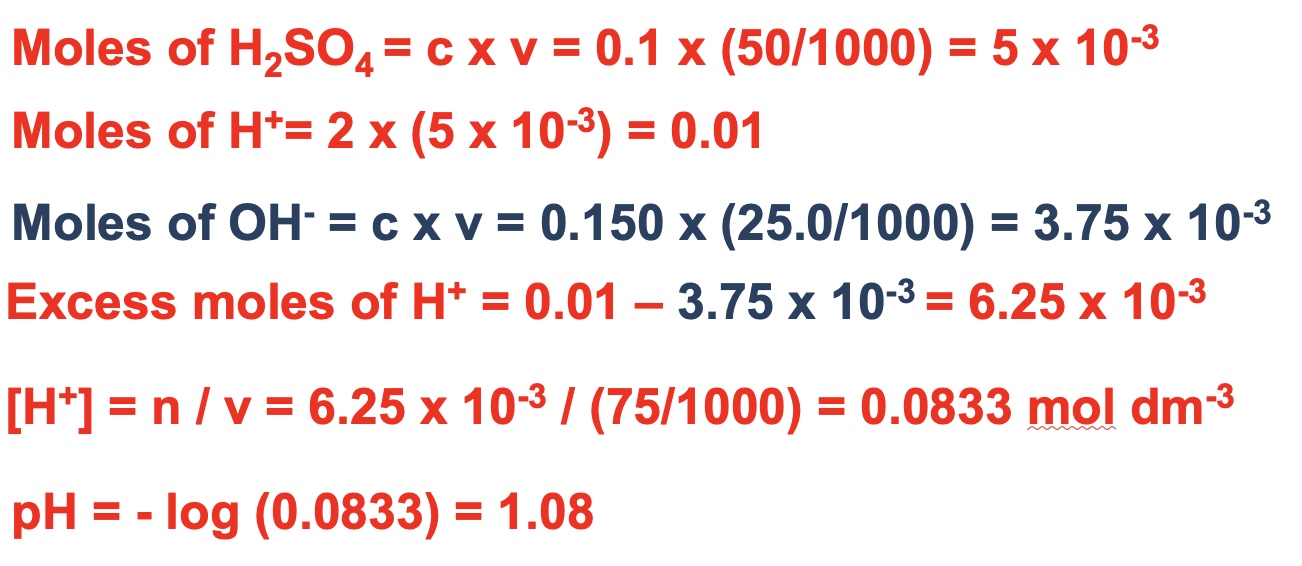3.3.12.3 the ionic product of water, Kw
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
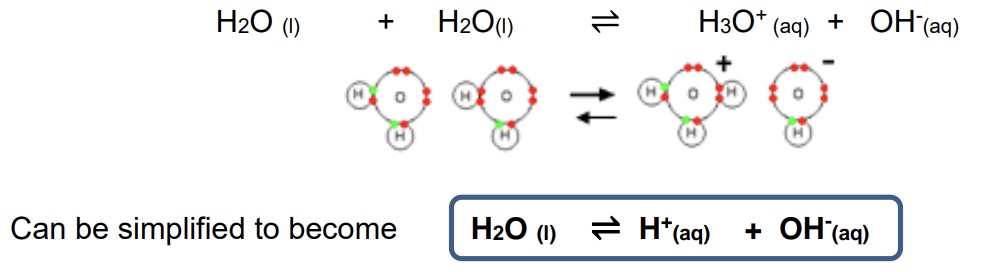
because water can act as both an acid + base, what does this mean?
that both hydrogen ions + hydroxide ions exist simultaneously in water according to the equilibrium
give the full + simplified equation of the equilibrium of hydrogen + hydroxide ions
as this is an equilibrium reaction, what can be derived?
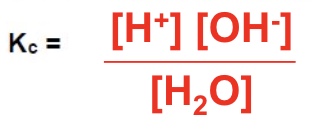
an equilibrium constant, Kc
give the expression of Kc
rearrange the Kc expression
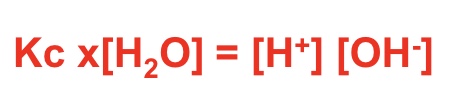
where does equilibrium lie to + why?
as water is only weakly dissociated, the equilibrium position lies far over to the left
explain what Kw is + how it is formed
due to the equilibrium lying to the left, the concentration of water is very high + can be taken to be constant + its value is incorporated into Kc
the new constant resulting is called the ionic product of water, Kw
give the Kw expression, its conditions + units
Kw = [H⁺][OH⁻]
where at 298K (25℃), Kw is 1.00 ×10⁻¹⁴
units: mol²dm⁻⁶
outline why pure water is neutral
because the hydrogen ion concentration is equal to the hydroxide ion concentration → for every hydrogen ion formed, there is a hydroxide ion formed as well
[H⁺] = [OH⁻]
because in pure water [H⁺] = [OH⁻], what does this mean you can do to the Kw expression?
this means you can replace the [OH⁻] term in the Kw expression by another [H⁺]
so give the Kw expression used for pure water
Kw = [H⁺]²
example:
calculate the pH of pure water at 298K when Kw = 1.00 ×10⁻¹⁴ mol²dm⁻⁶

the value of Kw is _______ dependent
temperature
how is Kw affected by temperature?
it increases with temperature
explain why Kw increases with temperature

what is meant by monoprotic + diprotic bases?
monoprotic base: base that can accept one proton eg NaOH, KOH
dirprotic base: base that can accept two protons eg Ba(OH)₂
give the steps for calculating the pH of a strong base
use concentration of base to find [OH⁻]
use Kw to find [H⁺]
[H⁺] = Kw/[OH⁻]
convert [H⁺] into pH
pH = -log[H⁺]
for example, calculate the pH go a 1.00moldm⁻³ solution of NaOH at 298K

for example, calculate the pH of [Ba(OH)₂] which has a concentration of 0.0500 moldm⁻³ at 298K
because barium hydroxide produces 2 OH- ions, the concentration of the base should be multiplied by 2 to find [OH⁻]
![<p>because barium hydroxide produces 2 OH- ions, the concentration of the base should be multiplied by 2 to find [OH⁻]</p>](https://knowt-user-attachments.s3.amazonaws.com/24b8f31b-7750-4c14-ab0b-5cfec399fe19.jpg)
give the steps of finding the [OH⁻] from the pH (+ so the conc of the base)
use pH to find [H⁺]
[H⁺] = 10⁻pH
use Kw to find [OH⁻]
[OH⁻] = Kw/[H⁺]
use [OH⁻] to find the concentration of the base
for example, calculate the concentration of a solution of NaOH at 298K with pH = 12.80

for example, find the concentration of Ba(OH)₂ with pH 13.30 at 298K
[OH⁻] is divided by 2 to find concentration of base
![<p>[OH⁻] is divided by 2 to find concentration of base </p>](https://knowt-user-attachments.s3.amazonaws.com/91ebc604-260f-4ee1-84c9-a3eb6fc7fab6.jpg)
calculate the pH of a solution formed by a dded 5.82g of NaOH to 250cm³ water
find moles
use moles/volume to calculate concentration of solution + so [OH⁻]
find [H⁺]
find pH
![<ul><li><p>find moles </p></li><li><p>use moles/volume to calculate concentration of solution + so [OH⁻]</p></li><li><p>find [H⁺]</p></li><li><p>find pH </p></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/5cf095bc-6ca6-46c5-bf9e-b9fa94152bab.jpg)
what does dilution refer to?
the addition of water
give the steps for calculating the pH of a strong acid after it has been diluted with water
calculate moles of H⁺
find new [H⁺] by dividing moles by new volume (volume of acid + water)
pH = -log[H⁺]
for example, calculate the pH of the solution when 50cm³ of water is added to 50cm³ of 0.20 moldm⁻³ HCl
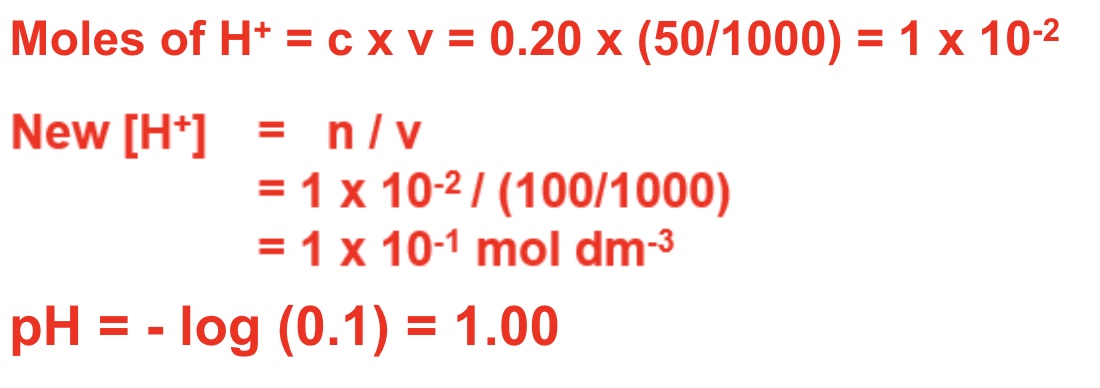

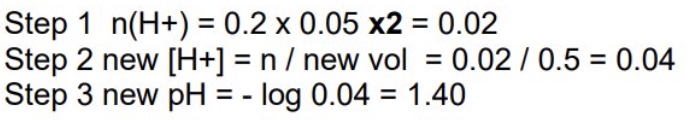

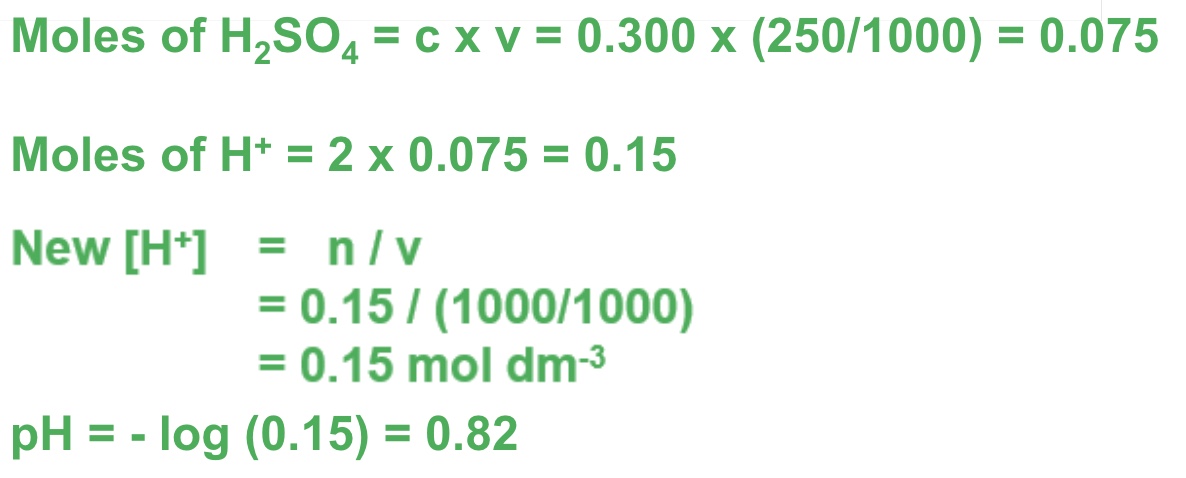
give the steps for calculating the pH of a strong base after it has been diluted with water
calculate moles of OH⁻
find new [OH⁻] by dividing moles by new volume (volume of hydroxide + water)
[H⁺] = Kw/[OH⁻]
pH = -log[H⁺]
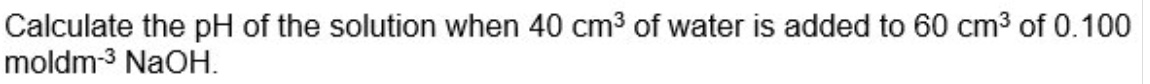
for example


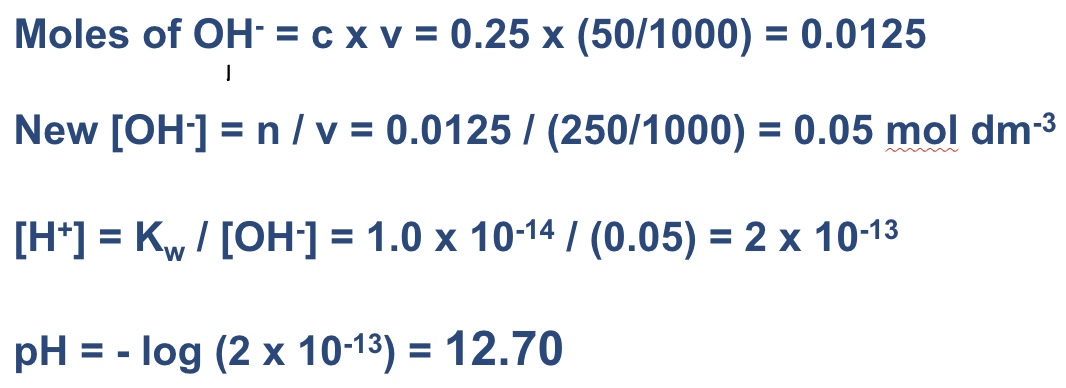




outline the steps for working out the pH of a solution from a neutralisation reaction where either the acid or alkali is in excess (mixtures)


example 1


example 2


example 3