Core Idea I - Lipids
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
What elements do lipids contain, and do they contain more oxygen than hydrogen?
Lipids contain carbon, hydrogen and oxygen; lipids contain MUCH LESS oxygen than hydrogen!
What are the 3 types of lipids, and what is one example for each type?
SIMPLE lipids → Fats and oils
COMPOUND lipids → Phospholipids
DERIVED lipids → Sterols
What are the 2 constituent components of lipids?
Fatty Acids and Glycerol.
What do fatty acids contain?
A fatty acid is a molecule containing a long hydrocarbon chain (made up of ONLY hydrogen + carbon), with a carboxyl group (—COOH) at one end.
What is the general formula of fatty acids?
General formula is R-COOH, where R is the long hydrocarbon chain and -COOH is the carboxyl group!
What is the type of molecule fatty acids are classified under? (solubility in water)
The fatty acid is an AMPHIPATHIC (sound like amphibian→some regions are philic, some are phobic) molecule as the hydrocarbon chain is NON-POLAR and HYDROPHOBIC, while the carboxyl group is POLAR and HYDROPHILIC
[Since water is POLAR, it wants to react with other polar molecules!]
Hence, are fatty acids soluble in water? Why or why not?
No, fatty acids are largely insoluble in water. This is because of fatty acids’ long hydrocarbon chains that are HYDROPHOBIC!!!!!!!
What are some differences between the different fatty acids?
They have hydrocarbon chains of different lengths, and saturation. [They usually have 14-22 carbon atoms] [Presence/Absence of C=C]
How does the length of the fatty acids chain affect their melting point?
Fatty acids that have ⬇shorter hydrocarbon chains⬇ have 🌊less hydrophobic interactions🌊 and therefore they have 🫠lower melting points🫠. [less surface area between the shorter chains!!]
What are the differences between saturated and unsaturated fatty acids?
Saturated fatty acids:
Saturated fatty acids are 🧑🤝🧑NOT KINKED (straight)🧑🤝🧑 and thus pack more tightly [🔼more hydrophobic reactions🔼], therefore they tend to have higher melting points!
❌No C=C bond❌!!
Unsaturated fatty acids:
Unsaturated fatty acids are 👬KINKED (not straight)👬, resulting in 👎loose packing of the fatty acids👎, thus they tend to have 🥶lower melting points🥶!
These kinks are caused by the C=C bonds! [every C=C bond has a BEND!!!!]
What is the formula of Glycerol?
C3H8O3.
Is glycerol soluble in water? Why or why not?
Glycerol is soluble in water, because it contains 3 hydroxyl groups (—OH) that can form HYDROGEN bonds with 🌊water🌊. (—OH groups are polar → interact with water (polar))
What does each triglyceride consist of?
Each triglyceride consists of 3 hydrocarbon chains [⭐no longer fatty acids in triglyceride⭐] and 1 glycerol molecule.
How is a triglyceride formed?
3 fatty acid molecules will react with 1 glycerol molecule via the condensation reaction.
Afterwhich, the hydroxyl group (—OH) of glycerol will react with the carboxyl group (—COOH) of each fatty acid.
This forms 3 ➡ester bonds⬅, and the 🗑removal of 3 water molecules🗑.
What is involved in the breakdown of triglycerides?
The 💔breakage of 3 ester bonds💔 via hydrolysis reaction, with the ➕addition of 3 water molecules➕.
At room temperature, what state do triglycerides exist in? Is there a difference between saturated and unsaturated fats?
They can exist in different physical states at 🌡room temperature🌡, such as solid (e.g. butter, Fats), or liquid state (e.g. oils)
Saturated → Soild and Unstaurated → Liquid
What is the melting point of a triglyceride determined by?
👆Longer hydrocarbon chains👆, they have more 🥹hydrophobic interactions🥹 between the chains; leading to a higher melting point.
Saturated fats have a higher melting point as compared to unsaturated fats; given that saturated fats are ➡packed more tightly➡ as they are not KINKED!
Are triglycerides soluble in water? Are there any exceptions?
No they are ❌insoluble in water❌! [However, they are soluble in nonpolar solvents, like ether and chloroform!]
Hence, how are tryglerides stored?
They are stored as 💧droplets in cells💧 → Given that they do not affect the water potential in cells!
As they are ❌insoluble in water❌, fats are prevented from ➡diffusing out of the cells➡.
[NOTE: Their insolubility in water makes triglycerides a GOOD energy store]
Does oil float on water? Why or why not?
Yes it does; given that triglycerides have ⬇lower density than water⬇.
How is having triglycerides beneficial to animals that are:
Aquatic
Living in cold tundra
Living in a hot desert
Triglycerides are ⬇less dense than water⬇, hence this provides 🦭aquatic animals🦭 with buoyancy in water.
Triglycerides that are stored in 👅animals’ adipose tissue👅 are 👎poor conductors of heat👎, providing these animals with thermal insulation.
When ☁oxidized☁, triglycerides can produce 🥈twice as much metabolic water🥈 per unit mass, as compared to an equivalent mass [must state the CONSTANT variable] of carbohydrates when ☁oxidized☁. Hence, this can help provide a useful source of water to animals living in areas that are 🥵water-scarce🥵.
[HOWEVER, lipids are a long-term source of energy; carbohydrates are still the most immediate source of energy as they can be easily hydrolysed!!]
Why are triglycerides known as a compact energy source? Why is this so? What is the energy released used to make?
Triglycerides release 🔢twice as much energy🔢 per unit mass as compared to an equivalent mass [must state CONSTANT variable] of carbohydrates, as it has 🔼more C—H bonds🔼 than carbohydrates.
This energy released is used to ⚡make ATP⚡.
How can using triglycerides as a energy source be beneficial for animals that move by speed/flight, and plants that have seeds that are to be dispersed by wind?
Triglycerides have ➗about half the mass of carbohydrates➗ for an equivalent amount of energy stored [RMB TO STATE CONSTANT VARIABLE]; it is therefore a 🪶LIGHTWEIGHT🪶 energy source for these organisms.
How are triglycerides benificial in protection of our organs?
Triglycerides form a 🛡PROTECTIVE layer🛡 around our 🥰delicate internal organs🥰, serving as ⚡ shock absorbers⚡, protecting organs from mechanical damage.
How are phospholipids formed?
It is formed via the condensation reaction between ⛓2 hydrocarbon chains⛓, joined to a glycerol backbone via ➡ester bonds⬅. The third hydroxyl group of the glycerol backbone is joined to a ➖ negatively charged phosphate group➖.
It is basically a triglyceride - hydrocarbon chain + phosphate group.
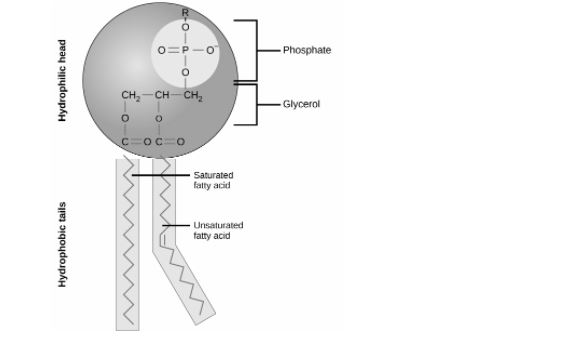
Are phospholipids amphipatic? Why or why not?
They are amphipatic. This is because of its 😄hydrophilic phosphate head😄, and its 😥2 hydrophobic hydrocarbon chains😥.
Why is being amphipatic such an important property for phospholipids?
This is because it is crucial in its role of 🧱forming cell membranes🧱, which is supposed to be a STABLE bilayer and ❌not easily disrupted❌.
What is the fluidity of phopholipid bilayers influenced by?
It is influenced by the factors of 🌡temperature🌡and degree of saturation.
Unsaturated fatty acids have ⛓💥kinks, increasing fludity⛓💥.
What structure has phospholipids as its main component?
(Cell) membranes.
What does the amphiphatic nature of phospholipids give rise to?
It gives rise to the selective permeability of the 🧱cell membrane🧱. The 😭hydrophobic core😭 of the phospholipid bilayer ❌prevents the free passage❌ of ⚡polar molecules and charged ions⚡. This regulates the entry and exit of substances.
The memberane provides a 🛡boundary🛡 between the internal and external environements.
The 🧱membranes of cellular organelles🧱 allow compartmentalisation within the cell. [protect the cell and increase efficiency]
What does the fluid nature of phospholipid bilayers allow for?
It allows 🧍individual phospholipid molecules🧍 to 🏃move freely🏃. Along with its flexibility and fluidity, this allows for 💪cell growth [++ phospholipids], exocytosis and endocytosis💪.
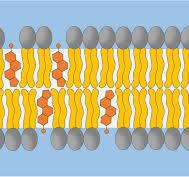
What does cholesterol consist of?
It consists of 💍4 interconnected hydrocarbon RINGS💍 with a hydroxyl group at one end, along with one more ⛓hydrocarbon chain⛓.
What can cholesterol be found in? What is it needed to synthesize?
It is a crucial component of 🧱cell membranes🧱. It is also required to synthesize bile acids, vitamin D and steroid hormones.
Is cholesterol amphiphatic? How is this property useful?
Yes, it is 🤯amphipatic🤯. It is crucial for regulating the 🌊fluidity and stability🌊 of the cell membrane.
It aligns itself with phospholipids, where the 💍hydrophobic rings💍 interact with the ⛓ hydrophobic hydrocarbon chains⛓ of phospholipids.
On the other hand, the 👯hydroxyl group👯 faces outward to interact with the 🚿hydrophilic phosphate head groups🚿 of phospholipids, and the surrounding aqueous environment.
[Hydrophobic + Hydrophobic, and vice versa!]
What does cholesterol maintain in the membrane?
It helps to maintain membrane 👍fludity and stability👍, as it can interact with the phospholipids!
a. At 🥶low temperatures🥶, cholestrol prevents phospholipids from ➡packing too closely⬅, preventing the membrane from becoming too rigid.
b. At 🥵high temperatures🥵, cholesterol reduces the ➡lateral movement (move sideways)➡ of phospholipids, preventing the membrane from becoming too fluid.
What does cholesterol help to improve in the membrane?
Cholesterol helps to ✨improve membrane impermeability✨, by preventing leakage of the small molecules.
What else does cholesterol help to improve in the membrane?
Cholesterol helps to improve ☮stability of the membrane☮, due to the 💪RIGID hydrocarbon rings💪.