3.1.5.2 the Maxwell Boltzmann distribution
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
when will particles only react?
if, on collision, they have at least the minimum amount of energy, which is known as the activation energy
describe + explain the energies of the particles in a reaction
the particles in a a reaction undergo random collisions, in which energy is transferred between the colliding particles → as a result there will be particles with different energies
what does the Maxwell Boltzmann distribution curve show?
the spread of molecular energies
what the four important points about the Maxwell-boltzmann curve?
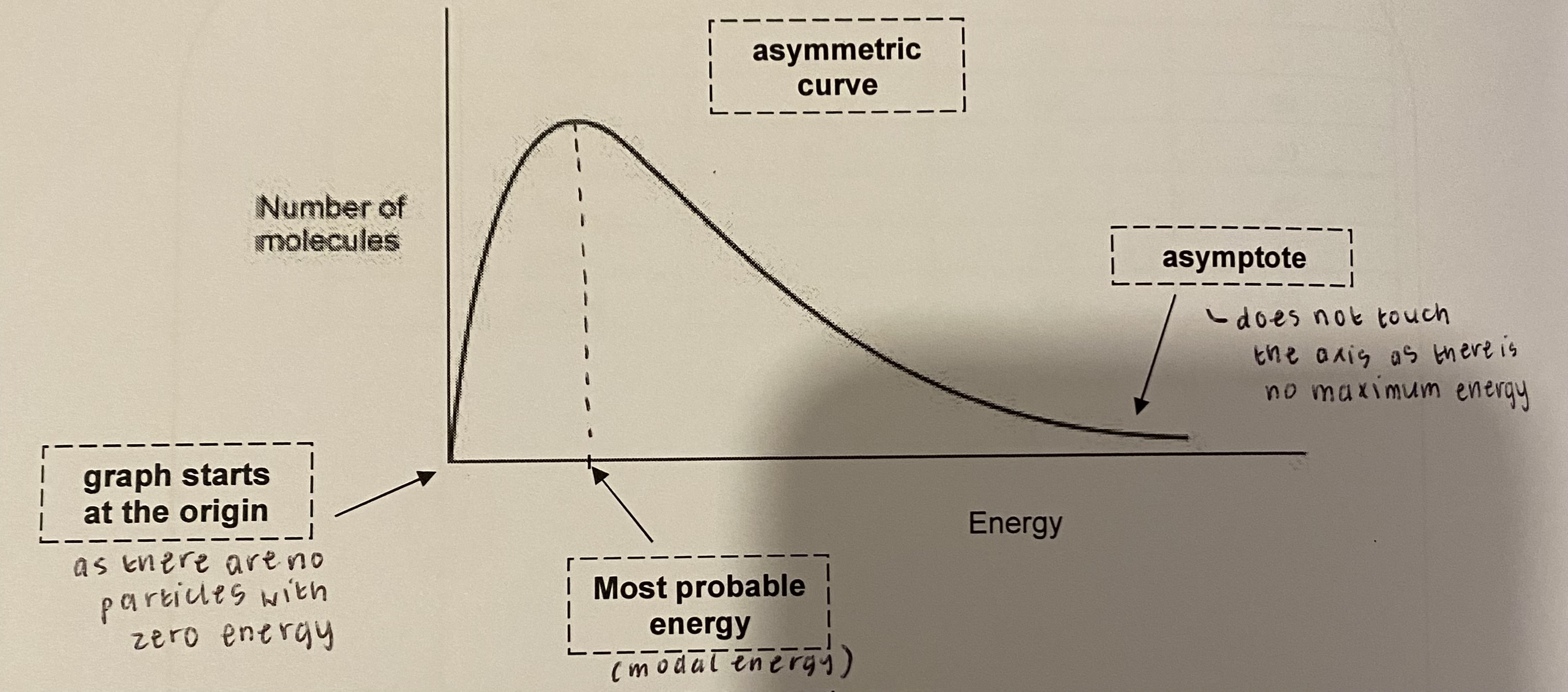
there are no particles with zero energy → graph starts at the origin
the curve does not touch the x-axis at higher energy because there will always be some particles with very high energies (there is no maximum energy); asymptote
the area under the curve is equal to the total number of particles in the system
the peak of the curve indicates the most probable energy
what type of curve is the Maxwell-boltzmann distribution?
an asymmetric curve
draw + label the Maxwell-boltzmann distribution curve
label the activation energy on the Maxwell-boltzmann distribution curve
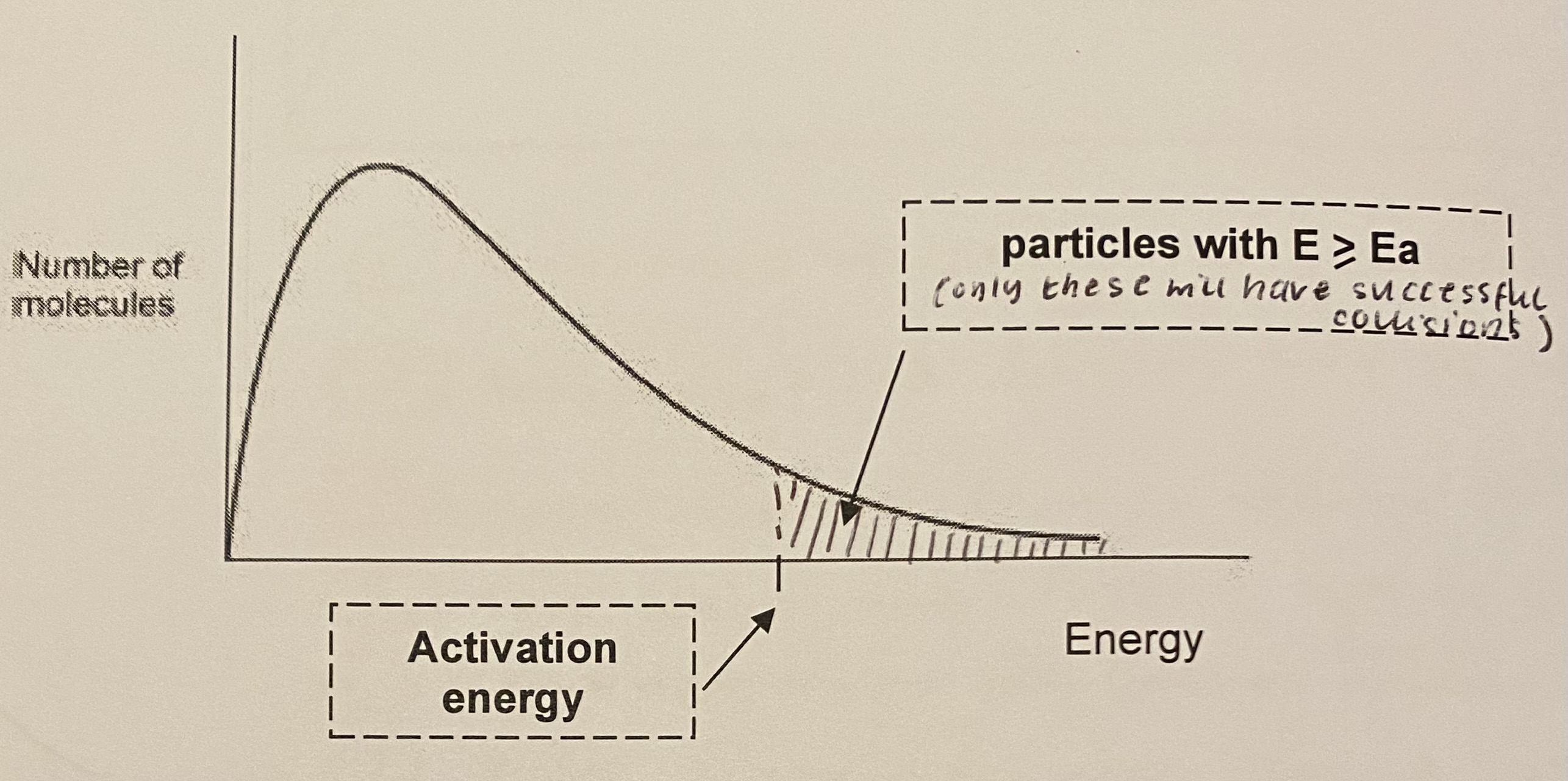
what are the only particles that can react when a collision occurs?
only those with energy greater than or equal to the activation energy, Ea
show (draw) the change in the Maxwell-Boltzmann graph as the temperature a reaction is increased
value of modal energy increases
number of molecules with modal energy decreases
number of molecules with Ea increases
area under the graph stays the same
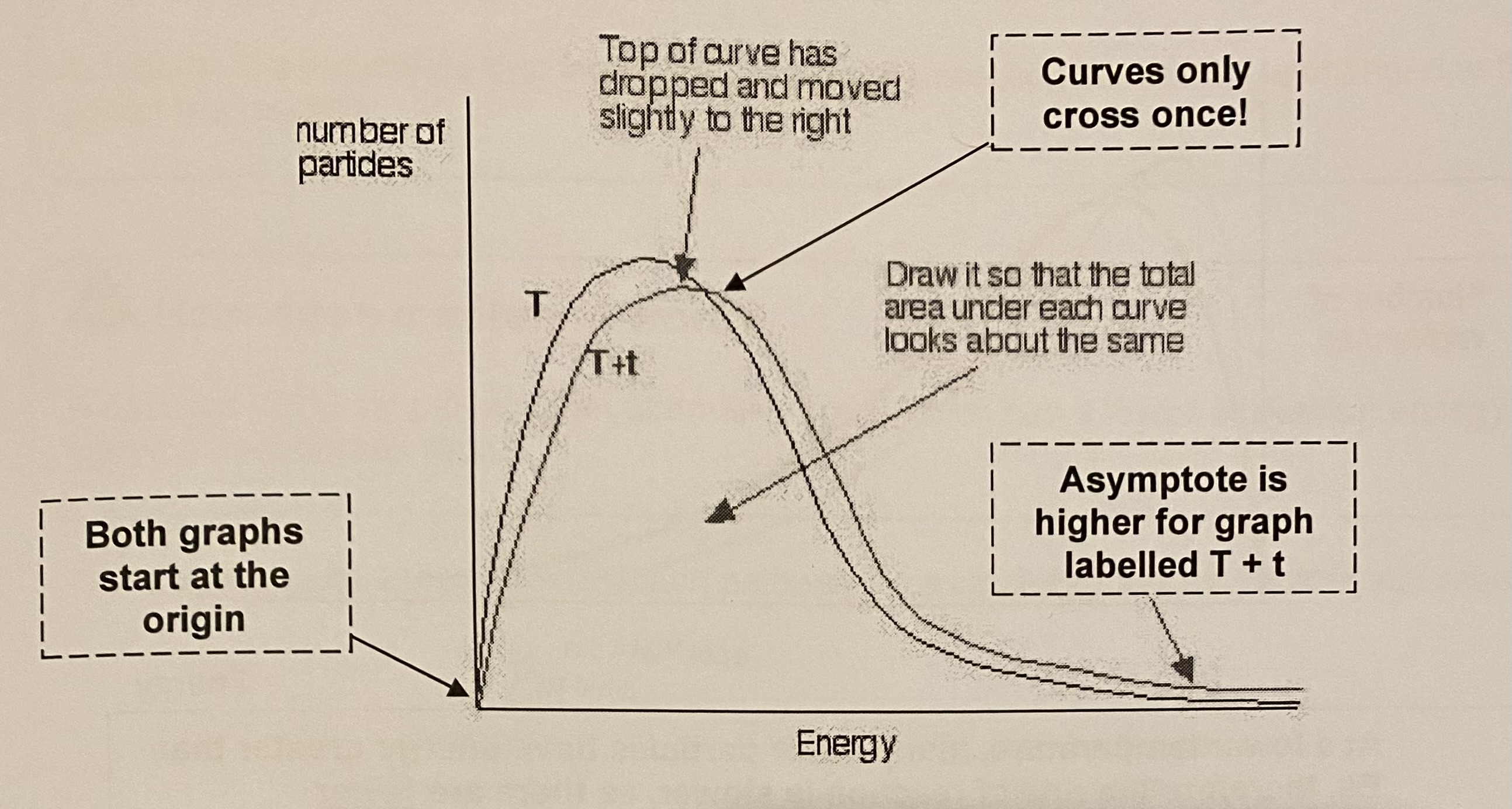
why are there more successful collisions at higher temperature?
many more particles have energy greater than Ea, therefore the rate of reaction is greater
therefore a small increase in temperature can lead to…
a large increase in rate because there are many more molecules with energy greater than Ea
what does the effect of fewer moles of gas have on the Maxwell-boltzmann distribution curve?
the modal energy does not change but the height of the peak does
what is a catalyst?
a substance that increases the rate of a reaction but itself is unchanged at the end of the reaction
how does a catalyst work?
by providing an alternative route which has a lower activation energy than the uncatalysed route
what effect does a catalyst have on a reaction profile?
it has no effect on the overall enthalpy change, therefore △H is unchanged
activation energy, Ea, is lowered if a catalyst is used
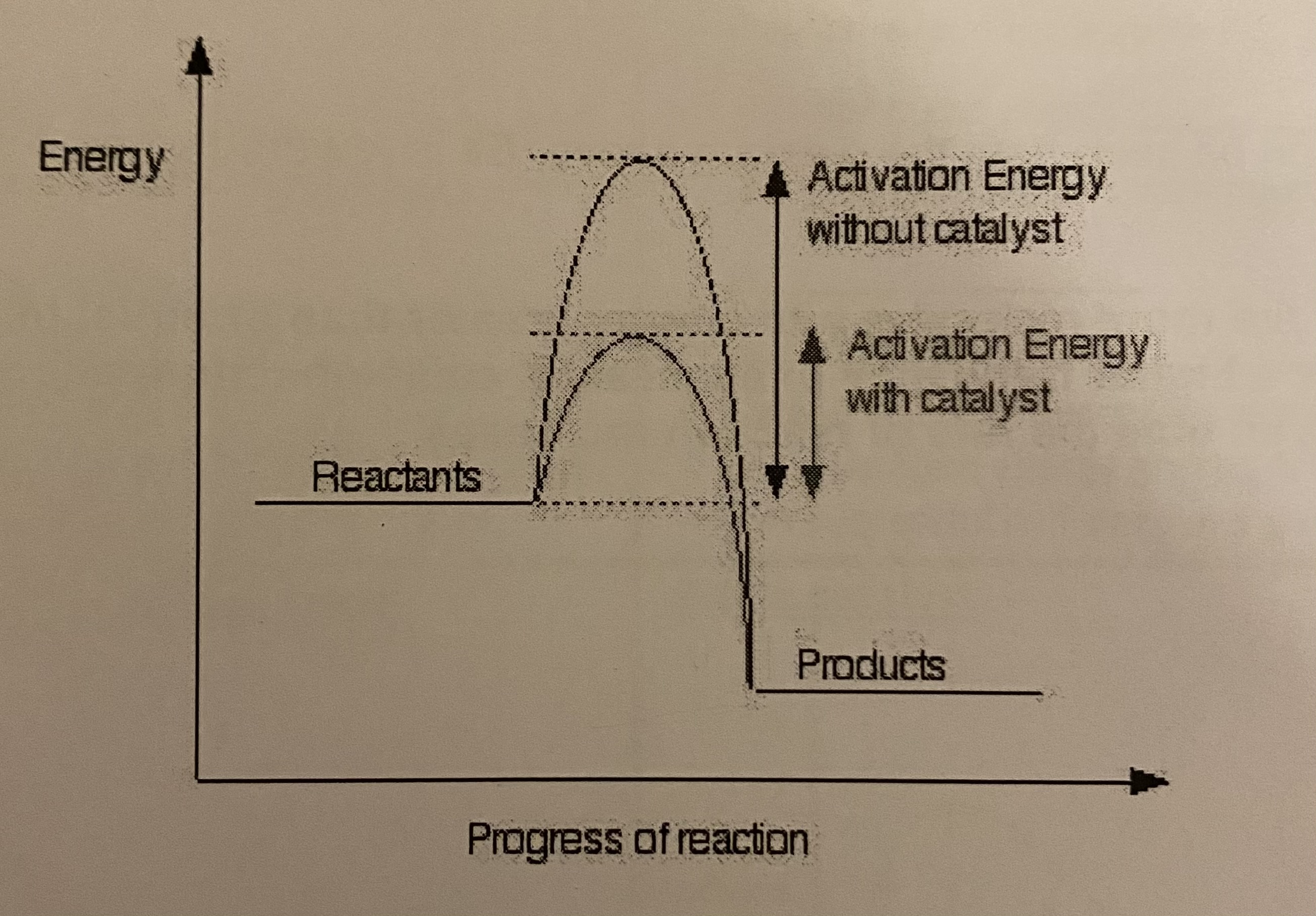
what effect does a catalyst have on the Maxwell-Boltzmann curve? + therefore explain the effect on the rate of reaction
Ea is lowered when a catalyst is used therefore:
there are more molecules with energy greater than or equal to activation energy
so there is a higher frequency of successful collisions in a given time the rate reaction increases