U4: Electrochemistry
1/72
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
ways to define oxidation/reduction
oxidation:
addition of oxygen atoms
remove H atoms
remove e-
reduction:
removal of oxygen atoms
addition of H atoms
addition of e-
difference in writing ionic charges vs oxidation numbers
ionic: 2+
oxidation number: +2
what is an oxidation number
In simple terms, an oxidation number (or oxidation state) is a value assigned to an atom to represent how many electrons it has gained or lost compared to its neutral state.
Think of it as the "apparent charge" an atom would have if all its bonding electrons were assigned to the most electronegative element.
Rules for assigning oxidation numbers
Pure elements and molecules of only 1 element (ex. Na, O2, O3 etc) have an oxidation number of zero (0)
Ions by themselves (ex. Ca2+, Cl-) have an oxidation number equal to their charge
group 17 elements have (-1) unless bonded to another more electronegative halogen
oxygen usually has an oxidation number of (-2) EXCEPT when:
combined with fluorine (F) => +2
In a peroxide (H2O2) => -1
H in compounds => +1
H in certain metal hydrides (NaH) => -1
these are the “fixed” elements
Some elements are so predictable that they almost always have the same oxidation state when they are in a compound.
Group 1 (Alkali Metals): Elements like Li, Na, K, Rb, Cs. They have one valence electron that they are "desperate" to get rid of. In a compound, they are always +1.
Group 2 (Alkaline Earth Metals): Elements like Be, Mg, Ca, Sr, Ba. They have two valence electrons they want to lose. In a compound, they are always +2.
Zinc ($Zn$): Even though it's a transition metal (which usually have multiple possible charges), Zinc is an exception that is almost always +2.
Why? These atoms are much more stable once they lose those specific electrons to reach a "noble gas" electron configuration.
Rule 7: Binary Compounds & Electronegativity
A binary compound is just a fancy way of saying a molecule made of exactly two different elements (like $NaCl, MgO,$ or $PF_3$).
When these two elements bond, they "fight" over the electrons. The rule says: The more electronegative atom wins.
Look at the two elements.
Determine which one is further to the top-right of the periodic table (that's the more electronegative one).
Give that atom the charge it would have if it were an ion (its "normal" group charge).
Calculate the other atom's number so the total equals zero.
2 overall rules for assigning oxidation numbers
● Oxidation state of all atoms in a neutral compound (ionic,
molecular or both) must sum up to give zero overall.
● The sum of the oxidation numbers of all atoms in polyatomic ions
must equal the overall charge of the ion.
oxidation numbers of PCl3
Example: $PCl_3$
Step 1: We have Phosphorus ($P$) and Chlorine ($Cl$).
Step 2: Chlorine is further right; it is more electronegative.
Step 3: If Chlorine were an ion, it would be -1 (it's in Group 17). So, its oxidation number is -1.
Step 4: There are three Chlorines ($3 \times -1 = -3$). To make the whole molecule neutral (0), the Phosphorus must be +3.
why does oxygen in a peroxide have an oxidation state of -1?
a peroxide is a specific group of compounds that contains a single bond between 2 oxygen atoms (O-O)
these 2 oxygen atoms are identical in electronegativity, so they share the electrons in that middle O-O bond equally, they only steal one e each from the atoms they are attached to
average oxidation states
this occurs in the compound Fe3O4
where Fe has an oxidation number of +8/3
but you cant actually lose 8/3 electrons, hence the fraction 8/3 is just an average oxidation state which represents 2 different iron oxides mixed together in one crystal lattice
FeO + Fe2O3 → Fe3O4
iron (II) and iron (III) oxide
this FE with +8/3 is Fe (II, III) oxide which tells you that within this single substance some iron atoms have +2 and some have +3 charge

biochemical oxygen demand (BOD)
BOD is the amount of dissolved oxygen required to decompose organic matter in a biological sample over a set time period, measured in mg/dm3 or ppm
what does BOD measure
BOD can measure the health of an aquatic ecosystem
bc polluted water with a high BOD cannot sustain life bc it has no means of replenishing oxygen
what is electricity
its the movement of electrons to generate a current (electrons move from - to +)
what is a galvanic cell
basically a battery that converts chemical energy into electrical energy to do work
composition of a galvanic cell
made of 2 half cells (beakers)
each half cell has an electrode (metals, metals conduct electricity)
what are the 2 types of electrodes? what occurs at each
anode = oxidation = lose e
cathode = reduction = gain e
half reactions at each electrode
electrons are being released at the anode (bc ox is happening) so the anode is negatively charged
electrons flow from anode to cathode
cathode is positively charged
what does it tell you if oxidation number decreases/increases
oxidation number decrease = reduction
oxidation number increase = oxidation
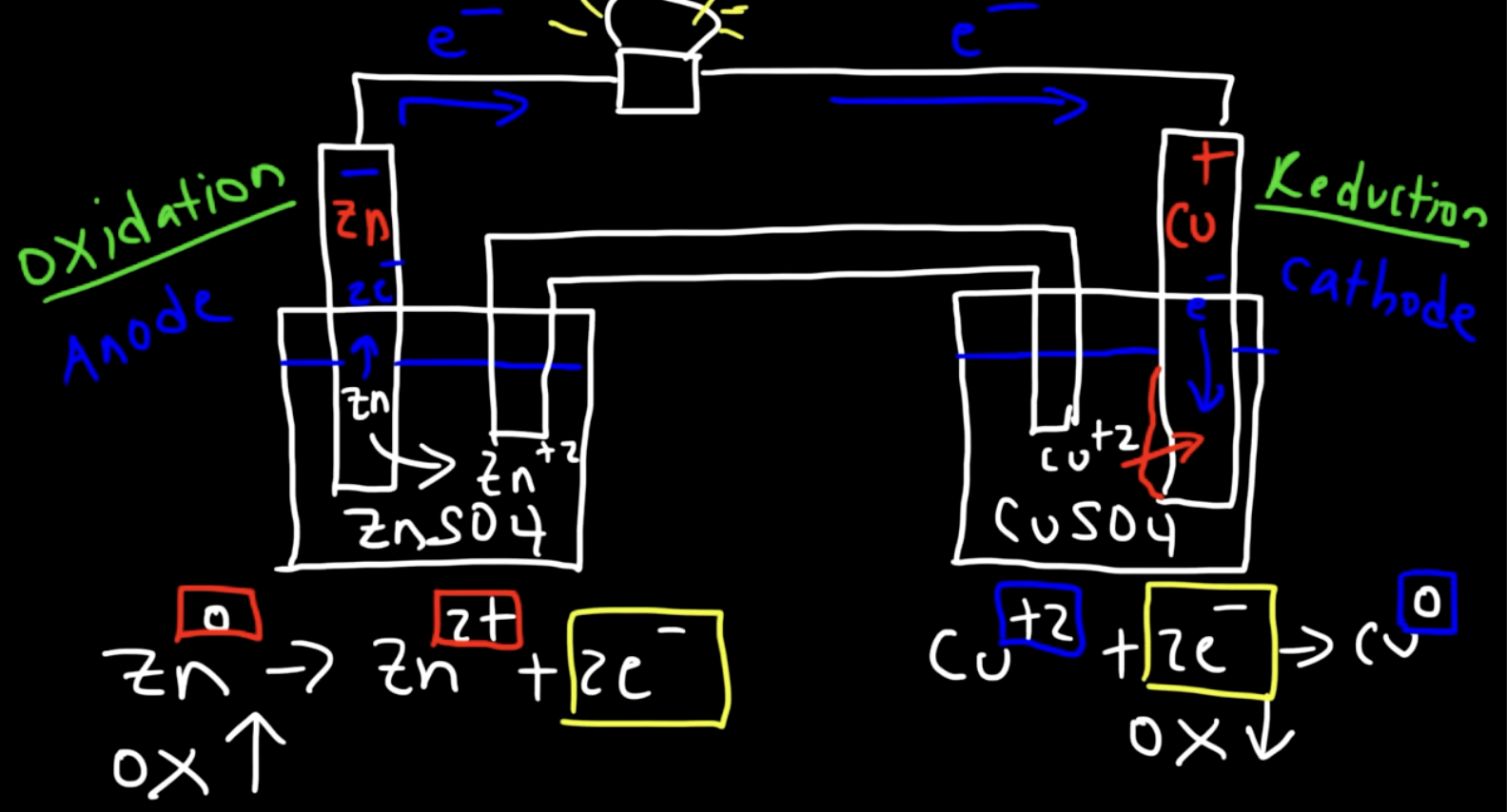
explain whats happening for the states of Zn and Cu at the interface
the Zn in the electrode is a solid, but when it gets oxidized it becomes an ion and dissociates into the solution
these electrons travel to the cathode
the Cu2+ ions in the solutions of the cathode are attracted to the incoming electrons and get reduced to make Cu (solid metal) which is deposited on the Cu electrode
mass changes in anodes and cathodes
anodes lose mass
cathodes gain mass
why do we need a salt bridge
you need a salt bridge to prevent a build up of charge on etiher electrode
anode loses e, so there will be a buildup of positive charge
cathode gains e, so there will be a buildup of negative charge
if the anode gets + charge, then the electrons will reverse directions, instead of moving from anode to cathode, itll mode from cathode to anode, creating a state of equlibirum → no electron flow!
salt bridge keeps a steady flow of electrons by keeping the solutions around the electrodes neutral
anions from cathode go to anode
cations from anode go to cathode
for the redox reaction:
Zn + Cu2+ → Cu + Zn2+
write the half reactions
what is the overall cell potential? how do we calculate?
how do you represent this galvanic cell system using line notation?
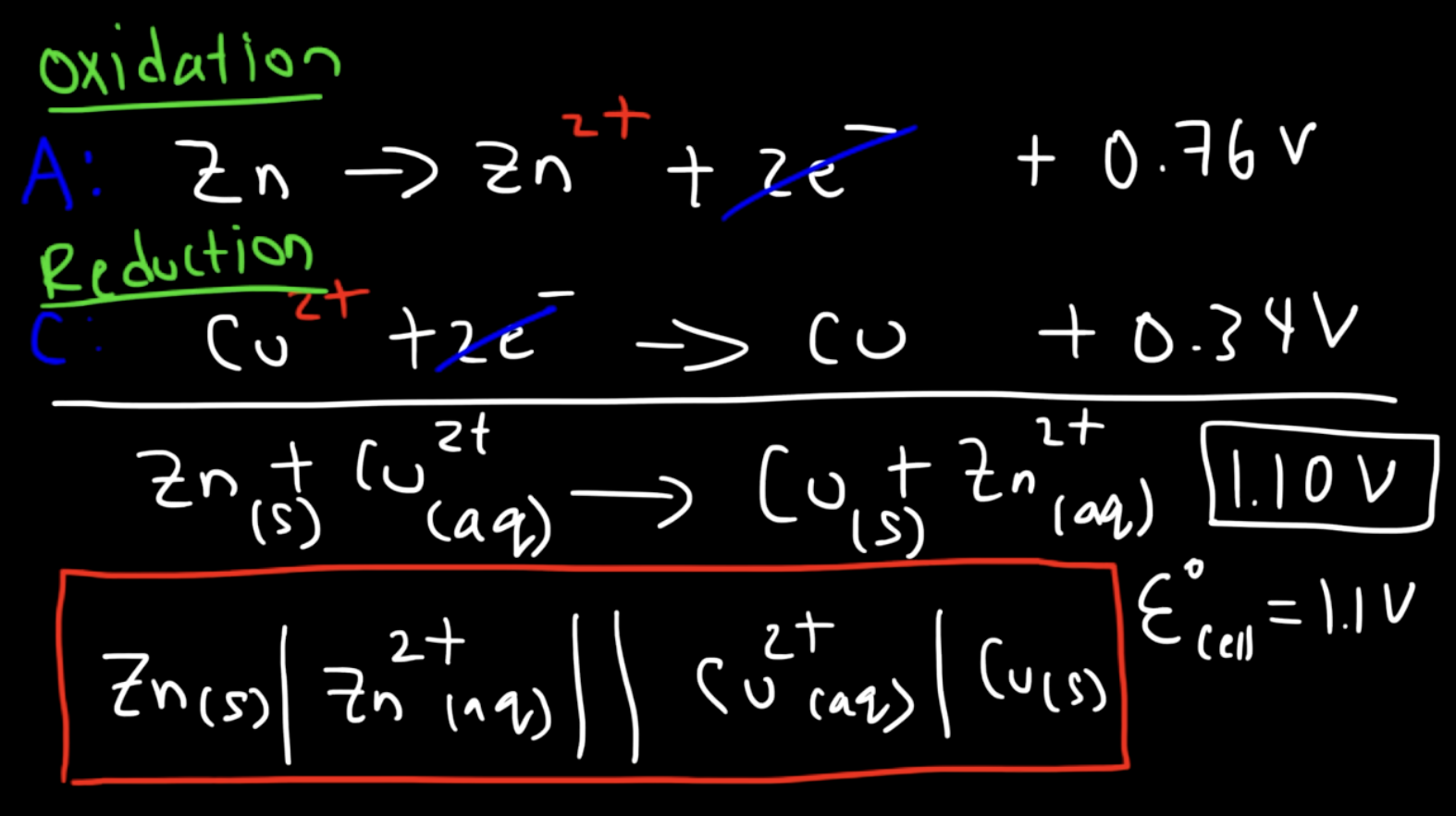
what does the cell potential tell you about the spontaneity?
what about galvanic cells?
galvanic cells convert chemical energy into electrical energy, and you cant derive energy from non spontaneous processes (these take energy!) so galvanic cells use spontaneous reactions (release energy) therefore they always have a + cell potential

the lowest cell potential a cell can have is 0
this is when its fully depleted (discharged) and its in a state of equlibiurm, can do no more work
units of cell potential
V (Volts)
KEY difference between electrolytic cell and galvanic cell
you have to put energy into electrolytic cells to get them to work
galvanic cells geneate their own energy
another name for galvanic cells
voltaic cells
what are the components of a half cell
a metal electrode immersed in a solution of its own metal ions called the electrolyte
the electrode is metal because metals lose electrons
what happens in each half cell of a galvenic cell
in each half cell, the atoms of the metal will form ions by releasing electrons
this makes the electrode more positive than the electrolyte solution
the difference in charge between the electrode and electrolyte
charge separation is called the electrode potential
the electrode potential denotes an equilibirum between the metal and its ions in the electrolute
explain the components of cell disgram/line diagram convention
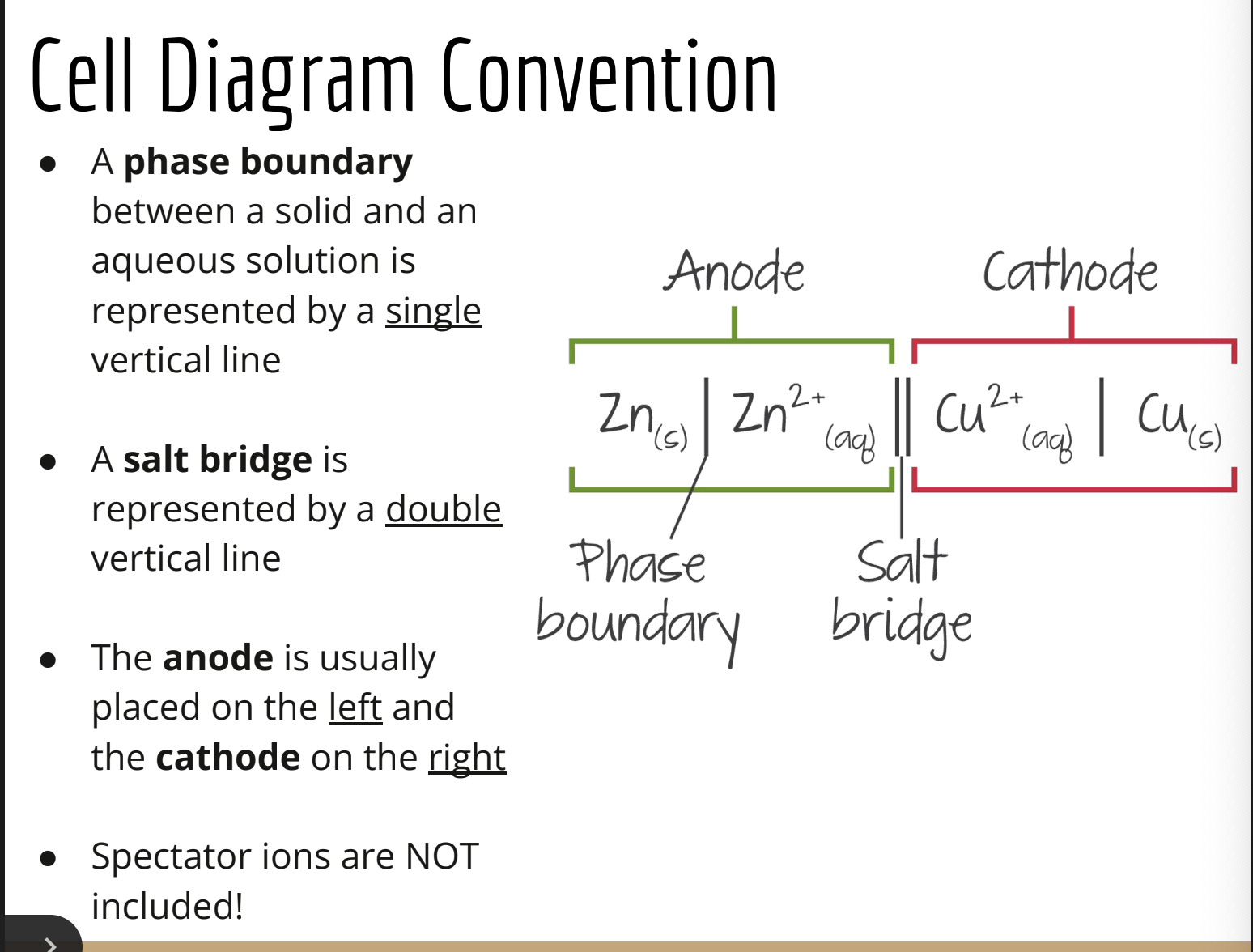
what is a salt bridge
a tube containing an electrolyte solutions that connects 2 half cells in a galvanic cell
how are voltaic cells spontaneous
This slide explores the mechanics of Voltaic Cells (also known as Galvanic cells), which are devices that convert chemical energy into electrical energy through spontaneous redox reactions.
The fundamental concept is that you can generate electricity without an external power source by physically separating the oxidation and reduction half-reactions.
voltage produced in voltaic cells depends on what?
voltage in voltaic cells depends on the difference in reactivity between 2 metals
what is a SHE
standard hydrogen electrode
this quantifies whether one element will more readily undergo reduction or oxidation compared to another element
SHE is the standard value other electrode potentials are compared to
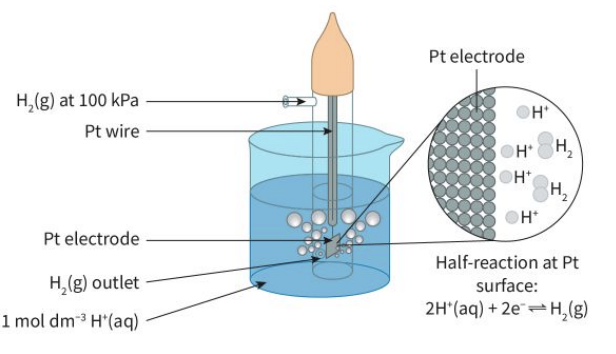
SHE is a platinum electrode submerged in a solution of 1 mol dm^-3 H+ (aq) ions
hydrogen gas is bubbled over the platinum electrode and a redox reaction occurs with hydrogen
pt. is used because its inert, so it doesnt react and wont undergo reduction over hydrogen
Hydrogen ions are reduced to form hydrogen gas and hydrogen gas is
oxidised to produce hydrogen ions in solution.
The hydrogen half-cell is assigned a E° = 0.00 V
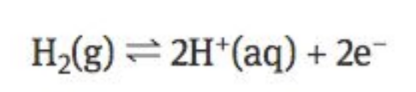
how can the SHE be used with a metal electrode?
depending on the reducing power of the metal electrode, the SHE will either be forced to act as the cathode or an anode when connected to the other half cell
this helps determine the reducing power of a different half cell compared to platinum
what is the SHE
the SHE is a reference electrode that scientists have agreed has an electrical potential of exactly 0V.
the components:
it uses H2 gas bubbling over a platinum electrode submerged in an acidic solution of H+ ions
purpose: It is used with other metal electrodes to determine their relative "reducing power"—essentially, how badly they want to give up or take electrons compared to Hydrogen.
how does do you determine the reducing power of a metal? how does this help create the standard electrode potential table?
How the Experiment Works
The Comparison: You connect a "mystery" metal (like the Zinc in your diagram) to the SHE using a wire and a salt bridge.
The "Push-and-Pull": The two sides fight over electrons.
If the metal is better at losing electrons than Hydrogen, it becomes the Anode (where oxidation happens).
If the metal is worse at losing electrons, the SHE is forced to act as the anode instead.
The Result: This allows us to rank every element on a "Standard Electrode Potential" table, showing which are the strongest at being oxidized or reduced.
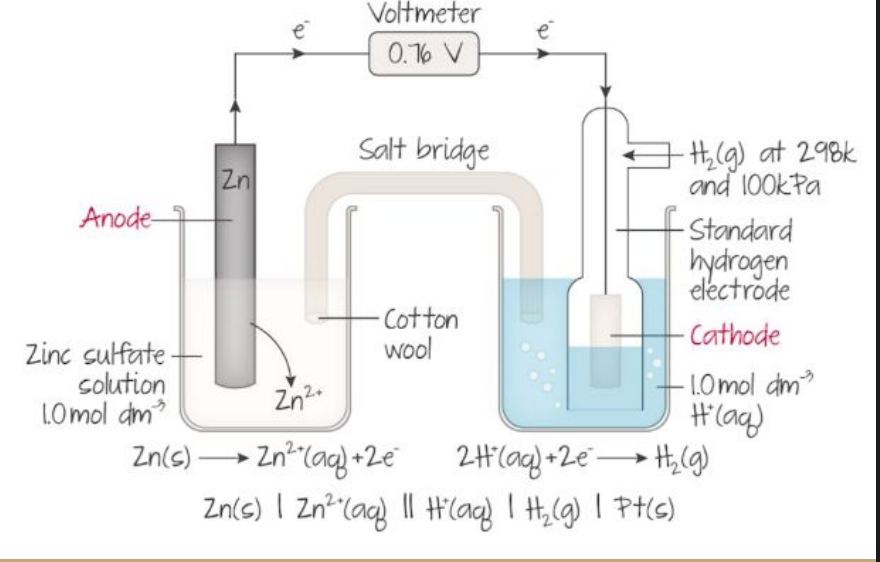
what is the voltmeter measuring in this photo?
the voltmeter is measuring the cell potential (Ecell) which is also known as the electromotive force (EMF)
standard electrode potential tables always lists values of _____
standard electrode potential tables always list values as reduction potentials
what is a reduction potential
what does a negative reduction potential tell us?
this is how much an atom wants to be reduced by gaining electrons
a negative reduction potential like Zn (-0.76) means that zinc is bad at gaining electrons compared to hydrogen
in fact zn would rather lose electrons (oxidation)
standard means its at standard conditions: 298K, 100KPa and 1 mol/dm3
why do we need a platinum electrode in SHE
Platinum (Pt): Used in the SHE because it is inert; it carries the electrons without reacting itself.
what does a + or - standard electrode potential tell you?
the standard electrode potentials reported are for reduction potentials
so the ability of an atom to gain electrons and be reduced
(+) values = wants to be reduced = very strong oxidizing agent
(-) values = dont want to be reduced, want to give e = very strong reducing agent
what is the electrochemical series
another name for the list of standard electrode potentials
its a list that ranks different species based on how much they want to be reduced
Why the Name Matters
Standard Electrode Potentials: This is the technical term for the specific voltage numbers assigned to each half-reaction.
Electrochemical Series: This is the name for the ordered list itself.
what is the SHE
its a platinum electrode submerged in a solution of 1 mol/dm3 of H+ aq ions
what happens at the SHE
at the hydorgen electrode, H2 gas is bubbled over the pt. electrode and a redox reaction occurs:
H+ are reduced to form H2 gas AND (equilibrium!) H2 is oxidised to produce H+
standard conditions
explain what happens if the half cell connected to the SHE is higher or lower on the electrochemical series

what is the SOA and SRA
SRA = higher on table, more negative = oxidized at anode
SOA = lower on table, more positive = reduced at cathode
what is the direction of electron flow when 2 half cells are connected?
when half cells are connected, the electrons flow from the half cell with the more negative SEP, Eº to the more positive Eº
the more negative Eº wants to give away electrons, oxidized = anode
the more positive Eº wants to get eelectrons, reduced = cathode
the difference between these 2 electrodes is called the standard electrode potential of the cell Eºcell = Eºcathode - Eºanode
aka. Eºcell = Eºred - Eºox
Ecell is also known as electromotive force
The difference in standard electrode potentials of the half-cells in a
galvanic (voltaic) cell
○ Measured in volts (V)
how is Eºcell related to gibbs?
the cell potential is an indicator of whether a redox reaction will occur spontaneously
Ecell is related to ∆Gº (the standard change in gibbs free energy) by the following equation
∆Gº = -nFEº
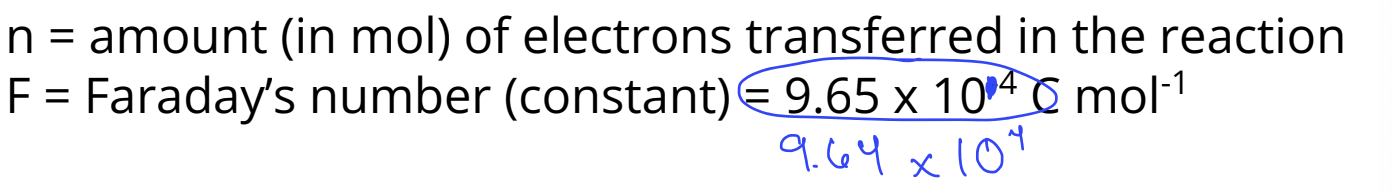
signs of ΔG° & E°?
ΔG° = - nFE°
Note the negative sign! This means that ΔG° & E° will have
opposite signs.
units of cell potential Eº
V (volts)
1 V = J/C
when do the coefficients affect the calculations

electrolysis
the process by which an ionic compound is broken downinto its constituent elements using electricity
*keep diatomic elements in their diatomic states

these reactions are examples of what
electrolysis
what are some uses of electrolysis
electrolysis is very important industrially:
its used to
extract metals from its ores, aka purify compounds
electroplating = adding a metal coating to a solid substrate
manufacturing of NaoH
hydrogen fuel purification
molten state
liquid
components of an electrolytic cell
molten or aqueous electrolyte
battery (power source)
2 electrodes, usually inert/non reacting → made of graphite or platinum
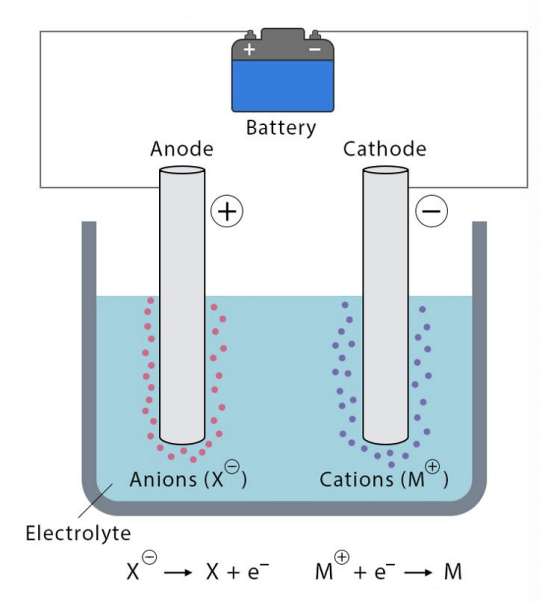
in an electrolytic cell, what are the charges on the:
anode
cathode
Hence what ions do they attract? why is this important?
anode = +
cathode = -
the anode will attract anions which have e to lose. this is important because oxidation (loss of e) occurs at the anode
the cathode will attract cations which can gain e. this is important because reduction occurs at the cathode
do electrolytic cells use a spontaneous or nonspontaneous redox reaction
E and G values?
electrolytic cells use a nonspontaneous reaction because it requires energy → a continous external electrical current from the battery
G>0 => Ecell <0
direction of electron flow?
anode → cathode
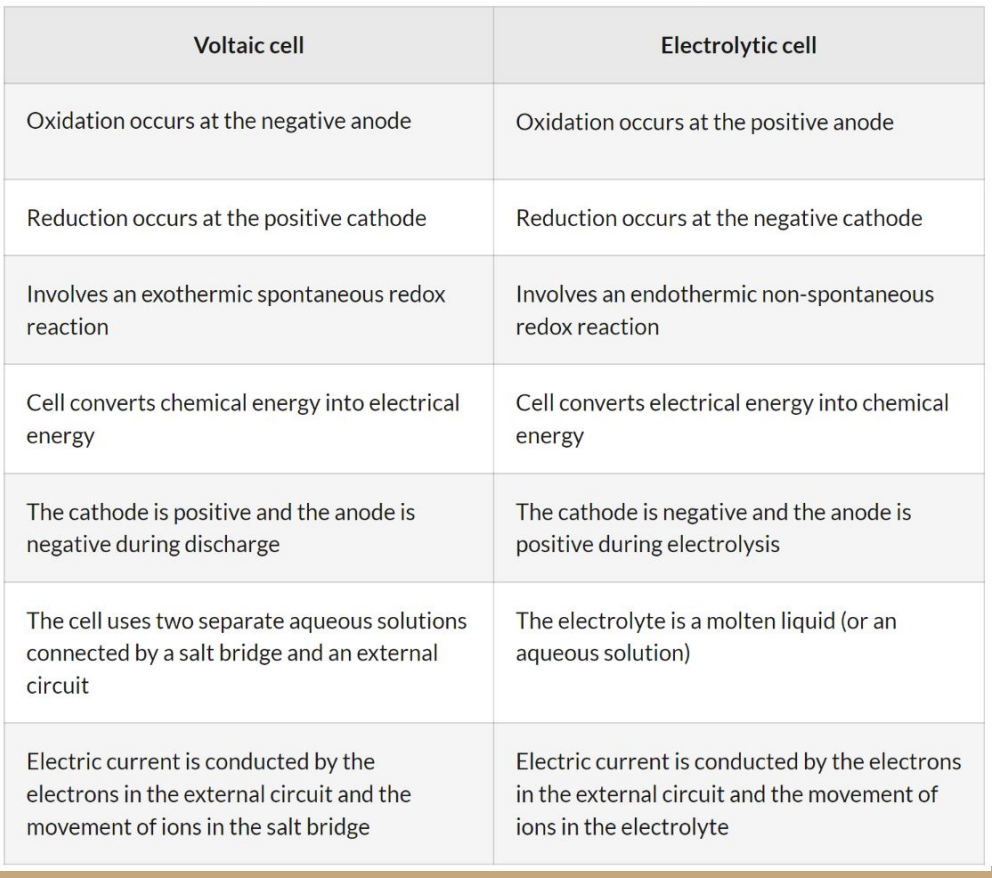
compare a voltaic cell with an electrolytic cell
electrolysis of water:
performed using____
overall chemical reaction
reduction and oxidation half reaction

what is added to the electrolysis of water to increase conductivity
H2SO4, sulfuric acid
what happens to the pH at the anode? cathode?
at the anode, the pH decreases as H+ ions are produced. at the cathode, the PH increases as OH- ions are produced
how do fuel cells generate electrical energy
electrical energy is derived directly from a chemcial reaction without the need for multiple transformations of energy like it does in the combustion of fossil fuels, which makes it more efficient compared to the combustion of fossil fuels.
A fuel cell consists of what?
a fuel cell uses a spontaneous exothermic chemical reaction that is divided into 2 regions (not half cells!!!)
these 2 half reactions occur separately in 2 different locations, separated by a membrane but connected by a wire
the electrons that are oxidized off the species in one half reaction travel across the wire to reduce the species on the other half
the movement of electrons creates electricity
methanol fuel cells
overall and half reactions

fuel, products and reactants on a fuel cell diagram?
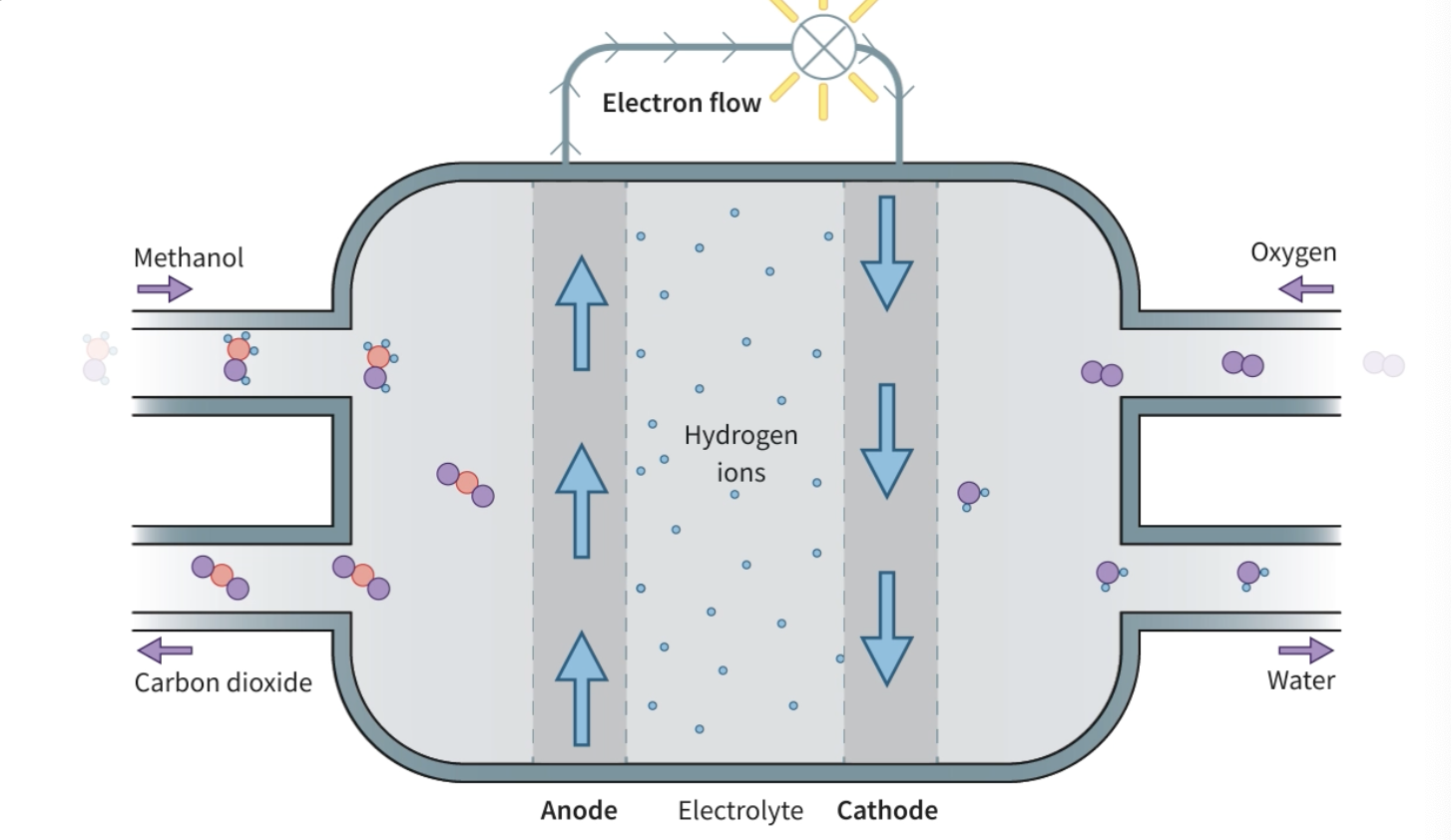
fuel cells are supplied with a ____ flow of ____
fuel cells are supplied with a continuous flow of reactant molecules, which are pumped into the cell. Unlike stand-alone batteries, fuel cells do not need to be charged over long periods. Methanol fuel cells can be refilled from a liquid methanol reservoir, which makes them convenient to use.
how much energy does the combustion of methanol and H2 release
711 KJ
286 KJ
environmental impact of hydrogen fuel cells
even though only H2O is produced as a result of hyrogen fuel cells, they still can have a negative impact on the environment:
takes a lot of energy to obtain hydrogen gas because most H2 is in the form of water, so to remove H2 from water it needs to be electrolysed
H2 can also be produced via coal gasification
this is where coal is heated in the presence of steam to produce H2 and CO, but CO is the same atmospheric pollutant as the incomplete combustion of fossil fuels and it also takes a lot of energy ebcause it occurs at high temperatures and pressures
how do rechargeable batteries compare to primary cells
rechargeable batteries use chemical reactions to provide energy to power devices just like primary cells, but it can be used more than once by recharging
recharching is when the chemical reaction is reversed by the use of an electrical current to drive the reaction back to its orginal state (turn products back into reactants)
what is battery life?
battery life is a measure of the time that the battery will continue to provide energy
what are the components of a lithium ion battery made of LiCoO2 (lithium cobalt oxide)
cathode = made of a lithium metal oxide like LiCoO2 (lithium cobalt oxide)
anode = made of lithium atoms absorbed into a lattice of gaphite
electrolyte = non aq solution (no water sol!) containing Li+ ions which flow between the cathode and anode during charging and discharging
when the lithium battery is discharged waht half reactions take place
what happens with Li+ and e- when battery is recharged?

Note that when being recharged, the two half equations above are reversed. During charging, lithium ions flow from the cathode to the anode through the electrolyte. This process stores energy in the battery. During discharging, the lithium ions flow back from the anode to the cathode through the electrolyte, generating an electric current that can be used to power devices.

