Ch. 28- Atomic Physics
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
rest mass energy
-Mass is just another form of energy!
-E is total relativistic energy (needed for speeds greater than about 1% the speed of light to avoid large errors)
-K is relativistic kinetic energy (it has its own equation we are not going to talk about)
-from Einstein’s relativity
*if convert all electron mass into energy, get energy to hold nucelus together → nuclear power plants/fusion
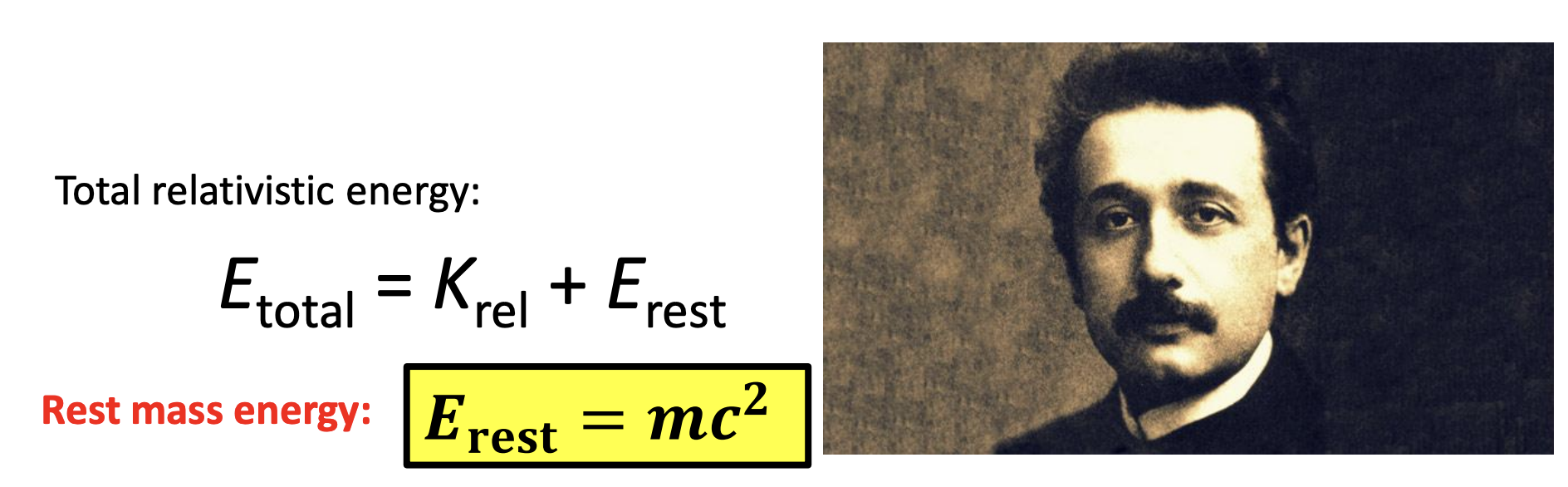
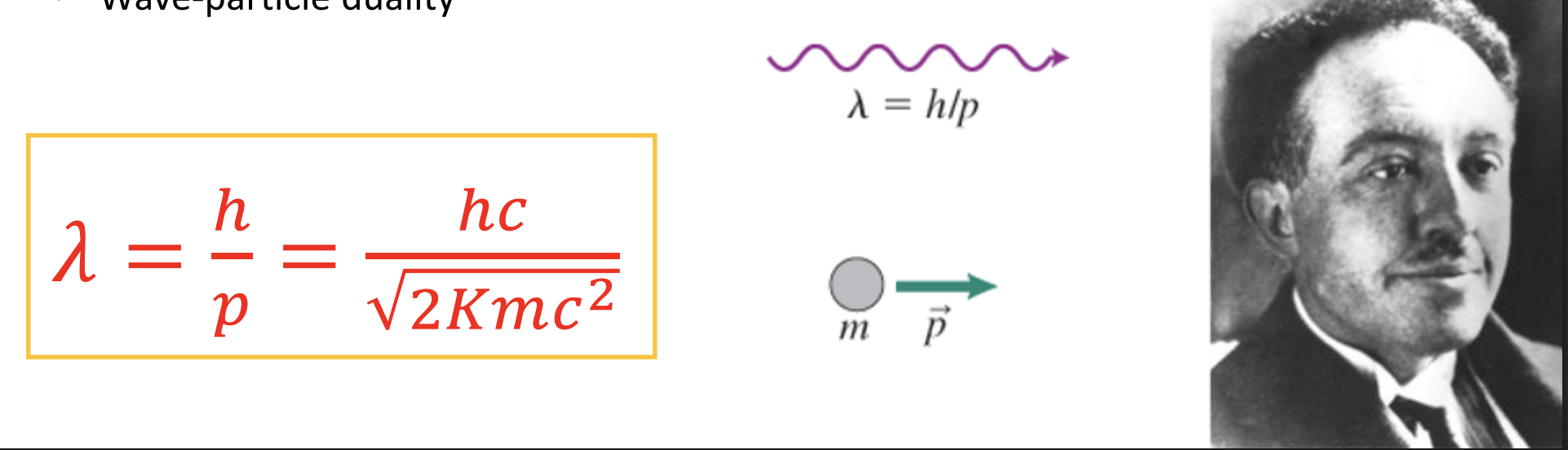
De Brogile wavelength
-In 1924, de Broglie postulated that because photons have wave and particle characteristics, perhaps all forms of matter have both properties. Nobel Prize: 1929
-Recall the momentum of a photon: 𝑝 = ℎ/𝜆 = h/mV
-The de Broglie wavelength of a particle is related to it’s momentum and therefore kinetic energy
-“Wave-particle duality”
what does and doesn’t de Brogile’s wavelength apply to?
-does apply to microscopic objects
-does NOT apply to macroscopic objects
pico
10^-12
Any particle can be diffracted with an appropriate
speed and appropriate slit size!
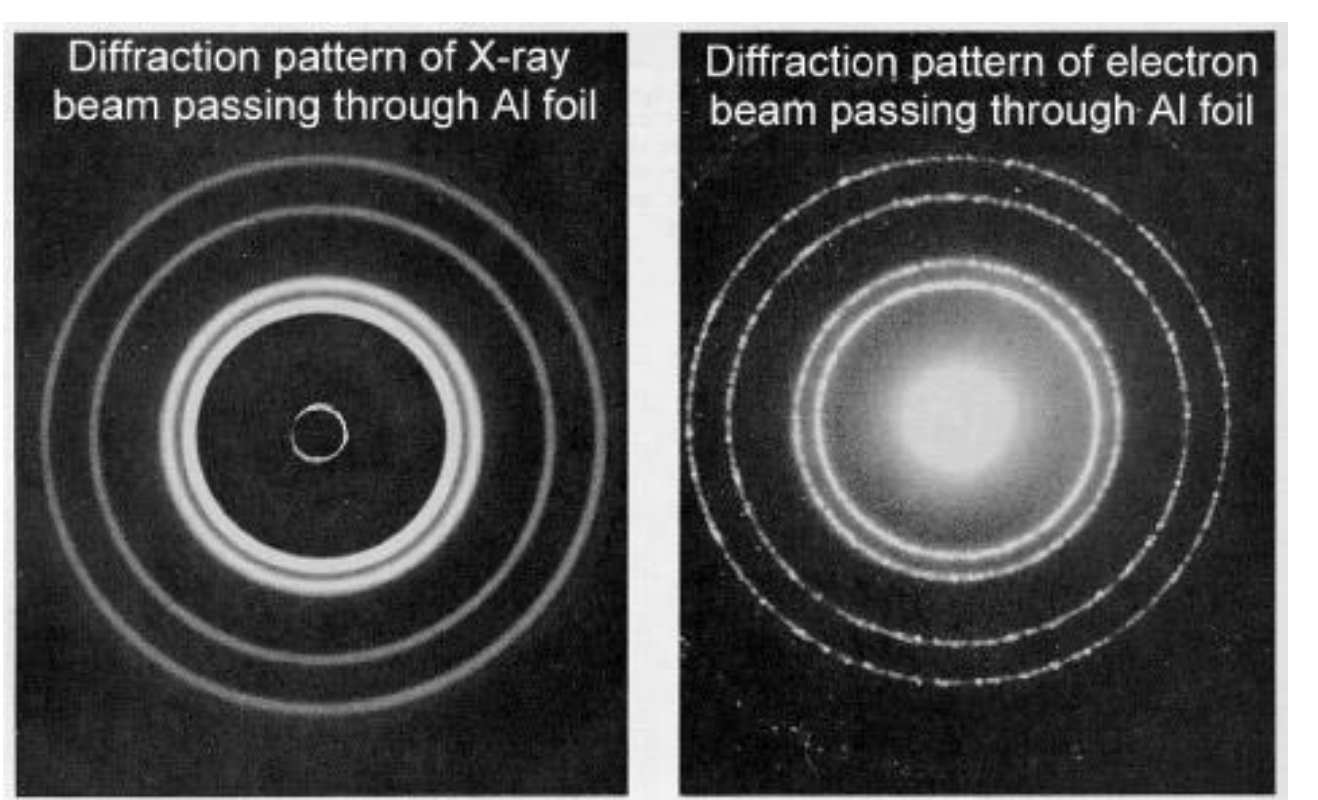
-Davisson and Germer (1925) first showed this with electrons while studying crystalline nickel.
bright rings=CI; dark rings=DI
rings same location, so electrons act like waves
uses: microscopes w/ electrons, use space between atoms (like distance between slits); atomic level detail
electron microscopes
-Can probe sizes of order of a pico-meter (pm = 10-12 m) without using harmful x-rays (*bc use electrons)
-Create surface maps of tissue or inorganic materials down to atomic level detail!
-Not much voltage required to get the elections to the needed speed. (*much safer)
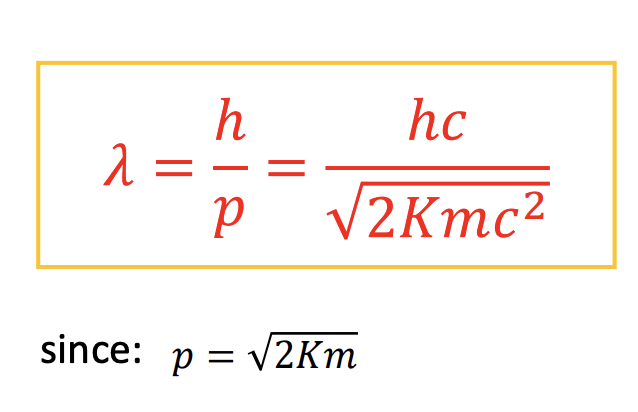
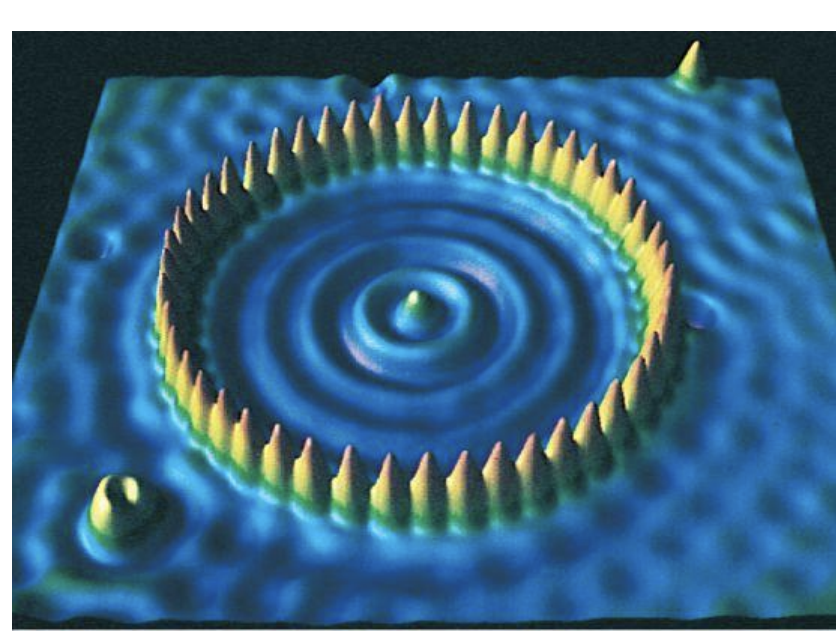
box model
-1993, IBM: 48 iron atom “fence” is 7.13 nm in radius
-can move individual atoms
-peak=single Fe atom
-1 electron trapped in middle creates wave; trapped particles with similar de Brogile wavelengths=acts like a wave, various things become quantized (ex=nucleus)
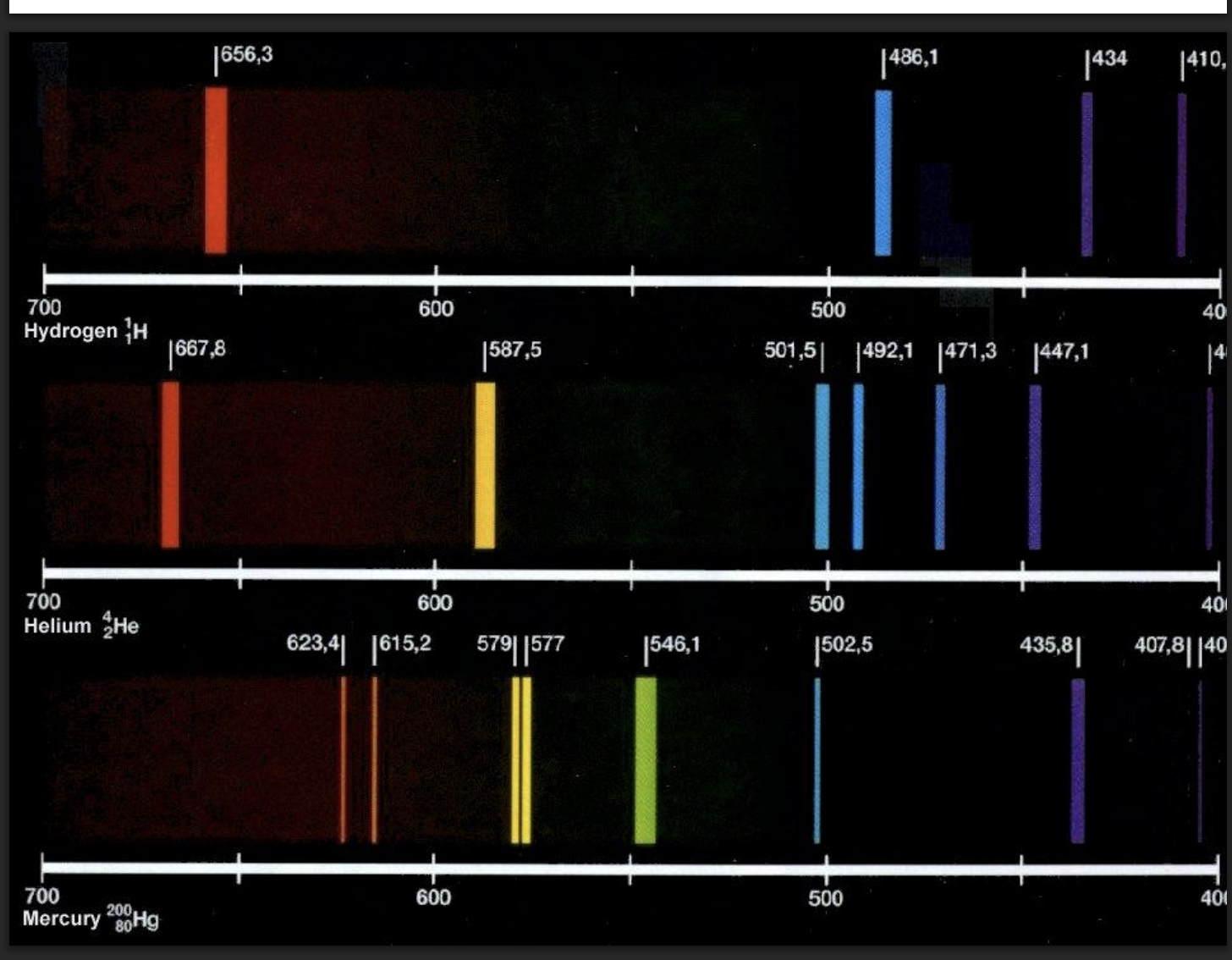
relationship number of electrons and emission lines
-increase electrons=increase emission lines (from electrons transitioning between orbits)
Bohr’s model of a 1 electron atom
-hydrogen or any atom singly ionized
-a negatively charged electron moves in a stable circular orbit about a positively charged nucleus
-when an electron transitions from one stable orbit to another, the atom’s energy changes and a photon is emitted or absorbed
-electron’s de Brogile wavelength must satisfy (equation)
-quantized

Bohr model electron orbits
-a0=Bohr Radius=0.0529nm=r1 (*smallest orbit; doesn’t increase linearly)
-The Bohr Model correctly explains the properties of the electron in the hydrogen atom on average (*varies based on where electron is and radii; reality=not perfect circle and actually electron cloud with probability to be in certain places)

emission and absorption
-Spontaneous emission of radiation (light) can occur if an electron is in a higher level with an unoccupied level below: 𝐸𝑖𝑒 = 𝐸𝑓𝑒 + 𝐸𝛾.
-The visible light emitted by hydrogen gas corresponds to four transitions to the n=2 state.
Transitions to n=1 have small wavelengths (ultraviolet)
Transitions to n=3 have larger wavelengths (infrared)
-If a photon comes in with the exact right wavelength (energy) it an be absorbed by an atom: 𝐸𝑖𝑒 + 𝐸𝛾 = 𝐸𝑓𝑒.
-The emission/absorption spectrum can be used to identify what element is in a gas.
ground state energy of hydrogen
-13.6eV
-increase n=energy closer to zero; at zero=ionized and add 13.6eV
-lowest energy AND most likely state/where electron spends most time
- -3.4eV=first excited state; -1.5eV=2nd excited state, etc.