Phase 1 Reactions
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
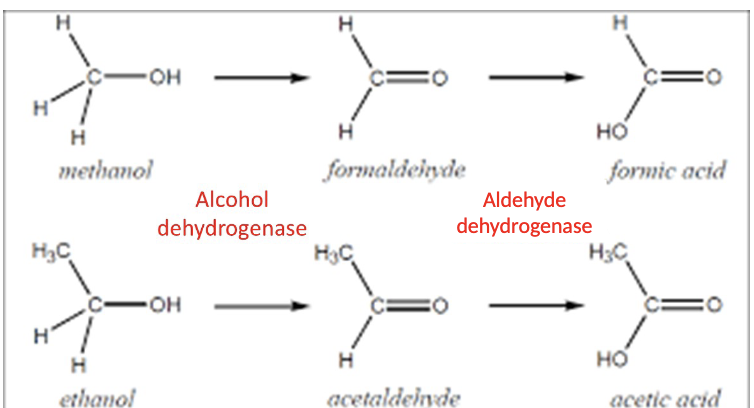
Oxidation
same enzyme can catalize different substances
for a given substances there can be a sequence of multiple reactions, with intermediate metabolites in Phase 1
multiple enzymes may act on the same compound
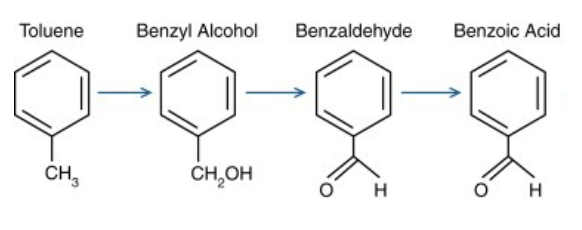
toulene phase 1 reactions
• From methylbenzene (toluene) to benzenecarboxylic acid
(Benzoic acid):
-The P450 enzyme CYP2E1 is the most active in oxidizing toluene
to forming benzyl alcohol. Other P450 enzymes, CYP2B6,
CYP2C8, CYP1A2, and CYP1A1, also contribute to the benzyl
alcohol formation.
- From alcohol to aldehyde and then to carboxylic acid, by ADH
and ALDH enzymes
Benzene
It is not metabolized into toulene but metabolized in the liver into hydroxylated and ring opened products some of which are toxic
oxidation of para position on benzene
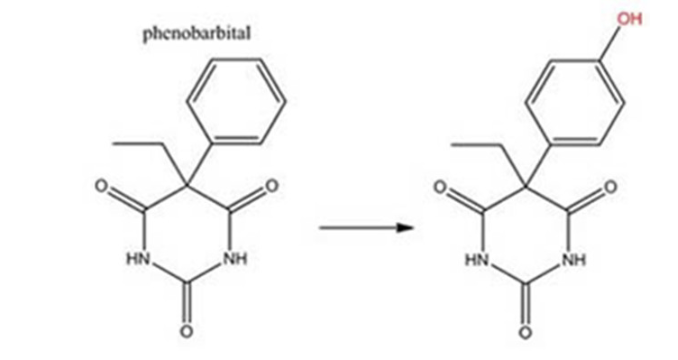
Phenobarbital
Reductive Reactions
Carbonyl, nitro, and azo groups can be
reduced by reductases.
1. Carbonyl groups (C=O) alcohols
2. Nitro ( -NO2) is reduced to amino
derivatives.
3. Azo groups (-N=N-) is reduced to amino
derivatives.
hydroxul and amino functional groips can go onto Phase II
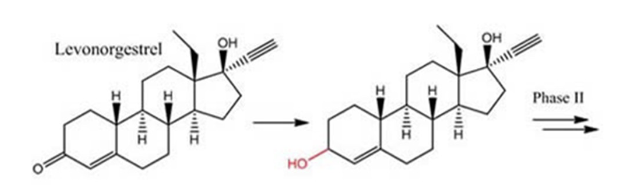
Reduction of Levonorgestreal
Phase I reductive biotransformation of the oral contraceptive
levonorgestrel – the morning-after pill. A Form of progestin used to stop
or delay ovulation and thickens the cervical mucus. It may prevent
implantation.
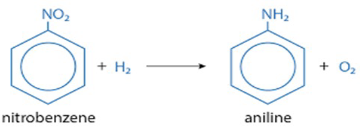
Nitrobenzene
• The most common exposure is at workplaces that use nitrobenzene to
produce dyes, drugs, pesticides or some types of rubber. Exposure can
occur through the air you breathe or through touch.
• Exposure pathways:
1. Breathing it in air at work, or near factory sites where nitrobenzene is used or
made, or near hazardous waste sites.
2. Drinking water with nitrobenzene in it. This can occur near hazardous waste sites.
3. Touching materials made with nitrobenzene.
4. Eye Contact by splashing the chemical in your eyes if you work where nitrobenzene
is used
• Toxicity, all routes: It can cause skin and eye irritation, decrease blood's
ability to carry oxygen (methemoglobinemia), cause liver injury and
reproductive toxicity. It is possible carcinogenic in humans.
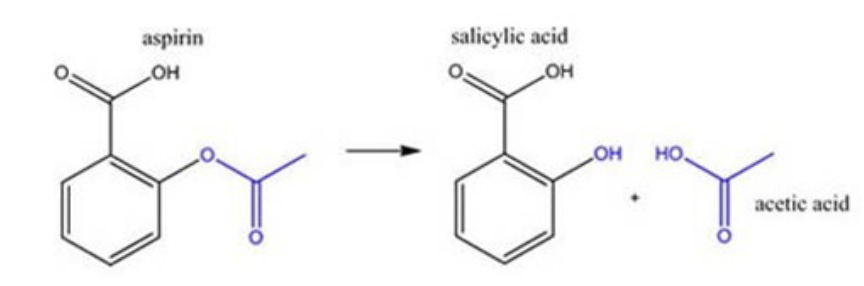
Aspirin Hydrolysis
Aspirin (acetylsalicylic acid) is hydrolyzed into salicylic acid in the
stomach, in the intestinal mucosa, in the blood and mainly in the liver.
• Salicylic acid is the active metabolite responsible for most anti-
inflammatory and analgesic effects.
• Acetylsalicylic acid, the parent compound, is active for the antiplatelet-
aggregating effect. Acetylsalicylic acid may also have anti-inflammatory
effects.
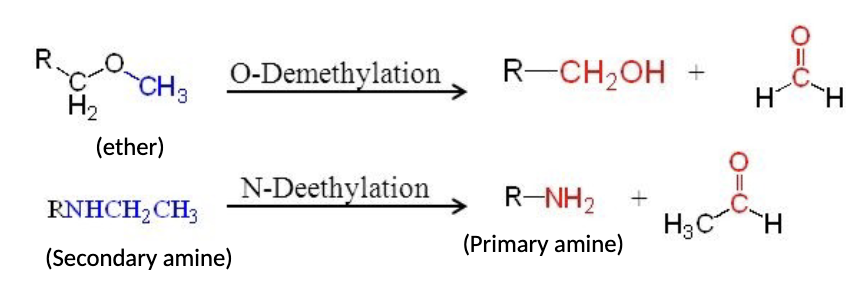
Unmasking
P450 Enzyme System
• The human body has 57 genes encoding cytochrome P450 (CYP)
enzymes, which are a group of enzymes primarily involved in drug
and xenobiotic metabolism, with some also playing roles in
endogenous processes.
• Majority of P450 enzymes catalyze oxidation reactions, others also
catalyze reductions, rearrangements and other non-redox reactions.
CYP450 in Oxidative Reaction (1)
are the most common. These enzymes are hemoproteins (i.e. they contain
heme and iron) and involve molecular oxygen (O2). CYP450 enzymes belong to a class of enzymes known as monooxygenases, also called mixed function oxidases.
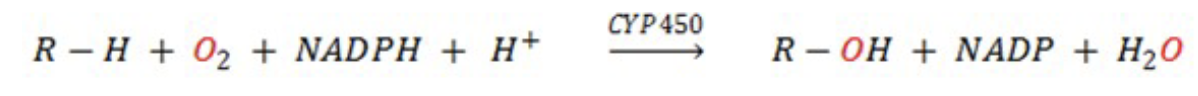
CYP45- in Oxidative Reactions (2)
• Accessible carbons are readily oxidized. For
instance,
1. Unhindered methyl substituents are often oxidized
to form alcohols, which can undergo further
oxidation to the carboxylic acid.
2. Other carbons such as carbon atoms in the alpha
position to a heteroatom (any atom that is not
carbon or hydrogen) can also undergo oxidation
(alpha oxidation).
3. On a benzene group, para position is often favored.
• Note:
• Epoxygenaseses derive The three-membered ring
epoxide from alkenes.
• Epoxides are hydrolyzed by soluble epoxide
hydrolase (sEH) in the liver into corresponding
diols.
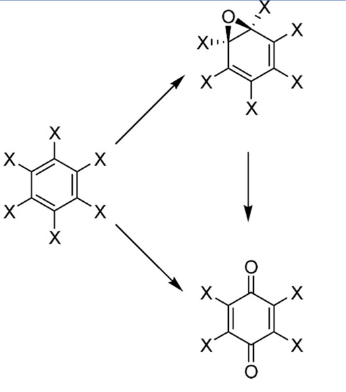
CYP450 in Oxidative Reaction (3)
Hexachlorobenzene was used as a fungicide in the
United States from 1960s to 1984.
• Oxidative degradation Intermediates,
hexachlorobenzene epoxide and 1,4-
tetrachloroquinone can rapidly arylate cellular
macromolecules
• Benzoquinones may generate reactive oxygen
species.
• Tetrachloro-p-benzoquinone (TCBQ) is one of the
most electrophilic quinones because the four
chlorine substituents strongly withdraw electron
density from the quinone ring.
Adducts to CYP3A4
TCBQ forms covalent adducts with accessible cysteine residues in
CYP3A4
• Structural disruption
• Loss of catalytic activity
• Note: This typically does not involve direct binding to Cys442 at the
active center, but rather other accessible cysteines.
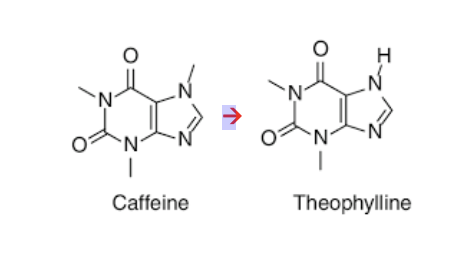
Oxidative demethylation of caffeine
• Oxidative N-demethylation reaction catalyzed by cytochrome P450
(CYP) enzymes
• Caffeine is a more potent central nervous system (CNS) stimulant,
while theophylline is more specialized for relaxing bronchial muscles
(treating asthma)
CYP450 in reductive reactions
• Under certain conditions, particularly anaerobic conditions, many cytochrome P450s can
function as a reductases. The most well-recognized reaction in this regard is probably
reductive dehalogenation.
• Cytochrome P450 could catalyze the reductive removal of halogens from
polyhalogenated alkanes such as hexachloroethane (188), to yield the corresponding
carbon-based radical. The radical would then undergo a second one-electron addition,
to yield the carbanion. Elimination of a chloride ion or the addition of a proton yielded
the observed products, tetrachloroethylene (189) and pentachloroethane (190),
respectively.