M2L3 Cytoskeletal regulation
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Outline the different types of cytoskeleton
Microtubules
Long structures involved in organelle and vesicular transport, cell polarity, cell division, and structural support
Made of α and β subunits, forming a tubule stucture
Intermediate filaments
Found everywhere, varying in shape and size depending on location
Provides mechanical strength, resistance to sheer stress, cell shape maintenance, anchorage of organelles
Many types - neurofilaments, vimentin, keratin, lamins, desmin etc
Actin filaments
Involved in cell movement/migration, muscle contraction, membrane organisation, organisation of membrane receptors/proteins
Cell migration in tumour invasion and metastasis is facilitated by the constant turnover of the actin cytoskeleton
What is the importance of actin depolymerisation?
Actin filament depolymerisation ensures turnover of actin filaments and maintains a pool of monomers to recycle for polymerisation
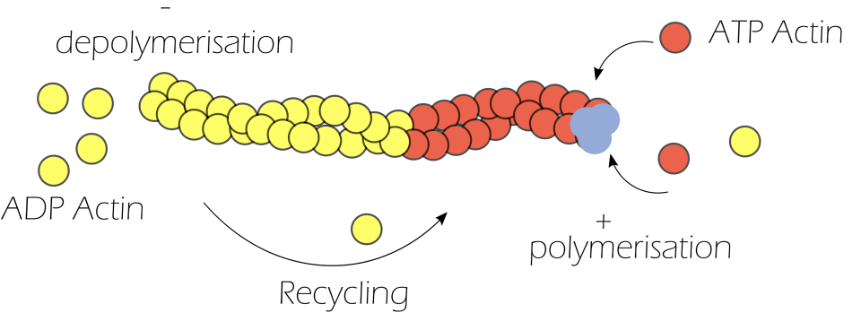
How does actin polymerise/depolymerise?
Actin is made of monomeric globular actin, which is highly energy dependent
Globular actin binds to ATP to form ATP-actin
Three ATP-actin monomers come together to form the nucleation site, from which polymerisation occurs
ATP hydrolysis converts ATP-actin to ADP-actin for depolymerisation
Actin polymerisation coupled with depolymerisation is known as actin treadmilling, resulting in a highly dynamic cytoskeleton
Compare the role of profilin vs cofilin
Profilin promotes the assembly of actin filaments, by catalysing the exchange of ADP for ATP, and inhibits spontaneous nucleation
Cofilin severs actin filaments so that they do not need to get released one by one, accelerating depolymerisation
What is the role of Arp2/3 in actin polymerisation?
Arp2/3 complexes are controllable nulceating structures which bind together G-actin monomers to initiate polymersation or bind to existing actin subunits at the ‘mother’ filament to form ‘daughter’ filament branches
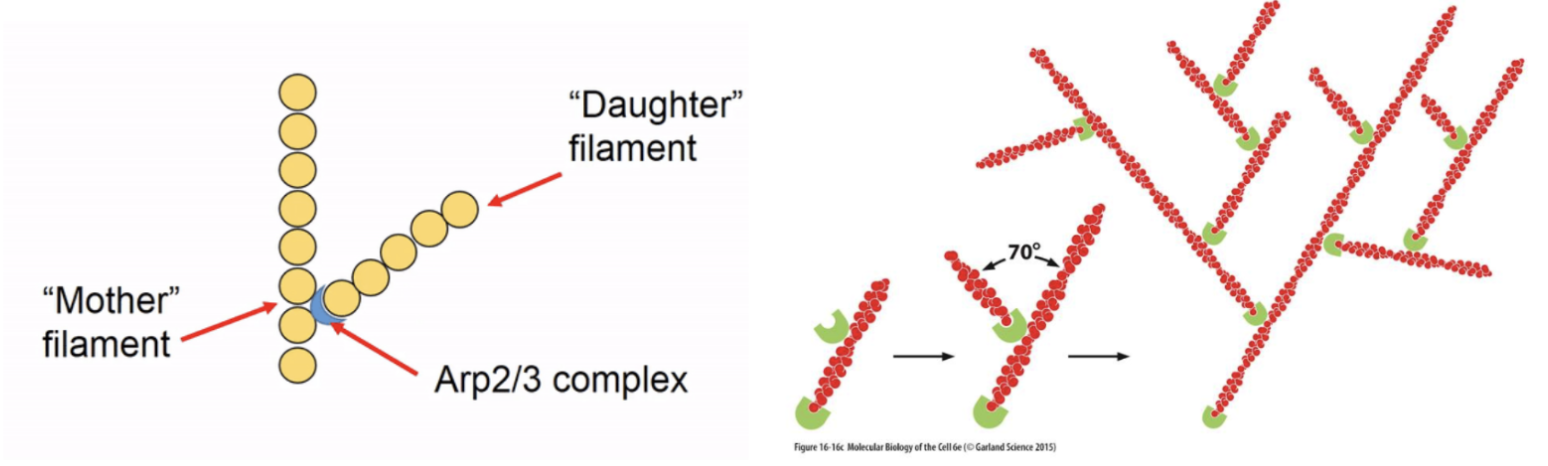
What is the role of formins in actin polymerisation?
Formins also initiate linear subunit addition at the + end of actin and its FH2 domain stabilises a spontaneously formed actin dimer to nucleate a new filament, elongating it by recruiting more actin monomers using its FH1 domain and profilin
What is the role of elongation factors in actin polymerisation?
Elongation factors (eg. VASP) help to formed actin filaments and stabilise the structure to aid in elongation
Good at allowing actin to polymerise, responding to extracellular signals, regulating cell migration, and are expressed as five different isoforms depending on where they are expressed (WASp, N-WASp, WAVE1, WAVE2, WAVE3)
What is the role of thymosins in actin polymerisation?
Thymosins sequester actin monomers to which they bind and block filament assembly and release them when filament growth is needed
Outline the steps in mesenchymal movement of cells
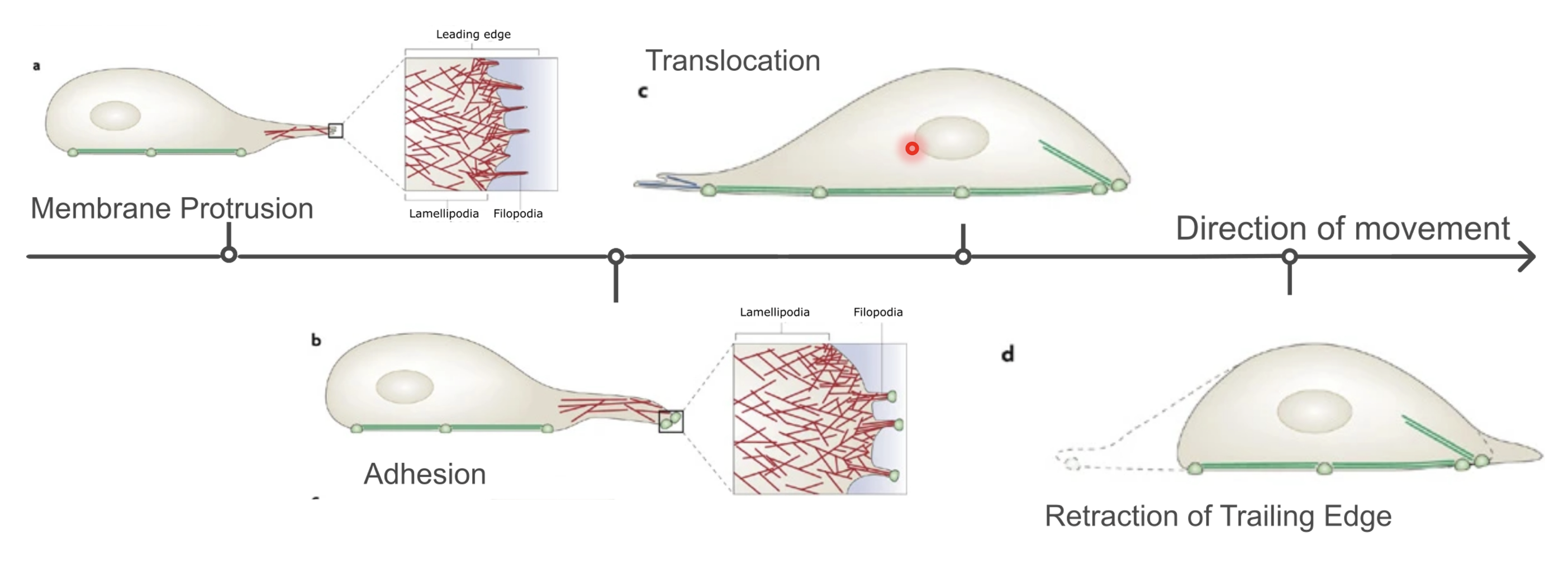
Membrane protrusion (extension of filopodia and lamellipodia) → focal adhesion → translocation (loosening of focal adhesions at the rear) → retraction of trailing edge
What structures are formed at the leading edge of crawling cells?
Filopodia - finger-like, made of parallel F-actin bundles
Lamellipodia - flat regions with highly branched actin network
Invadopodia - projection into the ECM for remodelling (eg. in carcinoma)
Podosomes - projections into ECM (but do not necessarily remodel) (eg. monocytic cells)
What are the members of the Rho family of GTPases which mediate cell movement?
Cdc42, Rho and Rac
Explain translocation and retraction mediated by Rho
Active Rho (Rho-GTP) activates Rho trigger (ROCK), which phosphorylates downstream targets to activate myosin and trigger actomyosin contractile forces for locomotion
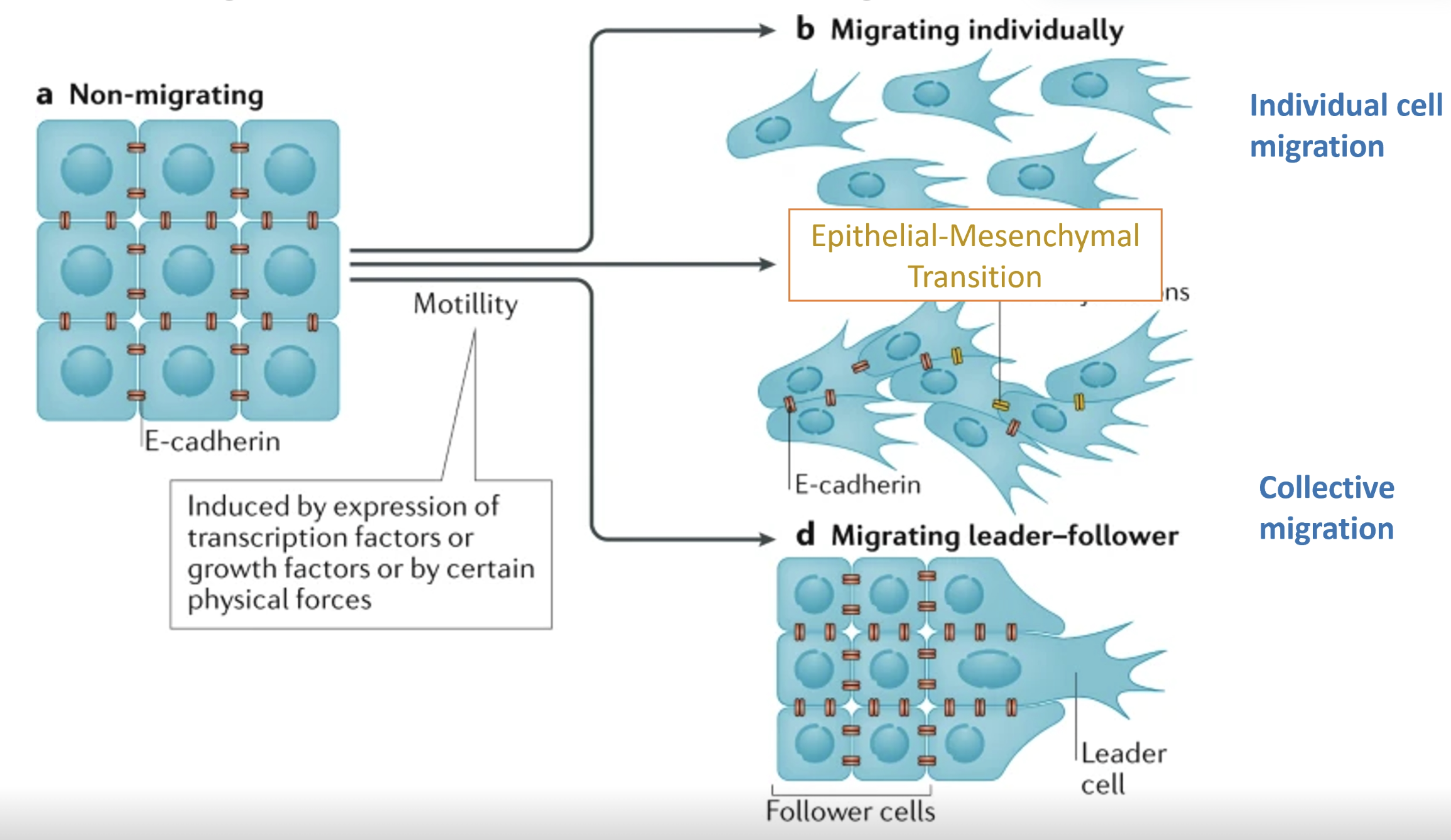
Distinguish collective vs individual migration in cancer
In collective migration in cancer, sheets of cells can move together
Migrating adherent vs migrating leader-follower models
Cells can also migrate individually
The migrating adherent model and individual migration model involve a cadherin shift (EMT)
Distinguish between mesenchymal and amoeboid movement
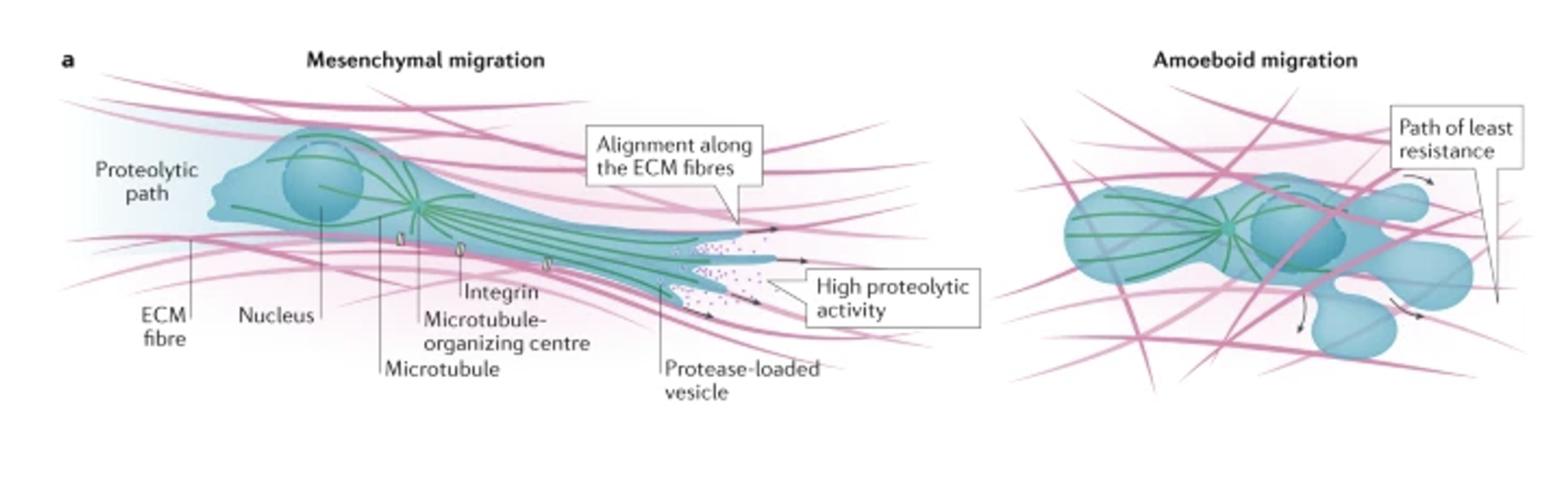
Mesenchymal (cells take on an elongated. fibroblast-like morphology)
More common in dense matrices
Involving extension of invadopodia and proteolytic ECM remodeling using MMPs in an integrin-dependent manner
Creates a proteolytic path for migration and invasion
Amoeboid (cells have rounded morphology)
More common in less dense matrices, involving contraction of cortical actomyosin cytoskeleton to create hydrostatic pressure needed to squeeze the cell body through gaps in the matrix
Integrin-independent process
Eg. non-neoplastic cells (lymphocytes, neutrophils) or neoplasmic cells (eg. lymphoma, leukaemia, small cell lung carcinoma)
How can the balance between Rac and Rho determine the mode of migration used by cancer cells?
Mesenchymal - Rac dependent
Amoeboid - Rho/ROCK signalling dependent, actin cytoskeleton reorganised at PM with dynamic blebbing
Describe the stiffness of the ECM in solid tumours
Tumours are highly stiff compared to surrounding tissue due to highly fibrous ECM
ECM can signal to cells to produce cytokines and chemokines to help maintain its stiffness
TGFβ and IL-15 are particularly good at signalling to the cells
TGFβ in particular is notable for inducing EMT and cytoskeletal reorganisation
How do cancer cells respond to mechanical signals?
Activate mechanosensors - detect cues such as force, stress, strain etc.
Detected by integrins, GPCRs, TRP ion channels, mechanosensitive Piezo ion channels, YAP/TAZ regulators of mechanotransduction
Initiate mechanotransduction - triggers signalling pathways
PI3K/AKT - cell survival and metabolism
JAK/STAT - transmits signals from mechanical stimuli to alter gene expression
FAK - mediates cell adhesion and signalling
Hippo - regulates growth and organ size
RhoA - activates responses to mechanical stress via GTP-binding proteins
Alter cell functions (altering gene expression, cytoskeleton organisation, membrane traffic)
What is the role of Hippo signalling in cancer?
Involves the kinase MST2 and SAV1 and RASSF1A apaptors which help activate MST2
When Hippo pathway is active MST2 phosphorylates LATS1/2 and MOB1A/B
LATS1/2 phosphorylates YAP/TAZ which get retained in the cytoplasm or targeted for degradation
When there is Hippo signalling dysregulation (common in cancer), LATS1/2 is not phosphorylated and YAP/TAZ is able to enter the nucleus
YAP/TAZ are well known mechanosensors
When they are active (stiff ECM, cell spreading, high contractile force), YAP/TAZ enter the nucleus and activate TEAD TFs to promote cell proliferation in cancer or osteogenic differentiation
When inactive (confined cell adhesion, soft ECM, low contractile force), YAP/TAZ are proteasomally degraded, causing apoptosis, growth arrest, or adipocyte differentiation
What is the link between the actin cytoskeleton and Hippo signalling>
Triggering Rho/Rac pathways and F-actin production can prevent LATS1/2 phosphorylation, allowing YAP/TAZ-mediated activation of proliferation and survival genes by interacting in the Hippo pathway
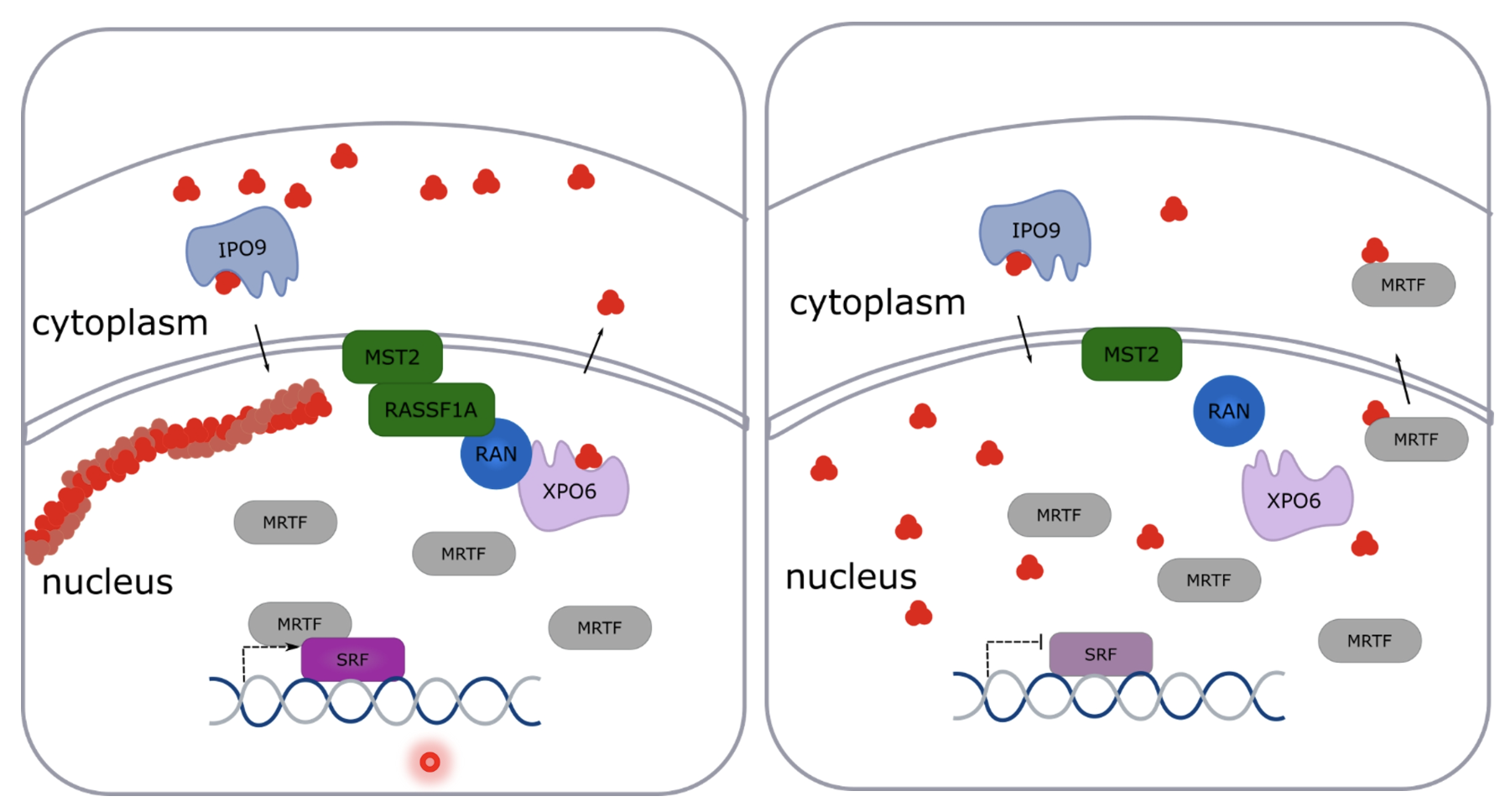
What is the role of RASSF1A in regulating nuclear actin?
Loss of RASSF1A —> matrix stiffness, collagen deposition, metastasis,
Also higher levels of stem cell TF Nanog
Increased stiffness activates Wnt/β-catenin and stemness
MST2 recruits RASSF1A to the nuclear envelope and RASSF1A interacts with XPO6/RAN to export G-actin, so loss of RASSF1A leads to increase in G-actin in the nucleus
XPO6 involved in shuttling G-actin from nucleus to cytoplasm and IPO9 brings it back
Therefore RASSF1A regulates nuclear actin (by binding to Filamin A)
How can mechanical signals get transmitted to the nucleus to cause actin polymerisation?
In the nuclear envelope there are LINC complexes, made of SUN proteins and KASH proteins (nesprins) which act as bridge to bind to different cytoskeletal elements depending on the type/location of the nesprin
Mechanical signals get transmitted to the nucleus via LINC complexes
When the signal is transduced, formins are recruited or coordinated to promote localised actin polymerisation in the nucleus
How is nuclear actin related to cancer?
Nuclear actin can regulate chromatin accessibility, EMT, DNA repair and CD4+ T cell effector functions
T cells and NK cells have very high RASSF1A expression and activated T cells have increased nuclear actin, responsible for Ca2+ signalling and effector responses (antibody production, antigen recognition)
RASSF1A loss impairs T cell migration and reduces T ccell tumour killing efficiency in CD8+ T cells due to downregulation of CD69 and nuclear actin
T cells have many microvilli to scan surfaces and they are rich in adhesion molecules and TCR, they are used to established contacts and initiate signalling
Microvilli shape is disrupted in T cells with RASSF1A mutations which impairs its function, not making contacts with tumour cells