Apologia Module 14
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
Reaction rate—
The rate of change of a product in a chemical reaction
Reaction rate equation
R= ∆[products] / ∆t = - ∆[products] / ∆t
In an experiment, what is the “control”?
The control is the test that is done under standard conditions and all other test are compared to it.
What must chemicals do to react?
To react chemicals must collide with one another so that their electrons can be either transferred or rearranged
Higher temps do what to a chemicalsReaction Rate and why?
Higher temperature increases chemical reaction rate because the reactant molecules (or atoms) move faster, increasing the chance for collision.
Decreasing concentration does what to the reaction rate and why?
Decreasing concentration decreases the chemical reaction rate because there are fewer reactant molecule (or atoms) to collide with one another.
What does increasing the surface area of a reactant do to the reaction rate and why?
Increasing the surface area of a reactant increases the reaction rate because the molecules (or atoms) of the reactant can more easily mingle with the molecules (or atoms) of the other reactants.
What is the Rate equation
R = k[A]x[B]y
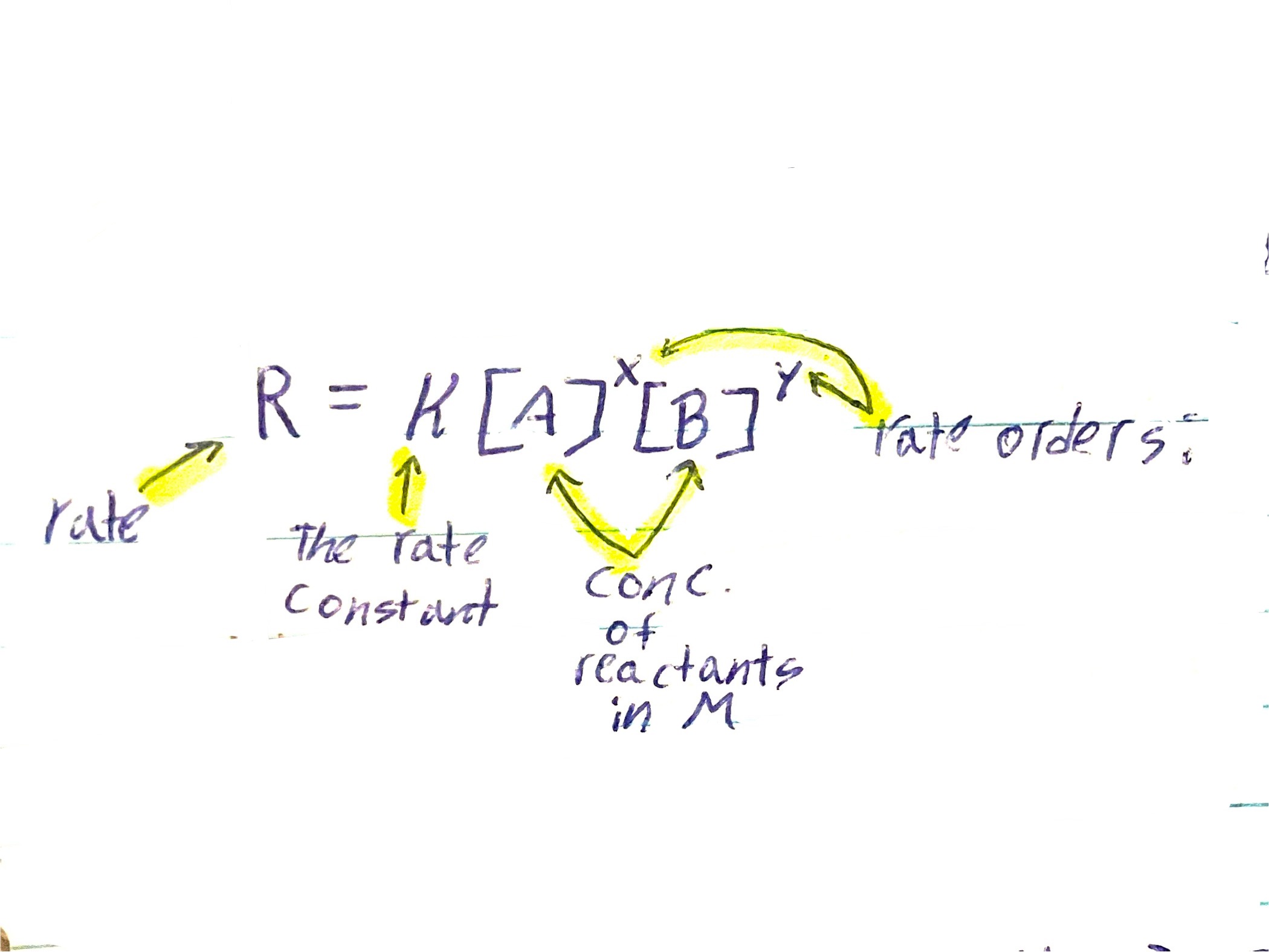
Rate equation meaning
R is the rate, k is the rate constant, The brackets are the concentration of reactants in M (molarity), X and Y are the rate orders
When is the exponent of a rate 0 for a reactant?
Sometimes, the rate of a chemical reaction will be unaffected by the concentration of one of its reactants.
In this case, the exponent of the rate equation is 0 for that reactant
How MUST rate orders be determined?
The reaction rate orders for a chemical reaction must be determined experimentally; they are NOT equal to the stoichiometric coefficients in the chemical equation.
Do chemical reactions ever finish?
Chemical reactions never actually finish. They just keep getting slower and slower until they proceed imperceptibly slowly. This means that for any chemical reaction, no matter how long we wait, there will always be some reactants left.
Heats affect on reaction rate-
The temperature dependence of reaction rate is determined by the variation of the rate constant with temperature. Since the rate constant increases dramatically with increasing temperature, the reaction rate does as well.
The rate of many chemical reactions- doubles for every—
Doubles for every 10°C increase in temp.
Do Catalysts get used up in a chemical reaction?
No Catalysts speed up the reaction rate without actually getting used up in the chemical reaction.
What does decreasing the activation energy of a reaction increase?
it increases its rate
How do catalysts spread up reaction rates?
Catalysts speed up reaction rates by lowering the activation energy of the reaction.
Heterogeneous catalysts-
Catalysts that are in a different phase than the reactants.
Homogeneous catalysts-
Catalysts that have the same phase as at least one of the reactants.
Reaction mechanism-
A series of chemical equations that tells the step-by-step process by which a chemical reaction occurs.