P3: Chemical Kinetics
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms

What is the rate coefficient k, Integrate it from t=0 corresponding to A0, to t corresponding to concentration A, draw the van’t hoff plot and state what order is this rxn, rearange to A
State the :
1st,Reaction, rate law and integrated rate equation

State the :
the 2nd A=B
Reaction, rate law and integrated rate equation

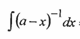
solve
yh with the (-)ive
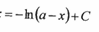
what do these equal to:
lnx + lny
lnx - lny
=ln(xy)
=ln(x/y)
e^0 = 1
State the :
2nd for A does not equal B
Reaction, rate law and integrated rate equation

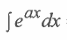
i
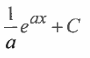
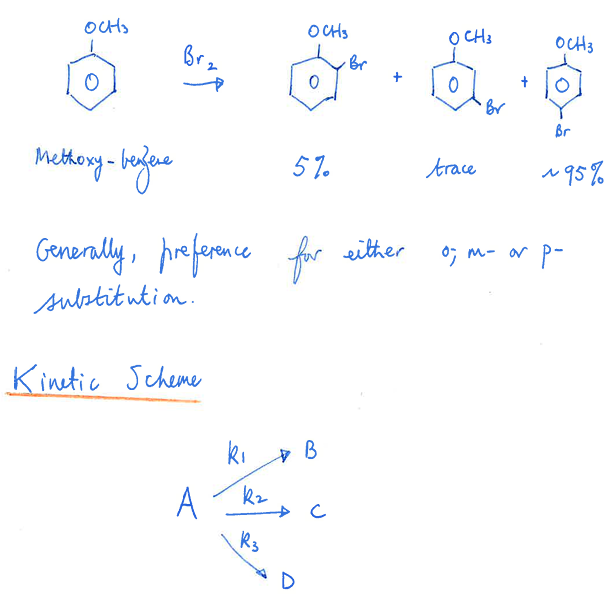
Give the rate law , integrated equation , what kind of rxn is it
concurrent reaction , electrophelic substitution rxn


what order is a consecutive rxn, writ ethe method to finding the rate of change and do the 1st step here
1st order usually so use its equation for integrated
the rxn goes A → B → C with the arrows being k1, k2, and k3 if not given.
after the step in the image we will sub in 7 and 8 into 6

What is the key feature of telling the difference between protic and aprotic solvents by only looking at the molecule
Protic solvents contain NH and OH (acidic Hs), dissolve both charges
Aprotic solvents DO NOT CONTAIN (OH) NOR (NH), only dissolves positive charges
state Eyring equation for TS
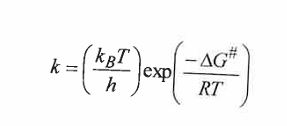
Calculate the Free E of activation for 2 different solvents
we isolate G from eyring
Kb( boltzmann constant in JK-1
room T in K , 25celicius → 298K
h plank constant in Js
we know ln(x/yz)= ln(x/y) - lnz
we can sub in for k (rate coeffecient) in M-1s-1
for eg. acetone and then for benzene
then delta delta G dash = delta G dash (Benzene)- delta G dash (acetone)
answer in Jmol-1 we should turn it to KJmol-1
We find rxn favours Polar solvents such as acetone coz they reduce TS free E and stabilise it

