VBSC 402W
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
Oxyurids – Pinworm Morphology
Oxyurids – Pinworm Morphology
Tail – sharp, pin
Esophageal bulb
Lips – 3
Male – 0 or 1 spicule
Flat egg – 1 side
Oxyurids – Pinworms
Oxyuris equi –
Enterobius vermicularis –
Enterobius gregorii –
Skrjabinema ovis –
Skrjabinema caprae –
Oxyurids – Pinworms
Oxyuris equi – horse
Enterobius vermicularis – human
Enterobius gregorii – human
Skrjabinema ovis – sheep
Skrjabinema caprae – goat
Oxyuris Equi
___ life cycle, ___l transmission
PPP
Migrate to ____- reproduction
Lay eggs
Unique:
Infective stage –
Oxyuris Equi
Direct life cycle, fecal oral transmission
Definitive host: horse
PPP 5 months
Migrate to large intestine - reproduction
Lay eggs perianal area, carried out with feces
embryonate and L1 --> L3 in environment
+ Retroinfection
Unique: because eggs are sticky, eggs may hang out in environment while staying on the perineum. Can crawl back in, go up to the colon, and go back through sexual reproduction
Infective stage – L3, in egg
Enterobius vermicularis
Definitive host:
Infective stage –
Diagnostic stage –
Enterobius vermicularis
Direct fecal-oral life cycle
Definitive host: human
Infective stage – L3, in egg
Diagnostic stage – Egg (in perianal folds)
scotch tape test - medical/clear tape, touch perineum and put on microscope
Oxyurids – Pinworm Disease Pathogenesis
Severe –
Oxyurids – Pinworm Disease Pathogenesis
Most asymptomatic
Pruritis ani (itchy perineum)
Severe – colonic mucosa inflammation
Susceptible to most anthelmintics
Spiruids
2 Suborders
Suborder Camallanoidea
Superfamily Camallanoidea
Superfamily
Suborder Spirurida
10 Superfamilies – all animals, humans, zoonotic
1. Gnathostomatoidea –
2. Physalopteroidea – vertebrate stomach/intestinal worm
3. Rictularoidea
4. Thelazoidea
Thelaziidae - Eye/lacrimal/conjunctival worm
Pneumospiruridae – carnivore lung parasite
5. Spiruroidea –
6. Habronematoida – equine stomach, cutaneous Habronemiasis
7. Acuarioidea – birds
8. Aproctoidea
9. Diplotriaenoidea
10. Filaroidea –
Spiruids
2 Suborders
Suborder Camallanoidea
Superfamily Camallanoidea
Superfamily Dracunculoidea – Guinea Worm
Suborder Spirurida
10 Superfamilies – all animals, humans, zoonotic
1. Gnathostomatoidea – carnivore stomach worm
2. Physalopteroidea – vertebrate stomach/intestinal worm
3. Rictularoidea
4. Thelazoidea
Thelaziidae - Eye/lacrimal/conjunctival worm
Pneumospiruridae – carnivore lung parasite
5. Spiruroidea – esophageal/stomach worm
6. Habronematoida – equine stomach, cutaneous Habronemiasis
7. Acuarioidea – birds
8. Aproctoidea
9. Diplotriaenoidea
10. Filaroidea – human, animal filarial worm! Public health concern!
Spiruida, Superfamily Dracunculoidea
Spiruida, Superfamily Dracunculoidea
Largest nematode – 2.5 ft female (Guineaa worm, females larger)
No lips, underdeveloped buccal area
SQ tissues of carnivores, humans (but oral exposure)
Guinea Worm
Dracunculus medinensis (Guinea Worm)
Infectious stage –
Diagnostic stage –
Life cycle:
No ___. Release ___
___consumed by ___
Larval stage molts to ___
Stays in ___, ___with ___
___in ___, ___, ___released
___ to ___
PPP
Dracunculus medinensis (Guinea Worm)
Infectious stage – L3
Diagnostic stage – Adult worm in blister
Ovoviviparous
Indirect life cycle
Intermediate host – Copepod
Definitive host: human
Life cycle:
No eggs. Release L1 filariform directly from subcutaneous blister bursting when exposed to water
L1 consumed by copepod
Larval stage molts to L3 larvae (infective form)
Stays in copepod, humans consume copepod with L3 larvae in water (copepods are too small to be visible)
L3 in stomach, copepod digested, L3 released
L3 migrate out of stomach and intestinal wall to lower leg, mature and create a blister
PPP 12-14 months
Spiruida – Dracunculoidea – Guinea Worm signs
Treatment -
3 conditions necessary for disease
1.
2.
3.
Challenge is
Spiruida – Dracunculoidea – Guinea Worm
2ndary bacterial infections
Aberrant migration
Dying nonemergent worms
Treatment - must physically remove the adult worm across week (do not come out on their own). No anthelmintics or treatments (killing 2.5 ft worm would cause hypersensitivity immune response, hyper shock, overwhelming immune system)
3 conditions necessary for disease
1. Infected individual in water
2. Copepods present
3. Drink water
Challenge is 12-14 month PPP. Have to wait full lifecycle to determine an area is negative
Spiruida – Dracunculoidea – Guinea Worm Control
Control strategies
___ - biggest progress
Spiruida – Dracunculoidea – Guinea Worm Control
Slow removal
No medications
Control strategies
Education
Safe drinking water supply
Clean water from copepods (does not remove other bacteria/protozoa). Copepods are not visually able to be seen but big enough that simple filtration eliminates (straw, weave) - biggest progress
Early case containment
Vector control (not elimination)
Spiruida, Suborder Spirurida
L1 –
Esophagus -
Some oviparous (__) some ovovivparous (__)
Spiruida, Suborder Spirurida
Lots of species and variation!
Intermediate host – Arthropod
Indirect lifecycle
Deep tissue/intestinal parasites
L1 – dorsal cuticular hooks/spines
Pseudolabia – 2 lateral lips
Esophagus - anterior muscular, posterior glandular
Some oviparous (thick-shelled) some ovovivparous (filaroidea)
Suborder Spirurida, Superfamily Gnathostomoidae
Females – 11-54 mm length
Parasite
Suborder Spirurida, Superfamily Gnathostomoidae
2 lateral lips
Swollen head – 4 circles of spines
Fat spiny body
Females – 11-54 mm length
Parasite of carnivores
Suborder Spirurida Superfamily Gnathostomoidae
Life Cycle
Indirect lifecycle
1st Intermediate host –
2nd intermediate host –
Paratenic host –
Human -
Definitive host –
Infective stage –
Diagnostic stage –
Suborder Spirurida Superfamily Gnathostomoidae
Life Cycle
Indirect lifecycle
1st Intermediate host – copepod
2nd intermediate host – fish, amphibian
Paratenic host – snakes, birds, humans, pigs
Human - dead end host
Eat IH/PH —> aberrant migration, dormant, abscess (visceral larval migrans)
Definitive host – carnivore (dog, cat, pig)
Oviparous. egg hatches in water, L1 in water is eaten by copepod (shallow water), molt to L2
2nd intermediate ingest copepod, mature and ensyst in smaller fish/amphibian as L3
Definitive host (usually dog, sometimes cat) eat, mature and sexually reproduce
Infective stage – L3
Diagnostic stage – L3
Suborder Spirurida, Superfamily Spirurodea
Spirocerca lupi
Suborder Spirurida, Superfamily Spirurodea
Spirocerca lupi
6 lips, developed buccal capsule
Stout worm
Eggs – embryonated when laid
Esophageal worm
Suborder Spirurida, Superfamily Spirurodea
Spirocerca lupi
Adults – esophagus
Eggs – shed in feces – embryonated!
Indirect lifecycle
Intermediate hosts – dung beetles
Paratenic hosts -birds, reptiles, mammals
Definitive host – dog
Spirocerca lupi Life cycle
Esophagus –
Eggs -
Spirocerca lupi Life cycle
dung beetles ingest egg, egg develops from L1-L3, dog ingests dung beetle with L3 larva
L3 enter, adults nodulate in esophagus, sexual reproduction, will release eggs
Paratenic may also ingest dung beetle with L3, paratenic host then eaten by definitive
Juveniles penetrate stomach wall to gastric artery to aorta
Esophagus – reproduction
Eggs - lumen
Suborder Spirurida, Superfamily Spirurodea, Spirocerca disease
Unique to Spirocerca lupi -
Suborder Spirurida, Superfamily Spirurodea, Spirocerca disease
Aberrant migration
Vomiting – stomach wall lesions
Aortic lesions – aneurysms, hemorrhage
Spondylosis, hypertrophic pulmonary osteoarthropathy, spondylitis
Unique to Spirocerca lupi - create spondylitis of the skeletal system via an immune system response, cause most common clinical sign bringing in dogs (limping, pain). "arthritic lesions"
Esophageal sarcomas
Europe, Africa, and Asia mostly. Sometimes US
Ascarid Morphology
Ascarid Morphology
Large stout nematodes – 45 cm!
3 lips surround small buccal capsule
No copulatory bursa
Eggs w thick shell, last long in environment
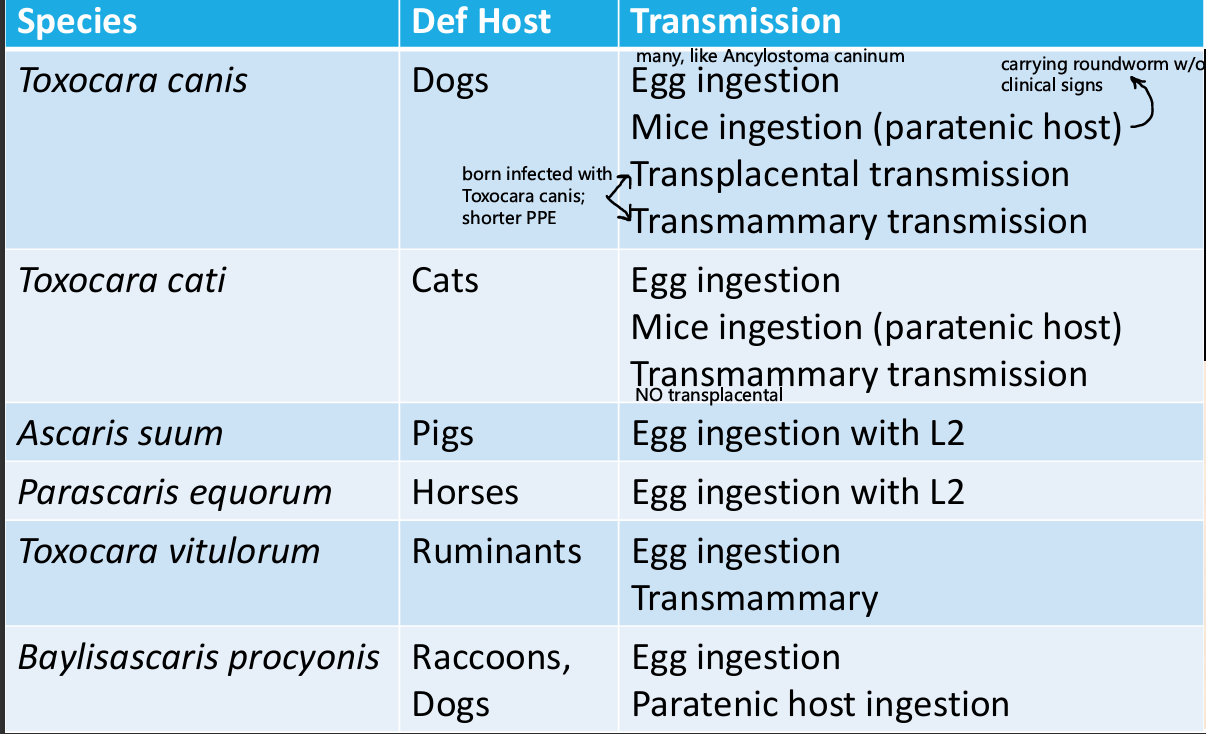
Toxocara spp.
Modes of Infection
Direct ingestion of eggs (Egg + ___) –
egg hatches in ___, migrate to ___, ___, ___(__), ___ (adult)
PPP canis –
PPP cati –
Transplacental –
__ in Mom___, ___, ___(__),___ (adult)
Transmammary –
__ in Mom to___, molt to __in___, ___
Ingestion of paratenic host –
Larva ___
L__ ___, ingested by ___/___
Toxocara spp.
Modes of Infection
Direct ingestion of eggs (Egg + L3) – T. canis, cati
Fecal excretion of unembryonated egg, embryonate, mature to L3 larvae in embryonated egg
egg hatches in stomach, migrate to liver, trachea (L3), cough/swallow, stomach (L4), small intestine (adult)
PPP canis – 14-28 days
PPP cati – 56 days
Transplacental – T. canis only
L2 in Mom crosses placental to fetal liver, trachea (L3), cough/swallow, stomach (L4), small intestine (adult)
Transmammary – T. canis, cati
L2 in Mom to mammary gland, molt to L3 in mammary gland, L3 ingested
Ingestion of paratenic host – T. canis, cati
Larva hatch and get into tissue
L3 encysts in tissues of mouse, ingested by dog/cat
Toxocara spp.
Diagnostic stage –
Also
Infective stage –
take ___ to mature/embryonate to __ in environment
PPP –___ (eggs)
PPP –___ (placental/mammary)
In humans; diagnostic stage is ___ ; infective stage is i___ or ingestion via___
In people: ___ host - ___ , larva___ ,
Toxocara spp.
Diagnostic stage – Eggs in feces
Also may see adult worms in feces or vomit
Infective stage – embryonated eggs
take 2-4 weeks to mature/embryonate to L3 in environment
PPP – 4-5 weeks (eggs)
PPP – 2-3 weeks (placental/mammary)
In humans; diagnostic stage is seeing larva in tissue biopsy of organs/conjunctiva; infective stage is ingestion of embryonated eggs or ingestion via undercooked paratenic
In people: aberrant host - visceral larval migrans, larva get into stomach, intend to migrate to liver and trachea to get to SI, instead get stuck in organs (liver, brain, skin, eyes)
Parascaris equorumi
PPP -
Parascaris equorumi
Foals <1 year
Not associate with VLM in humans
PPP - >10 weeks
Immature eggs in feces, eggs develop to infective stage (embryonate) in environment in 2 weeks, remain infective for years. Swallow, go to liver, go to trachea, cough up and swallow, small intestine
Ascarid Disease Pathogenesis
Contamination with eggs!
Eggs extremely resistant to ___
Eggs survive
Abnormal hosts –
Control:
Adult worms –
Inflammatory response in ___ – related to ___ and ___
Ascaris pneumonitis –
Ascarid Disease Pathogenesis
Contamination with eggs!
Eggs extremely resistant to disinfectants
Eggs survive months-years in soil, fomites
Abnormal hosts – could transport eggs
Control: Clean feces (eggs not infective until embryonated & matured), Anthelmintics targeting mothers (for Toxocaria canis and cati), Baylisarcarcis: stop pets from hunting, cook food thoroughly, wash hands around potential exposure in environment
Minimal GI damage to mucosa
Malnutrition, intestinal obstruction, Intestinal rupture
Adult worms – vomit or fecal excretion
Inflammatory response in liver, lungs – related to number of larva and organ affected
Milk spots – 2ndary fibrosis
white spots on liver, called that as these are young animals on milk. mostly in commercial and wild swine
Ascaris pneumonitis – inflammation in alveoli
Visceral Larval Migrans
Humans –
Accidental egg ingestion
Larvae
___ is Susceptible!
Treatment difficult –
Control parasite sources –
Visceral Larval Migrans
Humans – aberrant host of intestinal roundworm larvae
Accidental egg ingestion
Toxocara canis
Baylisascaris procyonis
Larvae burrow through small intestine; enter circulatory system
Any Organ is Susceptible!
Eye – blindness
Liver
Brain – neurologic problems
Treatment difficult – surgical removal
Control parasite sources – environment!
Filaroidea superfamily
____ reproduction
Filaroidea superfamily
All vertebrates except fish
Reduced buccal capsule, lips
Spiral flexure – male
Indirect life cycle
Intermediate host – blood sucking arthropod
Ovoviviparous reproduction
Microfilaria - pre-juveniles free in blood/lymph
Genus Onchocerca
Microfilaria – ___ & ___
Microfilaria –
Adults –
Parasite (adult mother) releasing microfilaria is___ and ___ dependent, releasing microfilaria ___ and ___
Genus Onchocerca
Rudimentary – no lips, spines, ring around mouth
Non-divided esophagus
No caudal alae
Microfilaria – sheathed & unsheathed
Microfilaria – time dependent
Adults – connective tissues, heart, arteries, body cavities
Parasite (adult mother) releasing microfilaria is time-dependent and temperature dependent, releasing microfilaria at times mosquitos are more active (dusk and dawn) and season
1. Onchocerca volvulus
___ life cycle
Microfilaria – ___– can live___
Adults – ___ - can live in ___for ___
Infective stage –
Diagnostic stage —
Life cycle
___-___ host, injects ___ into ___ . ___ hangs around in ___ (especially ___ ). Cause ___ from___ . Adults mature, sexually reproduce, release ___ , remain in ___ until ___ takes a bloodmeal. ___ --> ___ --> ___ --> ___ inside ___ host (___ )
1. Onchocerca volvulus
Indirect life cycle
Intermediate host – blackfly
Ovoviviparous
Microfilaria – unsheathed– can live 2 years!
Adults – SQ tissues - can live in SQ for 15 years!
Infective stage – L3
Diagnostic stage – microfilariae in skin biopsies
Life cycle
Blackfly - intermediate host, injects L3 larvae into human. L3 hangs around in subcutaneous tissues (especially eyes). Cause nodules from immune system. Adults mature, sexually reproduce, release microfilaria, remain in subq until blackfly takes a bloodmeal. Microfilaria --> L1 --> L2 --> L3 inside intermediate host (blackfly)
Onchocerciasis
associated with ___
___, ___. ___
___ – needs ___, ___
___ – needs ___, ___
Onchocerciasis –___
___ – benign ___, can cause ___
___– ___
___/Decreased ___– ___ invade ___, ___
Onchocerciasis
associated with rivers
Warm, wet. Central Africa
Black fly larva – needs clean, fast-moving water
Black fly adult – needs stream-side vegetation, high humidity
Onchocerciasis – River Blindness
Not fatal
Onchocercomas – benign collagenous SQ nodules, can cause elephantiasis
Dermatitis – Dying microfilariae in skin
Blindness/Decreased vision – microfilariae invade cornea, immune response damages retina
Knowing the lifecycle of Onchocerca volvulus, what would be a good control measure?
Knowing the lifecycle of Onchocerca volvulus, what would be a good control measure?
Mass drug administration/community-directed
Cleaning riverbanks of vegetation (destroy breeding ground)
Insecticide sprays
Community education
2. Wuchereria bancrofti & Brugia malayi
___ life cycle
Intermediate host – ___
___
Microfilaria – ___
Adults – ___
Microfilaria – shed into ___
Life cycle
___ inject ___ , ___ get into ___ and get into ___ . Adults sexually reproduce, release ___ into ___ and ___ ___ . ___ ingests ___ -->___ --> ___ in ___ ___ . Same for ___ and ___
2. Wuchereria bancrofti & Brugia malayi
Indirect life cycle
Intermediate host – mosquito
Ovoviviparous
Microfilaria – sheathed
Adults – lymphatic ducts
Microfilaria – shed into circulation
Life cycle
Mosquito inject L3, L3 get into subq and get into lymphatics. Adults sexually reproduce, release microfilaria into lymphatic and peripheral blood. Mosquito ingests microfilariae --> L1 --> L3 in intermediate mosqutio. Same for W. bancrofti and B. malayi
Wuchereria –
Both ___ and ___
Disease
1. ___ phase
2. ___ phase
3. ___ phase –
Control w: ___ , ___ , ___
Wuchereria – Elephantiasis
Both W. bancrofti and B. malayi
Lymphatic filariosis
Obstruction of lymphatic vessels/ducts. Balance btwn interstitial and circulating fluid. Fluid moves out of lymphatic vessels into tissues
Disease
1. Asymptomatic phase
2. Inflammatory/acute phase
3. Obstructive/chronic phase – host inflammatory immune response to adult worms
Even if parasite is killed, damage is permanent
Control w: bednets, insecticides, MDA,
3. Genus Dirofilaria
Microfilariae –
Adults –
3. Genus Dirofilaria
Thin worms
Simple mouth – no lips coiled tail
Caudal alae (male) – large papillae,
Non-divided esophagus
Microfilariae –unsheathed
Adults – connective tissues, heart
Dirofilaria immitis
Adults can live
Lifecycle of ___ from exposure (___ PPP)
Life cycle
Intermediate host - ___
Definitive host - ___
Infective stage - ___
Diagnostic stage - ___
Humans - ___
___ injects ___, migrate through wound into ___, ___ mature in ___ in ___ , ___ migrate through ___ and mature to ___ in ___ ___ (___ ). Immature migrate to ___ (early as___ post infection). Sexual maturity in___
___ to reproduce in blood
Dirofilaria immitis
Adults can live 5-7 years!
Lifecycle of 8 months from exposure (8 month PPP)
Life cycle
Intermediate host - mosquito
Definitive host - dog
Infective stage - L3 larvae
Diagnostic stage - adults in pulmonary arteries
Humans - aberrant host, other dirofilaria sp do not complete life cycle in humans, become cutaneous granulomas. L3 stuck in skin
Mosquito injects infective L3 larva, migrate through wound into dog host, L3 mature in subcutaneous tissues in 1-3 days, L4 larvae migrate through tissues and mature to sexually immature adults in subq 50-70 days (2 months). Immature migrate to heart and lungs (early as 70 days post infection). Sexual maturity in pulmonary artery.
Pulmonary artery takes deoxygenated blood form heart to lungs to be oxygenated
6 months to reproduce in blood
Dirofilaria immitis - Epidemiology
>60 ___species
Development ___dependent
___ to begin
Stops at __
Reservoir population –
___ in 2010 —> dec incidence
2019 forward higher incidence -
Dirofilaria immitis - Epidemiology
>60 mosquito species
Development temperature dependent
2 weeks >80 F to begin
Stops at <57 F
Reservoir population – wild canids
Heartworm preventatives developed in 2010 —> dec incidence
2019 forward higher incidence - Longer breeding seasons for mosquito, living longer, etc due to warming. More traveling
Dirofilaria immitis - Epidemiology
1.
2.
3.
4.
Dirofilaria immitis - Epidemiology
1. Susceptible host population
2. Stable reservoir of the disease
3. Stable mosquito population
4. Climate supportive of development
Heartworm disease
Clinical Signs – ___, ___
___disease
___ – ___
___ – ___(___), ___(___), ___(___)
Dirofilaria immitus –
___disease –
I
___ – ___, ___, ___, ___
Heartworm disease
Clinical Signs – coughing, exercise intolerance
Cardiopulmonary disease
Pulmonary hypertension – right heart enlargement
Heart failure – ascites (abnormal buildup of fluid in the abdomen), hydrothorax (fluid in the pleural space surrounding the lungs), hydroperitoneum (fluid in peritoneum)
Dirofilaria immitus – Caval Syndrome
2ndary hepatic disease – liver dysfunction
Acute disease of extreme adult worm burden
Increased venous pressure in liver
RBC fragility – hemolysis, anemia, hemoglobinuria (free hemoglobin in the urine), bilirubinemia (biliruibin in bloodstream —> jaundice)
Dirofilaria immitus – Diagnosis
Immunologic tests – ___
Dirofilaria immitus – Diagnosis
Clinical signs
Radiographic changes
Cardiac changes
Microfilarial tests – Knott test
Immunologic tests – ELISA
Dirofilaria immitus Treatment & Prevention
___– 1st treatment
1. Melarsomine dihydrochloride – “___”
2. Thiacetarsemid – “___”
___- 2nd treatment
Prevention – test 1st
1. ___– monthly
2. ___– monthly
3. ___ – daily
Dirofilaria immitus Treatment & Prevention
Surgical removal
Cannot just treat and kill, would overwhelm immune
Adulticide– 1st treatment
1. Melarsomine dihydrochloride – “immiticide”
2. Thiacetarsemid – “carparsolate”
Microfilarial- 2nd treatment
Prevention – test 1st
1. Ivermectin – monthly
2. Milbemycin oxime – monthly
3. Diethyllcarbamazine (DEC) – daily