triple physics paper 1 all flashcards
1/120
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
121 Terms
What is kinetic energy (Ek)?
The energy stored in moving objects. Stationary objects have none.
What is elastic potential energy (Ee)?
The energy stored in a stretched, squashed, or compressed object.
When is work done?
Whenever energy is transferred from one store to another.
What are the different types of energy transfer?
Mechanical - by a force moving an object
Electrical - by an electric current doing work
Heating - by a temperature difference between two objects
What is spring extension?
How much a spring stretches when a force is applied. The elastic potential energy equation only holds true providing the spring or elastic is not stretched past the limit of proportionality.
What is gravitational potential energy (GPE)?
The energy stored in an object due to its position above the Earth’s surface (due to gravity acting on an object).
What is thermal energy?
The energy stored in the particles of an object due to its temperature.
What is the law of conservation of energy?
Energy can be transferred usefully, stored or dissipated but it cannot be created or destroyed.
What is a system? What is a closed system?
An object or a group of objects. In a closed system, no energy can enter or leave so the mass and energy of the system never changes.
What does friction do?
Releases thermal energy / wastes energy.
What does it mean if energy is dissipated?
Energy is ‘wasted’ / spreads out to the surroundings (e.g. thermal from friction)
How do you reduce unwanted energy transfers / energy dissipation in a pendulum?
Using a lubricant (e.g. oil) on the fixed point
Remove the air particles from around the pendulum. Friction with air particles happens in many systems so keep this in mind.
What is power?
The rate at which energy is transferred or the rate at which work is done.
What is 1 Watt?
An energy transfer of 1 Joule per second
What is efficiency?
It tells us what fraction of inputted energy is transferred to useful forms of energy.
What are the factors that affect how quickly a building cools down?
Thermal conductivity: the higher the thermal conductivity of a material, the higher the rate of energy transfer by conduction across that material.
To reduce heat loss, we need to construct buildings using materials with low thermal conductivity (e.g. double glazed windows, insulated walls, loft insulation) / build the building with thick walls.
What is energy used for?
Transport, electricity generation and heating
Fossil fuels advantages + disadvantages
Reliable
Releases a lot of energy (so good for transport such as planes)
Cheap
Versatile (can be used for transport, electricity generation, and heating)
Burning fossil fuels releases carbon dioxide + other pollutants
Will run out
Nuclear power advantages + disadvantages
Once running, it releases no carbon dioxide
Very reliable + on demand
Contains dangerous radioactive materials - accidents disastrous
Dismantling a nuclear power plant takes years and is expensive
Generates radioactive waste that has to be stored for thousands of years
Describe the switch to renewables in the UK
In the 1970s, scientists began to realise that the carbon dioxide emissions from human activity were leading to climate change
However, there was political factors (politicians didn’t care), economic factors (fossil fuels were cheap + giving them up reduces jobs), ethical and social factors
From 2005, we’ve been reducing emissions and now the UK is using more wind power, solar power and biofuel
Issues with renewables
Bad reliability (e.g. wind farms rely on wind / solar power relies on sun)
Hydroelectric is reliable but destroys habitats when dams are built
Tidal is reliable but can be harmful to wildlife
Wave power is reliable but is experimental currently
Most (apart from biofuels) provide electricity, which is no good for most transport - as transport mainly uses fossil fuels
What is renewable energy?
An energy resource that is being (or can be) replenished as it is used.
What are the main energy resources available on Earth?
Fossil fuels
Nuclear power
Biofuel
Wind
Hydroelectric
Geothermal
Tidal
Solar
Waves
Future of the UK energy
Renewables providing the bulk of electricity
Nuclear providing our constant baseload
Gas providing electricity in emergency
Required Practical: specific heat capacity
Pour vegetable oil into a beaker and measure the mass of the oil using a balance.
Place a thermometer and immersion heater into the oil. Read the starting temperature.
Wrap the beaker in insulating foam.
Connect a joulemeter and power pack to the immersion heater. Ensure the immersion heater is fully submerged.
Time for 30 minutes with a stopwatch.
Read the number of joules of energy passed into the immersion heater and the final temperature of the oil. Avoid parallax error.
How are the particles arranged in a solid?
Very close together
Regular pattern
Particles vibrate but don’t move around
How are the particles arranged in a liquid?
Close together
Not arranged in a regular pattern
Can move around each other
How are the particles arranged in a gas?
Very far apart
Not arranged in any pattern
Moving very rapidly constantly
Explain the density of solids, liquids and gases.
SOLIDS
Very high density
Particles packed closely together so solids have a lot of mass for their volume
LIQUIDS
High density
Particles close together so liquids have a lot of mass for their volume
GASES
Low density
Particles very far apart so only have a small mass for their volume
Required Practical: calculating density
REGULAR OBJECTS
Find the object’s mass using a balance.
Use a ruler to measure the length, width and height of the object, then multiply them together to find the volume.
Use the equation density = mass / volume to find the density.
IRREGULAR OBJECTS
Find the object’s mass using a balance.
Put a beaker under a Eureka can spout and fill the can with water. Water should be dripping; wait for it to stop.
Now place a measuring cylinder under the spout.
Put the object into the Eureka can.
Measure the volume of the water displaced in the measuring cylinder.
Use the equation density = mass / volume to find the density.
What is internal energy?
The total kinetic energy and potential energy of all the particles (atoms and molecules) that make up a system.
How does heating affect internal energy?
Heating increases the energy of the particles in a system, which either raises the temperature of the system or produces a change of state.
Explain changes of state key points and name them.
Mass is always conserved during changes of state
They are physical changes not chemical changes - easily reversible to original properties
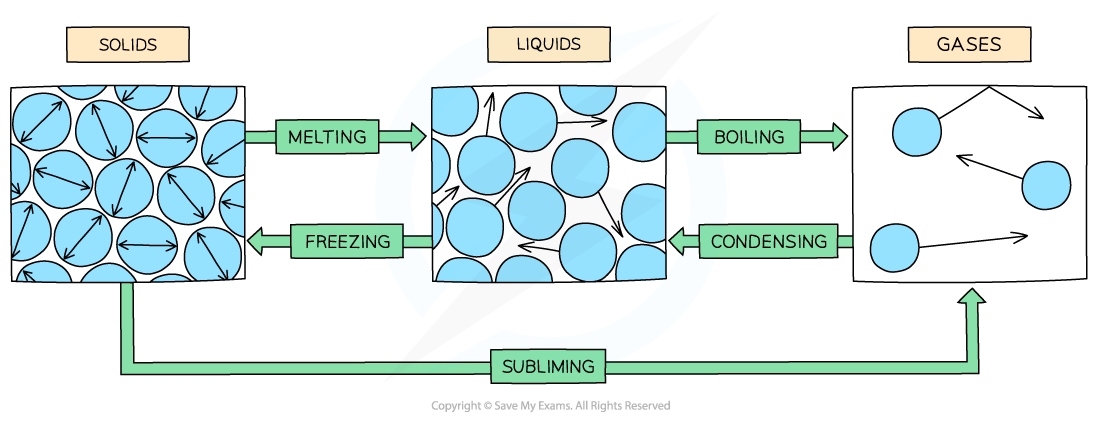
What is the specific heat capacity of a substance?
The amount of energy required to raise the temperature of 1kg of the substance by 1°C.
Why does the temperature of a substance remain constant when it changes state?
The energy supplied is used to weaken / break the forces between the particles rather than heat the substance. It changes the internal energy but not the temperature.
What is the specific latent heat of a substance?
The amount of energy required to change the state of 1kg of the substance with no change in temperature.
What is the specific latent heat of fusion?
The energy required to change 1kg of a substance from a solid to a liquid with no change in temperature.
What is the specific latent heat of vaporisation?
The energy required to change 1kg of a substance from a liquid to a vapour with no change in temperature.
What in a heating / cooling graph indicates a change of state?
When the temperature stops rising / the line is straight, a change of state is happening (see picture example).
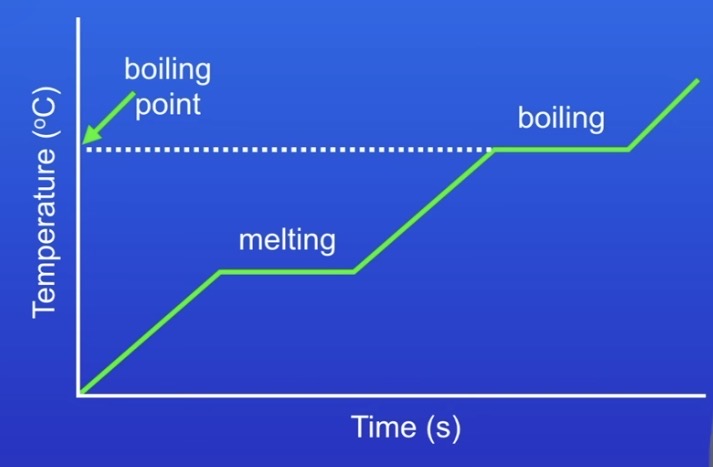
What causes pressure in a gas?
The particles colliding with the walls of the container that the gas is held in.
How can we increase gas pressure?
Increase the number of collisions per second
Increase the energy of each collision
Decrease container volume
How does a higher temperature affect gas pressure?
More kinetic energy therefore there’s more collisions per second with more energy, so higher pressure
How big is an atom and how big is a nucleus?
Radius of nucleus of atom: less than 1/10000 of an atom
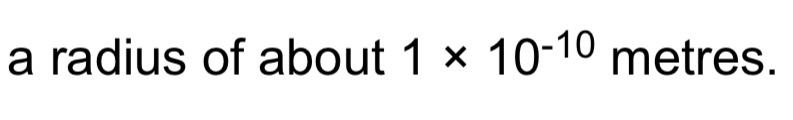
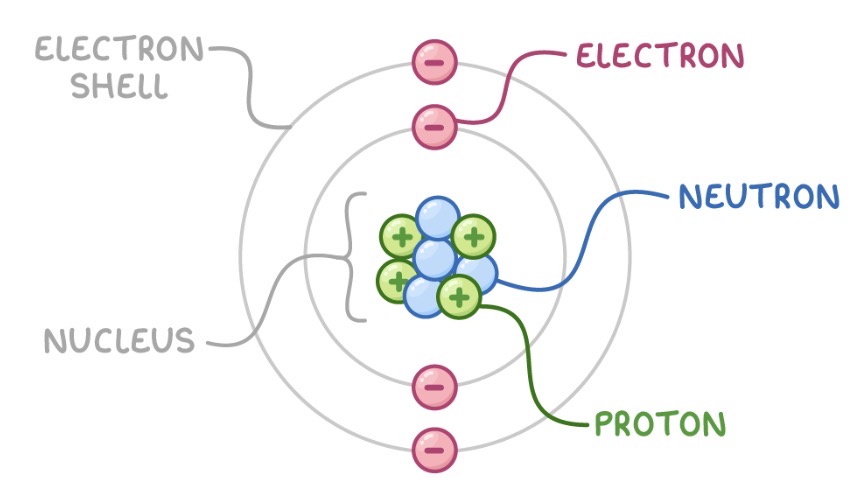
What is an atom composed of?
Nucleus - containing protons and neutrons, so positive
Electron shells
Electrons are arranged in different distances from the nucleus (different energy levels)
Energy levels furthest from the nucleus have the highest energy
How do electrons change energy levels?
If the atom absorbs electromagnetic radiation, an electron can move to a higher energy level. If it emits electromagnetic radiation, it will return to a lower energy level.
What charge does an atom have?
The number of electrons is equal to the number of protons in the nucleus. They cancel out so no charge.
What is a mass number and an atomic number?
MASS NUMBER
Total number of protons and neutrons added together.
ATOMIC NUMBER
All atoms of a particular element have the same number of protons. This is called its atomic number.

What is an isotope?
Atoms of the same element with different numbers of neutrons.
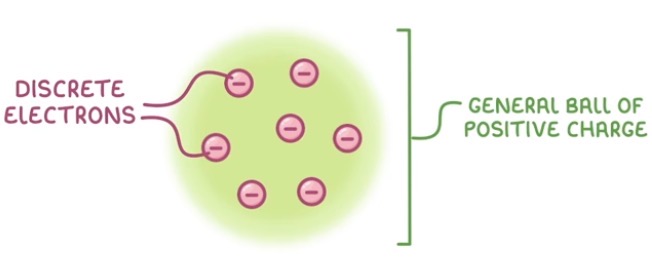
What did JJ Thompson discover?
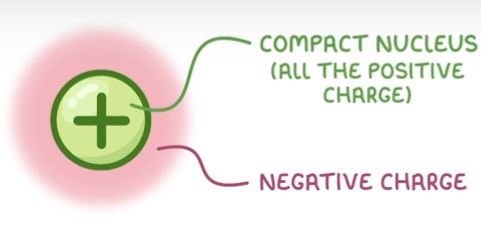
1897 - JJ Thomson found that atoms contain tiny negative particles (electrons). He proposed the plum pudding model - that the atom was a ball of positive charge, with negative electrons embedded it.
What was the nuclear model and how was it discovered?
Tiny alpha particles (+) were fired at a piece of thin gold foil (alpha scattering experiment). This led to the conclusion that the mass of an atom was concentration at the centre (nucleus) and that it was charged.
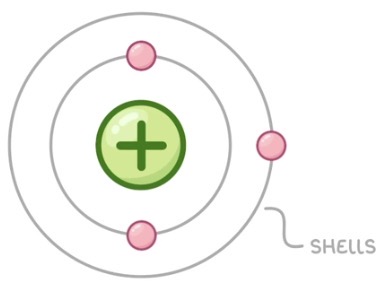
What did Niels Bohr propose?
Electrons orbit the nucleus at specific distances (energy levels). Bohr’s work agreed with other scientist’s experiments.
How was the discovery of protons and neutrons made?
Scientists found that the positive nucleus charge was due to tiny particles, protons. About 20 years after the nuclear model, James Chadwick discovered that the nucleus also contained neutrons.
What causes radioactive decay?
Some isotopes have an unstable nucleus. To become stable, the nucleus gives out unpredictable radiation. Nuclear equations are used to represent radioactive decay.
What is activity?
The rate at which a source of unstable nuclei decay. It is measured in becquerel (Bq). 1 Bq = 1 decay per second.
What is count-rate?
Number of decays per second
What are the 4 types of radiation?
Nuclear radiation emitted may be:
Alpha particle (α) - consists of two neutrons and two protons, same as a helium nucleus
Beta particle (β) - a high speed electron ejected from the nucleus, as a neutron turns into a proton
Gamma ray (γ) - a type of electromagnetic radiation from the nucleus
Neutron (n)
Range in air, penetrating power, and ionising power of different radiation
Alpha particles
Range in air: around 5cm
Penetrating power: stopped by a single sheet of paper
Ionising power: highly ionising
Beta particles
Range in air: around 15cm
Penetrating paper: a few mm of aluminium
Ionising power: quite ionising
Gamma rays
Range in air: several metres
Penetrating power: several cm of lead
Ionising power: weakly ionising
What happens to an atom when it emits radiation?
Atoms lose electrons from their outer energy level and form ions.
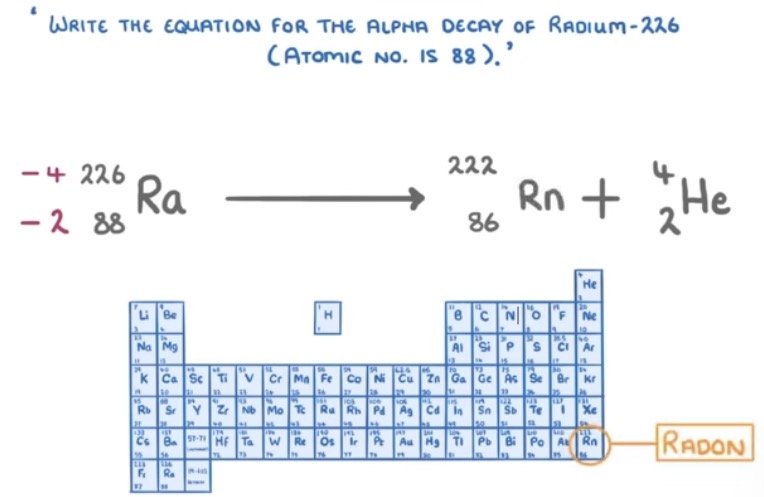
How is an alpha particle represented in a nuclear equation?

What are the rules of alpha decay?
The atomic number decreases by 2. The mass number decreases by 4.
How is a beta particle represented in a nuclear equation?
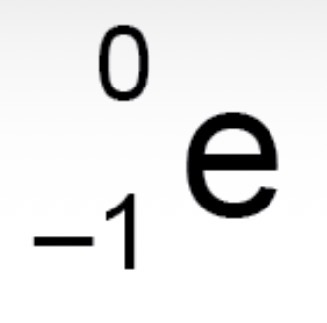
What are the rules of beta decay?
The atomic number increases by 1. The mass number does not change.
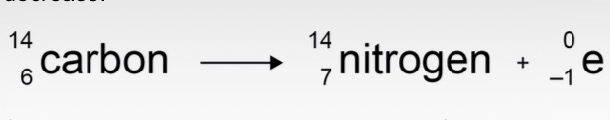
What are the rules of gamma decay?
The atomic and mass numbers have no change.
What is a half-life of a radioactive isotope?
The time it takes for the number of nuclei of the isotope in a sample to halve.
It is also the time it takes for the count-rate / activity from a sample containing the isotope to halve.
How to determine half life?
You will get given a half-life and an initial count-rate. From there, halve the count-rate every half-life to determine the final answer. See example picture.
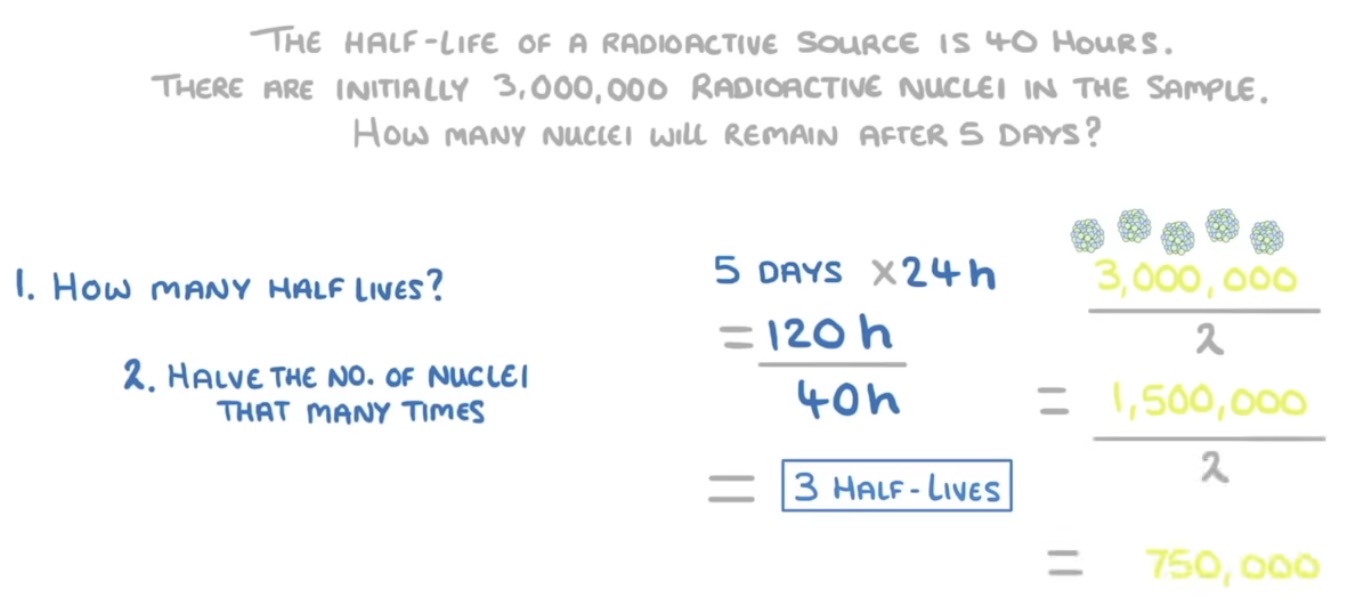
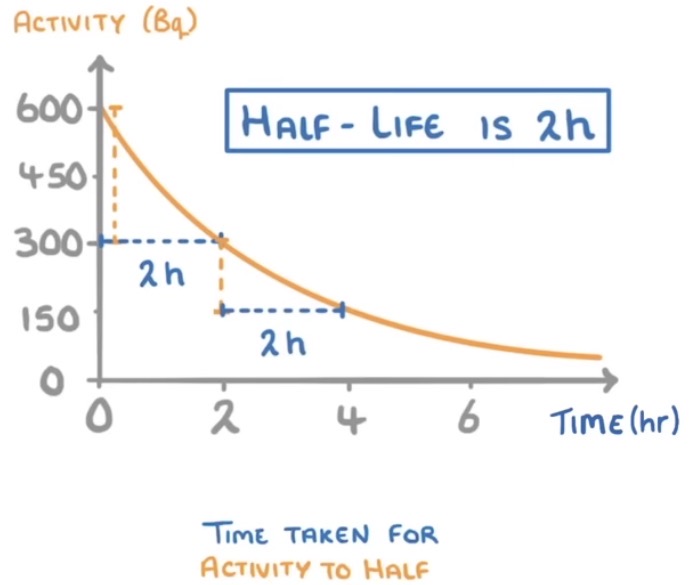
How to determine half-life from a graph?
See the time it takes for the activity to halve. See example picture.
What is irradiation?
Exposing an object to nuclear radiation. The irradiated object does not become radioactive, as it does not come in contact with the radioactive isotope itself.
What are the risks + precautions associated with ionising radiation?
Increases risk of cancer
Gloves can protect against alpha radiation
Beta and gamma radiation can usually be reduced by using a lead apron
What is radioactive contamination?
When unwanted radioactive isotopes end up on other materials.
What does the level of hazard depend on for radioactive contamination?
The type of radiation (e.g. alpha radiation are strongly ionising so very dangerous but easily stopped by skin, etc)
Cell circuit symbol
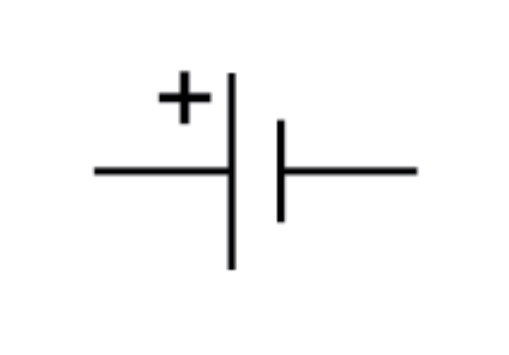
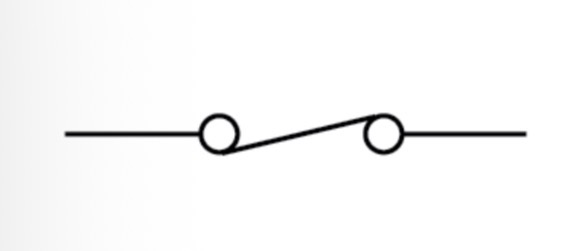
Switch (open) circuit symbol
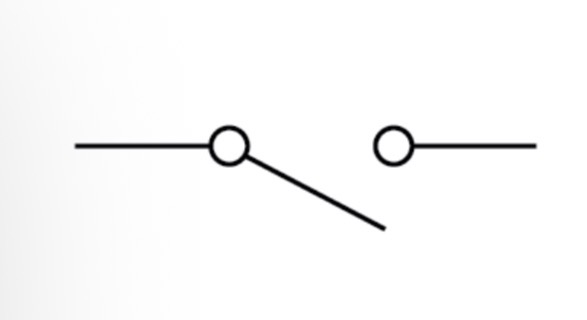
Switch (closed) circuit symbol
Battery circuit symbol
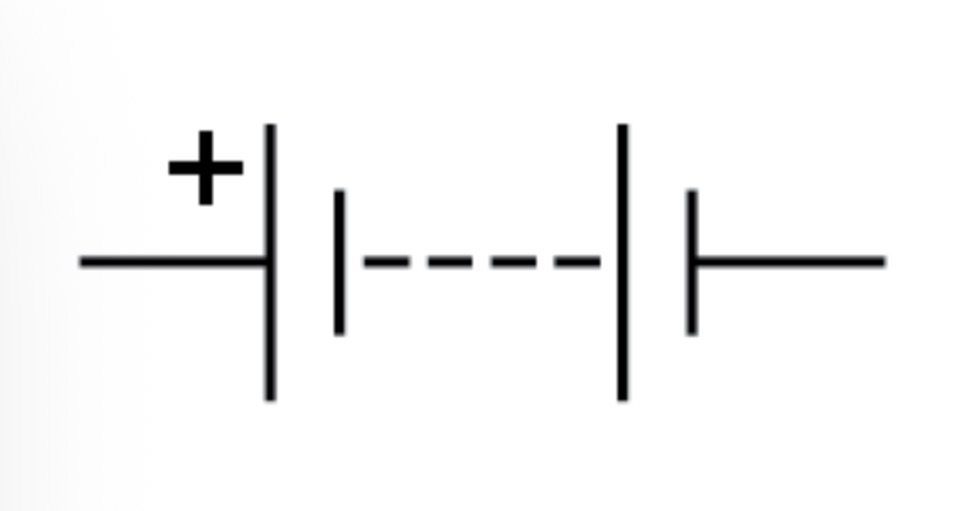
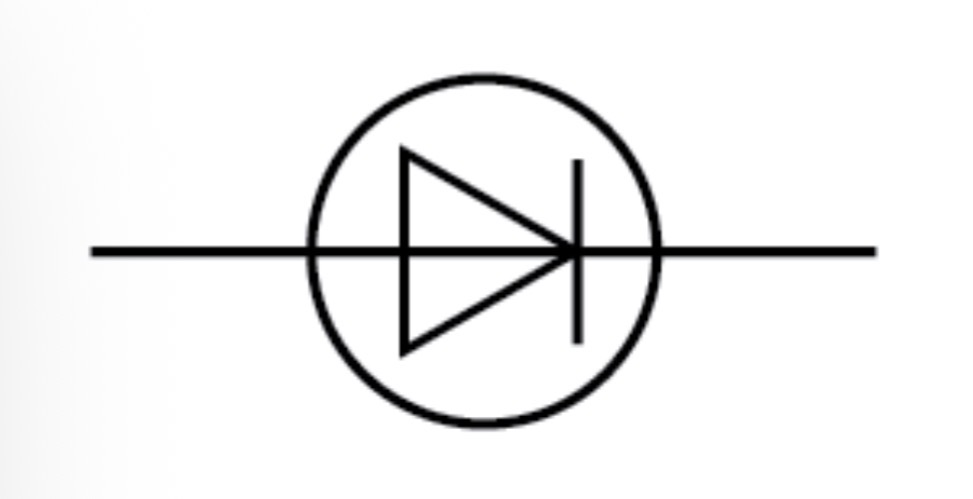
Diode circuit symbol
Resistor circuit symbol

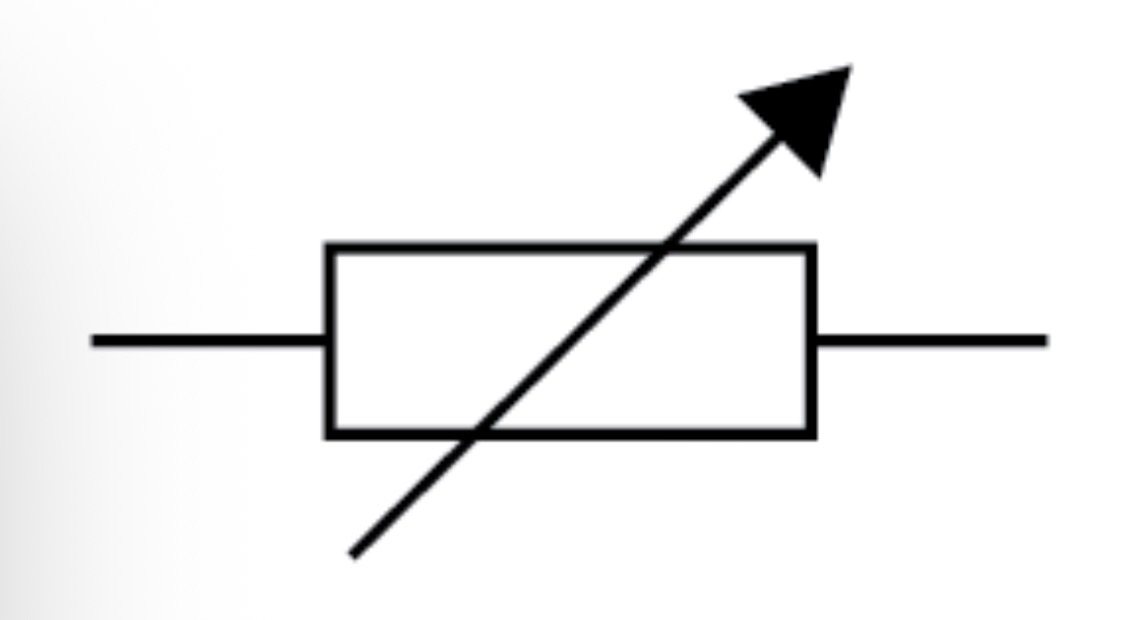
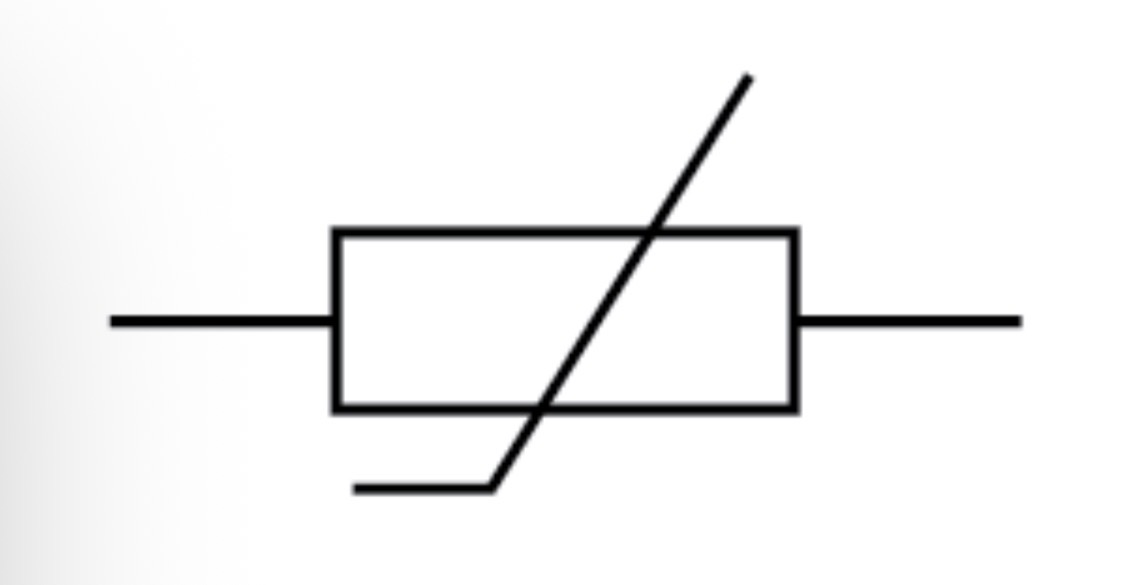
Variable resistor circuit symbol
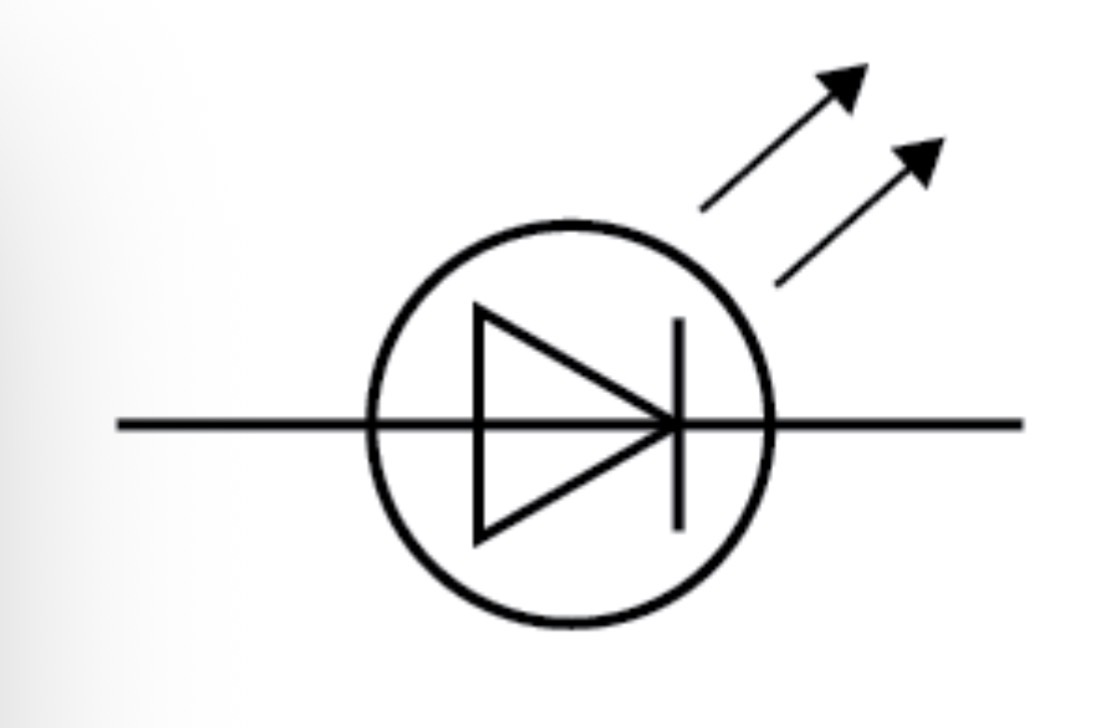
LED circuit symbol
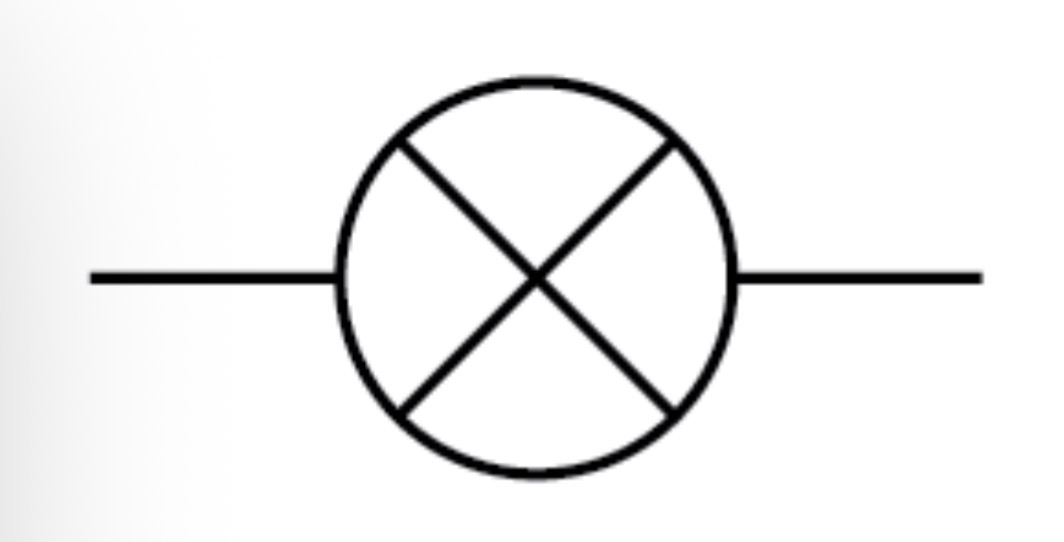
Lamp circuit symbol
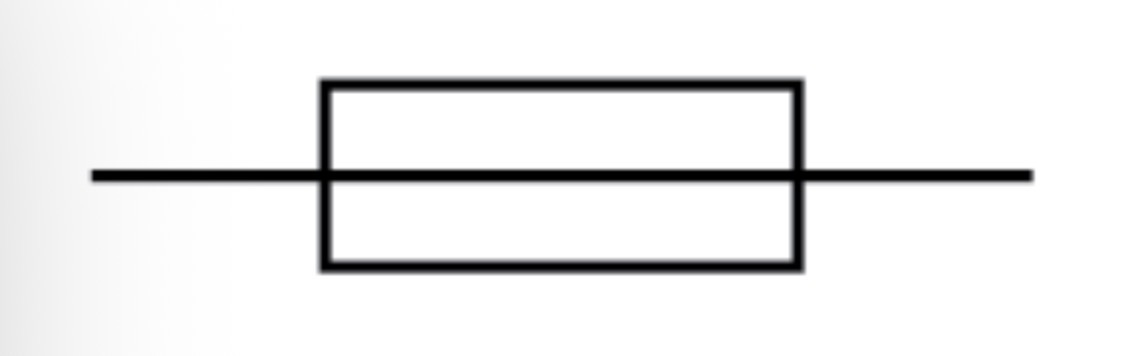
Fuse circuit symbol
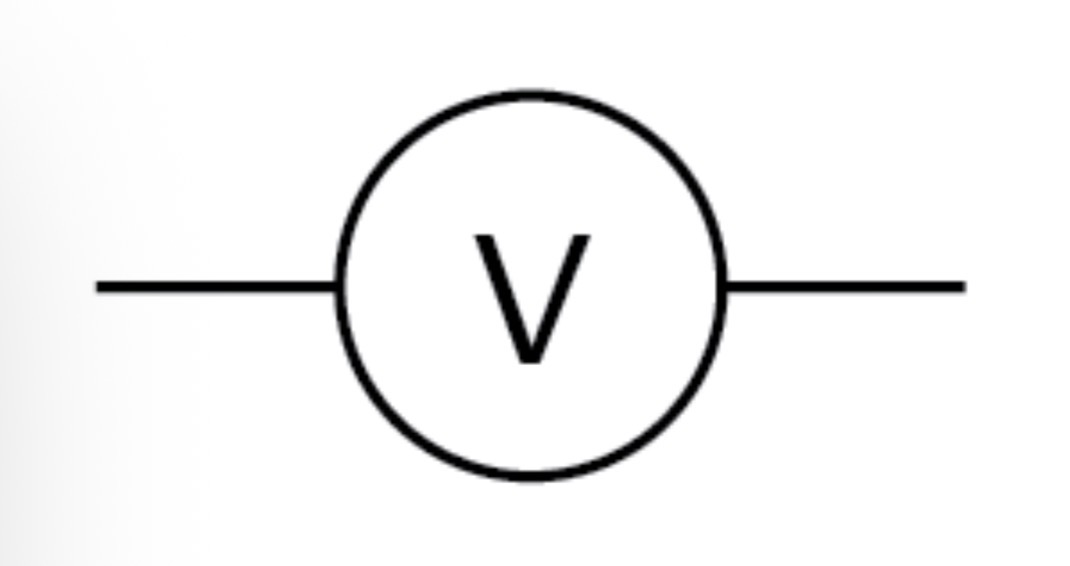
Voltmeter circuit symbol
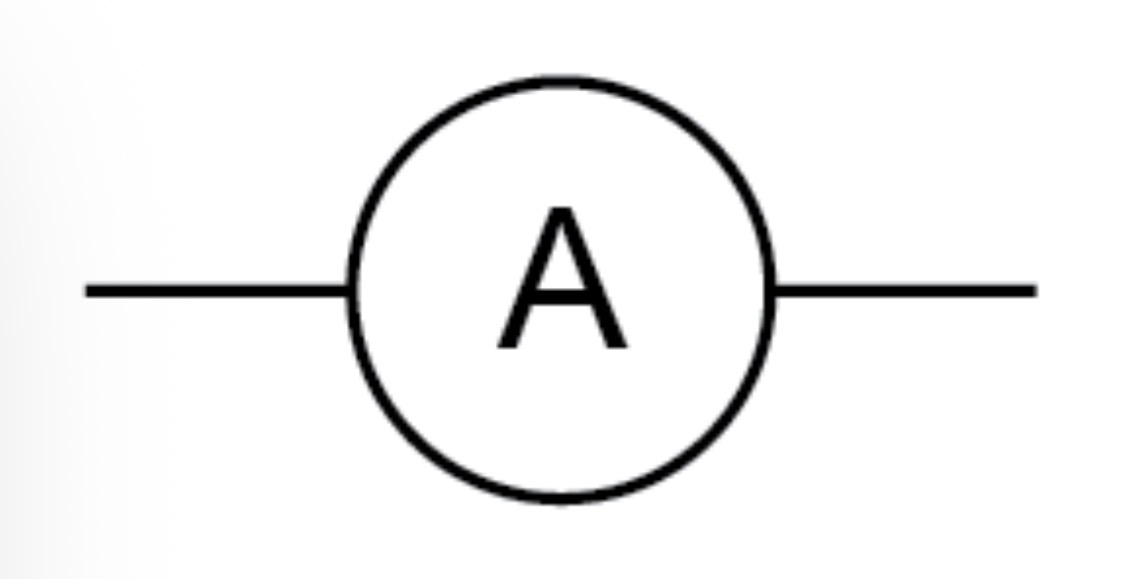
Ammeter circuit symbol
Thermistor circuit symbol
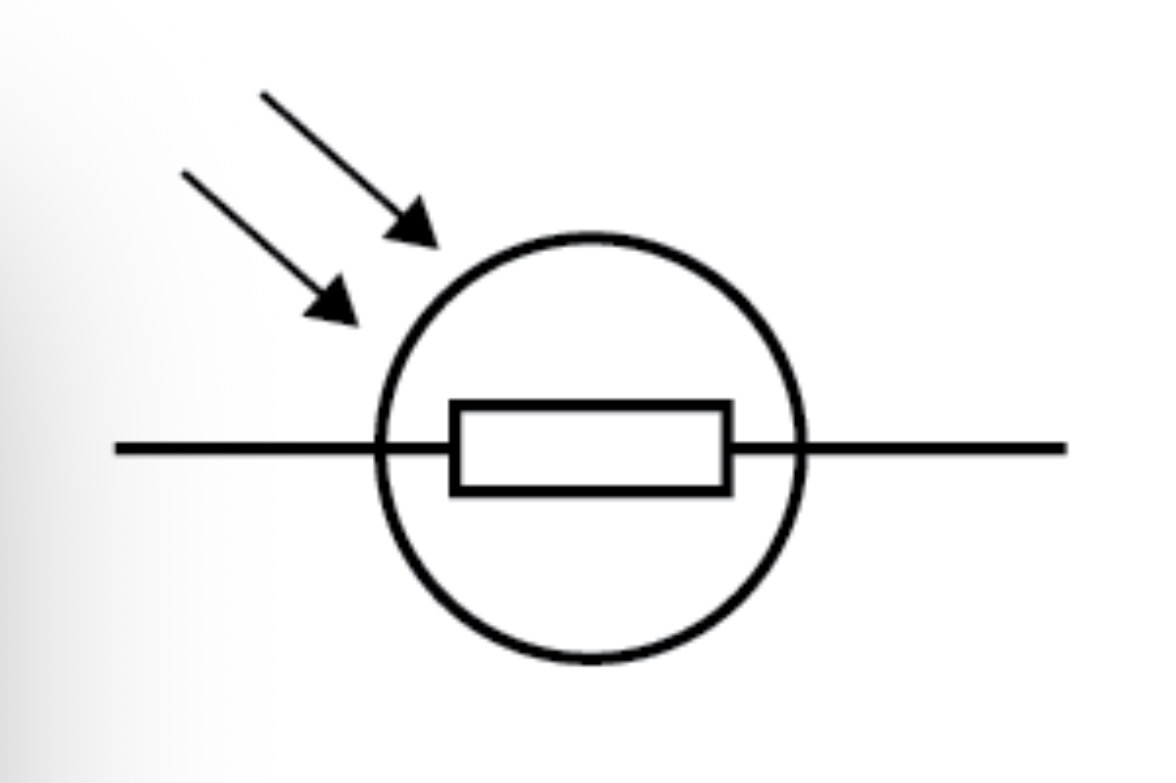
LDR circuit symbol
What is an electric current?
The rate of flow of electrical charge. It flows from negative end of the cell to the positive end in a circuit. However, it’s drawn in the opposite direction: conventional current.
What is a current of 1 Ampere?
1 coulomb of charge flowing per second.
What does a closed circuit need for electrical charge to flow?
A source of potential difference
What is resistance?
The potential difference required to drive a current through a component. The greater the resistance of the component the smaller the current for a given potential difference across the component.
What are the key points about series circuits?
Current is the same all the way around the circuit
Potential difference / voltage of the cell is shared between the components in the circuit - not always equally
Total resistance is the sum of the resistors in the circuit
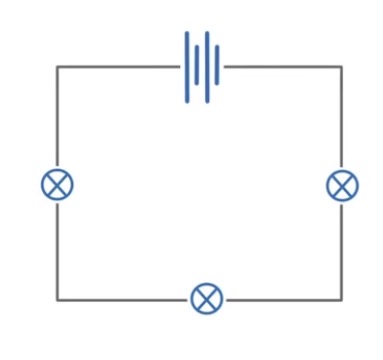
What are the key points about parallel circuits?
Total current in the circuit is the sum of the current in each branch
Potential difference / voltage is the same across each component
The total resistance of two resistors is less than the resistance of the smallest resistor in the circuit
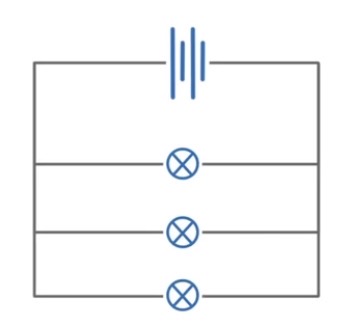
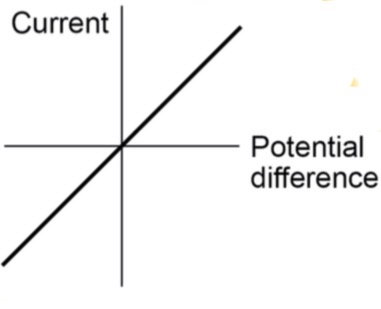
Explain what is meant by an ohmic conductor.
Current is directly proportional to the potential difference across the resistor. Resistance remains constant and as the current changes. The resistance will only remain constant if the temperature is constant.
Explain the graph of a filament lamp / anything that gets hot.
The current through the lamp isn’t directly proportional to potential difference. This is because the filament gets hot, which causes resistance to increase (at high temp, the atoms in the filament vibrate more - which means the electrons in the current collide more with the atoms, so more energy is needed to push the current through the filament). A filament lamp is not an ohmic conductor.
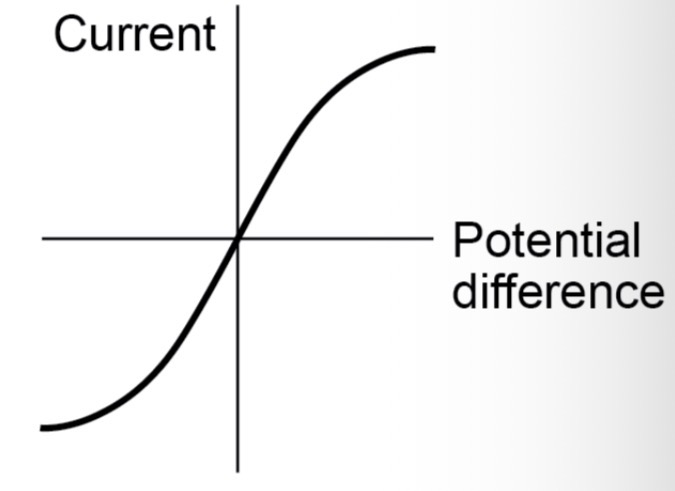
Explain the graph of a diode / LED.
The current through the diode flows in one direction only, because the diode has a very high resistance in the reverse direction. LEDs are like regular diodes, except they give off light when a current flows through.
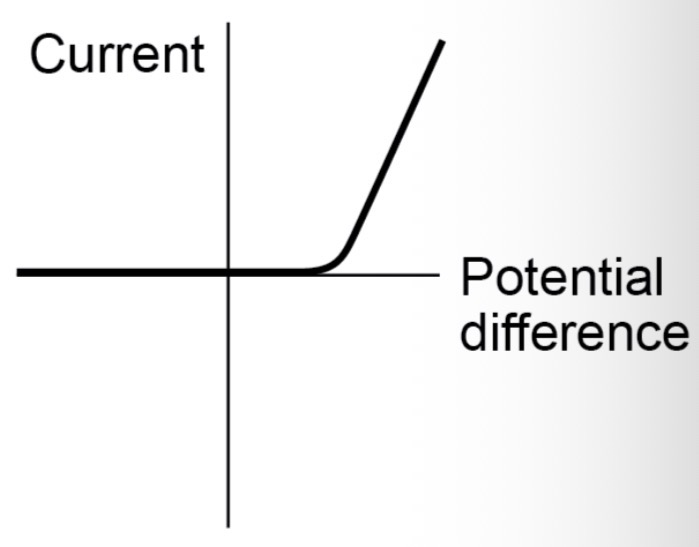
How does the resistance of a light-dependent resistor (LDR) change?
The resistance of an LDR decreases as light intensity increases. An LDR could be used for lights that switch on automatically when it gets dark (e.g. street lamps).
How does the resistance of a thermistor change?
The resistance of a thermistor decreases as temperature increases. Thermistors are useful as thermostats (which turn devices on / off depending on the temperature). For example, if computers get too hot, a fan cools it. A thermistor can change the speed of the fan depending on the temperature of the computer.
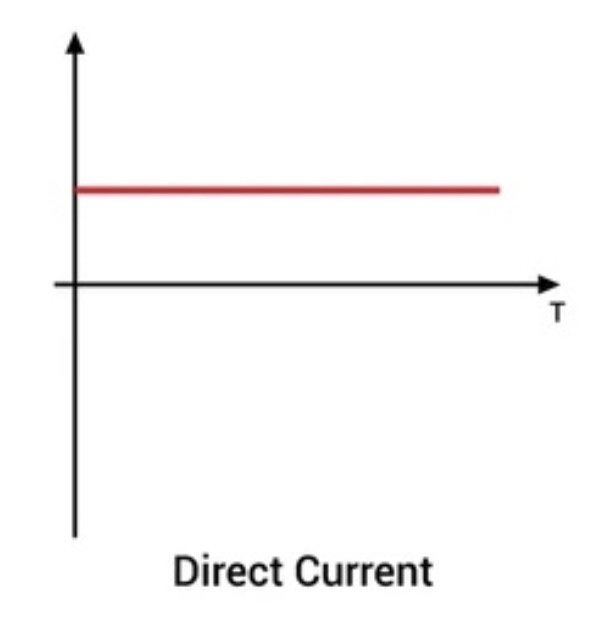
What is meant by Direct Current (DC)?
The current in a cell is DC - meaning the electrons only travel in one direction.
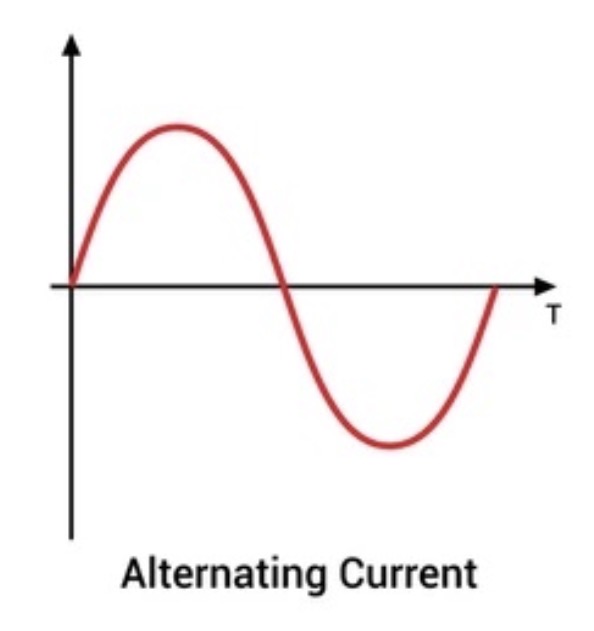
What is meant by Alternating Current (AC)?
The current is constantly changing direction. The benefit of AC is that it’s very easy to use a transformer to change the potential difference. We use transformers a lot when electricity is transferred to homes.
What is mains electricity in the UK?
An AC supply. In the UK the domestic electricity supply has a frequency of 50Hz and is 230V.
What is frequency of AC current?
The number of cycles per second (switching from positive to negative and back to positive again).
What two categories can we put most appliances into?
Electrical energy into kinetic energy of electric motors (e.g. blender)
Electrical energy into thermal energy of heating devices (e.g. iron) - these usually have a much higher power rating than kinetic appliances
Ones that do both (e.g. hairdryers / washing machines)