Biosynthesis of Neurotransmitters
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms


abnormal things found
Homovanillate is a metabolite of dopamine.
Vanillylmandelate is a metabolite of (epi/norepi)
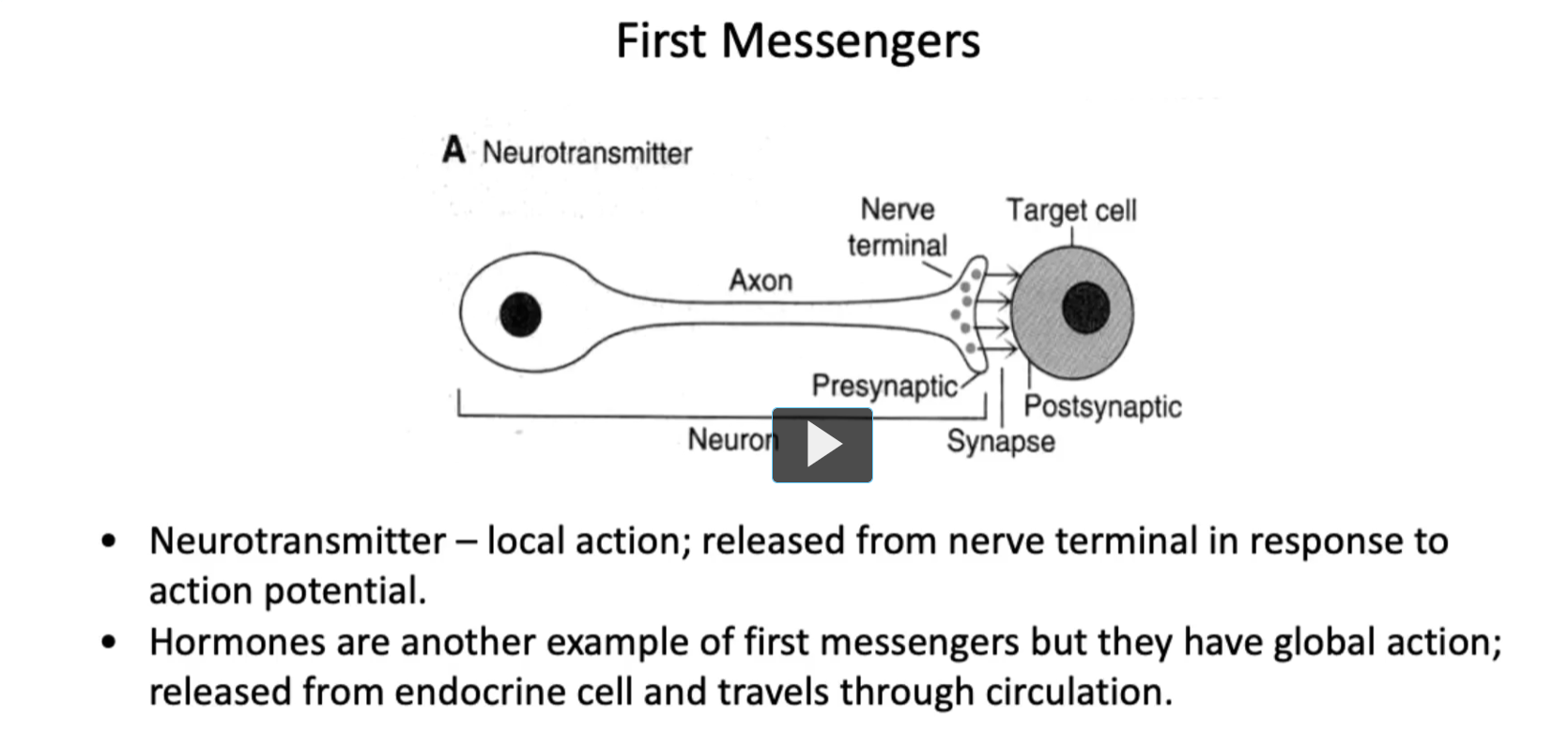
First Messengers
neurotransmitters are considered the local first messengers in the nervous system.
A Neurotransmitter
(A diagram labeling: Neuron, Axon, Nerve terminal, Presynaptic, Synapse, Postsynaptic, Target cell)
• Neurotransmitter – local action; released from nerve terminal in response to action potential.
Neurotransmitters specifically act at synaptic junctions. Therefore, understanding this distinction is key when evaluating drug targets and the neurophysiology.
• Hormones are another example of first messengers but they have global action; released from endocrine cell and travels through circulation.
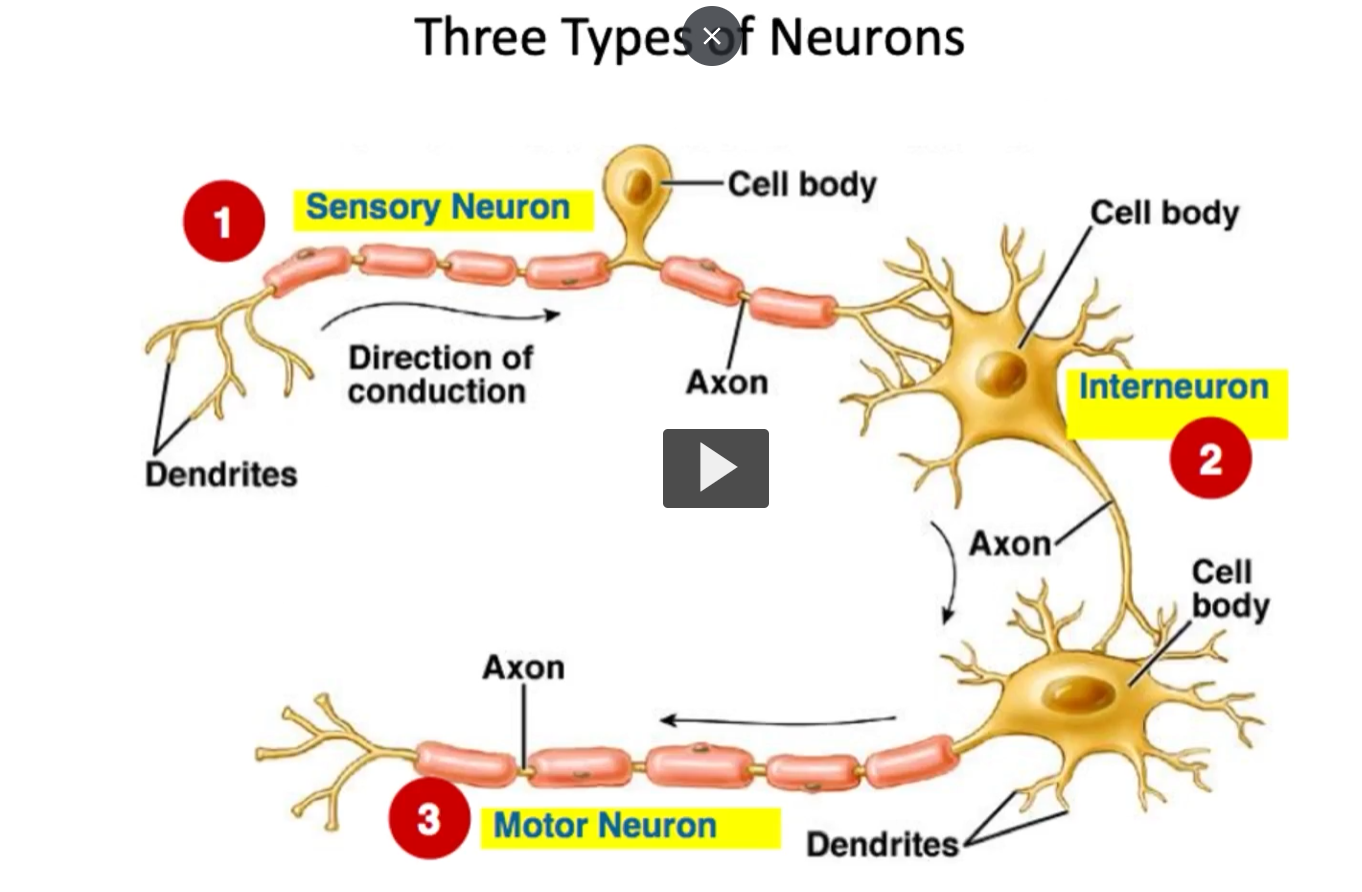
Three types of neurons
sensory neuron (afferent):
motor neuron (efferent)
Interneurons
Each of these play a unique role in signal transmission and many produce different neurotransmitters based on their function and target structure.

Basic types of Receptors
There are two types of receptors that we will be discussing. First, there is ionotropic, and second, metabotropic.
First, ionotropic receptors, such as those for glutamate or GABAa directly open ion channels “ionotropic”. Ionotropic are also known as ligand-activated ion gates.
• Ligand activated ion gates. (steps)
1. Sodium or calcium entry depolarizes (promotes action potential).
2. Chloride influx hyperpolarizes (leading to inhibition of action potential).
• Metabotropic receptors.
Some examples are the muscarinic acetyl-choline receptors.
-Metabotropic receptors work through second-messengers and influence gene expression or metabolism.
– Metabotropic receptors also alter the enzyme activities inside cells. Like the kinases.
General Principles of neurotransmitter biosynthesis
• The process of neurotransmitter synthesis is Neuron specific (through genes) – neurons express gene products for biosynthesis.
• Each neuron expresses the relevant enzymes are synthesized and transported to nerve terminal
• Raw materials (amino acids) are transported to or taken into nerve terminal by transporter proteins
• Occurs in presynaptic terminals
• Pool of neurotransmitter loaded into synaptic vesicles by specific transport proteins, to be exported through the process of exocytosis.
It is important to remember that peptide neurotransmitters made like any other protein
Peptide neurotransmitters are made using the normal protein synthesis machinery of the cell, just like any other protein (enzymes, receptors, etc.).
Here’s what that means step-by-step:
1. Made in the cell body (not the nerve terminal)
DNA → mRNA (in the nucleus)
mRNA → protein (on ribosomes in the rough ER)
2. Synthesized as a “prepropeptide”
They start as a large inactive precursor protein called a prepropeptide
Then it gets cut and modified → becomes the active neurotransmitter
3. Processed in Golgi and packaged
Modified in the Golgi apparatus
Packaged into vesicles
4. Transported down the axon
Vesicles travel from the cell body → nerve terminal (via axonal transport)

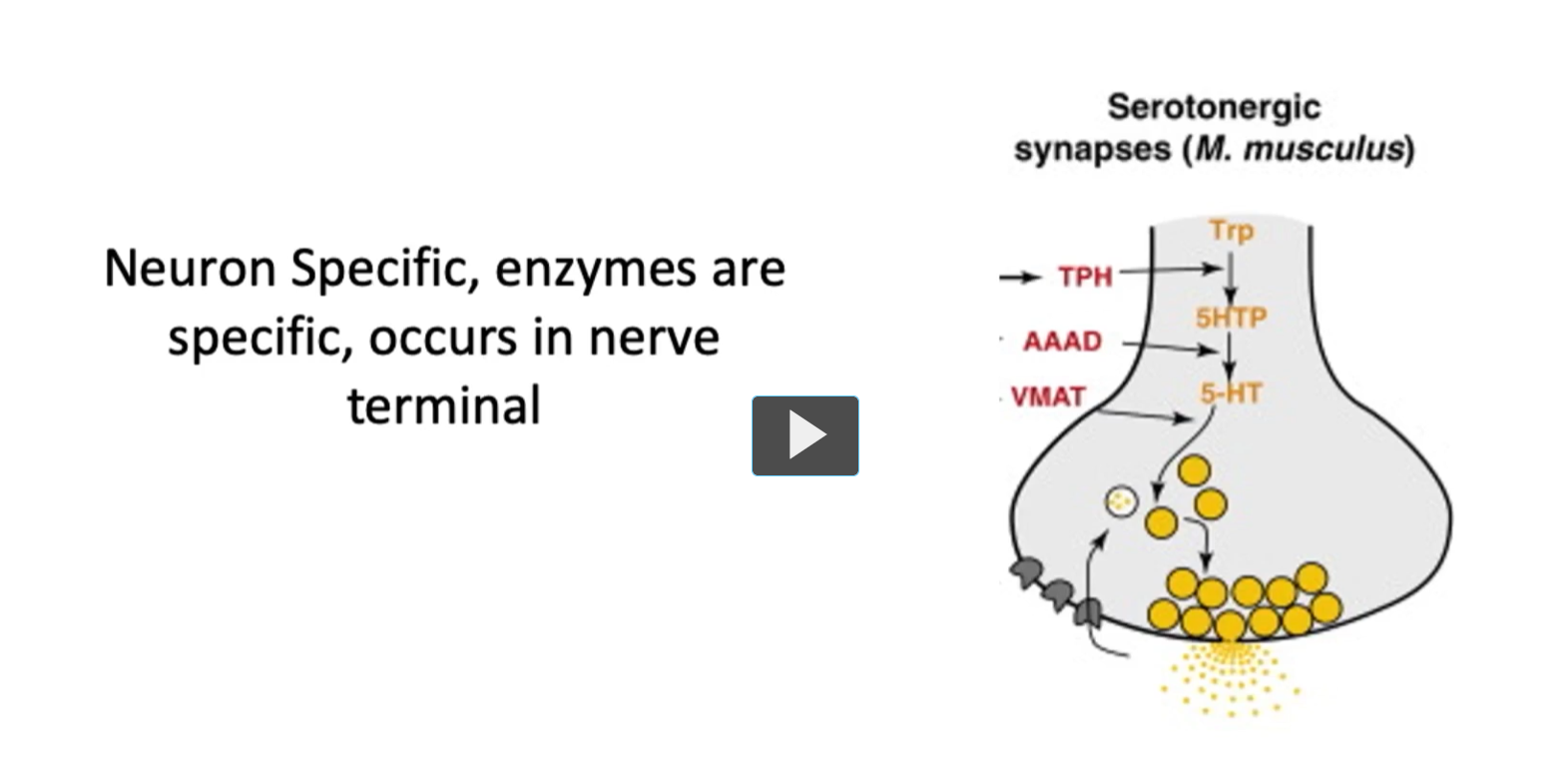
This concept of neural specific enzymes reinforces the fact that biosynthesis (of neurotransmitters) occurs locally within the pre-synaptic terminal, as we can see in this figure.
Enzymes (involved in neurotransmitter production) are synthesized in the soma and transported down to the axon of the neuron.
This compartmentalization will ensure that each neurotransmitter are made and released precisely when and where they are needed.
Biogenic Amines
Biogenic amines are small neurotransmitters derived from amino acids. The word breaks down as:
Bio = life
Genic = produced
Amine = contains an amino group (–NH₂)
So: “biologically produced amine neurotransmitters.”
• Five serve as neurotransmitters.
– Catecholamines (from tyrosine) (examples of catecholamines: dopamine, epinephrine, norepinephrine)
– Histamine (from Histidine)
– Serotonin (from tryptophan)
• Derived from tyrosine, histidine, or tryptophan
• Biogenic amines are important in a wide range of behaviors. Many drugs target synthesis, receptor binding/action, or catabolism.
Many psychiatric and neurological medications target the either the synthesis or degradation of these neurotransmitters.

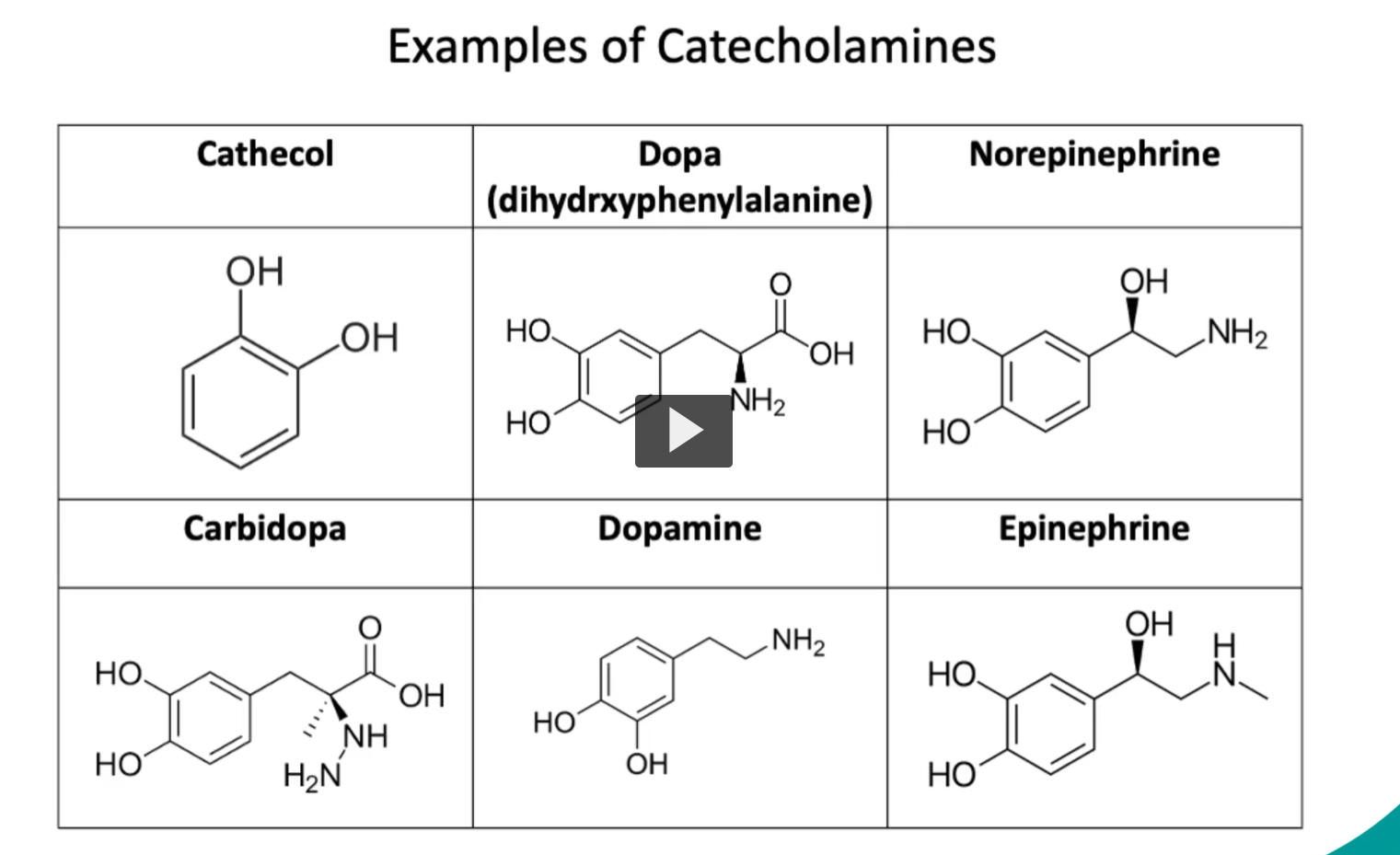
Synthesis of catecholamines (overview)
• Tyrosine → DOPA → dopamine → norepinephrine → epinephrine
-These neurotransmitters share a common synthetic pathway, from the amino acid Tyrosine.
• Key enzyme: tyrosine hydroxylase
Tyrosine hydroxylase catalyzes the conversion of tyrosine → DOPA (L-DOPA).
It adds a hydroxyl group (–OH) to tyrosine:
Tyrosine→ (tyrosine hydroxylase) L-DOPA
Uses O₂ (oxygen)
Requires a cofactor (tetrahydrobiopterin, BH₄ — high-yield detail)
• Key cofactors: pyridoxal phosphate (vitamin B6) for, vitamin C
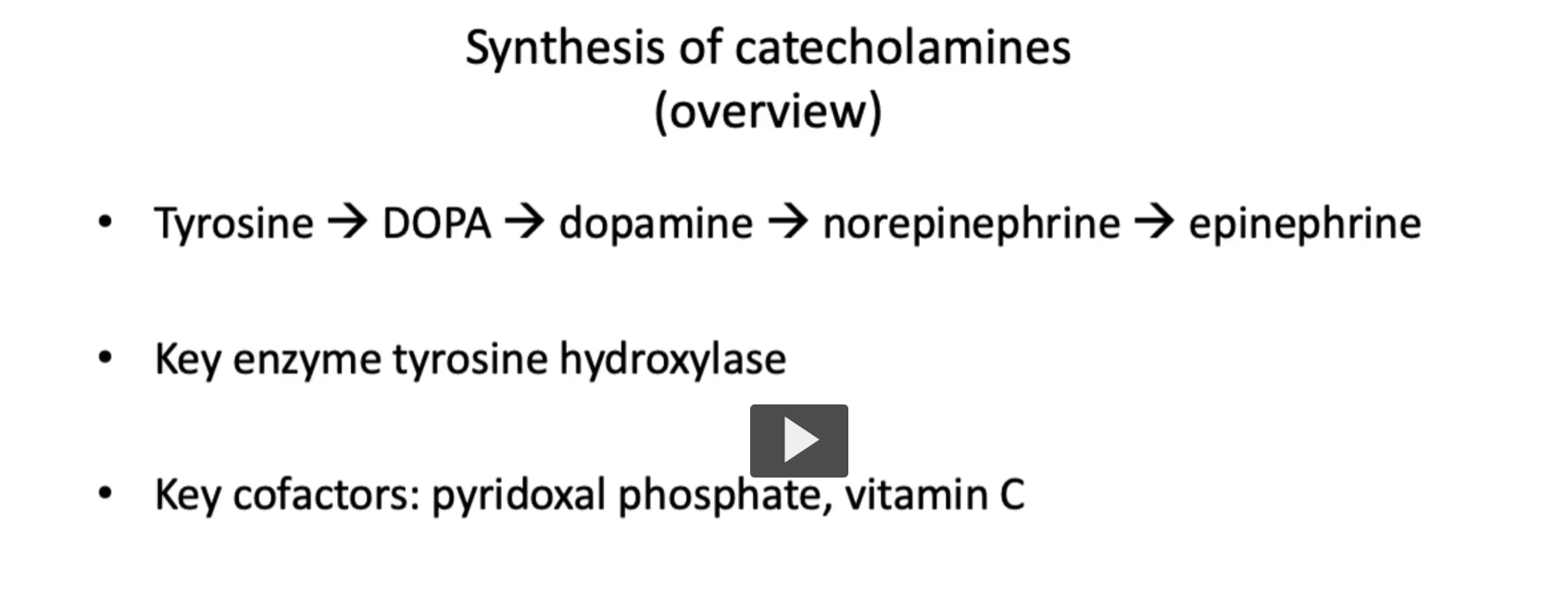
Tyrosine → Dopamine
Tyrosine (Tyrosine hydroxylase adds OH to tyrosine)→ Dihydroxyphenylalanine (DOPA)
Uses O₂ (oxygen)
Requires a cofactor (tetrahydrobiopterin, BH₄ — high-yield detail)
DOPA (DOPA decarboxylase takes a CO2 from DOPA, uses PLP or B6) → dopamine
The function of DOPA decarboxylase is to take a carbon away “decarboxylase” from DOPA, the function of PLP (B₆) is to stabilize intermediates during decarboxylation), which is what DOPA decarboxylase is doing.
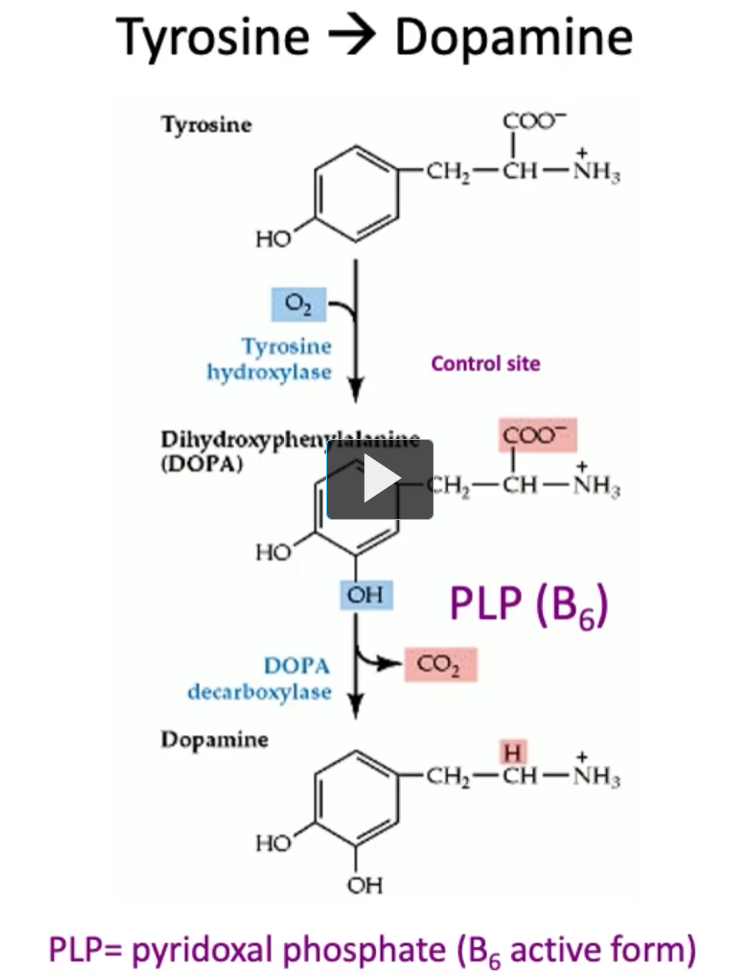
Then Norepi → Epi
Dopamine (Dopamine-β hydroxylase adds OH to Dopamine) → Norepinephrine
Vitamin C (ascorbic acid) is a water-soluble antioxidant that also acts as an enzyme cofactor.
Therefore, without adequate Vitamin C, noepinephrine production is impaired.
Works by maintaining the proper redox (electron) state of the enzyme.
Norepinephrine (Phenylethanolamine N-methyl-transferase It adds a methyl group (–CH₃) to norepinephrine) → Epinephrine
Methyl donor: SAM (S-adenosylmethionine)
Location: Mainly in the adrenal medulla
RCH₃ (this is the methyl group added)
locations
Step | Location in cell | Where in body |
|---|---|---|
Tyrosine → DOPA | Cytosol | Neurons + adrenal medulla |
DOPA → Dopamine | Cytosol | Neurons + adrenal medulla |
Dopamine → NE | Vesicles | Neurons + adrenal medulla |
NE → Epi | Cytosol | Adrenal medulla only |
Catecholamine Release and Removal
• Once synthesized, catecholamines are stored, contained, transported in synaptic vesicles.
• Depolarization causes Ca²⁺ to enter the cell.
• This in turn causes Ca²⁺-dependent exocytosis for release of catecholamines.
• Action mediated by neurotransmitter binding to specific receptor on postsynaptic cell.


Catecholamine Release and Removal
Excess neurotransmitter is either taken back by a pre-synaptic neuron by a sodium-dependent transporter OR the excess neurotransmitter will be metabolized by enzymes such as Monoamine oxidase (MAO), and catechol-o-methyl-transferase (COMT).
This mechanism will terminate the signal and prevent overstimulation.
Remaining is cleared from synapse by Na⁺-dependent transporter on presynaptic neuron.
Most is reused, some destroyed (MAO, COMT).
Monoamine Oxidase (MAO)
Definition:
Monoamine oxidase (MAO) is an enzyme that breaks down monoamine neurotransmitters "“monoamine oxidase” after they’ve done their job in the synapse.
What does MAO do?
It inactivates neurotransmitters like:
Dopamine
Norepinephrine
Epinephrine
Serotonin
This helps terminate the signal and prevents overstimulation.
Catechol-O-Methyltransferase (COMT)
Definition:
COMT is an enzyme that inactivates catecholamines by adding a methyl group (–CH₃).
What does COMT act on?
Mainly catecholamines:
Dopamine
Norepinephrine
Epinephrine
(Unlike MAO, it does NOT significantly act on serotonin)
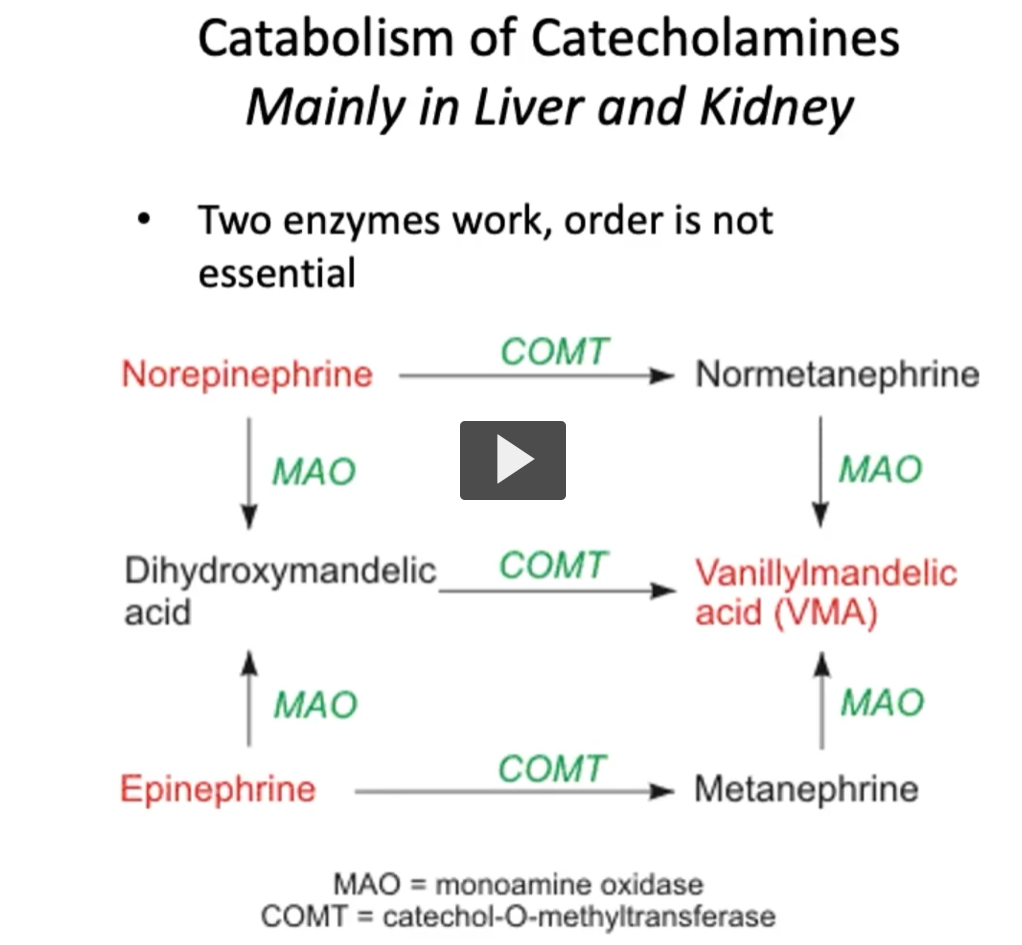
catabolism of catecholamines (mainly in liver and kidney)
Why are they located in the liver? They’re in the liver and kidneys for one main reason:
to detoxify and eliminate circulating monoamines (like catecholamines) from the body. (Chatgpt), the rest of the stuff is what the instructor said)
The primary sites of excess catecholamines breakdown is the liver and kidney. Both MAO and COMT degrade catecholamines through the process of oxidative deamination and as well as methylation.
The sequence of these enzymatic reactions is flexible and is not strictly linear, it does not follow a specific order, but it’s important to remember both processes.
It’s also important to remember the fact that both MAO and COMT enzymes are required for the full inactivation of these excess catecholamines.
Although the exact order of action may vary, both are crucial in maintaining neurotransmitter homeostasis.
A clinical relevant fact is that inhibitors of these enzymes are used therapeutically in Parkinson’s disease and depression.
Vanillylmandelic Acid (VMA)
VMA is the final breakdown product of catecholamines, specifically:
Norepinephrine
Epinephrine
How is VMA formed?
Catecholamines are degraded by MAO and COMT in sequence → ultimately producing VMA, which is excreted in urine.
Simplified pathway:
Norepinephrine / Epinephrine
→ (MAO + COMT)
→ VMA
Where is it found? Urine (measured clinically)
Clinical significance
High-yield test point:
↑ VMA levels → suggests excess catecholamine production

Clinical significance of metabolites
Metabolites such as Homovanillate (from dopamine) and vanillylmandelate (from epi/norepi) are secreted in urine.
High amounts of degradation products in urine may signal pheochromocytoma (catecholamine-secreting tumor) or neuroblastoma.
CSF levels of homovanillate may reflect dopamine activity and oxidative stress.
Levels of homovanillate in CSF reflect oxidative or metabolic stress as well as levels of dopamine.
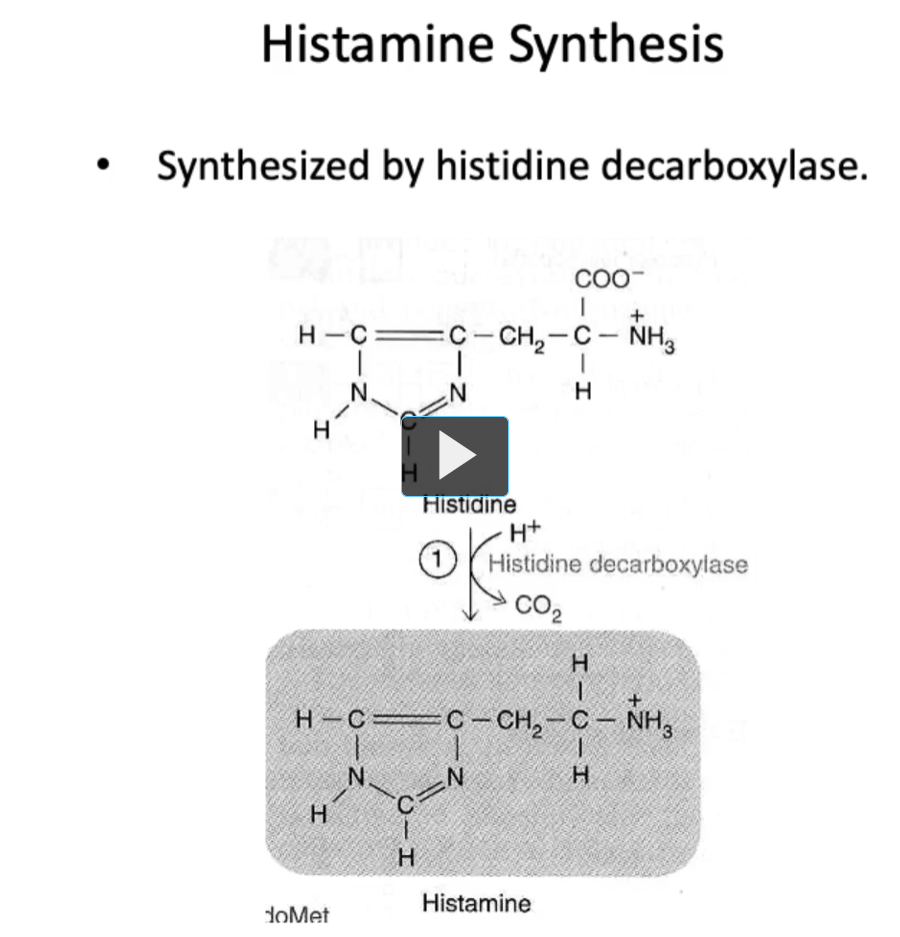
Histamine Synthesis
In the case of histamine, it is synthesized from the amino acid histidine by the enzyme histidine decarboxylase.
It plays a role in allergic reactions, gastric acid secretion, and neurotransmission.
Unlike the catecholamines, histamine is not recycled. It is metabolized after its release.
Histamine is not recycled because there is no efficient reuptake system and it is safer for the body to rapidly inactivate it than to reuse it.
In this figure, we can see the structure of histamine.

Histamine Degradation
Histamine is inactivated by methylation and oxidation via the enzyme Histamine N-Methyltransferase (HNMT) (intracellular) as well as diamine oxidase (DAO) (extra-cellular). These enzymes degrade histamine in a non-sequential manner. Primarily outside the brain.
Deficiencies in these pathways can lead to clinical examples of histamine intolerance or exaggerrated allergic responses.
Removed from synapse by transporter.
Histamine is metabolized rather than recycled.
Metabolized by action of methyltransferase and diamine oxidase (DAO); order independent.
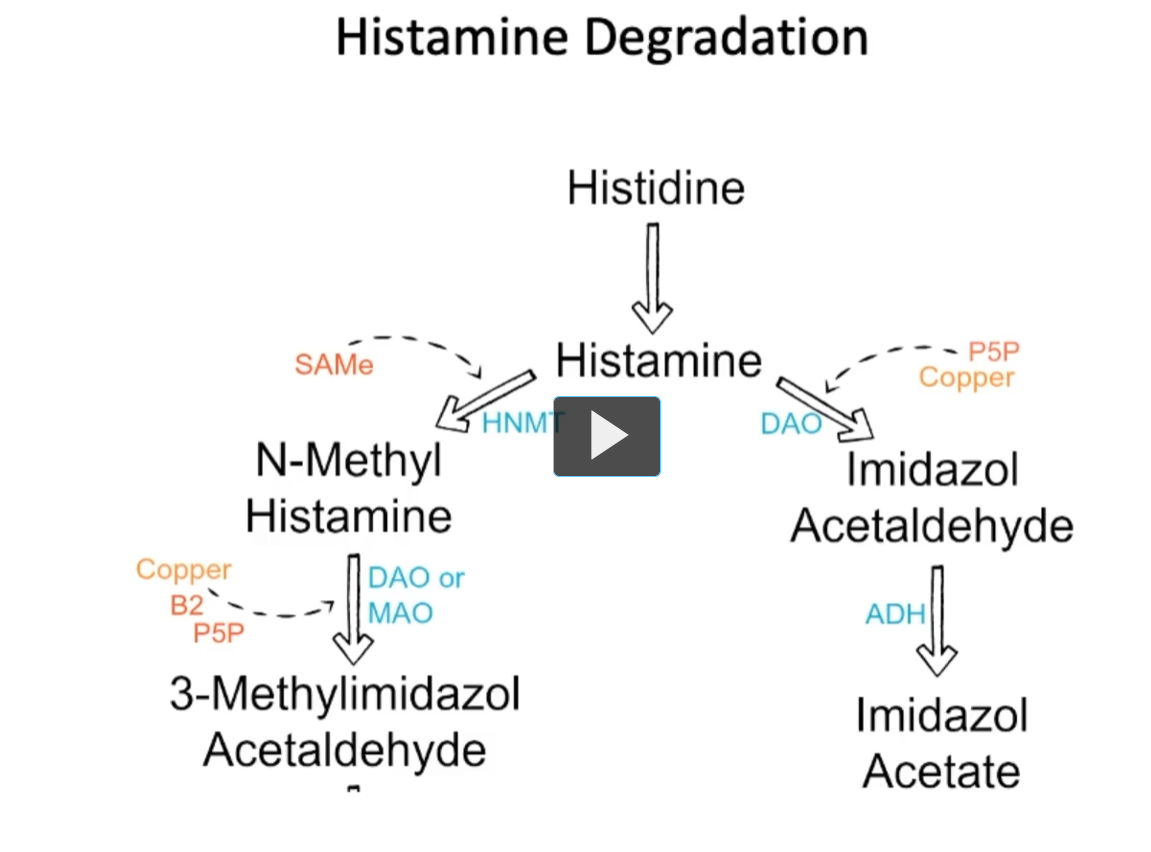
Histamine Degradation
Looking at this figure, we can see that Histamine is inactivated by methylation (Histamine N-Methyltransferase (HNMT) and oxidation (diamine oxidase (DAO), via
Histidine (Histine decarboxylase)
↓
Histamine
Left pathway:
(Histamine N-Methyltransferase (HNMT) inactivates Histamine (using SAMe as a methyl donor) → N-methyl histamine.
N-methyl histamine (DAO or MAO, Copper, B2, P5P) → 3-Methylimidazol Acetaldehyde
Right pathway:
HIstamine (DAO, P5P, Copper) → Imidazol Acetaldehyde
Imidazol Acetaldehyde (ADH) → Imidazol Acetate

Histamine Function & Clinical Importance
Four subtypes of G-protein-coupled receptors (H₁, H₂, H₃, and H₄).
H₁ is important to allergic responses.
why?
H₁ receptor
Location: Smooth muscle, endothelium, brain
Function:
Allergic responses (most important!)
Vasodilation
↑ vascular permeability → edema
Bronchoconstriction
Clinical:
Target of antihistamines (e.g., diphenhydramine)
Think: H₁ = “Allergy receptor”
Pharmocologically, or in terms of neurophysiology, there is a need for selective antihistaminic is critical to alleviate side effects.
Antihistaminics (Antihistamines)
What they are:
Drugs that block histamine receptors, mainly H₁ or H₂, to reduce histamine’s effects.
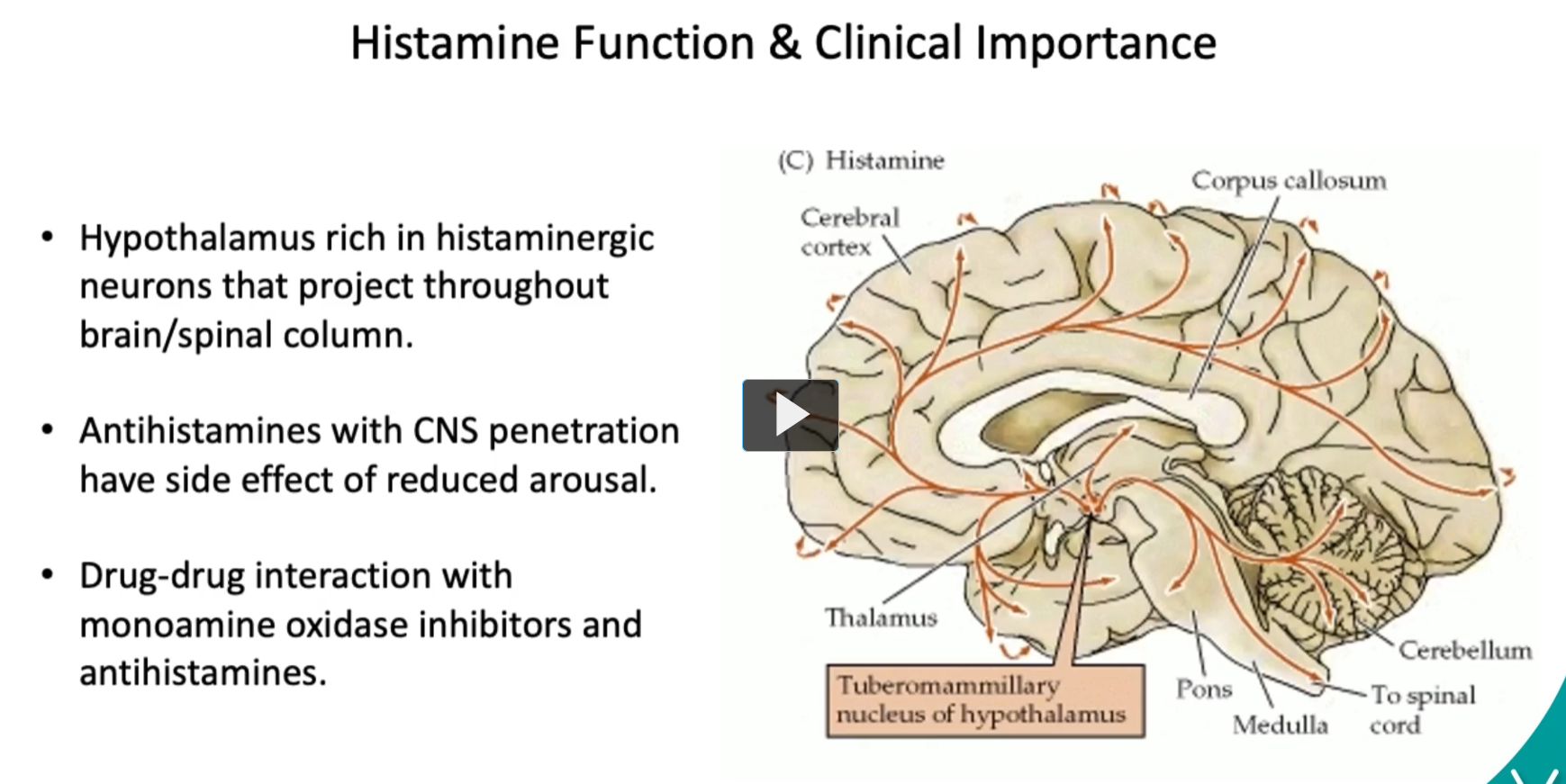
Histamine Function and Clinical Importance
The hypothalamus is rich in histaminergic neurons (the hypothalamus contains many neurons that release histamine) that project throughout brain/spinal column.
project: the axons of the histaminergic neurons of the hypothalamus travel long distances through the brain/spinal column.
Histaminergic neurons in the hypothalamus regulate arousal and wakefullness.
Histamine = direct “wake-up signal”
Serotonin = modulator of wakefulness + sleep cycles + mood
Antihistamines with CNS penetration (crossing of Blood Brain Barrier) can reduce alertness, have side effect of reduced arousal.
Anti-histamines reduce alertness because histamine promotes alertness.
Drug-drug interaction with monoamine oxidase inhibitors and antihistamines can exacerbate these effects.
Antihistamines (H₁ blockers) Block histamine in the brain Histamine normally promotes: Wakefulness Alertness. So blocking it → sedation / drowsiness
Monoamine oxidase inhibitors (MAOIs) Prevent breakdown of: Serotonin Dopamine, Norepinephrine These neurotransmitters affect: Mood Arousal Sleep–wake balance.
Therefore, it is important to be aware of the mechanism of action of histamine in the CNS.

Serotonin/5-hydroxytryptamine (5-HT)
Serotonin is synthesized from tryptophan (tryptophan must be in diet, meaning tryptophan is an essential amino acid).
Two step conversion of tryptophan to 5-HT.
Tryptophan hydroxylase is the rate limiting / commitment step for the conversion of tryptophan to serotonin.
Serotonin is involved in mood, sleep cycle, arousal, and depression.
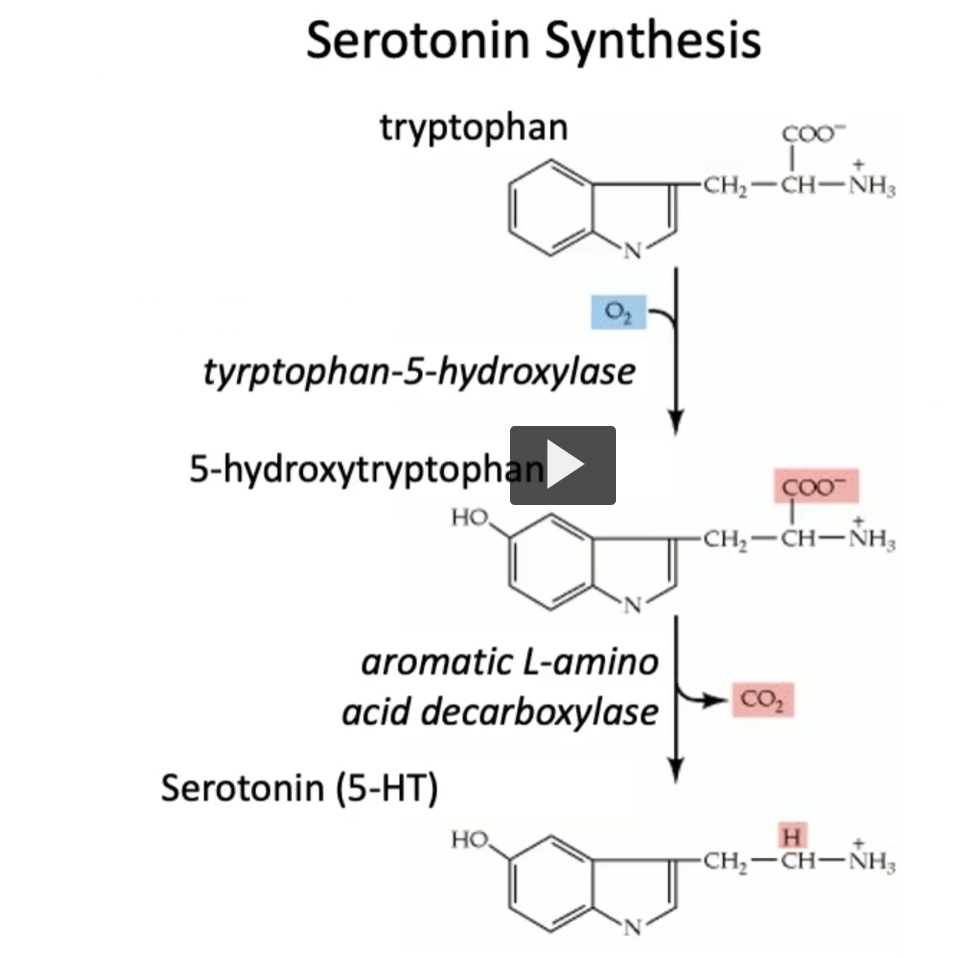
Serotonin Synthesis
In this figure, we can see the two-step process in the synthesis of serotonin.
tryptophan (tyrptophan-5-hydroxylase, adds a hydroxyl to tryptophan) → 5-hydroxy-tryptophan
OH is added.
5-hydroxytryptophan (aromatic L-amino acid decarboxylase) → Serotonin (5-HT)
Tryptophan is an aromatic amino acid.
CO2 is lost.
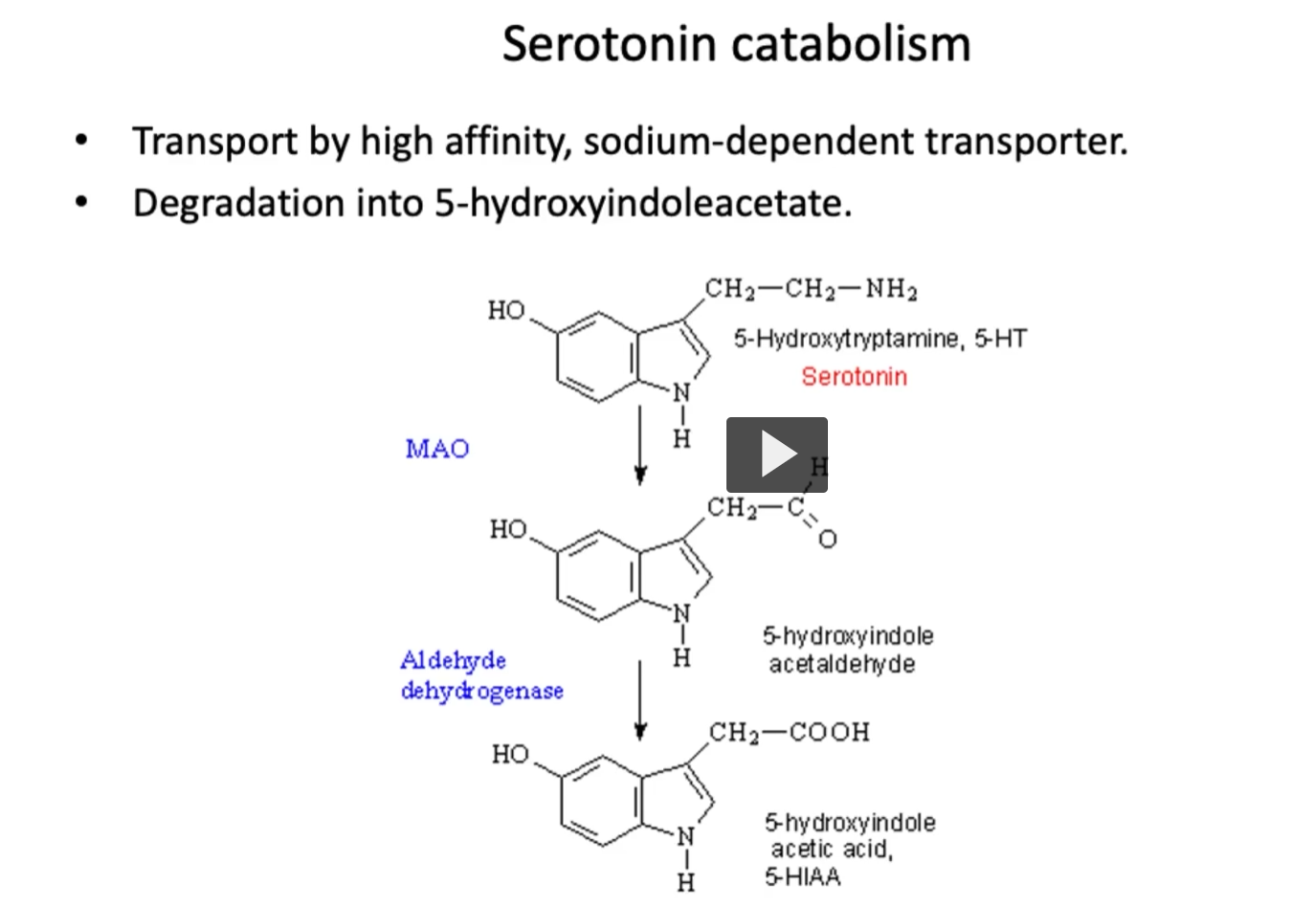
Serotonin catabolism
On the other hand, the process of catabolism of serotonin.
Serotonin will be re-absorbed by high affinity, sodium-dependent transporter.
Serotonin is degraded (by MAO) into 5-hydroxyindole acetaldehyde
5-hydroxyindole acetaldehyde is degraded (by aldehyde dehydrogenase) into 5-hydroxyindole acetic acid (5-HIAA).
5-hydroxyindole acetic acid (5-HIAA) will be excreted in the urine. Measurement of 5-HIAA is clinically useful for diagnosing any serotonin producing tumors such as seen in the carcinoid syndrome.
Serotonin roles and dysfunction
In CNS, serotonin controls mood, appetite and sleep.
In gut (enterochromaffin cells), serotonin modulates peristalsis, regulating intestinal movements
Excess serotonin may lead to adverse effects:
cardiomyocyte growth – right heart disease
tryptophan depletion and niacin deficiency (leading to the clinical condition of pellagra = dermatitis, dementia, diarrhea)
Defects in proper regulation of serotonin receptors cause psychotic disorders.
Too much serotonin can be caused by a carcinoid tumor detectable by excess 5-hydroxyindoleacetate (5-HIAA) in urine.

Excess serotonin may lead to adverse effects:
cardiomyocyte growth – right heart disease
Right Heart Disease (in the context of serotonin)
This refers to damage to the right side of the heart, especially the tricuspid and pulmonary valves, most classically seen in carcinoid syndrome
Why serotonin causes right heart disease
In carcinoid tumors:
Tumor releases large amounts of serotonin (5-HT) into the bloodstream
Serotonin effects:
Stimulates fibroblast and smooth muscle proliferation
Leads to fibrosis (scar-like tissue) on heart valves
What specifically happens
Thickening and stiffening of:
Tricuspid valve
Pulmonary valve
Results in:
Tricuspid regurgitation (most common)
Pulmonary stenosis
Why ONLY the RIGHT side?
This is very high-yield:
Blood from GI tumors → goes to liver first
Normally, the liver breaks down serotonin (via MAO)
BUT:
If tumor metastasizes to liver, serotonin enters systemic circulation
Still:
Lungs contain MAO → break down serotonin
So:
Right heart gets exposed first
Left heart is protected by lungs
Summary flow:
Carcinoid tumor → ↑ serotonin → fibrosis of right heart valves → right heart disease
Excess serotonin may lead to adverse effects:
tryptophan depletion and niacin deficiency (leading to the clinical condition of pellagra = dermatitis, dementia, diarrhea)
Niacin (Vitamin B₃)
A water-soluble vitamin essential for making NAD⁺ and NADP⁺, which are critical for energy metabolism.
Where it comes from
Diet (meat, grains, etc.)
Synthesized from tryptophan
Key connection (VERY HIGH-YIELD)
Tryptophan can go two ways:
→ Serotonin production
→ Niacin (B₃) production
If too much tryptophan is used to make serotonin:
↓ tryptophan available for niacin
→ niacin deficiency
Niacin deficiency = Pellagra Classic 3 D’s
Dermatitis
Diarrhea
Dementia
(+ 4th D = Death if severe)
“Pellagra” comes from Italian:
“pelle” = skin
“agra” = rough, harsh, or sour
So pellagra literally means “rough skin.”
Why that name makes sense
One of the hallmark features is dermatitis
The skin becomes:
Rough
Thickened
Hyperpigmented (especially in sun-exposed areas)
carcinoid tumor
A slow-growing neuroendocrine tumor that most commonly arises from enterochromaffin cells in the gastrointestinal (GI) tract.
“carcino-” = cancer
“-oid” = resembling
“Carcinoid” = “cancer-like”
(Originally named because it seemed less aggressive than typical cancers)
What they produce
Serotonin (5-HT) (most important)
Other vasoactive substances (e.g., bradykinin, histamine)
Carcinoid Syndrome
Occurs when tumor products reach systemic circulation (often after liver metastasis)
Symptoms:
Flushing
Diarrhea
Wheezing (bronchospasm)
Right-sided heart disease
Why right heart disease happens
Serotonin → fibrosis of heart valves
Affects:
Tricuspid valve (on right side of the heart)
Pulmonary valve (on right side of the heart)
Left heart is protected because lungs break down serotonin
Diagnosis
↑ 5-HIAA in urine
5-HIAA = serotonin breakdown product
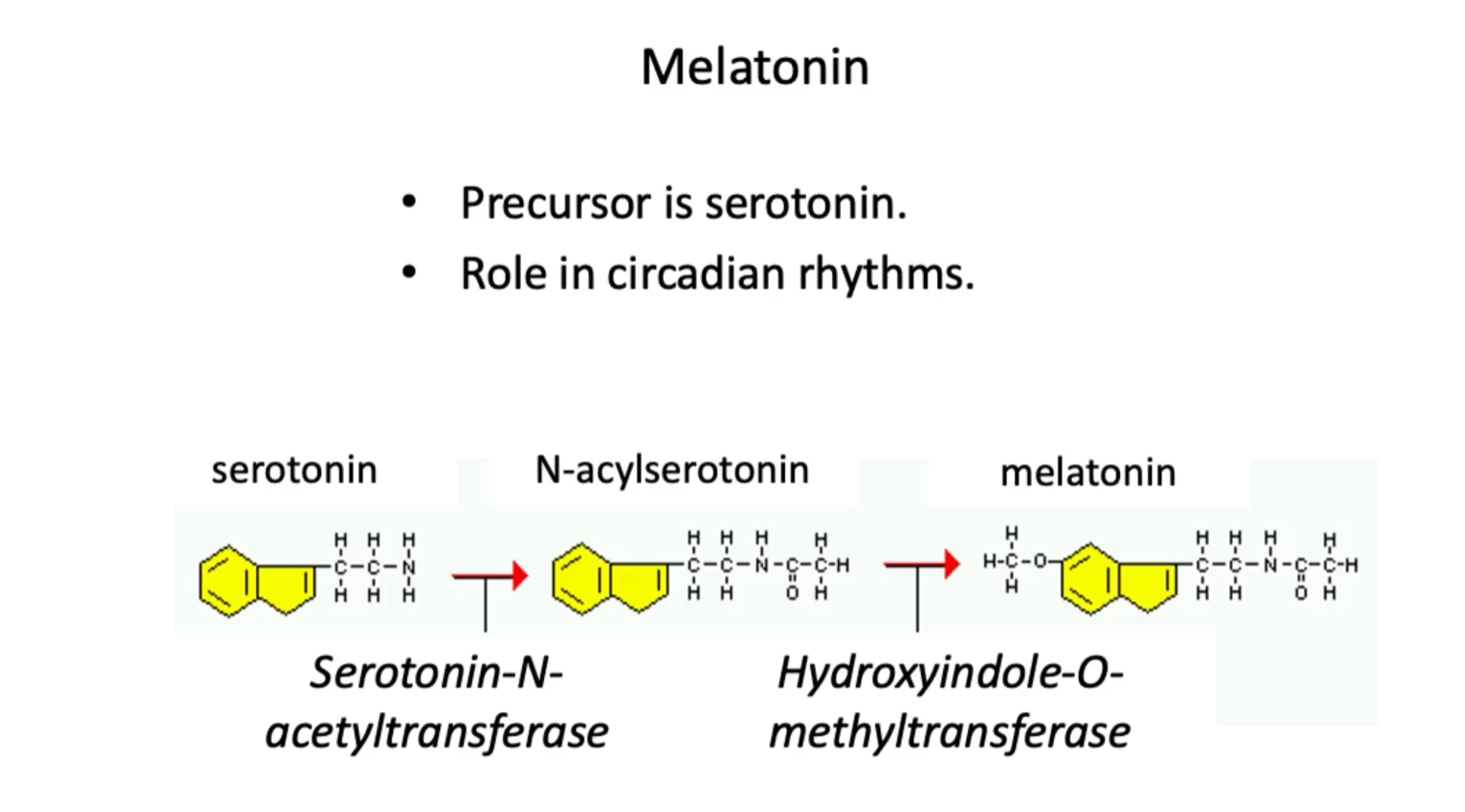
Melatonin
Serotonin is a precursor for melatonin.
Melatonin is synthesized from serotonin in the pineal gland.
Precursor is serotonin.
Role in circadian rhythms.
Production of melatonin will increase in darkness, thus promoting sleep
In this figure we can see the chain of reactions that lead to the conversion of melatonin from serotonin.
Pathway:
1. serotonin (Serotonin-N-acetyltransferase) → N-acylserotonin
Serotonin-N-acetyltransferase transfers an acetyl group TO serotonin.
2. N-acylserotonin (Hydroxyindole-O-methyltransferase) → melatonin
Hydroxyindole-O-methyltransferase transfers a methyl group to N-acylserotonin to get melatonin.
Hydroxyindole-O-methyltransferase =
An enzyme that transfers a methyl (–CH₃) group to the oxygen (O) of a hydroxyl group on an indole compound
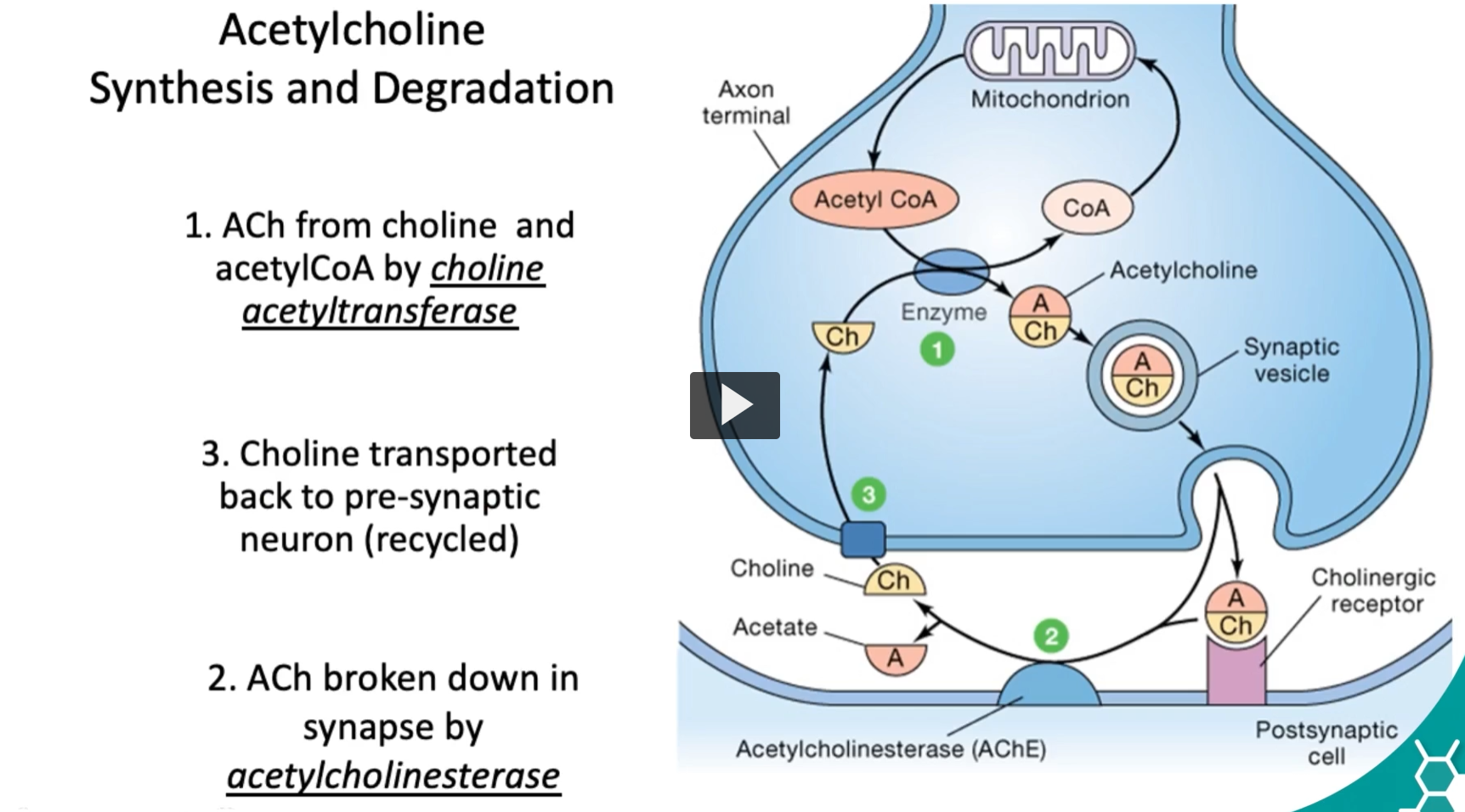
Acetylcholine Synthesis and Degradation
Now to discuss acetylcholine, it’s important to discuss the synthesis, and then we will discuss degradation.
ACh from choline and acetylCoA by choline acetyltransferase
Choline acetyltransferase (ChAT)
An enzyme that transfers an acetyl group (–COCH₃) from acetyl-CoA TO choline → acetylcholine.
ACh broken down in synapse by acetylcholinesterase
Choline transported back to pre-synaptic neuron (recycled)
This fast breakdown makes acetylcholine ideal for rapid, synaptic transmission.
We can remember that acetylcholine is one of the main messenger in neuromuscular-skeletal pathways for muscle contraction, leading to rapid access, release and degradation.
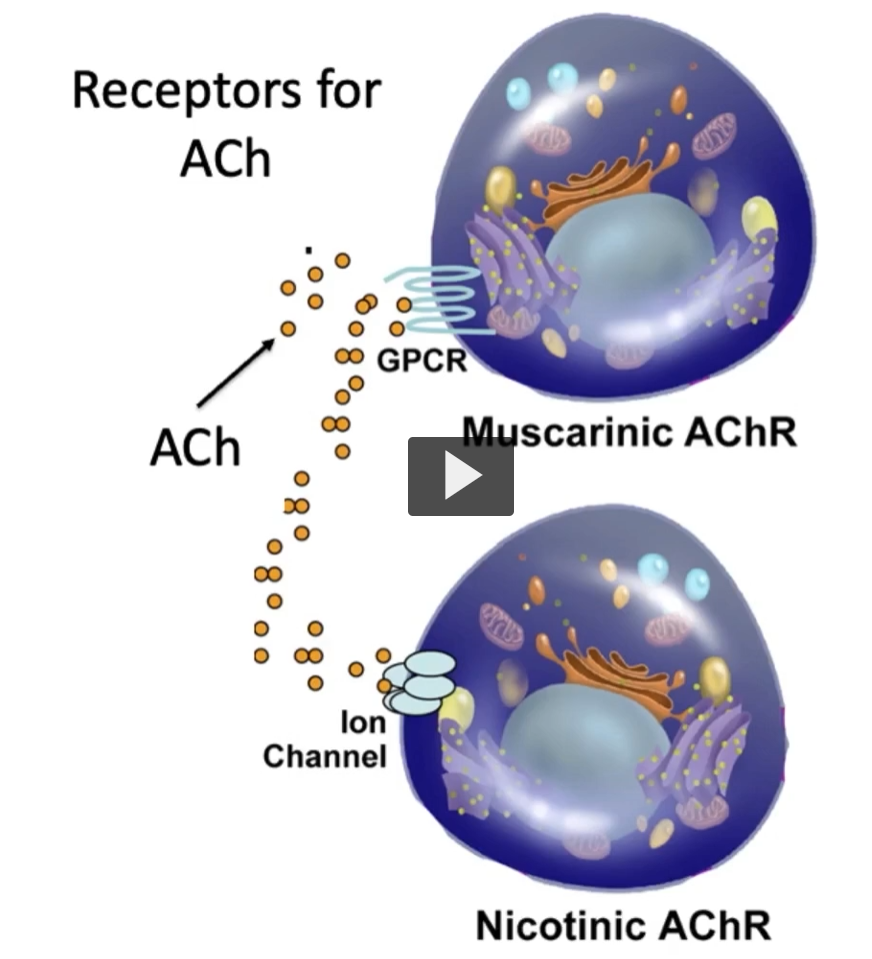
Receptors for ACh
There are two main acetyl-choline receptor types.
muscarinic acetyl-choline receptors, which are metabotropic and G-protein. The muscarinic acetyl-choline receptors mediate para-sympathetic effects.
-muscarine does NOT mean muscle, it means muscarine (a mushroom)
On the other hand, there are Nicotinic acetyl-choline receptors, which are ionotropic, regulating ion channels, facilitating skeletal muscle contraction.
Both muscarinic and nicotinic acetyl-choline receptors play roles in the autonomic and central nervous system function.
Scientists named receptor types based on which plant toxins activated them:
Muscarinic → muscarine (mushroom)
Nicotinic → nicotine (tobacco)

Muscarinic Receptors
Stimulated by the alkaloid muscarine and mediate slower, more modulatory effects via G protein.
Interacts with Gi protein (metabotropic).
Activates peristalsis but decrease heart rate. (parasympathetic)
Antagonist-scopolomine-used to treat motion sickness.Scopol-” → from the plant genus Scopolia
“-amine” → indicates a nitrogen-containing compound (alkaloid)
So scopolamine = an amine derived from Scopolia plants
Mechanism
Muscarinic receptor antagonist
Blocks acetylcholine (ACh) at muscarinic receptors
Clinical use of scopolomine
Motion sickness
Prevents nausea/vomiting by: Blocking signals from inner ear → brain
It is important to remember that All muscarinic receptors have seven transmembrane segments.
why seven transmembrane segments? Because all muscarinic receptors are G-protein–coupled receptors (GPCRs)—and GPCRs are built with a very specific architecture: Muscarinic receptors (M1–M5) belong to the GPCR family
All GPCRs have 7 transmembrane α-helices
That’s a defining structural feature of the entire receptor class.
Nicotinic Receptors
nicotinic receptors are activated by nicotine.
Ligand-activated ion channels (ionotropic).
Found at neuromuscular junction of somatic muscles.
Prone to desensitization upon overstimulation.
If nicotinic receptors are stimulated continuously or too strongly, they stop responding, even though the ligand (ACh or nicotine) is still present.
But people who smoke don’t get paralysis because
1. Dose and duration are very different
Paralysis (like with succinylcholine) happens when receptors are:
continuously flooded with agonist
at high concentrations
Smoking delivers lower, intermittent doses of nicotine
Not enough to keep receptors constantly activated


Glutamate
An excitatory amino acid.
Primary excitatory neurotransmitter in brain.
Considered most important neurotransmitter for brain function (learning).
Binds NMDA or AMPA ionotropic receptors.
Binds metabotropic glutamate receptor.
Glutamate plays an essential role in synaptic plasticity and this regulation is associated with excitotoxicity and in some cases, neurodegeneration.
NMDA receptor
NMDA = N-methyl-D-aspartate
A methyl group attached to nitrogen
“aspartate” = an amino acid (from asparagus, where it was first isolated)
“D” = specific stereoisomer (mirror-image form)
What is the NMDA receptor?
A type of glutamate receptor
Specifically an ionotropic receptor (ligand-gated ion channel)
Unique features (high-yield)
Requires 2 ligands
Glutamate + glycine (co-agonist)
Voltage-dependent
At rest → blocked by Mg²⁺
Needs depolarization to remove Mg²⁺ block
Lets Ca²⁺ in
Also Na⁺ in, K⁺ out
Calcium entry = key for learning
Why it matters
Central to long-term potentiation (LTP)
→ basis of learning and memory
Clinical relevance
Overactivation → excitotoxicity (neuronal damage)
Involved in:
Stroke
Neurodegeneration
AMPA Receptor
AMPA = α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid
Like NMDA, this is a synthetic agonist used to define the receptor.
What is the AMPA receptor?
A glutamate receptor
Specifically an ionotropic (ligand-gated ion channel)
What does it do?
When glutamate binds:
Channel opens immediately
Na⁺ flows in (mainly)
Some K⁺ flows out
Result = rapid depolarization (fast excitatory signal)
Key features (high-yield)
Fastest excitatory transmission in the brain
Responsible for EPSPs (excitatory postsynaptic potentials)
Does NOT require depolarization to activate (unlike NMDA)
Usually no Mg²⁺ block
Limited Ca²⁺ permeability (unless modified subunits
AMPA Relationship to NMDA (important!)
Think of them as a team:
AMPA fires first → depolarizes the cell
This removes Mg²⁺ block from NMDA
Then NMDA opens → Ca²⁺ enters → learning (LTP)
“glutamate binds metabotropic glutamate receptor.”
“Binds metabotropic glutamate receptor” — what does that mean?
It means glutamate can activate receptors that are NOT ion channels, but instead work through G-proteins and signaling pathways.
What are metabotropic glutamate receptors (mGluRs)?
Metabotropic = “changing metabolism”
These are G-protein–coupled receptors (GPCRs)
Unlike AMPA/NMDA, they do NOT directly let ions flow
How they work
Glutamate binds the receptor
The receptor activates a G protein
This triggers second messengers inside the cell (like cAMP, IP₃, DAG)
These signals:
Open/close ion channels indirectly
Change enzyme activity
Modify gene expression
Effects are slower but longer-lasting
Glutamate Synthesis
Glutamate is synthesized in neurons from glutamine via the enzyme glutaminASE.
GlutMINEASE removes an amine from glutamine to get glutamate.
Glutamate synthesis occurs in CNS.
Because glutamate cannot cross blood brain barrier; glutamate must be synthesized in neuron. (glutamate has COO- and NH3+ positive charges, at physiological pH, it exists as a polar, charged molecule, and therefore charged molecules CANNOT diffuse through the lipid membrane of the BBB)
Amino group removed by glutaminase.
Glutamate is stored in synaptic vesicles and released upon depolarization.
Transported into vesicles for release.

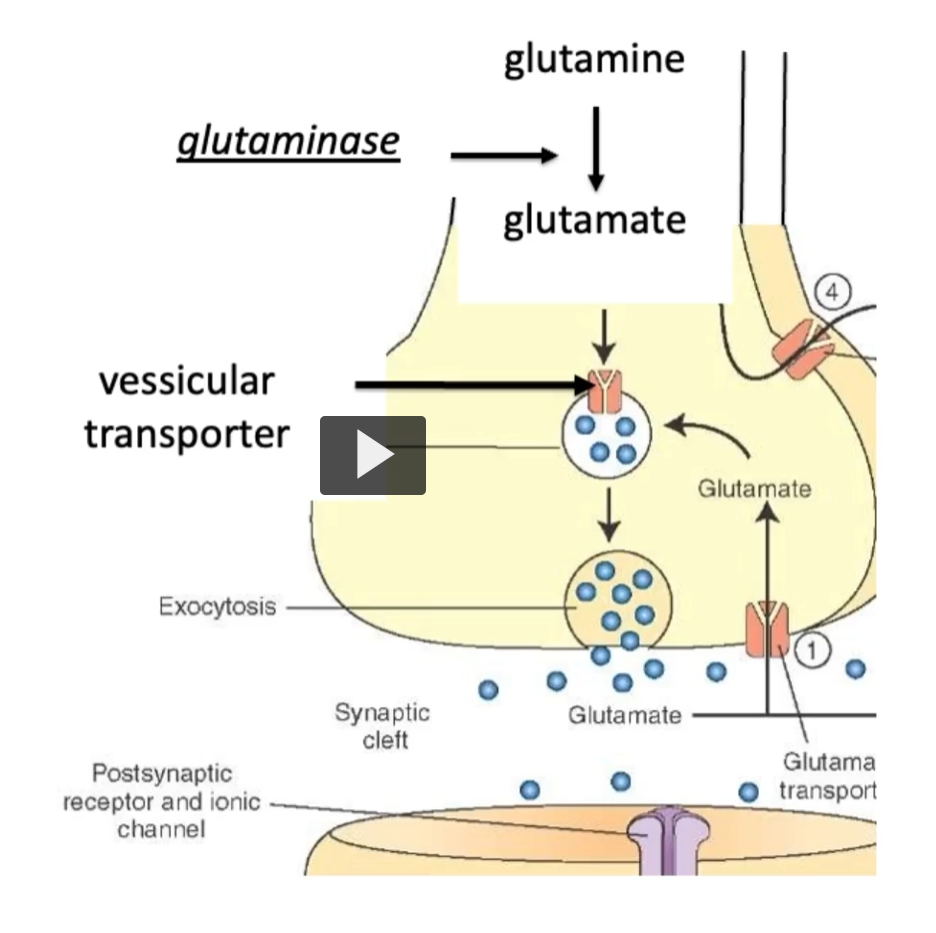
Glutamate
Excess glutamate must be rapidly cleared
Glial cells absorb glutamate via transporters and convert it back to glutaMINE, which is then recycled back to neurons.
In pair, glutamate clearance is implicated in diseases like ALS and Alzheimer’s disease.
In this picture, we can see the process of glutamine synthesis and clearance.
ALS — Amyotrophic Lateral Sclerosis
Etymology (helps you remember)
A- = without
Myo- = muscle
-trophic = nourishment
“Amyotrophic” = muscle wasting
Lateral = sides of the spinal cord
Sclerosis = hardening/scarring
“ALS” = degeneration (hardening) of lateral spinal cord pathways → muscle wasting
What it is
A neurodegenerative disease
Affects:
Upper motor neurons (UMN) → brain
Lower motor neurons (LMN) → spinal cord
What happens: Motor neurons die → muscles lose innervation → progressive weakness and atrophy.
Why glutamate matters (connect to your slide)
ALS is associated with excess glutamate → excitotoxicity
Too much stimulation → Ca²⁺ influx → neuron death
Alzheimer’s disease (AD)
Etymology
Named after Alois Alzheimer, who first described it (1906)
What it is
A progressive neurodegenerative disorder
Most common cause of dementia
Primarily affects:
Hippocampus → memory
Cerebral cortex → thinking, behavior
Core pathology (high-yield)
Amyloid-β plaques (extracellular)
Derived from APP (amyloid precursor protein)
“Sticky” deposits between neurons
Neurofibrillary tangles (intracellular)
Made of hyperphosphorylated tau protein
Disrupt microtubules
Loss of cholinergic neurons
Especially in basal nucleus of Meynert
↓ acetylcholine → memory impairment
Neurotransmitter changes
↓ acetylcholine (major)
↑ glutamate toxicity (excitotoxicity contributes to damage)
Glutamate removal/recycling
Excess glutamate is neurotoxic.
glutamate is rapidly removed by action of glutamate transporters on neurons and glia.
In glia, glutamate is converted to glutamine and recycled back into neurons.

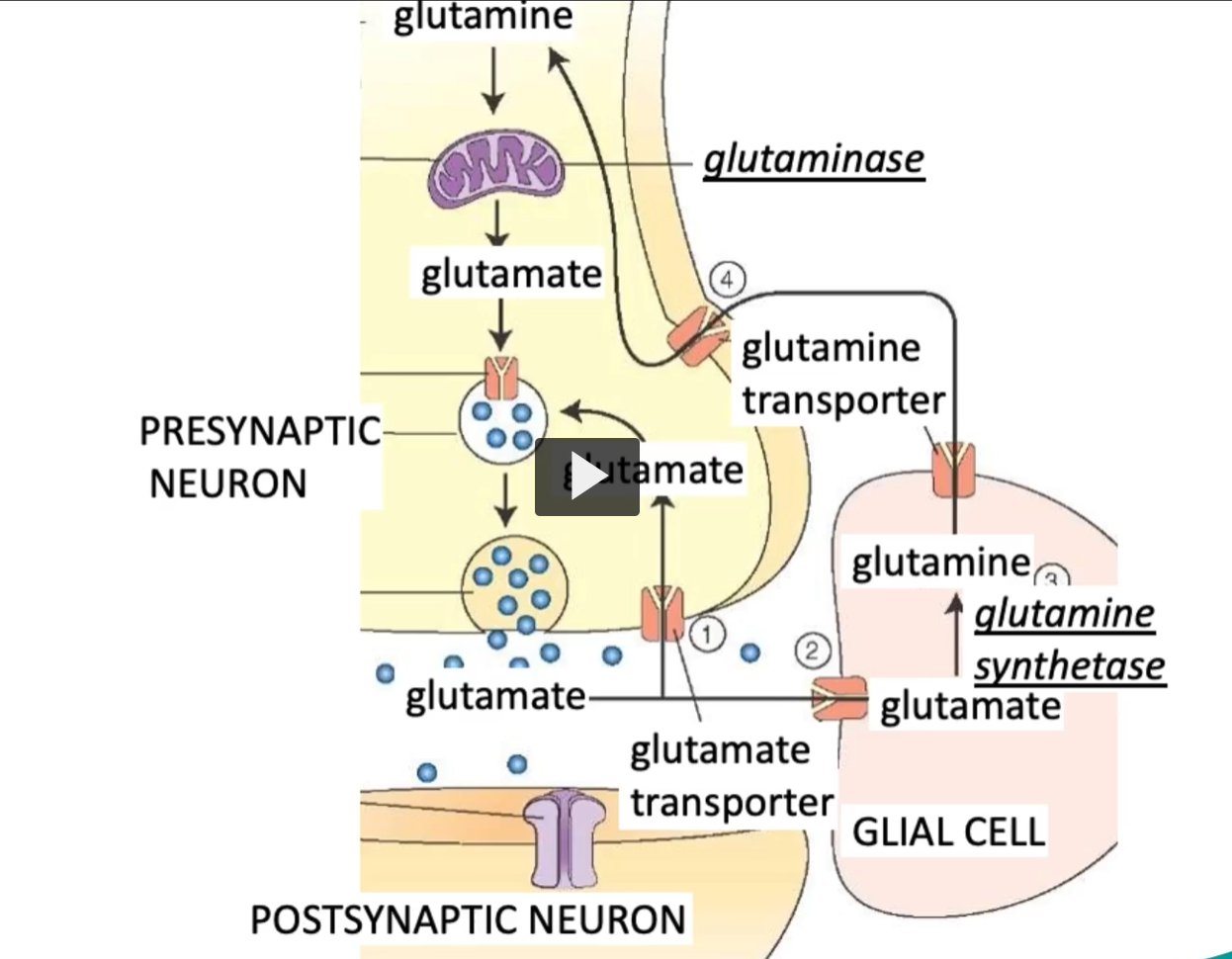
GlutaMINase: converts glutaMINE to glutaMATE
Glutamine Synthase: synthesizes glutamine
GABA and Glycine
On the other hand, there is GABA and Glycine, which are the main inhibitory neurotransmitters.
Main inhibitory neurotransmitters.
GABA is derived by decarboxylation of glutamate.
Two types of GABA receptors are GABAₐ and GABAᵦ.
GABA effects on GABAₐ receptors are allosterically increased by diazepam and barbiturates.
GABAₐ is a ionotropic ligand gated chloride channel.
GABAᵦ is a metabotropic receptor.
Glycine
Glycine plays a role in inhibiting signaling, Glycine is especially important in reflex pathways and spinal inhibition.

GABAₐ receptor
What it is
A GABA receptor subtype
Ionotropic (ligand-gated ion channel)
Mechanism
GABA binds → channel opens → Cl⁻ (chloride) enters the cell
This makes the inside more negative (hyperpolarization)
Result: inhibition (neuron less likely to fire)
Key features (high-yield)
Fast inhibitory signaling
Located throughout the brain (CNS)
Opposes excitatory neurotransmitters like glutamate
Pharmacology
GABAₐ receptors are enhanced by:
Benzodiazepines (e.g., Diazepam)
↑ frequency of Cl⁻ channel opening
Barbiturates
↑ duration of Cl⁻ channel opening
Alcohol
Also enhances GABAₐ activity
All lead to sedation, anxiolysis, anticonvulsant effects
GABAᵦ receptors
What it is:
A GABA receptor subtype
Metabotropic (G-protein–coupled receptor, GPCR)
Mechanism
Couples to Gi protein
Effects:
↓ cAMP
Opens K⁺ channels → K⁺ leaves cell → hyperpolarization
Closes Ca²⁺ channels → ↓ neurotransmitter release
Result: slow, prolonged inhibition
Key features (high-yield)
Slower than GABAₐ
Produces long-lasting inhibitory effects
Often:
Postsynaptic → hyperpolarization (via K⁺)
Presynaptic → ↓ neurotransmitter release (via Ca²⁺ inhibition)
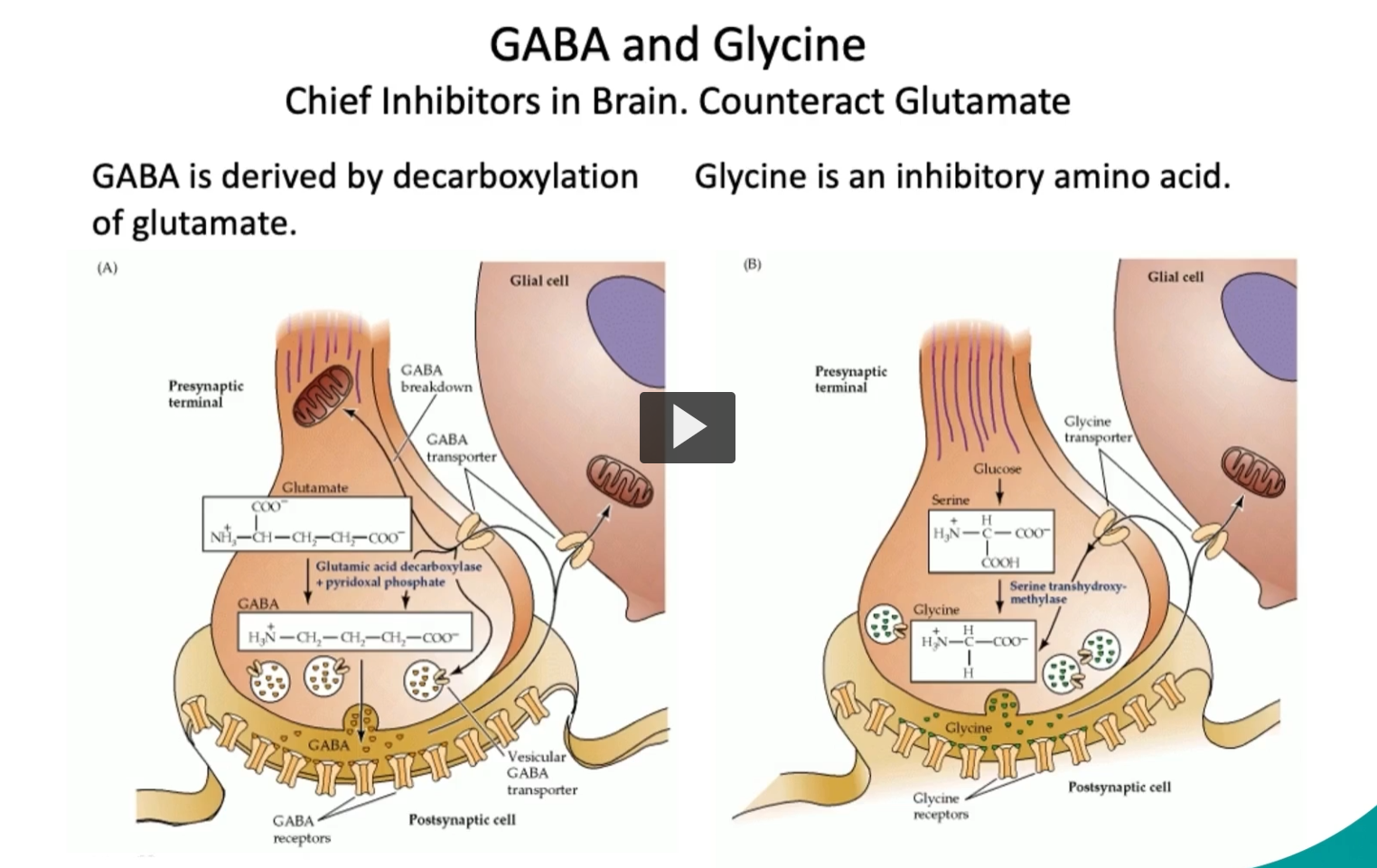
GABA and Glycine
Chief Inhibitors in Brain. Counteract Glutamate
In this slide, we can see both GABA and Glycine, which are the chief inhibitors in the brain, and they counteract glutamate activity.
On the left, we can see that GABA is derived by decarboxylation of glutamate.
On the right, we can see that Glycine is an inhibitory amino acid, important in reflex pathways and spinal inhibition.
Opioid Peptides
“Opium” → from Greek opion = juice of the poppy
“-oid” = “resembling”
Opioid = “opium-like substance”
Opioid peptides are endogenous, short peptides that bind to opioid receptors.
Opioids play important roles in motivation, pain, stress, food intake, reward system.
Opioids are derived from 3 homologous genes (All endogenous opioid peptides come from 3 precursor proteins (genes) that are later cut into smaller active peptides)
Proopiomelanocortin (POMC), POMC is cleaved to produce the beta-endorphins.
Enkephalins (Derived from proenkephalin, produce Met-enkephalin, Leu-enkephalin, function: Modulate pain (especially in spinal cord)
Dynorphins (Derived from prodynorphin, produce Dynorphin A, B, Function:
Act mainly on κ (kappa) receptors
Involved in pain and stress responses)
These peptides are produced through proteolytic cleavage of precursor proteins.


Opioid Receptors
The opioid receptors include the Mu, kappa, delta, nociceptin.
Mu is the pharmacologically important receptor. This is because the Mu receptor is the main pharmacological target of drugs like morphine.
The activation of the Mu receptor reduces pain perception
Morphine remains the most effective pain-killer in the world.
But prolonged use can lead to:
Tolerance
Addiction
Constipation
Withdrawal
Understanding receptor subtypes, can guide pain-management strategies.
Nitric Oxide
Nitric Oxide is a unique neurotransmitter made from arginine by the enzyme nitric oxide synthase.
Unlike other neurotransmitters, nitric oxide is a gas, a paracrine gas that diffuses freely.
The enzyme (nitric oxide synthase) contains a P450-type heme group.
Nitric oxide activates guanylate cyclase → increasing cGMP → activation of Protein Kinase G, and causing smooth muscle relaxation, especially in blood vessels.
Causes relaxation of blood vessels, via cGMP.

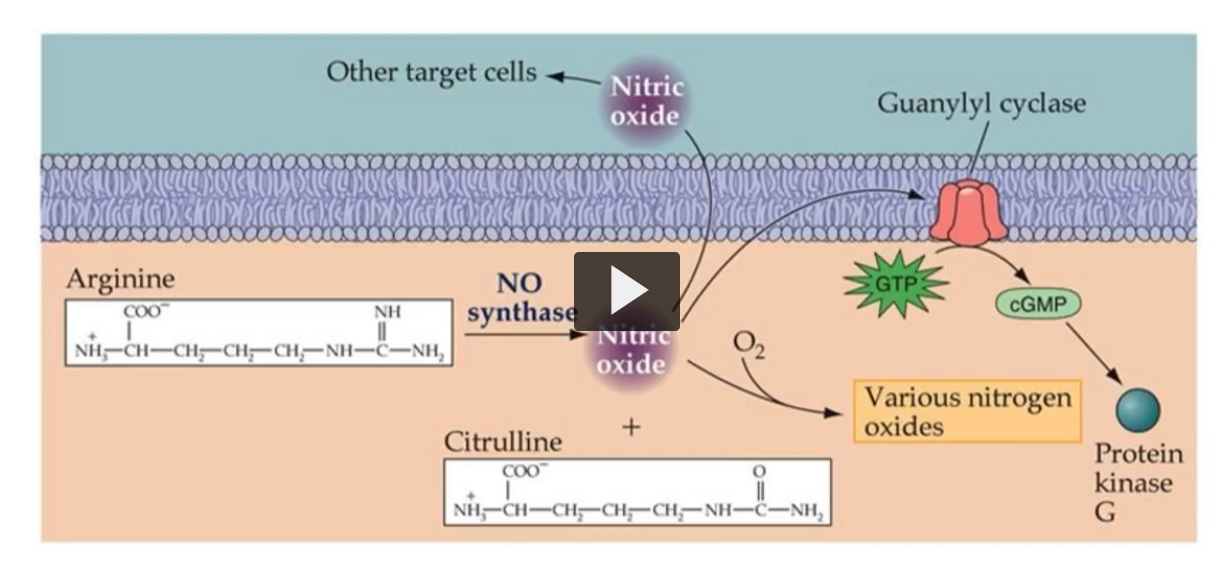
In this figure, we can see the production and its mechanism of action on Guanylyl Cyclase.
Which of the following amino acids is a precursor to the ‘reward’ neurotransmitter (dopamine)?
Please choose from the list below.
A. glutamine
B. glycine
C. histidine
D. tryptophan
E. tyrosine
