Isomerism
1/62
Earn XP
Description and Tags
Quick revision for isomerism, specifically for organic chemistry. Suitable for revision for IAT, JEE, NEET, etc. Question mode: Flashcards only. Answer mode: Answer with definition. Spaced Repetition recommended but not necessary.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
All isomers have the same molecular formula. True or false?
True
All isomers have same structural formula. True or false?
False, only stereoisomers have same structural formula
All isomers have the same degree of unsaturation. True or false?
True
What is the formula for calculating degree of unsaturation in a compound, given the number of carbons, nitrogens, halogens, and hydrogens?
DoU=22C+2+N−X−H
DoU=2(2C+2+N)(expected number of Hydrogens)−(X+H)(actual number of Hydrogens (and halides))
For a compound to be saturated, there is one more hydrogen in a molecule when nitrogen is present.
How do you calculate degree of unsaturation in a compound, given the number of double bonds, triple bonds, and rings in the structure?
Add them up
double bond = 1
triple bond = 2
ring = 1
All isomers have same physical and chemical properties. True or false?
False, isomers can have different chemical and/or physical properties
What is chain isomerism?
same functional group, different chain of carbons (parent + side chains)
What is position isomerism?
same functional group, same chain of carbons, different position of substituent/multiple bond/functional group
What is ring chain isomerism?
they have different modes of linking of carbon attom (open chain / closed chain)
What are functional group isomers?
different functional groups.
What are metamers?
same molecular formula, different arrangement of groups around polyvalent atom or functional group
polyvalent atom/group = not monovalent (connects with other people in more than one way)
What is tautomerism?
isomers keep converting into one another and remain in equilibrium
What are the two conditions for tautomerism?
electron withdrawing group must be present
at least one alpha hydrogen must be present at an odd numbered position
How do you check for the major product in a base catalysed tautomerism reaction?
check the stability of the reaction intermediate (remove alpha hydrogen and check stability)
How do you check for the major product in an acid catalysed tautomerism reaction?
check the stability of the double bond in the products
In space tautomerism, how can you count how many alpha hydrogens are replaceable?
think about mechanism of acid-catalysed space tautomerism, at each carbon that acquires negative charge, alpha hydrogens are replaceable (basically all odd-numbered carbons basically)
What does “enol content” mean?
the percentage of enol form in equilbrium mixture of keto and enol form tautomers
In keto and enol form tautomers, which is more stable, keto or enol?
keto
What are the two types of stereoisomers?
conformational
configurational
What is the condition for conformational isomerism to be present?
three continuous σ bonds must be present
What is Steric/Van der Waal’s strain?
repulsion between two non-bonded atoms or groups in conformational isomerism
What is the difference between conformational isomerism and geometrical isomerism?
In conformational isomerism, the energy barrier for free rotation of a bond is very low and achieved at room temperature, hence bonds can rotate easily, showing conformations.
In geometrical isomerism, the energy barrier for free rotation of a bond is much higher and is not achieved at room temperature, hence bonds cannot rotate, and thus we get geometrical isomers.
structure of oxime?
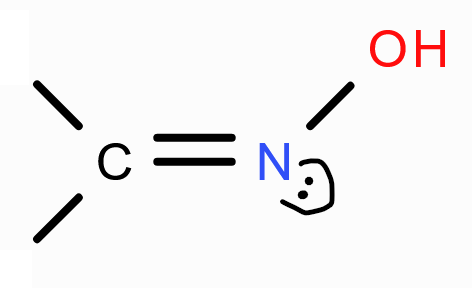
structure of imine?
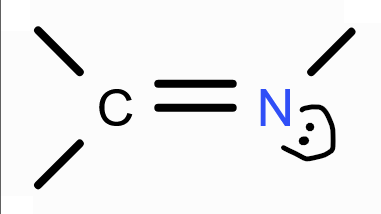
Structure of azo group?
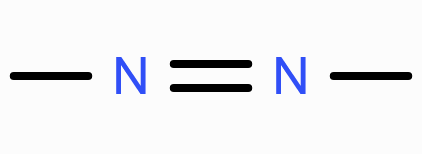
Which aldoximes exhibit geometrical isomerism?
All except formaldoxime
Which ketoximes exhibit geometrical isomerism?
All unsymmetrical ketoximes
Which azo compounds exhibit geometrical isomerism?
all
What is the condition for there to be geometrical isomerism in substituted cyclo alkanes?
At least two sp3 carbons of the ring individually must have two different atoms or groups
What is the condition for there to be geometrical isomerism in substituted cyclo alkenes?
3-7 membered ring: no GI
8-11 membered ring: yes GI, cis is more stable than trans
12+ membered ring: yes GI, trans is more stable than cis
In E-Z system of naming geometrical isomers, what does E mean?
E: Entgegen — high priority groups on opposite sides
In E-Z system of naming geometrical isomers, what does Z mean?
Z: Zusammen — Hi priority groups on same side
What is the next atom rule in CIP rule for determining priority of groups?
When all direct substituent atoms are same, list for each, the atoms they are next connected with, assign priority according to those atomic weights.
Multiple bonds means those atoms get counted that many more times in the list
What is the priority order of groups that are Entgegen and Zusammen?
Z > E
What is the syn-anti naming system of geometrical isomers used for?
azo compounds
aldoximes
ketoximes
What is the DOG/DEO rule for geometrical isomerism?
Different substituents on both carbons
Odd number of pi bonds / rings (in spiro compounds)
Geometrical isomerism shown only
Different substituents on both carbons
Even number of pi bonds / rings (in spiro compounds)
Optical isomerism shown only
How would you calculate the total number of geometrical isomers in a compound when the compound is unsymmetrical?
Total number of GI=2n
where n = the number of restricted rotatory systems about which GI is possible.
How would you calculate the total number of geometrical isomers in a compound when the compound is symmetrical?
Total number of GI=2n−1+2p−1
where n = number of restricted rotatory systems about which GI is possible.
If n is odd, p=2n+1
If n is even, p=2n
In general _______ form of the compound has greater dipole moment than _______ form.
(cis / trans)
In general cis form of the compound has greater dipole moment than trans form.
In general _______ form of the compound has greater boiling point than _______ form.
(cis / trans)
In general cis form of the compound has greater dipole moment than trans form.
Always, _______ form of the compound has greater melting point than _______ form.
(cis / trans)
Always, trans form of the compound has greater dipole moment than cis form.
In general _______ form of the compound has greater stability than _______ form.
(cis / trans)
In general trans form of the compound has greater dipole moment than cis form.
If an optically active compound rotates plane polarized light in the clockwise direction, it is _____________.
(dextrorotatory / leavorotatory)
dextrorotatory
If an optically active compound rotates plane polarized light in the anticlockwise direction, it is _____________.
(dextrorotatory / leavorotatory)
leavorotatory
What is the formula for specific rotation of an optically active compound?
specific rotation α=C⋅lθ
where,
θ is the angle through which PPL is rotated
C is the concentration of the solution
l is the length of the polarimeter tube
What is the condition for optical activity of a compound?
presence of chirality
In R-S configuration in Fischer Projection, what are the rules for determining configuration when the Lowest Priority Group is at vertical position?
VAS — Vertical, Anticlockwise, S
VCR — Vertical, Clockwise, R
In R-S configuration in Fischer Projection, what are the rules for determining configuration when the Lowest Priority Group is at horizontal position?
HAR — Horizontal, Anticlockwise, R
HCS — Horizontal, Clockwise, S
In R-S configuration in Wedge-Dash representation, what are the rules for determining configuration when the Lowest Priority Group is at dash position?
DAS — Dash, Anticlockwise, S
DCR — Dash, Clockwise, R
What are enantiomers?
Optically active isomers which are non-superimposable mirror images of each other
What are diastereomers?
Stereo-isomers (of each other) that are not mirror images of each other.
Diastereomers can be separated by fractional distillation. True or false?
Yes. They have different physical properties.
Enantiomers can be separated by fractional distillation. True or false?
False, they have the same physical properties so the same boiling points.
What are meso compounds?
Compounds with more than one chiral carbon but still optically inactive due to symmetry (internal compensation)
What is the rule(s) for determining D-L configuration of optical isomers?
Draw in fischer projection
put most oxidised group at the top of the chain
put CH2OH/R at the bottom of the chain
Identify the last (bottom-most) chiral carbon
If the OH or NH2 group is on the right side, it is D configuration
If the OH or NH2 group is on the left side, it is L configuration
What is the formula for calculating optically active optical isomers for unsymmetrical molecules?
2n
where n = the number of optical or chiral centres
What is the formula for calculating optically active optical isomers for symmetrical molecules?
n is the number of optical or chiral centres
if n is odd, p=2n+1, number of isomers =2n−1−2p−1
if n is even, number of isomers =2n−1
What is the formula for calculating meso optical isomers for symmetrical molecules?
n is the number of optical or chiral centres
if n is odd, p=2n+1, number of isomers =2p−1
If n is even, p=2n, number of isomers =2p−1
What is the formula for calculating meso optical isomers for unsymmetrical molecules?
0
What is the formula for calculating the total optical isomers for unsymmetrical molecules?
2n
(number of optically active isomers + number of meso compounds)
What is the formula for calculating the total optical isomers for symmetrical molecules with even number of optical or chiral centres?
2n−1+2p−1
where p=2n
(number of optically active isomers + number of meso compounds)
What is the formula for calculating the total optical isomers for symmetrical molecules with odd number of optical or chiral centres?
2n−1
(number of optically active isomers + number of meso compounds)
What is the rule for determining configuration of isomers using Erythro and Threo?
TOES
Threo — opposite sides
Erythro — same side