Biol 112 - McGill 2026
1/593
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
594 Terms
Atomic mass number?
Number of protons + neutrons
Atomic number?
Number of protons
Valence?
Number of unpaired electrons
3 properties of a covalent bond?
1. Covalent bond is made of 2 electrons
2. These 2 electrons are shared (one electron from each atom)
3. Atoms can form bonds until their outermost shell is filled
What properties of an element determine how that element can form bonds with others?
To link atoms together, only electrons are important
What is the difference between a polar and a nonpolar covalent bond?
Polar: Electrons are not share equally, so partial charges exist
Nonpolar: Electrons are shared equally
What about these properties (polar v.s. nonpolar covalent bonds) gives water it's biological properties?
Water (H20) consists of two polar covalent bonds (Hydrogen bonds) which gives it a high surface tension, and a meniscus when water meets a solid surface because of adhesion and cohesion.
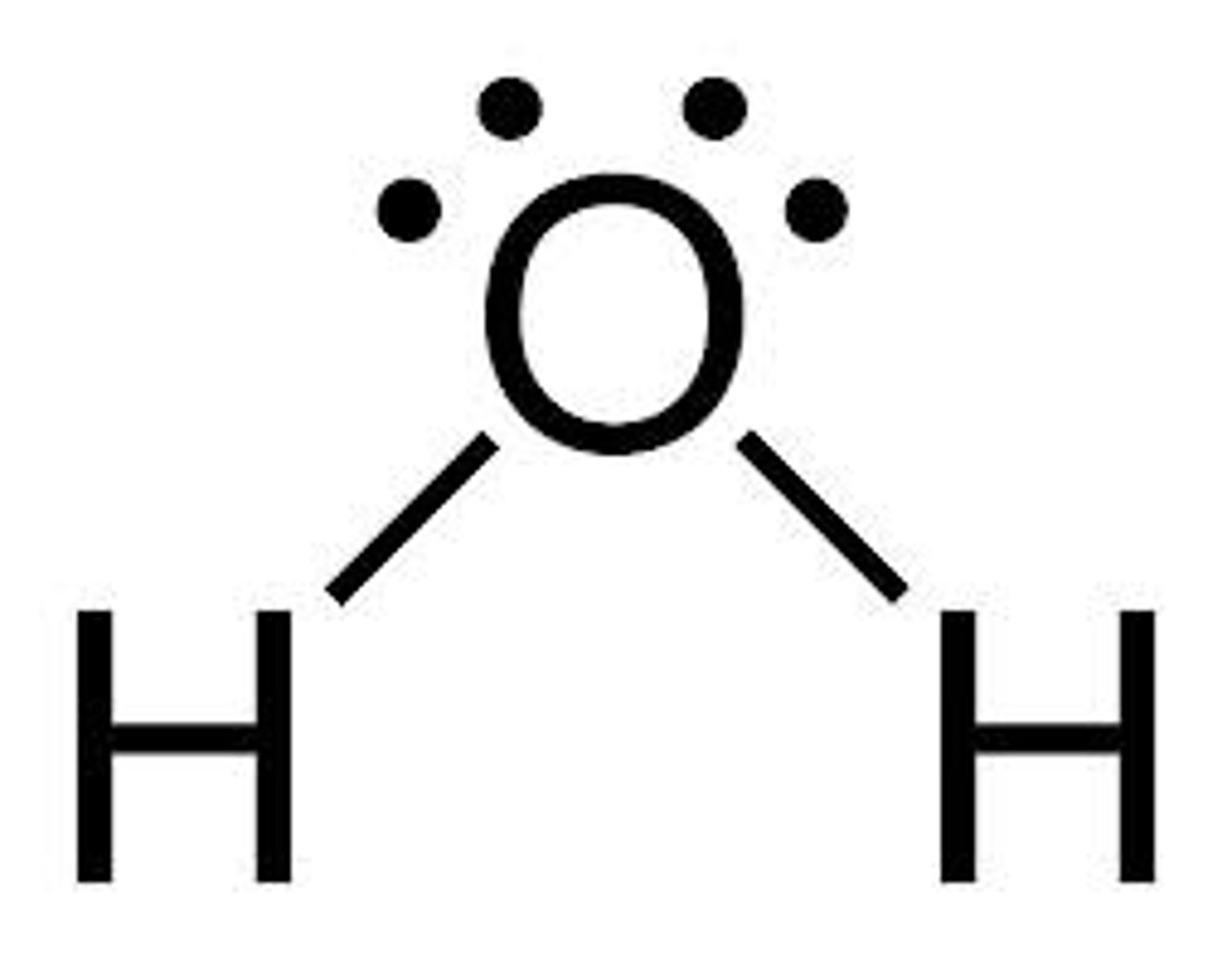
Place the atoms in increasing order of electronegativity (F, H, C, O, N)
H (2.1) < C (2.5) < N (3) < O (3.5) < F (4)
What are the most abundant elements found in organisms?
Hydrogen, Carbon, Nitrogen, Oxygen, Sodium, Magnesium, Phosphorus, Sulfur, Chorine
Explain adhesion and cohesion.
Adhesion: Water molecules that adhere to the glass resist the downward pull of cohesion.
Cohesion: Water molecules at the surface experience a net downward pull from hydrogen bonds with water molecules below.

What is a hydrogen bond?
Strong attraction between a slightly positive hydrogen atom (bonded to N, O, or F) and a slightly negative atom (like N, O, or F) with a lone pair.
How do ionic bonds differ from covalent bonds?
Ionic bond: Transfer of electrons to form ions that electrostatically attract oppositely charged ions
Covalent bond: Sharing of electrons
Hydrophilic?
Polar molecules and ions that dissolve readily in water
Hydrophobic?
Nonpolar molecules that do NOT dissolve in water
Explain hydrophobic interaction
Water forces nonpolar molecules together, because doing so minimizes their disruptive effects on the hydrogen-bonded water network.
Explain van der Waals interaction
Nonpolar molecules are attracted to each other via relatively weak attractions called van der Waals forces or transient dipoles (interaction due to fluctuating electrical charges)
What is an acid?
Acids release/donate H+ ions in solution
What is a base?
Bases accept H+ in solution (release OH-)
Which types of acids and bases are important in biological systems?
Acids: Carboxyl group (—COOH)
- Common in weak biological compounds.
- Functions as a weak acid because it dissociates PARTIALLY and REVERSIBLY (—COOH => —COO- + H+)
Bases: Amino group (—NH2)
- Functions as a weak base by PARTIALLY and REVERSIBLY accepting H+ (—NH2 + H+ => —(NH3)+)
What is pH?
Defined as the negative logarithm of the H+ concentration in moles/liter
What is a buffer? What do they illustrate?
Buffers make the overall solution resistant to pH change, because they react with both added bases and acids.
They illustrate the LAW OF MASS ACTION:
- Addition of reactants accelerates the reaction. Likewise, removal of products accelerates the reaction
Name the functional groups and their family of molecules
Hydroxyl: Alcohol (R-OH)
Phosphate (R-PO4) : Organic Phosphates
Amino: Amines (R-NH2)
Carbonyl: Aldehydes (R-C(=O)H) and Ketones (R-C(=O)R)
Carboxyl: Carboxylic acids (R-C(=O)OH)
What are macromolecules? What advantage do they have over other biochemical unities?
Macromolecules are very large biological molecules made by linking many smaller subunits (monomers) together, such as proteins, nucleic acids, carbohydrates, and lipids.
Macromolecules are made the same way in all living things and are present in all organisms in roughly the same proportions. An advantage of this biochemical unity is that organisms acquire needed biochemicals by eating otherorganisms.
Define polymerizaiton
It is a chemical process that links small molecules, known as monomers, together through covalent bonds to form long chains or three-dimensional networks called polymers.
Condensation reaction?
Monomer in, water out. (requires energy, anabolic)
Hydrolysis?
Water in, monomer out. (releases energy, catabolic)
What is a protein? What is its most crucial aspect?
Polymer made of amino acids.
Folding is crucial to the function of a protein and is influenced largely by the sequence of amino acids.
What do amino acids consist of? What is the difference between a non-ionized form and an ionized form?
Amino group (left) - CH - Side chain R (bottom) and Carboxyl group (right)
The primary difference is that ionized amino acids have charged groups, whereas non-ionized forms have neutral groups
What lies at the start and end of a polypeptide chain?
Start: N-terminus (amino terminus)
End: C-terminus (carboxyl terminus)
What properties of the peptide bond make proteins stable and functional?
Groups on either side of each peptide bond can rotate about single bonds, making polypeptides flexible.
Electron sharing between C-N and C=O makes the peptide bond double-bond-like (due to resonance), giving it stability.
What differentiates the formation of beta sheets and alpha helices?
Due to different hydrogen bonding patterns and polypeptide configurations (peptide backbone)
What amino acid does not conform to alpha helix nor beta sheet configuration? Why?
Proline! Its unique cyclic structure restricts the polypeptide backbone's flexibility.
What determines the tertiary structure?
Side chain interactions! (disulfide bonds, ionic bonds, hydrophobic interactions, hydrogen bonds, etc.)
What helps with the disruption of hydrogen bonding? What is an example of this?
The rearrangement of stretches of hydrophobic amino acids away from water.
Coiled coils arise when two a-helices have hydrophobic amino acids at every 4th position. Fibrous structural proteins consist mainly of ahelices arranged as coiled coils, such as the keratins in hair and feathers.
How would protein structure change if folding occurred in a hydrophobic environment?
They would rearrange so that non-polar (Hydrophobic) amino acids are away from water.
Explain why heat, pH, or ions denature proteins.
By disrupting the weak non-covalent bonds (hydrogen bonds, ionic bonds, and hydrophobic interactions) that maintain their specific 3D folded shape.
What occurs constantly in cells?
Protein turnover (breakdown and resynthesis)
Chaperones?
Specialized proteins that help keep other proteins (temporarily exposed hydrophobic regions) from interacting inappropriately with one another by sequestering some newly synthesized proteins to give them time to fold.
Can proteins fold by themselves? What experimental evidence shows this and is it the same in a live cell?
Many proteins fold spontaneously because the amino acid sequence contains all the information for the final 3D shape.
Anfinsen's experiment (where he used ribonuclease A) to show the denaturation of the enzyme and the refolding of the protein's structure.
Inside a cell the environment is more crowded, that's why the cells use chaperones to help keep proteins from interacting together; they sequester proteins to give them time to resynthesize
What makes up a Nucleotide?
Phosphate group (left, bonded to 5' carbon) - Sugar - Nitrogenous base (right, bonded to 1' carbon)
DNA v.s. RNA?
DNA: Deoxyribose, stores long-term genetic information as a stable, double-stranded helix
RNA: Ribose, acts as a temporary messenger, single-stranded
What are phosphates?
"Energy-rich bonds": Overall chemical reaction leading to their hydrolysis releases energy
What are the different Nitrogenous bases?
Pyrimidines: Cytosine (C), Uracil (U) in RNA, Thymine (T) in DNA
Purines: Guanine (G), Adenine (A)
A goes with T or U
G goes with C
How is the sugar-phosphate backbone formed?
Condensation reaction between 3' carbon with OH group and OH from phosphate (on the 5' carbon).
Creates a "Phosphodiester linkage"
Starts at 5' end of nucleic acid. New nucleotides are added to the unlinked 3' carbon.
Nitrogenous bases bond (glycosidic bond) to the 1' carbon.
What pairs fit inside the double helix? What bond forms between the pairs
ONLY PURINE-PYRIMIDINE PAIRS
Hydrogen bonds form between G-C and A-T/U pairs
What do single-stranded RNA form?
Forms a loop called a "hairpin" or "stem-loops" to protect messenger RNAs, guide the molecule's tertiary structure, and serve as recognition sites for proteins.
The short RNA strand is folded on itself in some regions to form double-stranded helices.
Explain or draw the structure of DNA including the nucleotide bases, backbone and bonds that form. Why do the bases point internally in the helix and what is the evolutionary advantage of this structure?
Bases point inward because they are hydrophobic. Useful for storing genetic information.
Define anabolic and catabolic
Anabolic: reactions link simple molecules together to make complex ones. These are energy-storing reactions. They REQUIRE energy.
Catabolic: reactions break down complex molecules into simpler ones. They RELEASE energy.
What drives energy conversions?
The drive of energy to become evenly distributed or dispersed.
What are the Laws of Thermodynamics?
First Law of Thermodynamics: Energy is neither destroyed nor created.
Second Law of Thermodynamics: Energy spontaneously disperses from being localized to becoming spread out if it is not hindered from doing so (entropy increases).
Describe two ways that a cell can release free energy to drive a chemical reaction?
1) With a chemical reaction creating disorder in the cell Ex: digesting a polymer --> turning into many monomers (ENTROPY change - ΔS)
2) With a chemical reaction that releases heat in the surroundings (ENTHALPY change - ΔH)
What does it mean when Δ𝐺 is + or - ?
+ Δ𝐺 = energy is REQUIRED (unfavorable reaction)
- Δ𝐺 = energy is RELEASED (favorable reaction)
What are the 4 types of reactions?
1) Heat is released and disorder is increased: always spontaneous (exergonic/exothermic) :
- Δ𝐺 = (ΔH) - (T)(Δ S)
2) Heat is released, but disorder decreases: only spontaneous below a certain temperature; e.g. denatured/native protein, lipid bilayer vs. individual lipid
Δ𝐺 = (- ΔH) + (T)(ΔS)
3) Heat is used, but disorder increases: spontaneous above a certain temperature; e.g. dissolving NaCl in water
ΔG = (ΔH) - (T)(ΔS)
4) Heat is used and disorder decreases: never spontaneous (endergonic/endothermic)
ΔG = (ΔH) + (T)(ΔS)
How is energy transferred in cells?
ATP (adenosine triphosphate), for capture, transfer and storage of energy.
Why is the concentration of ADP low in animal cells?
To maintain a high ATP:ADP ratio, ensuring that energy-demanding cellular processes remain energetically favorable.
ATP can hydrolyze to yield ADP and an inorganic phosphate ion (Pi)
The reaction is very exergonic (ΔG = -12kcal/mole) because ADP is constantly removed either by reforming ATP or by hydrolysis to AMP (ΔG° = -7.3kcal/mole)
What determines whether a reaction proceeds or not?
Overall AG of coupled reactions must be negative to proceed
Catalyst?
Any substance that speeds up a chemical reaction without itself being used up. (ex: enzymes)
Can enzymes change the ΔG of a reaction? Why or why not?
No. They do not alter the starting energy of reactants or the final energy of products.
What are the 3 main ways enzymes catalyse?
1) Orientation: Moves substrates closer to facilitate binding
2) Charge: electrostatic interaction
3) Strain: helps breaking covalent bond
What is a cofactor?
Anything that is not an amino acid. These can be metal ions (zinc, copper), small organic molecules temporarily binding, and small organic molecules that are permanently bound to the protein (heme).
When are enzymes saturated?
When all binding sites are occupied. As substrate concentration increases, rate of product formation slows down.
Lysozyme: 1 molecule/sec
Catalase: 40 million molecules/sec
What are the major categories of carbohydrates?
Monosaccharides
Disaccharides (2 x monosaccharides)
Polysaccharides (3 or more monosaccharides)
Aldose?
Carbonyl group at end of carbon chain
Ketose?
Carbonyl group in middle of carbon chain
How does the glucose ring structure form?
Oxygen from the 5-carbon bonds to the 1-carbon, resulting in a ring structure. (alpha or beta structure)
Describe how two sugar molecules can bond together in our bodies? What general chemical property allows for the formation of multiple types of molecules?
Two sugar molecules can bond by forming glycosidic linkages (ether bond formed between two sugar molecules through condensation reaction).
The isomeric property of sugar molecules is what allows for multiple (very different molecules) to form from the same monomers, as well as the presence of reactive functional groups, allowing for peptide, ether, glycosidic bonds, etc.
Compare the structures and function of the polysaccharides amylose and cellulose. What is the biological significance of their different chemistries?
Amylose: formed by alpha 1-4 bonds, their structure is coiled because the ligands repell eachother since they are on the same side. This makes amylose less soluble in water because the coil prevents molecules from interacting with the hydroxyl groups that are inside the coil. Allows storage of a lots of sugars, making it great for energy reserves.
Cellulose: very rigid structure due to its beta 1-4 bonds, making it ideal for structural support or in cell walls of plants.
Why does a beta 1-4 bonded molecule flip?
To allow a straight beta 1-4 covalent bond.
What is an oligosaccharide?
Carbohydrates composed of a short chain of 10-20 monosaccharides. Usually attach to protein on outside of cell.
True or False: lipids are soluble in water
False. Lipids are NOT soluble in water
What are some roles for lipids in organisms?
1) Energy storage (fats and oils)
2) Cell membranes (phospholipids)
3) Hormones and vitamines (steroids and modified fatty acids)
4) Electrical insulation of nerves (brain is mostly fat)
5) Water repellency (waxes and oils for animals and plants)
What is a fatty acid?
An amphiphilic (both hydrophobic and hydrophilic), hydrocarbon chain with an oxygen-containing carboxyl group, serving as the fundamental building block for fats, oils, and lipids.
Ex: soap! It is lipid soluble and brings it into solution
What is a triglyceride?
Glycerol linked by ester linkages to three fatty acids.
What is a phospholipid? What properties of this molecule allow it to form a lipid bilayer and how is its fluidity regulated?
Water-loving (hydrophilic) phosphate "head" and two water-fearing (hydrophobic) fatty acid "tails"
Phospholipids arrange themselves so the hydrophobic tails don't come into contact with water and seal to form an enclosed space (energetically favorable BELOW certain temperature)
Phospholipids are in constant lateral motion, but rarely flip to the other side of the bilayer. Fluidity of the bilyar is regulated by forming double bonds in the hydrocarbon chain to form "kinks". This allows for more room to move and maintains fluidity of bilayer. (ex: survival of fish and plants in the winter!)
Saturated v.s. Unsaturated fat?
Saturated fat: all single bonds (ex: butter, has lower permeability and is less fluid --> solid)
Unsaturated fat: at least one double bond (ex: oil, higher permeability and is more fluid --> liquid)
Lipid bilayer with no unsaturated fatty acids
Lower permeability, less fluid
Lipid bilayer with many unsaturated fatty acids
Higher permeability, more fluid
Proteins can be... (how it interacts with water)?
Amphiphilic
Describe what properties allow a protein to be amphiphatic. What does this allow the protein to do?
At the end of the protein: The polar and charged amino acids are hydrophilic
In the middle of the protein: The nonpolar amino acids are hydrophobic
Amphipathic proteins can integrate into lipid bilayers.
What is a fluid mosaic? What does it consist of?
Fluid mosaic: Lipids + Proteins making up a dynamic and flexible structure (phospholipid bilayer with embedded proteins)
Consists of:
- Peripheral membrane protein (Extra cellular)
- Integral/ Trans membrane protein (inside the phospholipid bilayer)
- Peripheral membrane protein (Intra cellular)
True or False: Membrane proteins can flip.
False! Membrane proteins have a unique orientation. Flipping is energetically unfavorable (requires massive energy to move)
During freeze fracture EM, why does the lipid bilayer split in half (splits into individual leaflets)?
The weakest interactions (van der Waals interactions) are in the interior membrane of the lipid bilayer, meaning they are easier to separate than the hydrogen bonds between the polar molecules of the exterior membrane in the lipid bilayer.
Name the functions of membranes.
1) Barrier
2) Transport
What is an experiment to test permeability?
Ex: Planar bilayers: Artificial membranes. Tests how rapidly different solutes cross the membrane (if at all) when different types of phospholipids are used to make the membrane.
What type of cellular transport is associated with low v.s. high permeability?
Low Permeability: Active transportation. Large and/or polar molecules (ex: Glucose, sucrose, Cl-,K+,Na+,etc.)
High Permeability: Passive transportation. Small nonpolar molecules or small uncharged polar molecules (ex: O2, CO2, N2, H2O, glycerol, etc.)
Describe diffusion. What is the reason behind it?
The passive mixing of substances resulting in net transport along a concentration gradient
"random walk" (Brownian motion) of individual molecules due to thermal motions and collisions
What determines diffusion rates? Explain how it works using Gibbs free energy.
Temperature, size of molecule, and steepness of concentration gradient.
The reaction starts with negative ΔG, as diffusion happens, it becomes less and less negative until it is at equilibrium and there is no more diffusion, delt G is zero.
What is osmosis? Explain the difference between osmosis and diffusion.
Osmosis: diffusion of water across a selectively permeable membrane
Diffusion relates to solutes, osmosis relates to the solvent (water)
What are three types of solutions? Explain each one.
1) Hypertonic solution: Higher solute concentration outside membrane. NET FLOW OF WATER IS OUT. Cell shrinks.
2) Hypotonic solution: Higher solute concentration inside membrane. NET FLOW OF WATER IN. Cell swells or even bursts.
3) Isotonic solution. Solute concentrations are equal on both sides of membrane. NO NET FLOW OF WATER.
What is facilitated diffusion? What does it depend on?
A passive transport process where molecules move across a cell membrane down their concentration gradient, but with the help of specific transmembrane proteins.
Depends on two types of membrane proteins: channel proteins (for ions) and carrier proteins (for sugars)
How does facilitated diffusion work?
Electrical charge outside the membrane triggers shape change that allows the ions to pass through the channel proteins along the concentration gradient.
How do carrier proteins differ from channel proteins?
A carrier protein binds the transported substance. Transition between these states is random and reversible!
True or False: the carrier/channel proteins can be saturated. What does this mean in terms of rate of diffusion?
When all carrier binding sites are occupied or the channel is full, the carrier/channel is saturated; therefore the rate of diffusion levels off.
How do active transport and passive transport differ?
In contrast to diffusion, active transport requires the expenditure of energy (directly or indirectly). Δ𝐺 is positive. Substances are moved across the membrane against the concentration gradient.
Why does the cell perform Primary Active Transport?
1) Control of osmolarity (net movement of one ion out). Reduces inflow of water.
2) Generation of membrane potential (electrical charge). Net transport of ions out. Important for neuronal cells.
3) Sets up a concentration gradient that allows other transport processes to occur.
Define Secondary Active Transport. How does it work in terms of Gibbs free energy?
Secondary active transport systems use established gradients to move substances.
How it works:
The highly negative Gibbs free energy (Δ𝐺) of 𝑁𝑎+ moving down its concentration gradient into the cell (high to low concentration) provides the necessary energy to drive the transport of glucose against its concentration gradient (low to high concentration).
Why does the cell undergo secondary active transport?
1) economical: 1 ATP : 3 Na+ : 3 glucose
2) concentration gradients are used by other biological processes to transport molecules in the body
How many cells are in the body?
40 trillion
Why are cells so small?
Because most chemical reactions in cells require diffusion. Diffusing molecules don't travel long distances because of their "random walk" (brownian motion).
If volume is too large, it constrains cellular transportation.
What are the two organizational patterns of cells? Explain them briefly.
- Prokaryotes: very small. Have no nucleus and few membrane-enclosed compartments
- Eukaryotes: have a membrane-enclosed nucleus and many other membrane-enclosed compartments (organelles).