Alkene reactions
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
How do you test for the presence of a C=C double bond (unsaturation), and what is the observation?
Add bromine water (orange/brown) to the sample. If a C=C double bond is present, the bromine adds across the double bond in an addition reaction and the orange colour is decolourised (the solution turns colourless). Alkanes do not react with bromine water, so their bromine water solution remains orange
Why are alkenes more reactive than alkanes?
The π bond in the C=C double bond has a relatively low bond enthalpy compared to the σ bond. It involves less effective sideways orbital overlap, making it easier to break. The high electron density of the π bond above and below the molecule also makes alkenes attractive targets for electrophiles (electron-pair acceptors). Alkanes have only strong σ bonds and no easily accessible electron density, so they are much less reactive.
What is an addition reaction?
An addition reaction is one where two molecules combine to form a single product, with nothing else produced. In alkenes, the π bond breaks and atoms from the adding molecule bond to each of the two carbons of the former double bond.
What happens when an alkene reacts with hydrogen, and what are the conditions?
Alkenes react with hydrogen (H₂) in a hydrogenation reaction to form an alkane.
The conditions are a nickel catalyst at approximately 150°C. Example: CH₂=CH₂ + H₂ → CH₃–CH₃ (ethene → ethane).
This reaction is used industrially to harden vegetable oils to make margarine.
What happens when an alkene reacts with a halogen (e.g. Br₂), and what is the product?
Alkenes react with halogens (Cl₂ or Br₂) at room temperature to form a dihaloalkane, where one halogen atom adds to each carbon of the former double bond. Example: CH₂=CH₂ + Br₂ → CH₂Br–CH₂Br (ethene + bromine → 1,2-dibromoethane). This is the basis of the bromine water test for unsaturation.
What happens when an alkene reacts with a hydrogen halide (e.g. HBr), and what is the product?
Alkenes react with hydrogen halides (HCl, HBr, HI) at room temperature to form a haloalkane. One H and one halogen atom add across the double bond. Example: CH₂=CH₂ + HBr → CH₃–CH₂Br (ethene → bromoethane).
If the alkene is a gas, the two gases are mixed; if the alkene is liquid, the HX is bubbled through it.
What happens when an alkene reacts with steam, and what are the conditions?
Alkenes react with steam (H₂O(g)) in a hydration reaction to form an alcohol. The conditions are a phosphoric acid (H₃PO₄) catalyst and high temperature and pressure.
Example: CH₂=CH₂ + H₂O → CH₃CH₂OH (ethene → ethanol). This is the industrial method for producing ethanol.
What is an electrophile?
An electrophile is an electron pair acceptor — a species that is attracted to regions of high electron density and accepts a lone pair or bonding pair to form a new bond.
Why are alkenes attracted to by electrophiles?
The π bond creates a region of high electron density above and below the C=C bond. This attracts electrophiles (electron-deficient species), which are drawn to this electron-rich region. The π bond then acts as the electron donor and breaks to form a new bond with the electrophile.
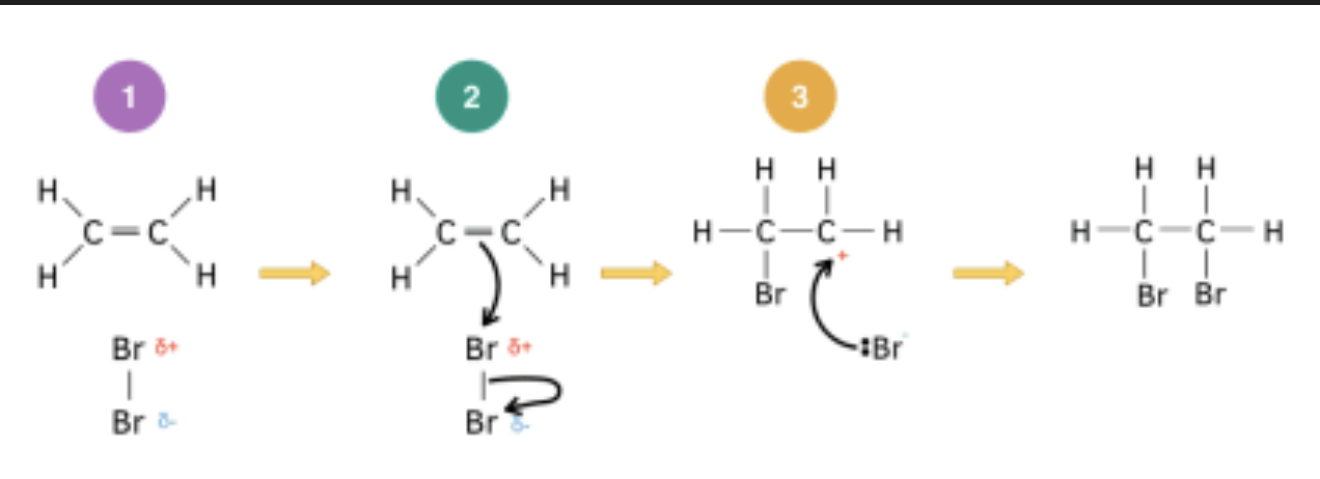
Describe the mechanism for the electrophilic addition of Br₂ to ethene, including the intermediate formed
As Br₂ approaches the electron-rich π bond, the electron density of the π bond induces a dipole in the Br₂ molecule, making the nearer Br atom δ+.
The π bond electrons attract the δ+ Br atom — a curly arrow goes from the C=C bond to the Br–Br bond, breaking the Br–Br bond by heterolytic fission.
This gives a carbocation intermediate (a positively charged carbon) and a :Br⁻ ion.
In the second step, the :Br⁻ ion donates a lone pair to the carbocation, forming the second C–Br bond. The product is 1,2-dibromoethane.
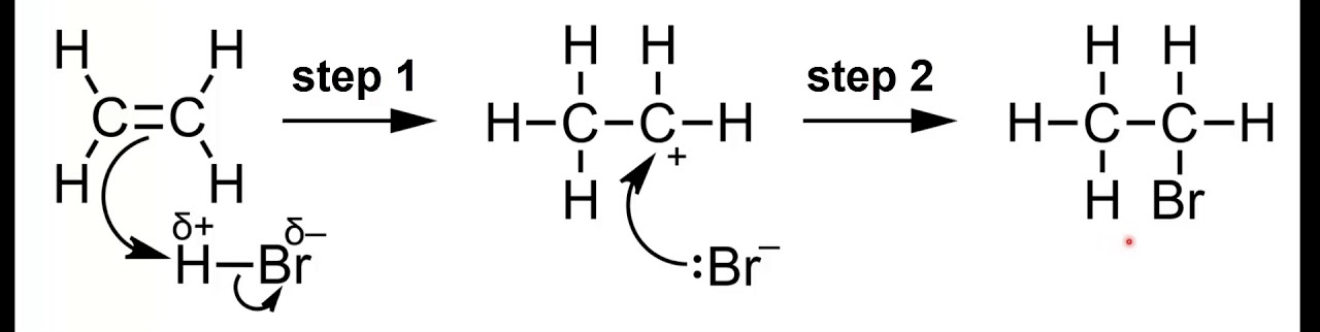
Describe the mechanism for the electrophilic addition of HBr to ethene.
HBr is a polar molecule (Hδ+–Brδ−). In the first step, a curly arrow goes from the π bond electrons to the δ+ H atom, breaking the H–Br bond heterolytically. This forms a carbocation on one carbon and :Br⁻. In the second step, :Br⁻ donates a lone pair to the carbocation to form the C–Br bond, giving bromoethane.
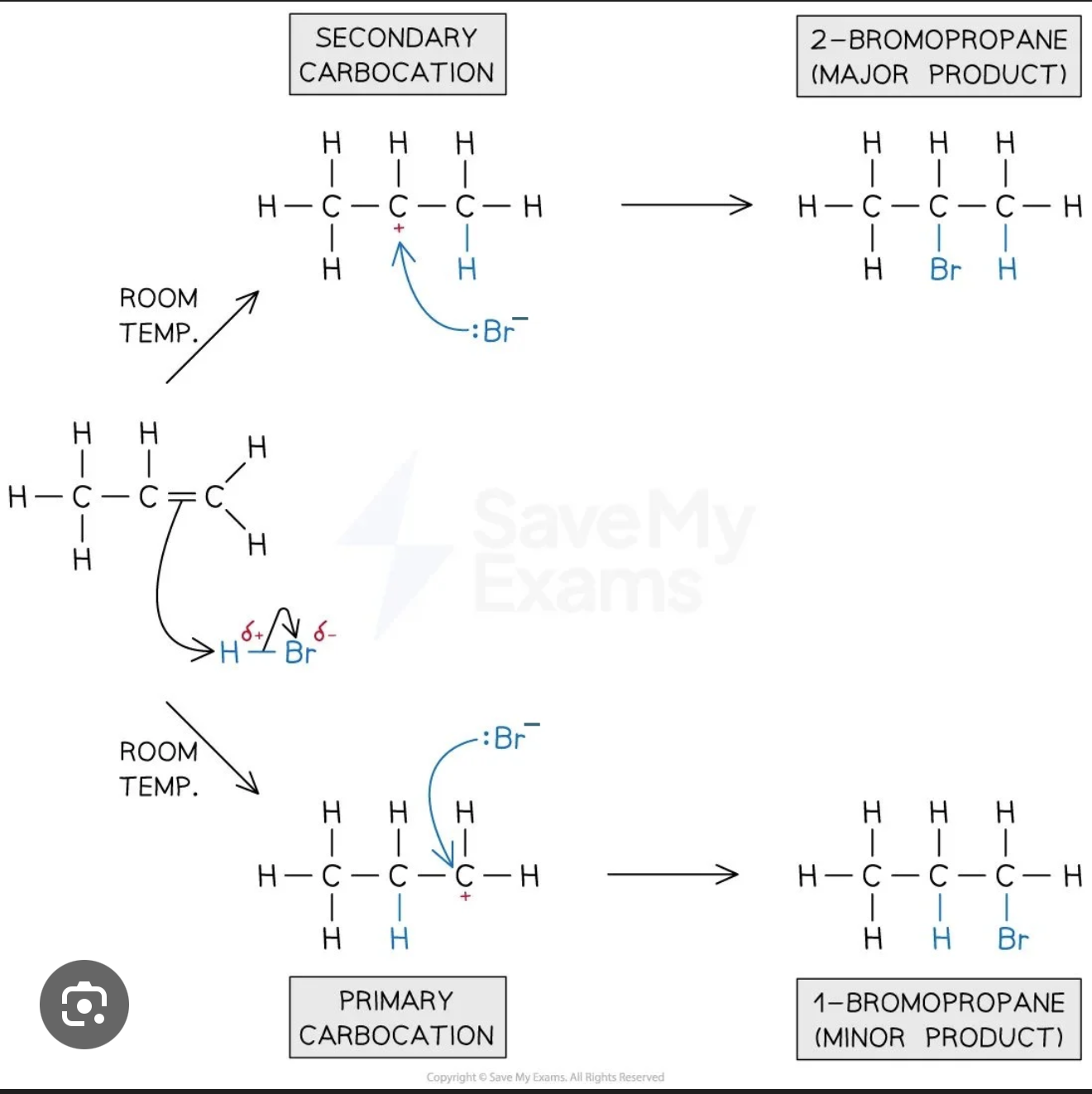
What problem arises when HBr adds to an unsymmetrical alkene such as propene, and what is Markovnikov's Rule?
With an unsymmetrical alkene like propene (CH₃–CH=CH₂), the H could add to either carbon of the double bond, giving two possible products: 1-bromopropane or 2-bromopropane. Markovnikov's Rule states that the major product is the one formed when H adds to the carbon that already has more hydrogen atoms — in other words, H adds to the less substituted carbon of the double bond.
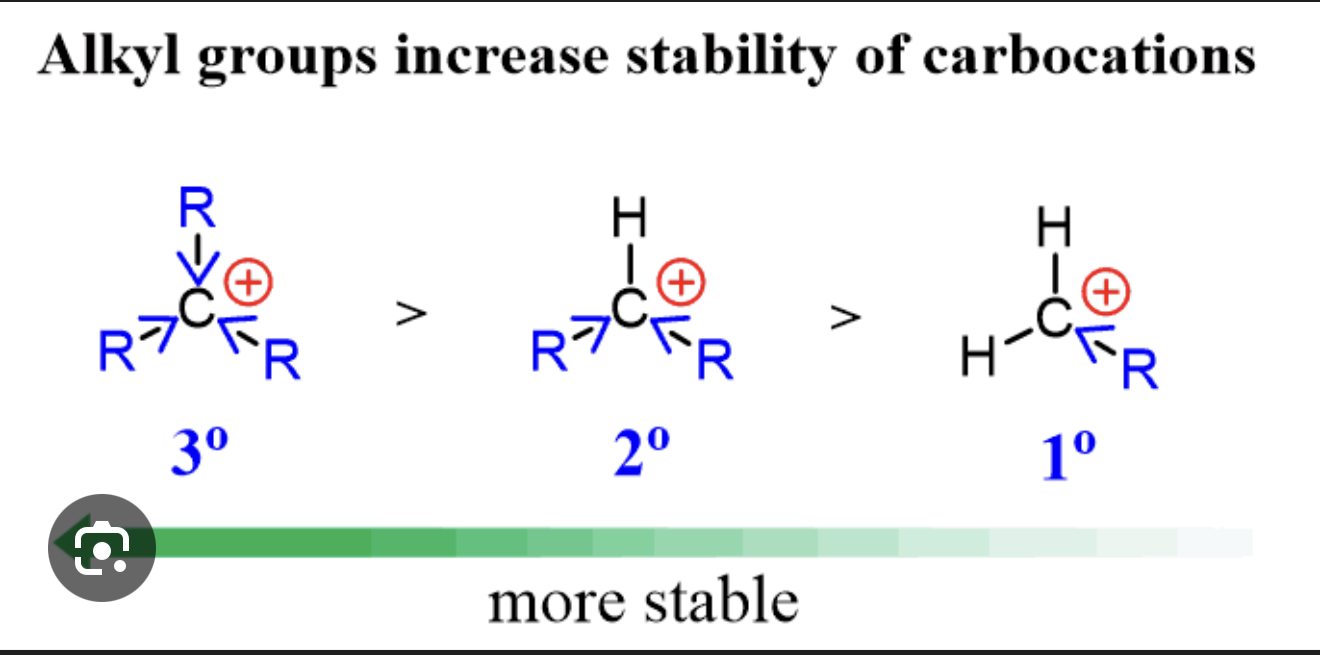
Why does Markovnikov's Rule hold — what is the reason in terms of carbocation stability?
When H⁺ adds to propene, it can form either a primary carbocation (on CH₂, the less substituted end) or a secondary carbocation (on CH–CH₃, the more substituted end). Secondary carbocations are more stable than primary ones because the extra alkyl groups attached to the positive carbon donate electron density towards it, helping to spread and stabilise the positive charge, same for tertiary to secondary. The reaction therefore preferentially proceeds through the more stable secondary carbocation, giving 2-bromopropane as the major product.