Alcohols
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
General properties of alcohols
General formula: CnH2n+2O
Alcohols can be classified as primary (1º), secondary (2º) or tertiary (3ºº depending on how many R groups (carbon chains) the carbon with the OH is attached to
Chain and position isomers based on position of OH or length of carbon chain
Functional group isomers of alcohols are ethers R—O—R
Physical properties of alcohols
Form hydrogen bonds between molecules so have high melting and boiling points
Bigger alcohols have more electrons and stronger van der waals forces between the molecules, requiring more energy and giving them a higher bp
Branched alcohols have lower bp as they have fewer points of contact between the molecules- so the van der waals forces are weaker and less energy is needed
Alcohols are soluble in water due to hydrogen bonding
what is the industrial preparation of alcohols
Hydration of alkenes
Reagent: H2O(g) (steam)
Conditions: H3PO4 catalyst, high temperature and high pressure
The starfish does the box splits and and OH is added to one leg and H to the other
Position isomers can be formed based on the position of OH
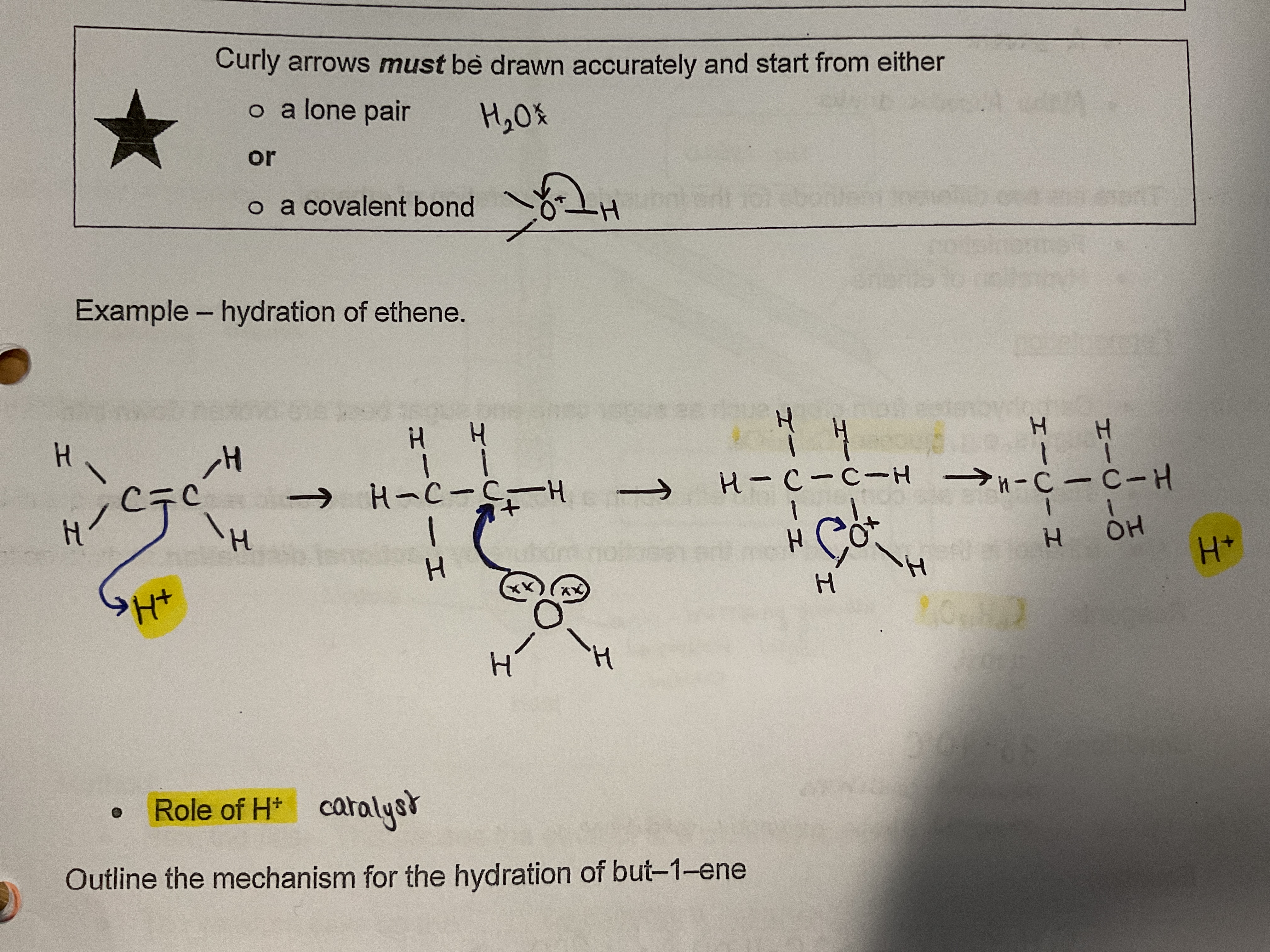
Hydration mechanism
H+ is taken by the double bond
Starfish does the splits and the H is added (be mindful of the position)
Water with micky mouse ears (curly arrow from lone pair on water to carbocation)
Arrow from hydrogen on water to oxygen carbocation
Product and H+ ions form
H+ role : catalyst
Uses of ethanol
Chemical feedstock (used to make other chemicals)
A solvent
Alcoholic drinks
What are the two methods for the industrial preparation of ethanol?
Fermentation
Hydration of ethene
Process of fermentation
Carbohydrates from crops are broken down into sugars
Sugars converted to ethanol through anaerobic respiration
Ethanol removed from the reaction mixture by fractional distillation
Anaerobic respiration
Reagents: C6H12O6, yeast
Conditions: 35-40ºC, aqueous conditions, anaerobic conditions
Equation: C6H12O6(aq) —→ 2C2H5OH(aq) + 2CO2(g)
Air is kept out of the reaction to prevent oxidation
Temp of 35ºC is used to increase reaction rate without denaturing enzyme (yeast)
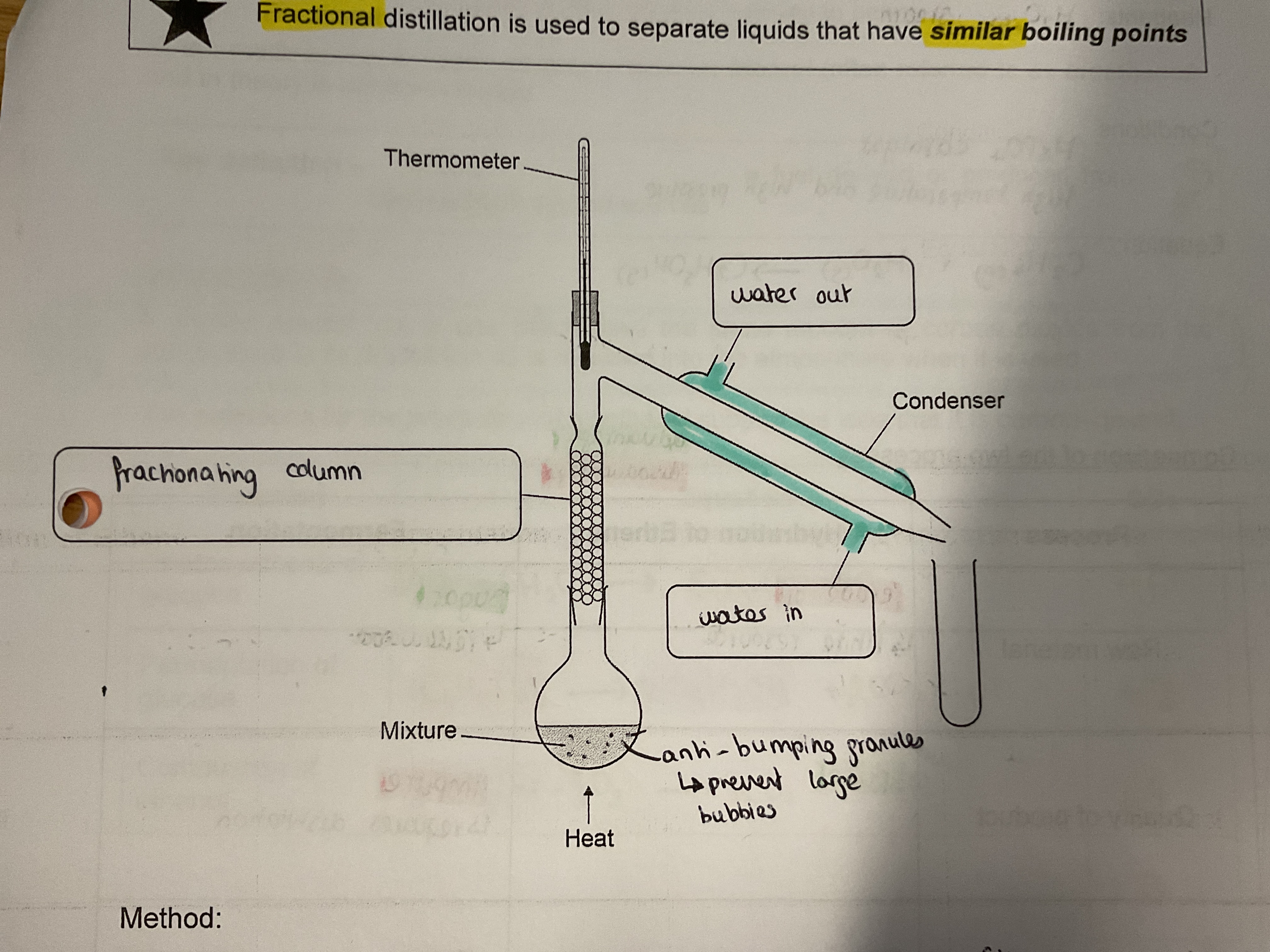
Fractional distillation
Used to separate liquids that have similar boiling points
Heat the flask (vaporising the ethanol and water)
Vapours pass up fraction in column so water and ethanol will separate
Water will condense back into flask as has higher bpt than ethanol
Observe thermometer and keep the temp at or just below bpt of ethanol
Ethanol vapour passes into condenser where water in outer jacket cools vapour which condenses into a liquid which is collected
Comparison of the two processes of the preparation of ethanol
Hydration uses crude oil which is a finite resource, fermentation uses sugar which is renewable
In hydration the product is pure, in fermentation it is impure and requires distillation
Hydration fast rate of reaction, fermentation slow rate of reaction
Very high energy requirements in hydration, very low in fermentation
Hydration continuous process, fermentation batch process
Hydration higher atom economy (100%) than fermentation
Biofuel definition
A fuel derived or produced from renewable biological sources
When is ethanol being called a biofuel?
When it is made by fermentation
What is a carbon neutral fuel?
One which uses the same amount of carbon dioxide from the atmosphere in its production as is released into the atmosphere when it is used

Why is it argued that bioethanol is not carbon neutral?
However, fossil fuels need to be burned to power machinery used to make fertilisers, harvest crops and refine and transport bioethanol
Burning fuel to power machinery produces CO2
Disadvantage to use of crops for production of fuel
Land used to grow crops for fuel cannot be used to grow food
Fertilisers added to soil to increase biofuel crops production
Deforestation to create land for biofuel crops to grow