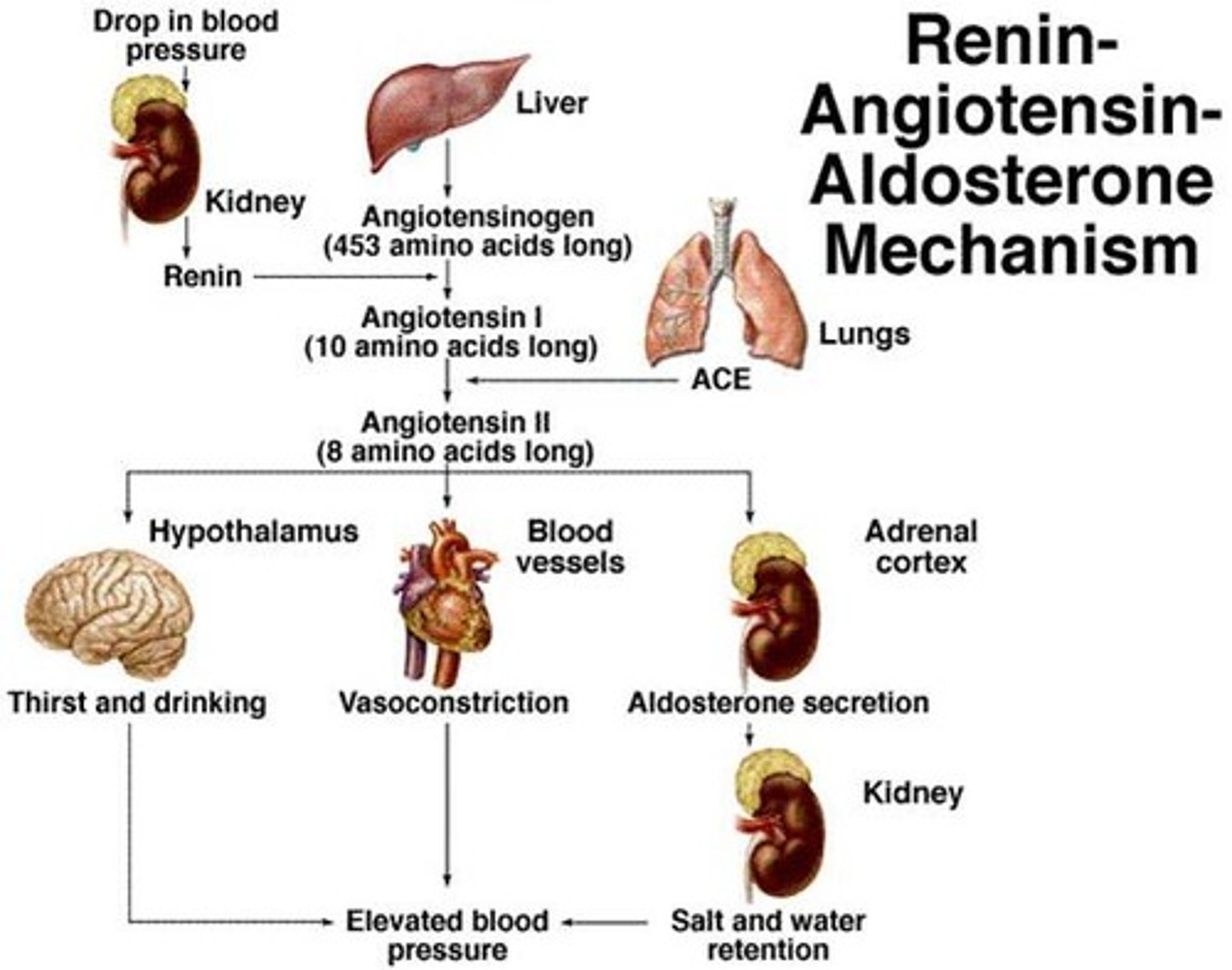
RAS in HF
- low cardiac output stimulates the renin-angiotensin system (RAS)
- helps maintain arterial pressure and retains sodium & water
- the sympathetic nervous system stimulates beta-1 receptors in the juxtaglomerular region of the kidney = release of renin
- renin → angiotensinogen → angiotensin I → angiotensin II
- angiotensin II: a potent vasoconstrictor
- promotes sodium & water retention, stimulates release of aldosterone, and causes pathological myocardial hypertrophy
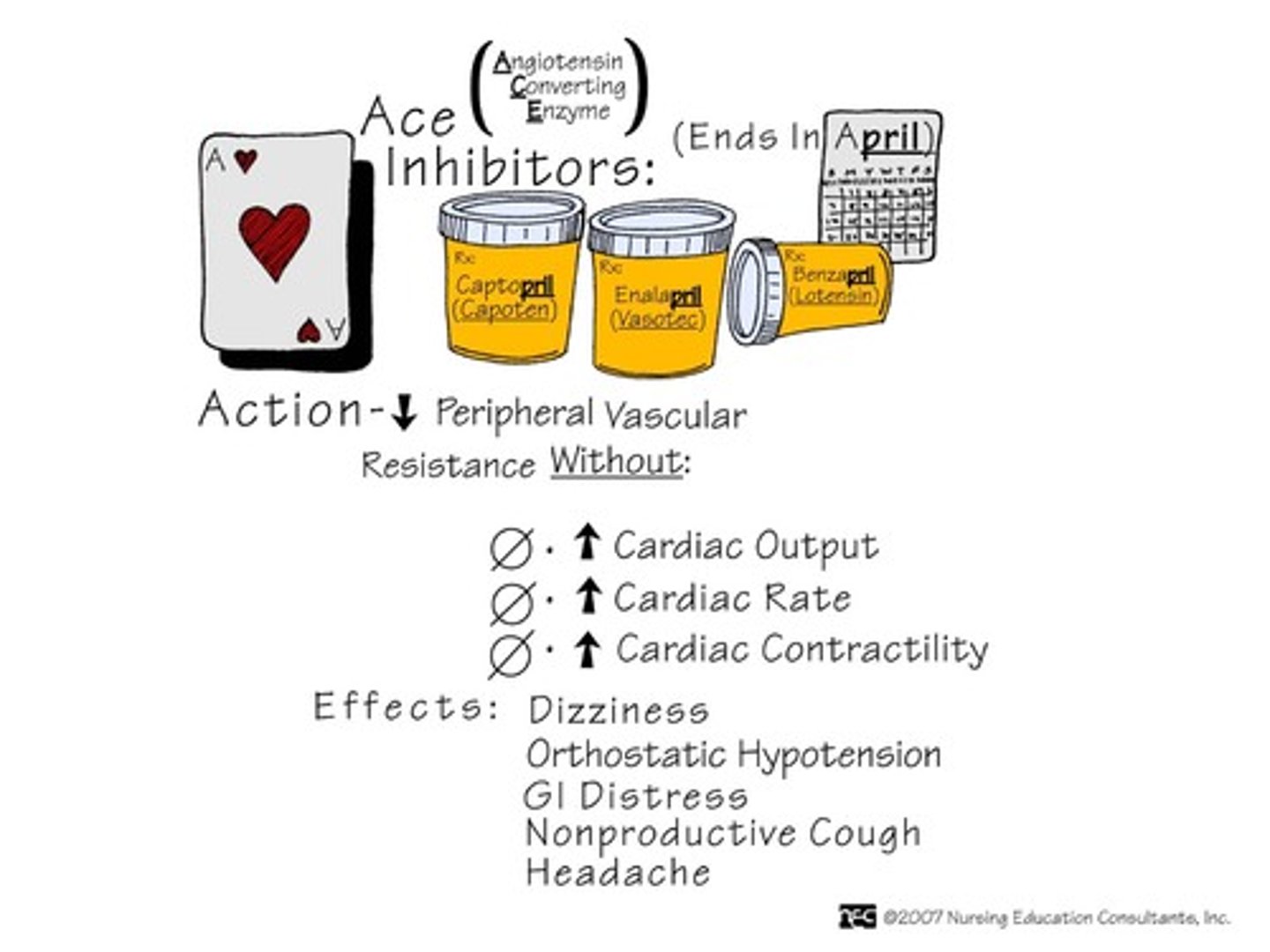
ACE inhibitors in HF
captopril, enalapril, lisinopril, quinapril, fosinopril
ramipril, trandolapril: only indicated for HF after an MI
- MOA: blocks action of ACE and prevents formation of ang I into ang II
> causes vasodilation, reduces pathological remodeling of the heart, reduces afterload & preload, decreases aldosterone, decreases LV hypertrophy
- used for: HFrEF
- ADE: hypotension, increases renal insufficiency; angioedema, cough (due to ↑ bradykinin levels
ARBs in HF
valsartan, candesartan
- AT1: primary angiotensin II receptor
> causes vasoconstriction, NE release, aldosterone release
- MOA: blocks AT1 receptor
- used for: HFrED when ACEi is not well tolerated
renin/ angiotensin drugs vs beta blockers
- in class II-III, beta blockers produce a greater beneficial effect than ACEis
- in class IV, ACEi or aldosterone inhibition + ACEi produce a greater reduction in mortality than beta blockers
(beta blockers decrease cardiac function too much)
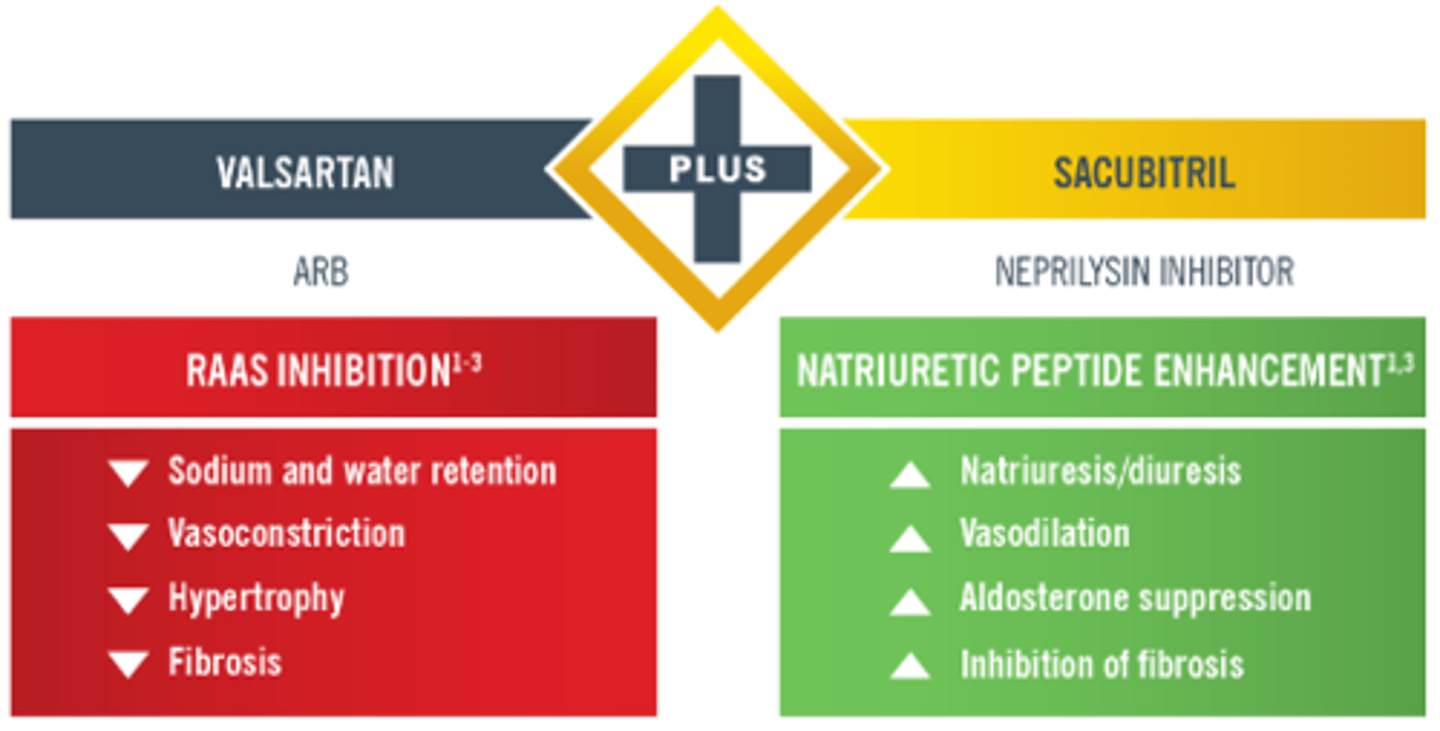
sacubitril and valsartan
Entresto
neprilysin inhibitor + ARB
- MOA:
> sacubitril: inhibits the enzyme neprilysin → prevents the breakdown of natriuretic peptides (ANP, BNP). these peptides promote vasodilation, natriuresis (sodium loss), and diuresis, reducing heart overload
> valsartan: blocks AT1 receptor
- used for: pts in class II-IV HFrEF in place of an ACEi or ARB

SGLT2 inhibitors in HF
dapagliflozin (Farxiga), empagliflozin (Jardiance), sotagliflozin (Inpefa)
- MOA: not clearly defined in HF; good for patients with and without hyperglycemia
> reduction of filtered glucose reabsorption, reduction of sodium reabsorption
- benefits: increases diuresis, decreases cardiac preload nad afterload
- ADE: urinary tract infections

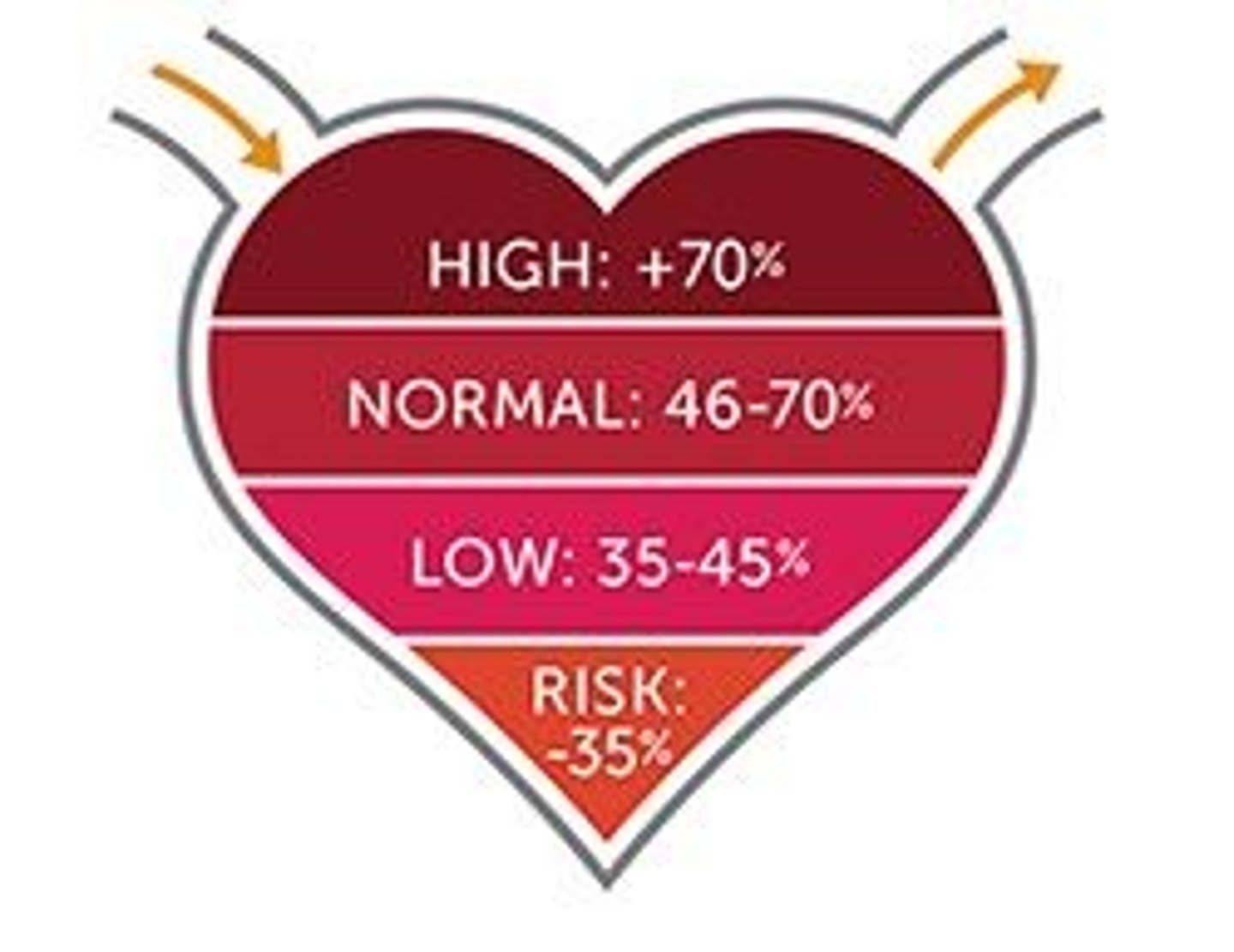
ejection fraction (EF)
The fraction of blood pumped out of the left ventricle with each contraction
- determined by dividing stroke volume by end-diastolic volume (expressed as a percentage)
causes of HF
HFrEF
- ischemic heart disease
- inflammatory damage
- toxic damage
both
- hypertension
- arrhythmia
- valve disease
- cardiomyopathy
HFpEF
- obesity
- diabetes
- pericardial disease
- chronic kidney disease
cardiotoxins
- anthracyclines (used in breast cancer)
- alcohol
- appetite suppressants (stimulants)
- clozapine
- infliximab
- antiparkinsonian drugs
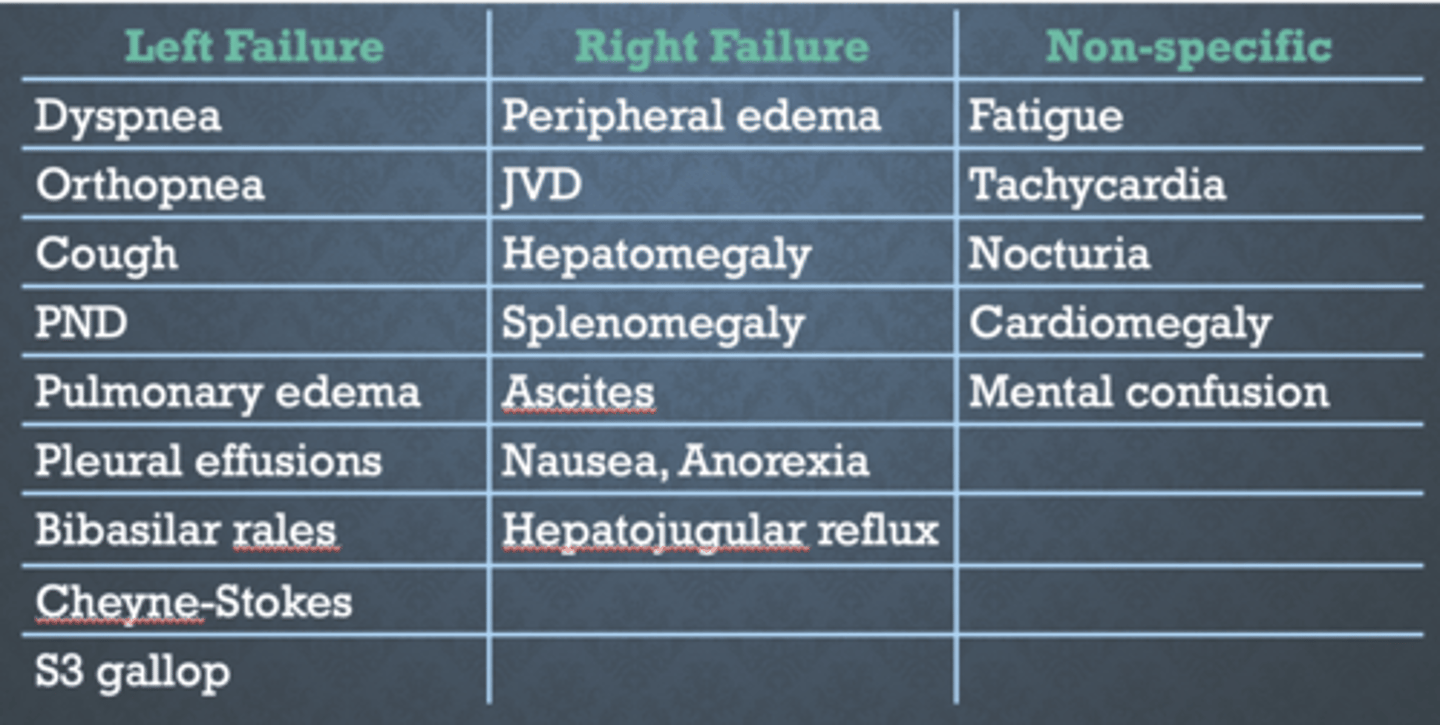
HF signs and symptoms
- paroxysmal nocturnal dyspnea (PND): causes shortness of breath while laying, need to sit up to sleep
- peripheral edema (PE)
- nocturia: waking to pee (avoid diuretic close to bed)
- jugular vein distension (JVD): due to pressure overload
- hepatojugular reflex (HJR): palpate liver = jugular vein distends
- anorexia: decreased appetite
- S3/S4: altered heart sounds
- shortness of breath
- fatigue
- chest pain
- cold feet/ hands
- poor memory
diagnosis of HF
assessment
- clinical history, physical exam, ECG, labs
- natriuretic peptide: NT-proBNP >125 pg/mL
- transthoracic echocardiogram: noninvasive ultrasound of chest wall, can see the EF on this
once HF diagnosis is confirmed, determine cause & classify
1. HFrEF: reduced ejection fraction
- LVEF ≤40%
2. HFmrEF: mildly reduced ejection fraction
- LVEF 41-49%
3. HFpEF: preserved ejection fraction
- LVEF ≥50% (normal)
evaluate for precipitating factors and initiate treatment
6 steps in HFrEF treatment
1. address congestion and initiate guideline directed medical therapy (GDMT)
- 4 pillars
2. titrate to target dosing as tolerated; get labs, health status, and LVEF
- if LVEF ≤40%: persistent HFeEF, escalate to step 3
- if LVEF >40%: HFimpEF (improved), continue treatment
3. consider patient factors and add therapy as appropriate
- class III-IV in african american pts → hydral nitrates
- class I-II with LVEF ≤35% and >1 year survival → ICD
- class II-III or ambulatory IV with LVEF ≤35% and NSR and QRS ≥150 ms with LBBB → CRT
4. implement additional GDMT and device therapy as indicated
5. reassess symptoms, labs, health status, and LVEF
- if refractory, escalate to step 6
- if improved, continue therapy
6. referral for HF specialty care
- durable MCS, cardiac transplant, palliative care, or investigational studies
addressing congestion
diuretic therapy
- benefits: decreases preload and congestive symptoms, increases exercise tolerance
- drawbacks: no mortality data, depletes electrolytes (arrhythmia risk), activates neurohormonal pathways
- do not use as monotherapy
> used w ACEis, beta blockers, vasodilators
- mainly used loop diuretics
loop diuretics
equal in efficacy if given in equipotent doses
furosemide 40 = torsemide 20 = bumetanide 1
- dosing: start with lower dose (furosemide 20-40) once daily and titrate based on weight/ symptoms/ tolerability
- ADE: ototoxicity (tinnitus), fluid and electrolyte abnormalities, skin reactions
monitoring
- response: daily weights, LE edema, urine output, CrCl, electrolytes, SOB, dyspnea, PND, chest xray
- excess dose: dizziness, lethargy, blood pressure, muscle cramping
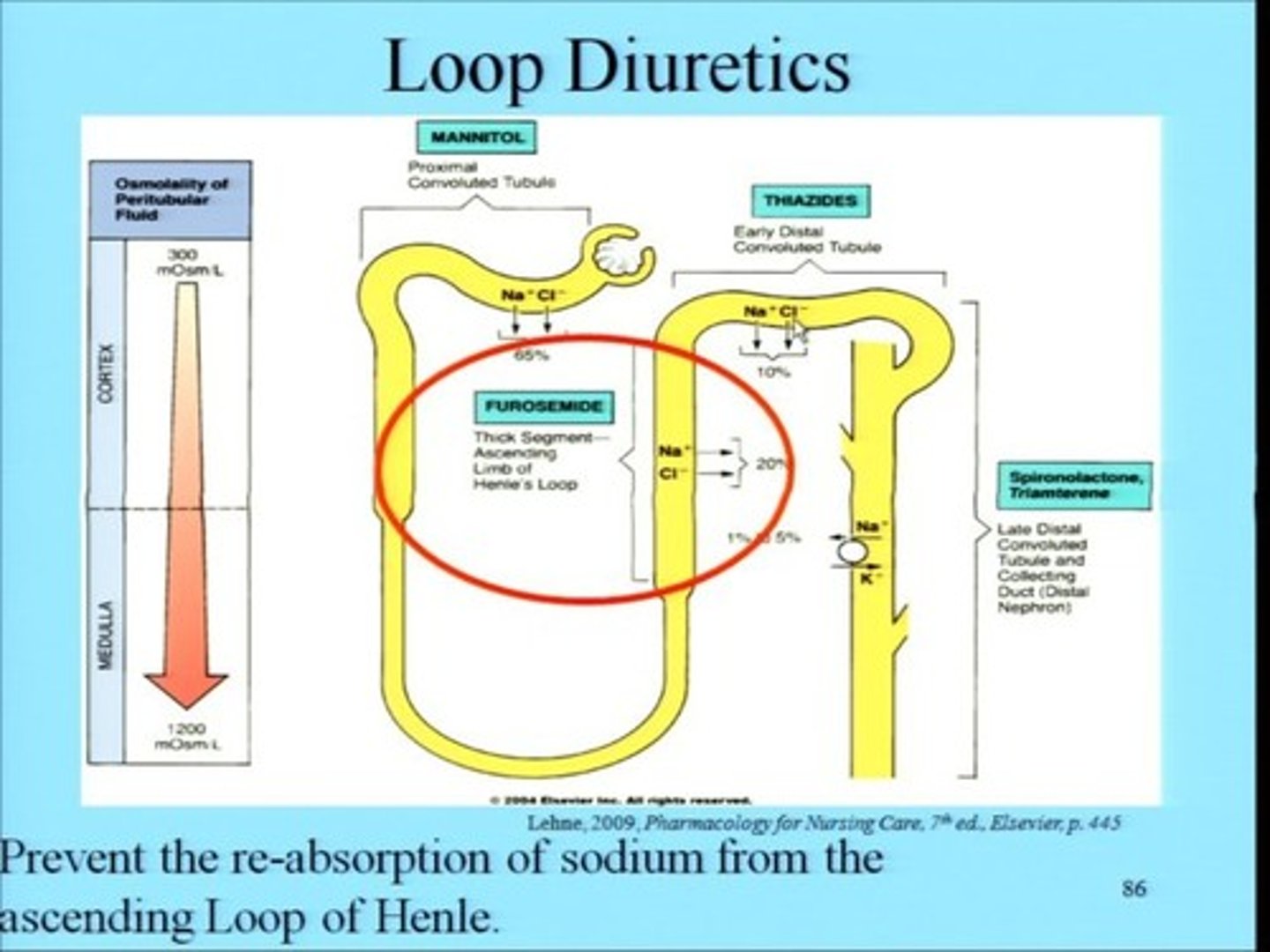
patient symptom scale
- may be used instead of NYHA scale, based on how patient is feeling that day
green: no action needed
- feel normal, breathing okay, no swelling, sleeping okay, no chest pain
yellow: need to call primary provider
- dizzy/ lightheaded, trouble breathing, gaining 2 pounds/ day or 5 pounds/ week, trouble sleeping flat, minimal chest pain
red: go to ER or call 911
- passing out, suddenly cannot breath, new chest pain even after resting for 10 mins
potassium replacement
goal: >4 and <5 mEq/L
- given with loop diuretics if needed
- give PO if possible, IV requires access and burns
- when giving PO, do not give more than 60 mEq at one time, more is irritating to stomach (N/V)
- give 10 mEq PO per every 0.1 mEq/L that K is below 4 mEq/L
> ex: pt's K = 3.6
4-3.6 = 0.4 ; 0.4 x 10 = 40 mEq ; supplement 40 mEq oral KCl
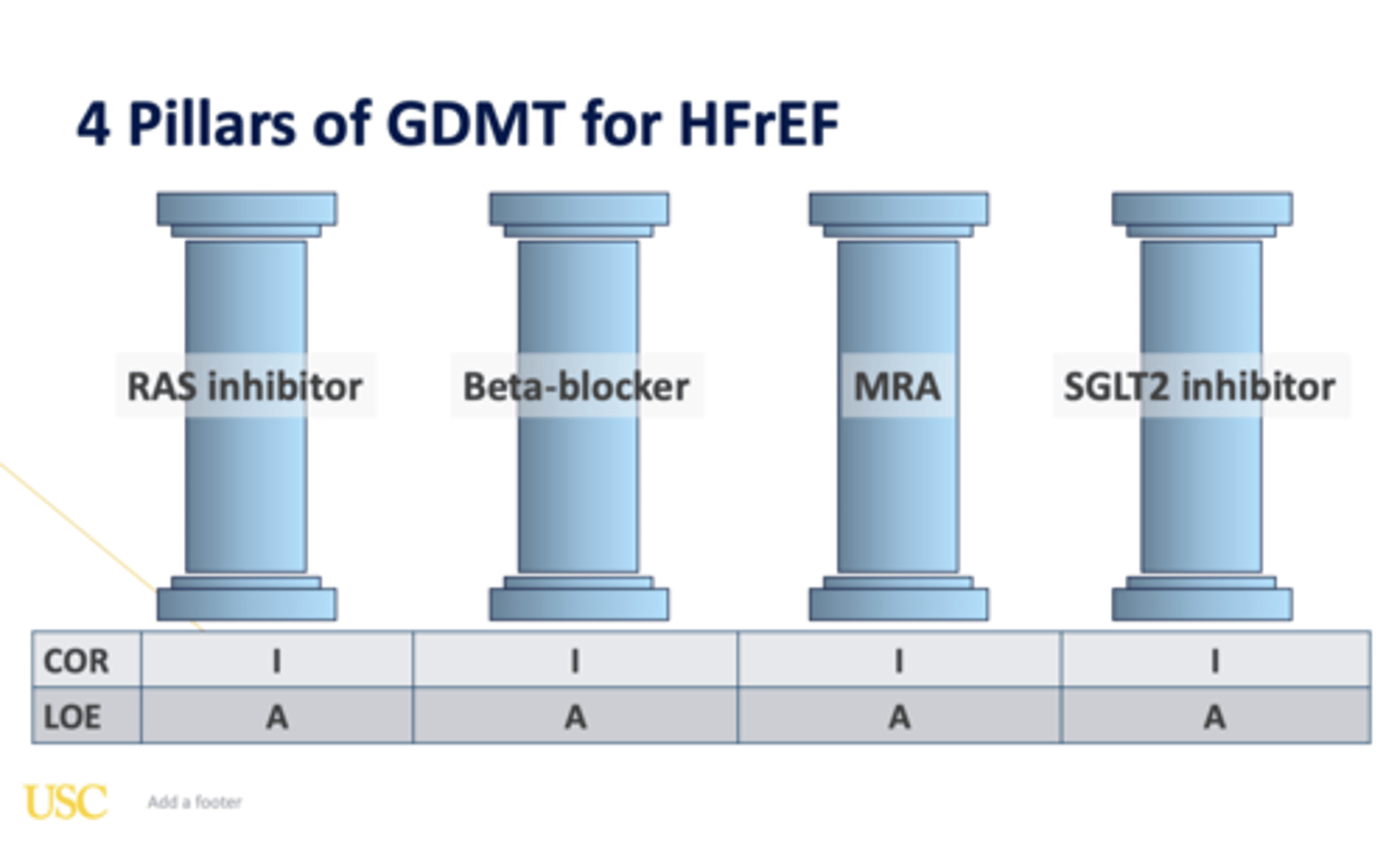
4 pillars of GDMT
1. ARNI (Entresto)
- alt: ACEi or ARB
2. beta blocker
3. MRA
4. SGLT2i
- for each: initiate, optimise (titrate), and reassess
- typically initiate all 4 at the same time
- consider additional therapies if needed
ARNI
angiotensin receptor neprilysin inhibitor
sacubitril/valsartan
- when to use: patients who were first approved and used an ACEi or ARB, first line as a RAAS inhibitor
- when not to use: history of angioedema, pregnancy/ lactation, severe hepatic impairment
- caution in: SBP <100 mmHg, renal artery stenosis, volume depletion
- dosing:
> 24/26 mg bid if low GFR or ACEi naive
> 49/51 mg bid if previously on moderate ACEi/ ARB
> must allow at least 36 hour washout between ACEi to prevent angioedema
> titrate dose every 2-4 weeks: double dose
ACEi
- CI: angioedema, anuric renal failure, pregnancy, bilateral renal artery stenosis, K+ > 5.5
- ADE: hypotension, ↑Scr, cough, K+ retention, angioedema
- PK:
> t1/2: lisinopril > enalapril > captopril
> food decreases oral bioavailability of captopril
> if liver dysfunction, can use captopril or lisinopril (enalapril is a prodrug)
- monitoring: Scr and K+ within 1-2 weeks, Scr is excpected to increase up to 30%
ARB
- when to use: alternative to ACEi in patients who are intolerance (cough or angioedema), first line in pts with borderline low BP to test plan to transition to ARNI
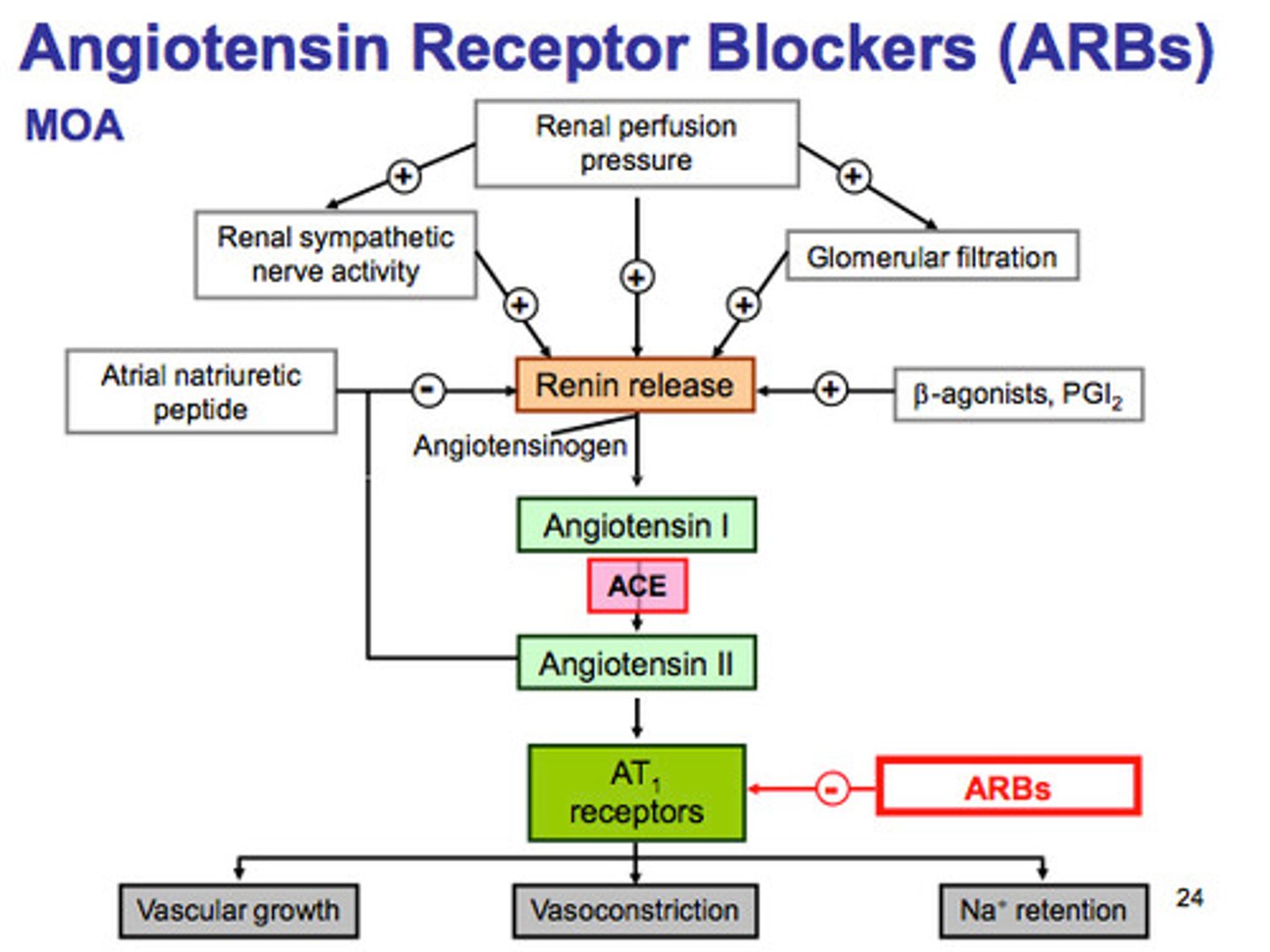
which RAAS inhibitor to use in HF
- ARNI is becoming preferred 1st line
- use shared decision making based on cost, BP, diuretic effect, other conditions
- best choice for patient overall is to titrate whichever option to target dose
> this will reduce risk of rehospitalization
beta blockers
carvedilol (Coreg), metoprolol succ (Toprol XL), bisoprolol (Zebeta)
- why do we use: inhibits sympathetic nervous system, reduce afterload, increases ejection fraction, reduces remodeling/ slows progression, reduces hospitalization, reduces mortality
- who gets them: ALL patients with HFrEF
> caution when initiating in class III-IV patients
- dosing: increase to maximum tolerated dose every 2 weeks
> target dose = reduced mortality
- ADE: ↑SOB, edema, weight gain, rales, ↓HR, depression, fatigue, impotence

MRA
spironolactone (Aldactone), eplerenone (Inspra)
mineralocorticoid receptor antagonist (aka K+ sparing aka aldosterone antagonists)
- used to reduce mortality in all chronic HFrEF patients
> including post-MI with new low EF (+ DM or + HF symptoms)
- eplerenone is selective, use if intolerance to spiro
- CI: Scr >2.5 (males) or >2 (females), K >5, eGFR <30
- ADE: gynecomastia (spiro), hyperkalemia, hypotension
- monitoring: serum K+ (must monitor closely)
> reduce dose if hyperkalemia develops: if >5, reduce to 12.5 mg
> if K+ is low or stable, consider titrating up to 50 mg
SGLT2i
dapagliflozin (Farxiga), empagliflozin (Jardiance)
- addition to optimal HFrEF medical therapy reduces mortality
- used for: patients already on RAAS inhibitor, BB, and MRA
> evolving practice: initiate all 4 at the same time
- CI: SBP <95, eGFR <30 (dapa) or 20 (empa), T1DM, recurrent or active UTI
- monitor: mycotic infections, volume depletion, ketoacidosis, AKI
> hold when sick and 72 hours before surgery
- dosing: 10 mg daily
> can decrease to 5 mg if hypotension
introducing quadruple therapy
chronic HF
- d/c ACEi/ ARB and initiate ARNI
- initiate SGLT2i
- continue BB
- initiate MRA after 2 weeks
de novo HF
- initiate ARNI & BB
- initiate SGLT2i & MRA after 2-4 weeks
- start ARNI and BB dose low and titrate to goal or max tolerated dose
- monitor: hypotension, eGFR decline (20%, will stabilize), hyperkalemia
isosorbide dinitrate/hydralazine
BiDil
- isosorbide is a venous vasodilator = reduces preload
- hydralazine is a direct arterial vasodilator = reduces afterload and prevents nitrate tolerance
- ADE: ↓ BP, N/V, headache, +ANA (lupus), tachycardia
- used for: african american patients with class III-IV HFrEF receiving optimal medical therapy (1A), people who cannot be given first line agents
ivabradine
Corlanor
- used for: symptomatic chronic HFrEF who are in sinus rhythm with resting HR ≥70 bpm and are on either max tolerated doses of BB or have a CI to BB
- has significant reductions in HF hospitalizations
- CI: afib
- titrate to target HR 50-60 bpm

digoxin
Lanoxin
- used for: pts on max BB that are still symptomatic/ have many hospitalizations
- benefits: ↑ contractility, ↑ vagal tone, ↑ FC, ↑ exercise capacity
- monitor: changes in renal function, K+
- similar to ivarbadine: to decide between the 2 look at cost, kidney function, afib

recommendation in HFimpEF
- HFimpEF >40% LVEF
- GDMT should be continued to prevent relapse of HF and LV dysfunction, even in patients who may become symptomatic (1 B-R)
- continue therapy + optimize dosing, adherence, patient education, and address goals of care
GDMT for HFmrEF
- HFmrEF 41-49% LVEF
- if symptoms appear to be more like HFrEF, can use the 4 pillars and reassess
- if symptoms appear to be more like HFpEF, manage like so (most patients fall here)
- typically use: diuretics as needed and SGLT2i
HFrEF vs HFpEF
HFrEF: HF with reduced ejection fraction; often due to a weakened muscle that can't pump enough
HFpEF: HF with preserved ejection fraction; a filling problem rather than a muscle damage or squeezing problem
- more often due to a long standing change in pressures and LV hypertrophy
> secondary due to: age, obesity, AF, OSA, HTN
- also has more mixed pathology
diagnosing HFpEF
- really need to rule out other causes here since EF is normal
- must demonstrate something is actually wrong with the heart
- scoring tool gives points for: BMI >30, on 2+ anti HTN medications, afib, pulmonary HTN, age >60, and elevated LV filling pressure
GDMT for HFpEF
diuretics as needed and SGLT2i
- diuretics decrease volume overload symptoms
- SGLT2i may lower hospitalizations, and extra impact if T2DM
other pillars (ARNI, ACEi/ARB, MRA, and BB) may be used
- ARNI: EF >45%, older, female, ischemic history
- ACEi/ ARB: reasonable 1st line antihypertensive
- MRA: consider for extra BP lowering, experiencing hypoK with other diuretics, or resistant HTN
- BB: elevated HR or necessary for afib